Abstract
Outpatient parenteral antimicrobial therapy (OPAT) has been widely used in clinical practice for many decades because of its associated cost savings, reductions in inpatient hospital days, and decreases in hospital-associated infections. Despite this long history, evolving practice patterns and new drug delivery devices continue to present challenges as well as opportunities for clinicians when designing appropriate outpatient antimicrobial regimens. One such change is the increasing use of extended and continuous infusion (CI) of antimicrobials to optimize the achievement of pharmacokinetic and pharmacodynamic targets. Elastomeric devices are also becoming increasingly popular in OPAT, including for the delivery of CI. In this article, we review the clinical evidence for CI in OPAT, as well as practical considerations of patient preferences, cost, and antimicrobial stability.
Keywords
Introduction
Outpatient parenteral antimicrobial therapy (OPAT) is the administration of parenteral antimicrobial therapy in at least two doses on different days outside of an acute hospital setting. 1 For OPAT in the home, administration is usually undertaken by the patient or a caregiver. Therefore, a simpler, less frequently administered antimicrobial regimen is most likely to garner adherence.1,2 However, several antimicrobials that may be selected for a patient requiring OPAT are traditionally dosed multiple times a day to meet the required pharmacokinetic and pharmacodynamic (PK-PD) targets to treat pathogens. One possible option to leverage less-frequent administration and maximize the chances of meeting PK-PD targets is continuous infusion (CI). In this article, we review the evidence supporting the use of antimicrobial agents often given by CI in OPAT, their stability in available delivery devices, patient satisfaction, and cost considerations. We also discuss clinical controversies in CI and evidence for CI use with newer antimicrobial agents.
Practical considerations
Patient/caregiver acceptance
In some cases, CI may be less burdensome than intermittent infusions for OPAT. This is particularly true for medications that are traditionally administered 4–6 times daily and cannot be administered via intravenous (IV) push. CI may allow for a dose administration just once daily, and medication delivery devices can allow patients to be fully ambulatory during the infusion. CI does require patients to be connected to an infusion all day, however, and may introduce compatibility concerns if other concomitant medications need to be administered.
Common delivery devices used for CI include ambulatory electronic infusion pumps and elastomeric devices. Ambulatory electronic infusion pumps (Figure 1), or
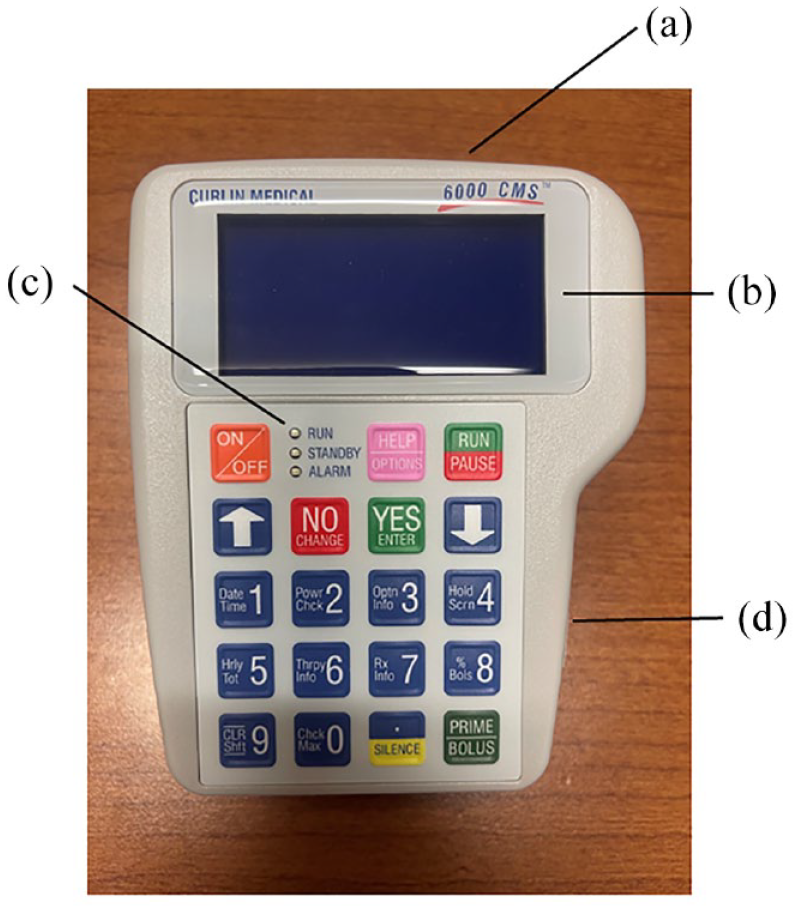
Ambulatory pump: (a) pump door for installation of tubing, (b) display screen, (c) LED light indicators, and (d) battery compartment on back (two size C batteries).
