Abstract
Liver cystic echinococcosis (CE), known as hydatid disease, is caused by the tapeworm Echinococcus granulosus sensu lato. Humans are accidental hosts in this zoonotic disease process, and hepatic infection accounts for over two-thirds of all cases. Since signs and symptoms are mainly non-specific, especially in early disease, clinicians should have a low threshold to include CE as a differential diagnosis in patients with positive serology and suggestive radiological findings, especially in endemic regions. The standard management for liver CE depends on the patient’s symptoms, the radiological stage, the size and location of the cyst, the presence of complications and the treating clinicians’ expertise. In this review, we discuss the lifecycle of Echinococcus granulosus sensu lato and its epidemiology and then focus on discussing the clinical features, diagnosis and treatment options of CE of the liver.
Keywords
Background
Human echinococcosis is a zoonotic disease caused by tapeworms of the genus Echinococcus. The lifecycle of Echinococcus granulosus sensu lato (s.l.) requires a definitive host, most commonly a dog, and an intermediate host, commonly a sheep. Humans are the accidental intermediate hosts after ingesting ova passed in dog faeces. Although six different species of Echinococcus have been identified, only four are considered relevant to humans and include E. granulosus s.l., E. multilocularis, E. vogeli and E. oligarthrus. E. granulosus s.l. is the most prevalent species worldwide and causes cystic echinococcosis (CE), also known as hydatid disease. Mitochondrial DNA sequencing has identified ten distinct genetic subtypes (G1–10), of which the sheep strain (G1) is commonly associated with human infection.1–5 Despite a low mortality rate (0.2/100,000 population with a case fatality rate of 2.2%), the morbidity associated with CE is significant, reflected in the 3.6 million DALYS (Disability-Adjusted Life Years) lost globally.6,7 Furthermore, the estimated annual livestock production loss has been reported at more than 2,190 million USD. 8 Despite these figures, the infection is grossly under-reported and does not receive the attention it deserves – especially considering it is largely a preventable disease. For these reasons, CE has been listed as one of the neglected tropical diseases that the World Health Organisation (WHO) aims to control or eradicate by 2030.3,6 CE can involve any visceral organ, but due to the blood supply of the portal venous system, the liver is most commonly affected accounting for over two-thirds of all cases.1,9 In this review, we discuss the lifecycle of E. granulosus s.l., and its epidemiology, and then focus on discussing the clinical features, diagnosis and treatment options of CE of the liver.
Lifecycle
The adult tapeworm resides in the small bowel of the definitive host (Figure 1), which is most commonly dogs. The adult tapeworms attach to the definitive host’s small intestinal mucosa and later shed gravid proglottids (the last segment of the mature worm that produces eggs) in the host’s faeces. Each proglottid contains numerous eggs that may be ingested by the intermediate host, where they mature into cysts and daughter cysts. Sheep, intermediate hosts, become infected by grazing in grass contaminated with egg-laden faeces from the definitive host. Humans are accidental intermediate hosts that become infected by faecal-oral transmissions, that is, accidental consumption of food or water contaminated with faeces-containing eggs or by direct contact while handling a definitive infected host. 1–3 Definitive hosts are infected by eating the offal of infected intermediate hosts, an example of which would be a dog eating the internal organs of slaughtered sheep, thus completing the lifecycle.1,2,4

Schematic diagram showing the lifecycle of Echinococcus granulosus sensu lato. The adult tapeworm resides in the small intestine of the definitive host (e.g. dog). The intermediate host (e.g. sheep) ingests the eggs by grazing in grass contaminated with egg-laden faeces. Humans are accidental hosts to the disease process. They are infected by accidental consumption of food or water contaminated with faeces-containing eggs or by direct contact while handling a definitive infected host. The definitive host may then consume infected sheep offal, thus completing the lifecycle (Original – Adobe Photoshop).
Once ingested by the intermediate host, the eggs release oncospheres, which attach to the intestinal wall. The oncospheres migrate from the intestine via the portal venous system to various organs, where they develop cysts and daughter cysts. 1–4 The most common site of occurrence is the liver (70%), followed by the lungs (20%). Infrequent sites include the spleen, kidneys, peritoneal cavity, heart, central nervous system and bones.2,4,10–12 Hydatid cysts grow slowly at approximately 1–10 mm per year, with liver cysts growing slower than lung cysts. 4
The CE cyst is characteristically a round cystic lesion that consists of an inner, germinal layer, referred to as the endocyst, and an outer, acellular, laminated layer, referred to as the ectocyst. The inner germinal layer produces small vesicles called brood capsules that divide via asexual division and produce many protoscolices (Figure 2). Around the echinococcal laminated layer lies a granulomatous, adventitial layer known as the pericyst. This layer is produced by the host’s immune system to wall off the cystic infection and is often seen in imaging studies.2,4,5,10,13

Representation of the liver hydatid cyst layers, which includes the outer acellular layer of the cyst (laminated layer), the inner nucleated layer of the cyst (germinal layer) and a granulomatous layer produced by the hosts’ immune system to wall off the cystic infection (adventitial layer). The protoscolex, the future head of the adult worm, is seen here budding from the germinal layer, and the daughter cyst is seen here floating within the main cyst. The hydatid sand is a sonographic finding representing a combination of cystic fluid and protoscolices. (Original – Adobe Photoshop).
