Abstract

Introduction
The use of outpatient parenteral antimicrobial therapy (OPAT) as a treatment strategy with the aim of dehospitalizing patients has been growing since its advent during the 1970s. 1 OPAT has become a safe and standardized practice for patients presenting with various infections who require long-term parenteral antimicrobial therapy. International consensus guidelines have determined that OPAT can be performed in ambulatory care clinics, specialized infusion centers, or at home.1–3 In Brazil, initiatives to implement OPAT regimens began in the 2010s, with the publication of national guidelines by the Brazilian Society of Infectious Diseases in 2017. 4
Healthcare structure in Brazil and OPAT
Brazil has a robust public health system, Sistema Único de Saúde (SUS), that provides universal health coverage for every person living in the country, with the Brazilian population estimated at 217 million people in 2023. 5 It is funded by the federal administration but has administrative responsibilities at all levels of government: federal, state, and municipal. The delivery of care is handled at the state and municipal level. Brazilian Constitution defines the universal right to comprehensive care at all levels such as primary, secondary, and tertiary. SUS offers many services free of charge such as prevention services, primary care, outpatient care, inpatient care, maternity care, mental health services, pharmaceuticals, dental care, vision care, and physical therapy for residents and visitors, including undocumented individuals. 6 Home care is also contemplated by SUS through a specific policy called ‘Melhor em Casa’ (Better at Home) that, although it is quite comprehensive, has not yet been implemented in all 5560 municipalities in the country. 7 Despite the scope of the SUS’s activities allowing the performance of OPAT, a specific health policy for its practice has not yet been established.
Private sector is also present at the financing and provision levels of healthcare. Private health insurance (PHI) is voluntary and can be classified as duplicate coverage as it covers medically necessary curative services that are also covered under SUS. In 2019, 24.2% of Brazilians had PHI, while in 2008, this proportion was around 22%, about 50 million people by the current projection of country’s population. 8
In Brazil, OPAT is a treatment option available in both public and private health systems. For services linked to SUS, there is a predominance of use of ambulatory care units and day hospitals for the infusion of antimicrobials. Usually, the patient or their caregivers are responsible for organizing transport to the healthcare unit for the infusion. In the private system, however, there is a predominance of home care as model of choice for OPAT.9–12
Organization of OPAT in Brazil and guidelines
In 2017, the Brazilian Society of Infectious Diseases published the recommendations for performing OPAT in Brazil. This document was prepared by a group of specialists and covers the guidelines for carrying out this treatment modality in the country, including the categories of health professionals necessary for its operation. Multidisciplinary team trained to make evaluations regarding patients’ eligibility for OPAT and to conduct follow-up on this type of therapy. These team should be led by a physician, preferably an infectious diseases specialist with experience in using long-term parenteral antimicrobials. In addition, each team needs to include a nurse with experience in manipulating central venous access and a social worker. A clinical pharmacist may also be included in the team, although this is still an uncommon professional in most Brazilian healthcare services (Table 1).
Professionals required for an OPAT program and their attributes according to the Brazilian guidelines.

OPAT, outpatient parenteral antimicrobial therapy.
These recommendations also provide information on venous access (with preference for using peripherally inserted central catheter) and care orientations (Table 2), as well as recommendations for appropriate dosages, dilution, reconstitution, and infusion of antimicrobials (Table 3), in addition to laboratory monitoring routines (Table 4). The elaboration of these guidelines considered the particularities of both healthcare services and patients in the country; therefore, only antimicrobials that can be administered once or twice a day were considered. Patients’ social conditions and vulnerability factors were also taken into account among the factors determining eligibility for OPAT. 4 Considering aspects of patient safety for the Brazilian reality, self/carer administered OPAT is not allowed in Brazil and only healthcare professionals trained in handling venous accesses can infuse antimicrobials.
Types of central lines indicated for OPAT in Brazil according to the Brazilian guidelines.

OPAT, outpatient parenteral antimicrobial therapy; PICC, peripherally inserted central catheter.
Recommendations and instructions for reconstitution, dilution, and infusion for the antimicrobials to be used within the OPAT regimen in Brazil according to the Brazilian guidelines.