Elastomeric devices (Figure 2) are medication containers made of polyvinyl chloride, and when filled with medication they resemble a ‘ball’ or ‘grenade’. These devices use a patented membrane technology that creates a positive pressure system, which allows for consistent and uniform drug delivery. The tubing is already attached and there is no need for gravity, which makes these devices relatively easy to use. 3 Use of elastomeric devices has been shown to be preferred by patients, 5 but this may come at the expense of a higher margin of error (15%) because flow rates depend on fluid viscosity and temperature. 4
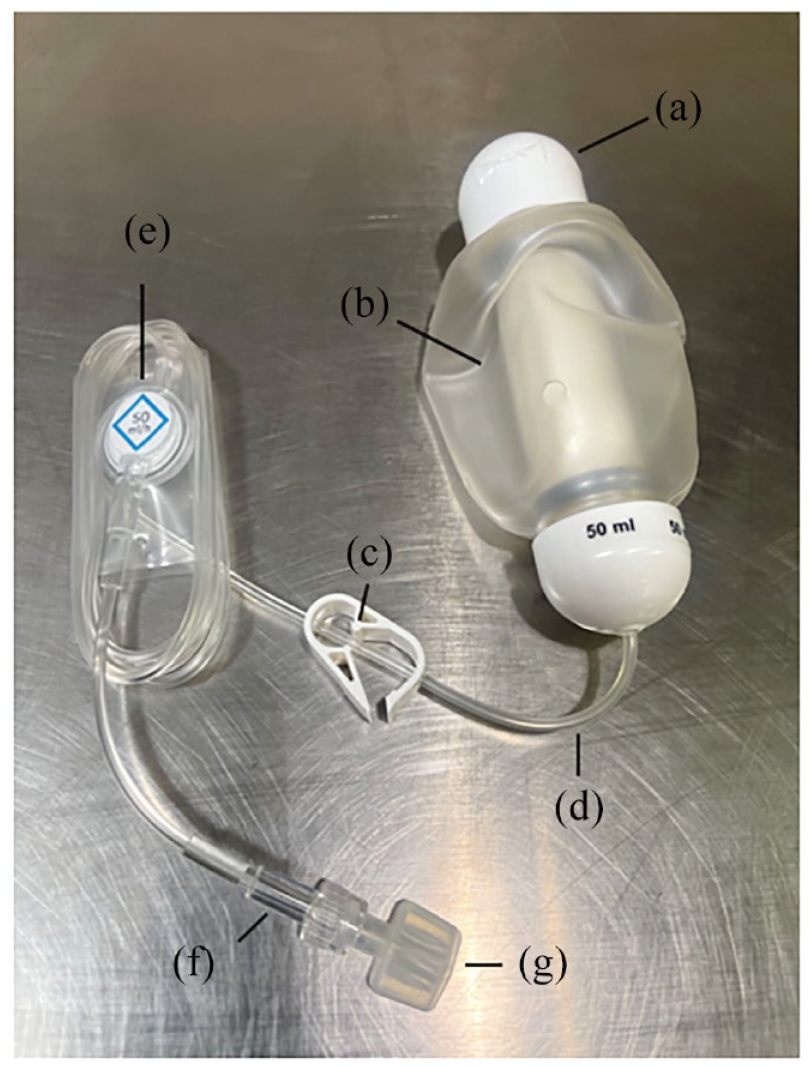
Elastomeric device: (a) filling port, (b) medication container with elastomeric membrane (deflated), (c) clamp, (d) tubing, (e) particulate- and air-eliminating filter with infusion rate indicator, (f) flow restrictor, and (g) Luer-Lok tip.