Epidemiology
It is estimated that 2–3 million cases of the disease exist globally. However, due to the paucity of epidemiological studies in all endemic areas, an overall prevalence underestimation of the condition exists.1,14,15 E. granulosus s.l. is endemic in India, Australia, Turkey, China, South America, Middle Eastern and Eastern European countries where incidence rates are as high as 50 per 100,000 person-years with an estimated prevalence of 10% of the population in highly endemic areas.2,15,16 Increased incidence in endemic areas may be due to factors, such as low socioeconomic status and unsanitary animal slaughtering practices. 2
Clinical presentation
Patients may be either completely asymptomatic or present with non-specific signs and symptoms in early disease. In intra-abdominal CE, patients present with symptoms late into the disease process. This may be due to the slow growth of the cysts or the host’s immune system to initially wall off the cystic infection via the granulomatous adventitial layer.2,3,10 According to Wen et al. 3 , cystic growth is faster in CE patients with AIDS, indicating that an immunosuppressive state may play a role in disease advancement.
Liver involvement usually presents with symptoms when the cyst size is large (> 10 cm) in diameter or when 70% of the organ volume has been taken up by the cyst(s). Common presenting complaints may include right hypochondrial pain, a hepatic mass, and nausea and vomiting.1,17,18 Obstructive jaundice may occur if an adjacent liver cyst compresses the biliary system.2,4 Physical examination findings may exhibit hepatomegaly or abdominal distension.2,3,19
Patients with complicated disease may also present with cystic rupture occurring spontaneously or infrequently after blunt abdominal trauma. Although rupture is a rare presentation, it is a potentially fatal complication. Cholangitis may be present due to biliary tree obstruction if ruptured cysts communicate with the biliary system. Free intra-peritoneal rupture may result in an immunological response resulting in an allergic reaction, the most fatal of which is anaphylactic shock.1,2,18 Cystic rupture can also cause ‘seeding’ of viable protoscolices in the peritoneum resulting in secondary hydatidosis.2,17 In rare cases, secondary pulmonary involvement may occur due to the rupture of a liver cyst through the diaphragm. 20
Diagnosis
A full clinical evaluation of the patient is paramount to diagnosing liver CE. This includes a thorough history, specifically identifying contact with dogs, wildlife, or any travel to endemic areas and a focused physical examination. In patients with suspicious clinical findings, other laboratory and radiological investigations can help to attain the diagnosis.1,2,17
Laboratory investigations