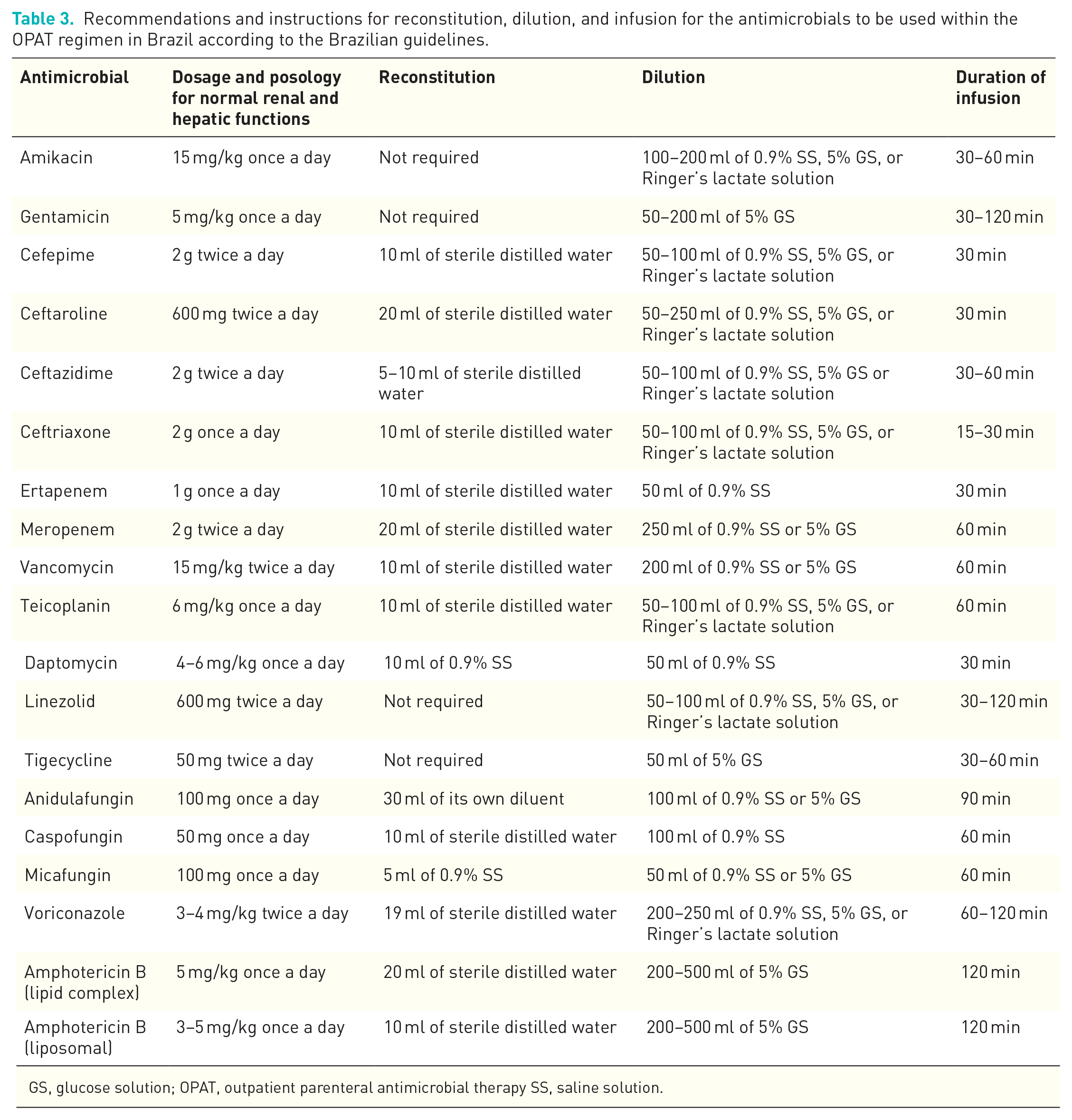
GS, glucose solution; OPAT, outpatient parenteral antimicrobial therapy SS, saline solution.
Recommendations on routine laboratory tests and monitoring of adverse events for OPAT in Brazil according to the Brazilian guidelines.

ALT, alanine transaminase; AST, aspartate transaminase; CPK, creatine phosphokinase; GT, glutamyl transferase; OPAT, outpatient parenteral antimicrobial therapy.
Experiences with OPAT with Brazil
The first experience of a structured OPAT program in Brazil was published in 2016 and reports the results of a 1-year partnership between a reference orthopedic hospital and the public (linked to SUS) municipal healthcare network in the city of São Paulo which started in 2013. This initiative made it possible to dehospitalize 116 patients, making 11,698 bed-days available for patients requiring hospitalization and few adverse events related to OPAT. This study motivated the development of national guidelines later published in 2017. 9 After the publication of the national guidelines, other studies were published reporting experiences with OPAT.
Two other studies that report the treatment of patients in SUS showed favorable results regarding clinical outcomes but were conflicting regarding the cost-effectiveness analysis. While a study carried out with 291 trauma patients in the south of Brazil showed that OPAT was effective in reducing costs, another study carried out with 23 kidney and liver transplant patients using carbapenems in the northeast region showed higher costs related to OPAT. In this case, the result was related to the higher cost of ertapenem, a carbapenem used on an outpatient basis as compared with meropenem, used for hospitalized patients.10,13 Another study with 39 patients designed specifically to evaluate the cost-utility of OPAT for SUS, however, showed that this treatment modality was effective in this regard by allowing overall savings of 31.86% from the hospital perspective and 26.53% from the SUS perspective, with favorable clinical outcomes and perception of quality of life. 14
Regarding OPAT in PHI, two other studies were published. Both studies analyzed a considerable number of patients (441 in one study and 278 in another) also reported favorable clinical outcomes, although they did not assess cost-effectiveness aspects. These two publications also report on the positive impact of antimicrobial stewardship programs in the context of OPAT.11,12 Table 5 provides a summary of studies on OPAT in Brazil.