Cost
Multiple studies have demonstrated the cost-effectiveness of OPAT compared with inpatient treatment. One study showed the cost-effectiveness of antimicrobials (including nafcillin for 32% of the cohort; penicillin G, 34%; piperacillin/tazobactam, 13%; cefazolin, 17%; and cefepime, 4%) administered via disposable elastomeric CI pumps for 91 patients treated in the outpatient setting. 6 The use of self-administered elastomeric CI pumps for a total of 1925 OPAT days resulted in cost avoidance of $2.3 to $3.5 million relative to an inpatient-only treatment course. 6 A study in Spain among patients receiving CI OPAT via elastomeric devices (with piperacillin/tazobactam, 43%; meropenem, 20%; ceftazidime, 25%; ceftolozane/tazobactam, 3%; and cloxacillin, 2%) showed a similar cost avoidance of €745,290.55 associated with 1409 OPAT days compared with inpatient days. 7
Data are limited regarding cost comparisons among various OPAT delivery methods. A recent cost-minimization analysis based on OPAT practice models in the United Kingdom showed similar costs associated with CI via elastomeric devices compared with bolus dosing via elastomeric devices.
8
However, the use of elastomeric devices had higher costs than bolus dosing via gravity infusion.
8
The CHID (Comparing Home Infusion Devices) study protocol included pump operating costs of electronic
Stability
The stability of sterile antimicrobial preparations is typically determined under controlled conditions at room temperature (25°C) or with refrigeration (5°C). The actual conditions encountered in a patient’s home environment may vary substantially, particularly in warm climates and with prolonged infusions during which elastomeric devices may be kept close to the body and reach temperatures between 30°C and 37°C.10,11 In a study that examined temperature variations associated with the use of CI antimicrobials via elastomeric devices, the use of a white carrying pouch prevented excessive temperature increases. 11
Data on the stability of medications under these warmer conditions (30°C–37°C) are currently limited, but some suggest that ceftazidime and meropenem may be physically or chemically unstable at these warmer temperatures during prolonged infusion.12,13 Although some data support the safety and efficacy of ceftazidime via CI, as described below, it does degrade to a potentially toxic by-product, pyridine, which requires careful safety consideration to limit its accumulation. Jones
Available stability data for agents included in this review are summarized in Table 1 (plastic containers) and Table 2 (elastomeric devices). Medications being considered for CI should ideally have data to support stability at 25°C or higher for no less than 24 h. Preparations that do not meet these criteria are highlighted in gray in the tables.
Stability in plastic containers. a

Shading indicates preparations that do not have data to support stability at 25°C or higher for ⩾24 h.
Theoretical or established risk of instability at temperatures ⩾30°C.12,13
d, day; D5W, dextrose 5% in water; EVA, ethylvinyl acetate; h, hour; NA, not available; NS, normal saline; PVC, polyvinyl chloride; SWFI, sterile water for injection.