Serology for CE is a valuable adjunct to radiological investigations in diagnosing liver hydatid disease.3,4 However, it is not without limitations which include the inability of serology to distinguish between active and inactive cysts when the ultrasound is equivocal. 21 Serological tests include enzyme-linked immunosorbent assay (ELISA), indirect hemagglutination assay (IHA) and Western blotting (WB), which may play a role in diagnosing liver CE.4,13,22 Enzyme-linked immunosorbent assay (ELISA) is considered the gold standard serological test for liver-only hydatid involvement. Immunoglobulin G (IgG) ELISA detects E. granulosus-specific antigen with a sensitivity of 93.5% and specificity of 89.7%.1,10,17,18 However, seronegative results are often found in early-stage cysts where the E. granulosus antigens are contained in the endocyst. The antigens are then sealed off from the host’s immunological response to the parasitic infection. The same lack of immune response occurs in the late stages of the disease process, where the cysts are calcified, often resulting in a seronegative result. 17 WB serology for liver CE has a high sensitivity of 80–100% and a specificity of 88–96%. Drawbacks with WB are that the test is expensive, and sensitivity rates reduce dramatically in extra-hepatic disease.4,13,22 IHA testing has a sensitivity of 90%; however, if the result is positive, it may remain positive for several years after that. 23
Routine blood tests may show non-specific changes. Only about 40% of patients have abnormal liver function tests. If derangement is present, alkaline phosphatase is often elevated with/without hyperbilirubinaemia. Differential white cell count may show eosinophilia in 25–40% of cases.2,4,10
Imaging
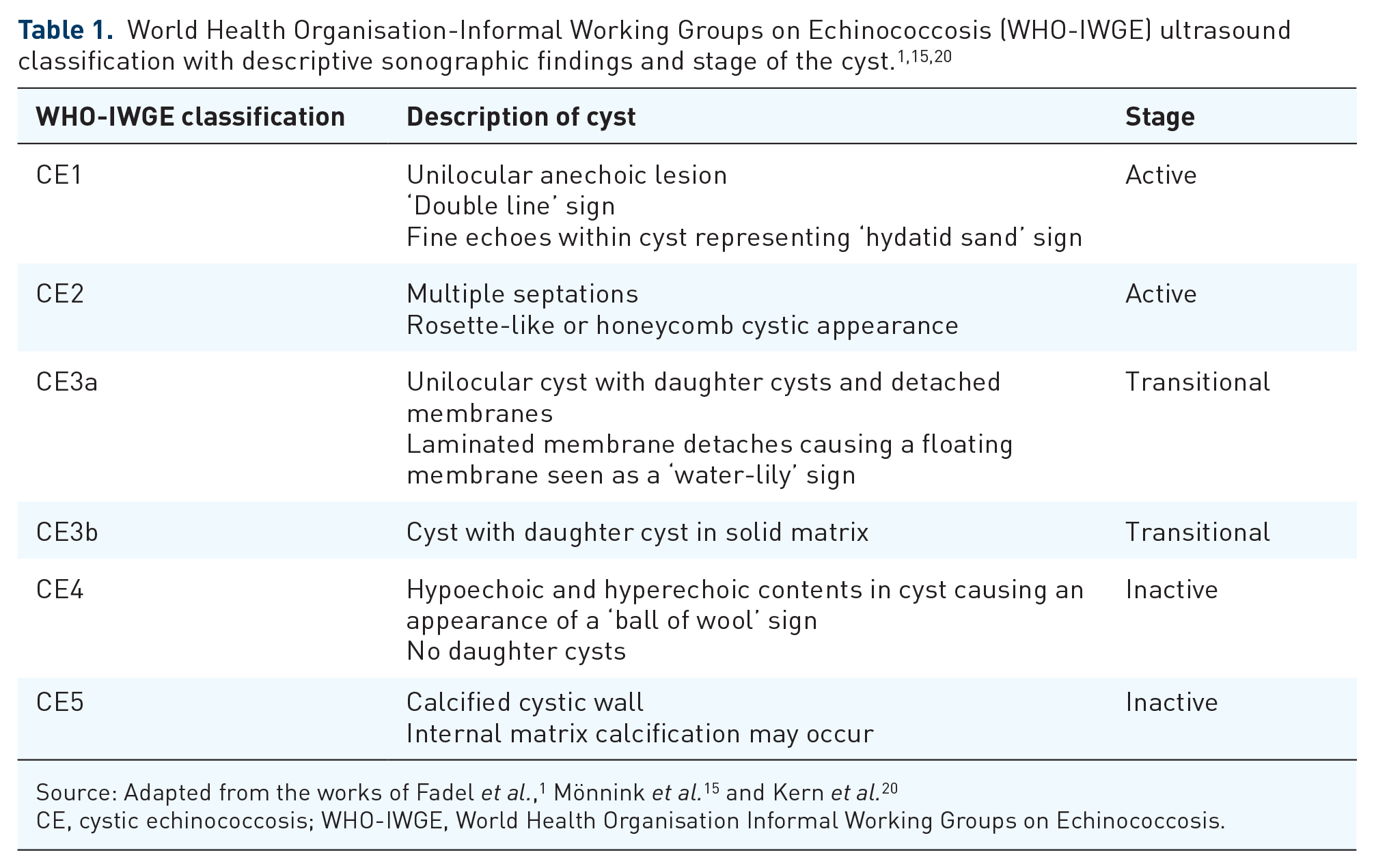
Ultrasound imaging is the diagnostic tool of choice for liver CE, with a sensitivity reported between 90% and 95%.1,10,11 On ultrasonography, the appearance of liver CE may vary considerably from a unilocular anechoic cyst that resembles a simple cyst to a pattern appearing as a solid mass. 20 As the patient changes position, a ‘falling snowflake pattern’ created by the multiple echogenic foci produced by the combination of cystic fluid and protoscolices, also known as ‘hydatid sand’, can be seen under real-time imaging. 20 The detachment of the endocyst from the pericyst gives an appearance of ‘floating membranes’.4,20 In addition, the multivesicular mother cyst with daughter cysts separated by radiating septae representing cyst walls and hydatid sand/matrix may give rise to a ‘wheel-spoke’ pattern. 24 World Health Organisation Informal Working Groups on Echinococcosis (WHO-IWGE) have classified hydatid cysts based on sonographic features.1,3 This classification categorises cysts from CE1 to CE5 based on various features seen on ultrasound (Table 1). The ‘active’ stage of the disease corresponds with CE1 and CE2 types. The ‘transitional’ stage pertains to CE3a and CE3b, and the ‘inactive’ stage corresponds to CE4 and CE5 (Figure 3). While this system helps to classify liver hydatid cysts universally, it also guides further management.1,3
CE, cystic echinococcosis; WHO-IWGE, World Health Organisation Informal Working Groups on Echinococcosis.