Summary of published studies on OPAT in Brazil.
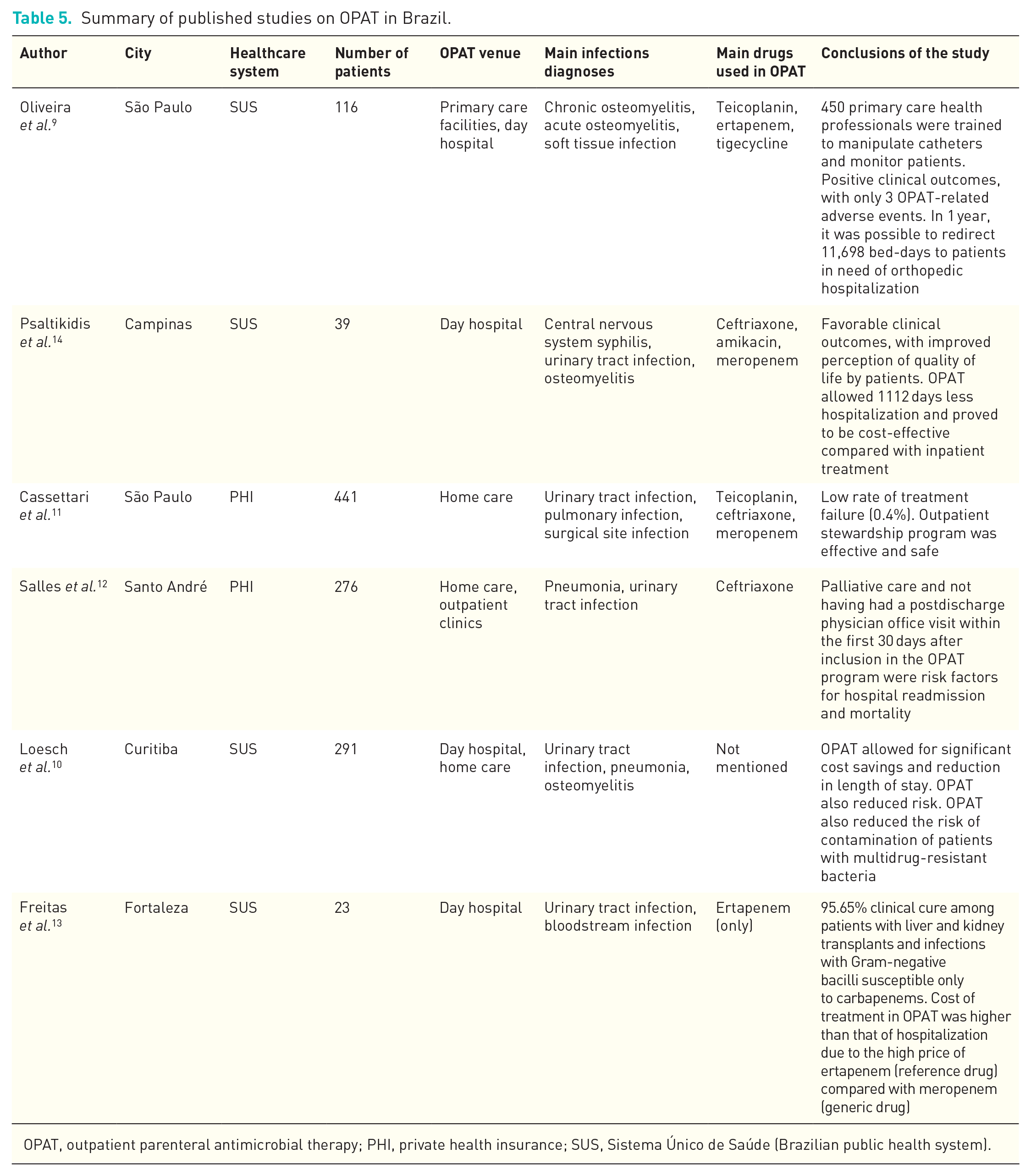
OPAT, outpatient parenteral antimicrobial therapy; PHI, private health insurance; SUS, Sistema Único de Saúde (Brazilian public health system).
General considerations and future prospects
Although OPAT was adopted late in Brazil, with the first large-scale experience initiated in 2013 and reported in 2016, published reports show that the country’s experiences with this type of treatment are favorable.9–14 Publication of national guidelines, which considered the particularities of the country, allowed the dissemination of OPAT services and the advancement of dehospitalization in Brazil. 4 It is still necessary, however, to make progress with these strategies, especially regarding financing of antimicrobials. While there is no specific public policy for OPAT in SUS, this treatment modality may be unfavorable from a financial point of view in some situations, such as shown in one of the reported studies. 12 Creation of this policy and strengthening of existing programs such as ‘Melhor em Casa’ would certainly contribute to an even greater expansion of OPAT in Brazil and the consequent optimization of beds and hospital resources.
Within the scope of PHI, OPAT also shows strong expansion in Brazil and proves to be effective from a clinical point of view, lacking specific studies that corroborate the cost-effectiveness of this treatment modality in this model of healthcare system.