Stability in elastomeric devices. a
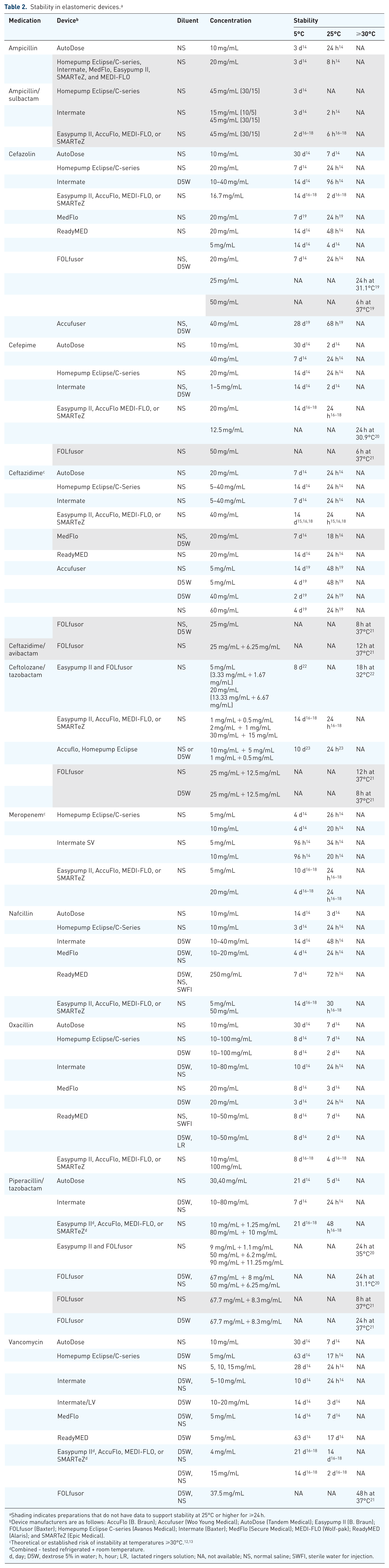
Shading indicates preparations that do not have data to support stability at 25°C or higher for ⩾24 h.
Device manufacturers are as follows: AccuFlo (B. Braun); Accufuser (Woo Young Medical); AutoDose (Tandem Medical); Easypump II (B. Braun); FOLfusor (Baxter); Homepump Eclipse C-series (Avanos Medical); Intermate (Baxter); MedFlo (Secure Medical); MEDI-FLO (Wolf-pak); ReadyMED (Alaris); and SMARTeZ (Epic Medical).
Theoretical or established risk of instability at temperatures ⩾30°C.12,13
Combined - tested refrigerated + room temperature.
d, day; D5W, dextrose 5% in water; h, hour; LR, lactated ringers solution; NA, not available; NS, normal saline; SWFI, sterile water for injection.
Outcomes associated with CI antimicrobial use
Penicillins
CI penicillins have been used for serious invasive diseases such as central nervous system infections and enterococcal endocarditis.24–26 In one study of patients with central nervous system infections (meningitis, brain abscesses, and neurosyphilis), cure was noted all for patients with penicillin delivered as CI via ambulatory electronic infusion pump. 26 More recently, penicillin was shown to be frequently prescribed and administered via elastomeric systems for neurosyphilis and other diseases, which resulted in substantial institutional cost savings. 6
Ampicillin, which has more limited stability than penicillin, has been less frequently used in elastomeric systems and may require the use of ambulatory electronic pump systems. Some stability data are available for ampicillin in an elastomeric device as well as in a plastic container (72 h refrigerated; 20 mg/mL concentration).27,28 The ability to achieve large desired daily ampicillin doses (8–12 g/day), however, is hindered by available elastomeric device sizes and the need to provide more frequent home deliveries. In contrast, IV amoxicillin (although unavailable in the US) has been shown to be stable in such devices and has successfully treated
Data indicate that ampicillin-sulbactam also possesses limited stability with elastomeric systems. The limited stability of both ampicillin and ampicillin-sulbactam formulations must be considered when making treatment-related decisions, given that it takes extra time for compounding and delivery of OPAT medications. This compounding issue has limited their routine inclusion in the OPAT home infusion models and formularies.
1
However, data on the extended stability of ampicillin with and without sulbactam may support its use with ambulatory electronic pump-based CIs.
30
This method, although currently lacking published efficacy data, may allow for the provision of OPAT to treat deep-seated enterococcal infections and multidrug-resistant (MDR)
Historically, antistaphylococcal penicillins such as nafcillin, as well as isoxazolyl penicillins (e.g. oxacillin, cloxacillin, and flucloxacillin), have been used for deep-seated methicillin-susceptible
Piperacillin-Tazobactam
The short interval of traditional piperacillin-tazobactam dosing makes this agent less desirable for routine use in OPAT other than with CI. Piperacillin-tazobactam has been widely included in published reviews of the efficacy and safety of elastomeric pump systems in OPAT programs.6,7,32 It is important to note that rates of use of piperacillin-tazobactam varied throughout these reports (8−43%), but clinical success rates were high, with these investigations demonstrating approximately 84–95% cure rates. One study demonstrated mean (SD) serum piperacillin concentrations of 25.8 (15) mg/L. This resulted in advantageous serum concentration ratios of piperacillin relative to the epidemiologic cutoff values of the identified bacteria, which were one or greater in 12 of 14 assessments (86%). 32 The specific antimicrobial doses and tazobactam concentrations were not reported.
The safety and efficacy of CI piperacillin-tazobactam has been reported for
Cephalosporins
Cefazolin, a first-generation cephalosporin, has recently become more prevalently used in lieu of traditional nafcillin or oxacillin-based therapies for the treatment of invasive MSSA infections, which has prompted multiple reviews and meta-analyses.35–39 Traditionally, cefazolin has been administered as an every-8-h infusion with dose adjustment for kidney dysfunction. It has demonstrated stability in both elastomeric and electronic pump-based systems (Tables 1 and 2). Given the favorable results of clinical studies, as well as OPAT-related data demonstrating reduced tolerability of nafcillin-based regimens, CI cefazolin may present clinicians with an evidence-based, better-tolerated treatment alternative. Cefazolin has comparable, if not improved, clinical outcomes regarding patient tolerability compared with nafcillin-based therapy.40–42 These clinical data are supported by PK data in 100 patients with difficult-to-treat bone and joint infections. 43 Cefazolin demonstrated excellent median (range) serum cefazolin concentrations [60.75 (13–203) mg/L] when compared with the 90% minimum inhibitory concentration (MIC) in this study (1 mg/L). The median bone-to-serum concentration ratio of cefazolin was 0.25 (range, 0.06–0.41) in eight patients.
Later-generation cephalosporins, specifically ceftazidime and cefepime, have also been used as CI in acute care settings, mostly to optimize PK-PD target attainment. Efficacy and safety data with ceftazidime when administered as CI have been available since the late 1980s for patients with cystic fibrosis and
Substantial effort has been expended toward optimizing the PK-PD parameters of cefepime, often with extended or CI regimens.
47
In the OPAT setting, CI cefepime has limited but promising efficacy data. In a study by Voumard
Clinical controversies in CI
Management of staphylococcal infections
In recent years, increasing reports of intolerance and adverse events with nafcillin, when compared with agents such as oxacillin or cefazolin, have prompted debate regarding preferred therapies for OPAT. One 5-year study showed significantly increased rates of premature antimicrobial discontinuation with nafcillin
Subsequently published studies, in both admitted patients with MSSA endocarditis and OPAT-treated patients, have reported similar findings.40,41 Interestingly, nafcillin had increased rates of nephrotoxicity, with nearly a threefold increased rate of acute kidney injury compared with oxacillin-based treatments (18%
Management of enterococcal endocarditis
The 2015 IDSA guidelines recommend ampicillin plus either gentamicin or ceftriaxone for up to 6 weeks for enterococcal endocarditis. 53 However, the frequency of administration and short stability at room temperature have presented challenges for the transition of ampicillin into the outpatient setting.
Ampicillin is a β-lactam antibiotic that exhibits time-dependent PD activity, and because β-lactams lack bactericidal activity for
Clinical data evaluating CI ampicillin for enterococcal infections are limited. In one study, patients with enterococcal endocarditis were treated with ceftriaxone 4 g administered via short infusion over 30 min in combination with ampicillin 12 g daily in 500 mL of NS solution delivered as 2 g over every 4 h via programmable pump. 57 For the four patients who met the inclusion criteria, with a median of 22.5 days of outpatient therapy, all patients achieved clinical and microbiologic cure without recurrence or complications in the subsequent year. 57 Although guidelines recommend a dosage of ceftriaxone 2 g every 12 h for the treatment of enterococcal endocarditis, the dosing strategy that best optimizes synergy with ampicillin CI remains unknown, which demonstrates the need for additional prospective studies. 53 When using ceftriaxone and CI ampicillin in combination, the ampicillin infusion may be paused for administration of ceftriaxone due to potential concerns of physical incompatibility.
Although the combination of ampicillin and ceftriaxone has emerged as an effective and possibly safer regimen for specific patient populations, a recent case report described successful administration of CI ampicillin in conjunction with once-daily gentamicin for the treatment of enterococcal endocarditis. 58 Ampicillin and gentamicin were compounded daily and administered in an infusion therapy center. After gentamicin administration, ampicillin was set to infuse over the next 22 h via ambulatory infusion pump with battery charging completed overnight. As a result, the patient achieved clinical cure and was without relapse or readmission in the subsequent year. Of 42 days of therapy, 34 days were administered in the ambulatory setting. 58
The utility of TDM for optimization of CI ampicillin has also been investigated. In a study by Gatti
Vancomycin
The first case reports of CI vancomycin were published in the early 1990s.59,60 Since then, interest has increased regarding the use of CI vancomycin to maximize its PK-PD properties, including a potentially faster time to a therapeutic steady state and less variation in levels. The 2019 IDSA update to the vancomycin dosing and monitoring guidelines specifically identified CI vancomycin as an appropriate modality to reach the area under the curve (AUC)-to-MIC targets.
61
Interestingly, for methicillin-resistant
Previous studies assessed CI vancomycin in various patient populations such as critically ill, pediatric, and outpatient populations. Improved clinical efficacy of CI compared with intermittent vancomycin regimens has not been demonstrated, which is expected because the two modalities obtain a similar AUC. The possible benefit of CI vancomycin is largely believed to be a reduced risk of nephrotoxicity. Multiple meta-analyses have evaluated the nephrotoxicity of intermittent infusion
The use of CI vancomycin has many practical aspects that are of value to the clinician. Historically, substantial coordination has been needed between the patient, treating clinician, outside facilities, home healthcare nurses, and infusion pharmacies to ensure that blood samples are collected at an appropriate time for measurement of vancomycin levels. Several factors could potentially create scenarios in which vancomycin trough levels are not captured or are not clinically valid. With CI vancomycin, any random level obtained after 24 h of initiation is assumed to be equivalent to steady-state concentration and can be measured whenever it is most convenient for the patient.
Several studies of CI vancomycin in the adult outpatient population have been performed. In a randomized clinical trial of the outpatient treatment of osteomyelitis in France, CI vancomycin regimens were associated with more predictable serum concentrations and were less likely than intermittent infusions to require dose adjustment. Additionally, patients were less likely to discontinue CI vancomycin because of adverse drug reactions
The increased safety and practicality benefits of CI vancomycin should be weighed against the potential work for the patient or caregiver. Patients receiving OPAT vancomycin in one study stated that receipt of vancomycin moderately affected their daily routines, which was not observed with receipt of daptomycin.
71
Of note, that study did not use CI vancomycin, and patient preference may have differed for CI
Meropenem
The appropriateness of using meropenem for CI has been a clinical challenge given the variable stability data at different concentrations, particularly at the higher concentrations potentially used to achieve typical daily doses.73,74 Data regarding use of CI meropenem demonstrate its stability in both polyvinyl chloride and elastomeric systems, at concentrations up to 20 mg/mL, which would allow for compounding and delivery for in-home administration. 75 Similar to the situation with ampicillin, the lack of sufficiently sized elastomeric systems can hinder the use of larger daily doses (e.g. 6 g/day). Recently, a case series described meropenem compounded as 1% and 1.25% solutions (w/v) used in four patients transitioning from intermittent infusions to CI for OPAT. 76 CIs were coupled with TDM, and all four patients achieved serum drug concentration at least two times the measured or presumed MIC of the pathogen. Meropenem CI is promising and may become increasingly needed in OPAT for MDR organisms, but more research is needed.
CI OPAT experience with select newer antimicrobials
Ceftolozane/Tazobactam
Ceftolozane/tazobactam (C/T), a combination cephalosporin/β-lactamase inhibitor, is an option for certain MDR
PK analyses, case reports, and case series support that C/T can be safe and effective when given as CI. A multicenter prospective cohort study evaluated the percentage of patients reaching PK-PD targets of 100% time-free drug concentration maintained above the MIC (
The first clinical report of C/T administered by CI in the outpatient setting was for a 71-year-old woman with MDR
Two case series from 2020, both including outpatients, describe the use of CI C/T.82,83 One was a single-center retrospective analysis of CI C/T from December 2016 to June 2019 that included inpatients and outpatients.
83
Seven unique CI C/T regimens were used, primarily for deep-seated infections caused by MDR
Furthermore, C/T has been investigated for CI via elastomeric devices (FOLfusor LV10; Baxter Healthcare and Easypump II; B. Braun Medical Ltd) in the UK. 22 C/T, diluted in NS at 5 mg/mL or 20 mg/mL (combined active drugs), degraded during in-use storage at 32°C, with less than 95% of the drugs remaining at 24 h at both concentrations in both devices. Extended storage up to 8 days at 2°C–8°C plus 12 h at 32°C in use with both devices was supported. The authors concluded that C/T can be administered via CI for 12 h at 32°C and as 24-h CI in countries where a 10% loss of C/T is acceptable. 22
Ceftazidime/Avibactam
Ceftazidime/avibactam (C/A), a combination cephalosporin/β-lactamase inhibitor, may be a necessary treatment for complex infections with
In 2021, a case series including PK data detailed the use of C/A via CI for OPAT at a single center in Belgium.
84
It included 10 patients with infections mainly caused by MDR
Antimicrobial CI dosing.

Source: Wilson
Existing evidence is extremely limited for renal dose adjustments of CI antimicrobials. When no evidence exists for drugs with standard renal dose adjustment, a proportionate dose adjustment to the total daily dose when using CI dosing is generally recommended.
AUC, area under the curve; CI, continuous infusion; ClCr, creatinine clearance (mL/min); HAP/VAP, hospital-acquired pneumonia/ventilator-associated pneumonia; IV, intravenous; max, maximum; MDR, multidrug-resistant; MIC, minimum inhibitory concentration; NS, normal saline (0.9% sodium chloride); NTM, nontuberculous mycobacteria; UTI, urinary tract infection.
Ceftaroline
Ceftaroline is a cephalosporin, administered as a prodrug (ceftaroline fosamil), that is US Food and Drug Administration approved for complicated skin and skin structure infections and community-acquired bacterial pneumonia at a dosage of 600 mg IV every 12 h infused over 5 min to 1 h for those with creatinine clearance greater than 50 mL/min.
89
Ceftaroline exhibits gram-negative antibacterial and broad gram-positive antibacterial activity, including against methicillin-resistant
With an every-12-h bolus dose regimen option, CI ceftaroline is less compelling from a practical standpoint for OPAT. One study has reported on the stability of ceftaroline in elastomeric infusion devices for CI. 90 Ceftaroline 6 mg/mL in NS or D5W was stable for 144 h at 4°C, 24 h at 25°C, and 12 h at 30°C. At 35°C, ceftaroline was stable for 12 h in NS and for 6 h in D5W. The results supported a 12- or 24-h elastomeric CI of a ceftaroline-NS admixture. However, no known clinical reports of CI ceftaroline use in OPAT exist to date.
Conclusion
New data continue to emerge supporting the use of CI antimicrobials as a safe and effective administration strategy for OPAT; however, its use should be individualized by patient preference, feasibility, cost, and medication stability and supported by clinical outcome data. Multidisciplinary teams including pharmacists, physicians, advanced practice providers, nurses, and others are needed to determine when CI is the preferred OPAT option. On the basis of this review and author experience, CI antimicrobials that are the best fit for the OPAT setting include penicillin, piperacillin/tazobactam, and vancomycin. Ampicillin and meropenem, in contrast, have important stability issues that create a barrier to their use for CI. Areas for future research include the in-use stability of medications in elastomeric devices under various storage conditions and clinical outcome data between CI and traditional intermittent infusions. As demonstrated by recent reports of CI methods with newer antimicrobial agents, CI in the OPAT setting remains an exciting and relevant field.
Footnotes
Acknowledgements
The Scientific Publications staff at Mayo Clinic provided editorial consultation, proofreading, and administrative and clerical support. The authors have authorized Scientific Publications to submit the article on their behalf, and the authors have approved all statements and declarations.
Correction (December 2025):
article updated to include footnote “d” in Table 2 and to correct the links in References 16 and 18.