Imaging of liver cystic echinococcosis.
Abdominal computed tomography (CT) is a helpful adjunct to ultrasound, and it confers numerous benefits, with sensitivities reported as high as 95%.10,11 It is especially useful when the sonographic examination is limited by patient factors or the imaging findings are equivocal. In addition, it is useful for pre-operative planning. It may provide a road map regarding the accessibility to the cyst(s) and ascertain the location and depth of the cysts, especially in relation to their proximity to adjacent bile ducts.1,2,18,25 It can also help to rule out concurrent extra-hepatic CE. 18
Magnetic resonance imaging (MRI) is deemed a superior imaging modality to CT for identifying biliary involvement and observing the internal composition of hydatid cysts. Diffusion-weighted MRI has the added benefit of accurately differentiating simple liver cysts from hydatid liver cysts.18,20
Treatment
The standard management for liver CE depends on the patient’s symptoms, the radiological stage, the size and location of the cyst(s), the presence of complications and the treating clinicians’ expertise.1,15,26
Management options include medical therapy, a ‘watch and wait’ approach, percutaneous therapy and surgical treatments.1,2,4,11
Medical therapy
Medical management with anthelmintic therapy confers numerous benefits for the patient. It can be used as monotherapy for the uncomplicated CE1 and CE3a liver cysts less than 5 cm in diameter and multiple liver cysts or multiorgan involvement in the inoperable candidate.3,18,20 It is also used as a neoadjuvant agent before percutaneous or surgical therapy to chemically sterilise the parasite to render the cyst inactive, reduce the cystic wall tension and reduce the risk of intra-operative viable cyst rupture.2,20 Due to the increased bioavailability and improved absorption from the gut, albendazole is the preferred anthelmintic agent of choice for liver CE.3,14,18,26 It is administered at a 12-hourly dosage of 10–15 mg/kg/day.3,20 There has been no consensus on the schedule and length of anthelmintic in the neoadjuvant and adjuvant setting, with various authors making different recommendations. 3 Akhan et al. 27 recommended albendazole therapy be given for Puncture, Aspiration, Injection and Re-aspiration (PAIR), 1 week before and 1 month after treatment. Velasco-Tirado et al. 26 recommended that albendazole be used pre-operatively anytime from 3 months to 1 day before surgical or percutaneous intervention and continued 1–3 months after the procedure. Wen et al. 3 suggested initiating albendazole therapy 1 week before intervention and continuing for up to 2 months after the procedure. A comprehensive regime to administer for uncomplicated cysts is albendazole therapy, administered at least 3 days pre-operatively and continued post-operatively for 4–8 weeks. In complicated cysts or intra-operative spillage, the post-operative duration may be extended to 3–6 months.2,4,20
A continuous treatment regime should be administered with the previous regime of cyclical administration avoided in current practice.4,20 Clinicians must be prudent about the side effects of albendazole, such as hepatic dysfunction, alopecia and agranulocytosis. Patients should be monitored regularly with white cell count and liver function tests.17,20 Blood tests should be done on days 5, 14 and 28 after initiation, biweekly and monthly, if therapy is ongoing.4,17 It should be noted that albendazole is contraindicated in early pregnancy as it is teratogenic in rats and rabbits.10,20
Some authors have postulated that combination therapy with praziquantel increases the albendazole metabolite’s serum and cystic fluid levels compared with patients receiving albendazole monotherapy.4,26 However, due to the lack of more robust evidence with further randomised control trials, it is not a widely accepted practice.14,20,26
‘Watch and wait’ approach
Inactive, degenerating CE4 and all CE5 cysts can be observed with interval ultrasound monitoring without pharmacological or surgical intervention.1,3,15
Percutaneous therapy
Percutaneous approaches for the treatment of liver CE include PAIR, standard catheterisation and modified catheterisation technique (MoCat).4,28–33
PAIR therapy is successfully used for single-compartment cysts, such as CE1 and CE3a cysts less than 10 cm in diameter. Deterrence of PAIR use includes CE2 and CE3b cysts, cases where safe percutaneous cyst localisation with ultrasound guidance is unavailable, and when there is evidence of cysto-biliary communication.1,20,28,30,34
Standard catheterisation technique is also used for CE1 and CE3a cysts but includes cysts with a diameter greater than 10 cm or those with cystic fluid content of more than 1,000 ml.20,35 If a cysto-biliary communication is found during PAIR, a standard catheterisation technique may be utilised to control biliary drainage. 35 One of the drawbacks is the increased duration of hospital stay. Akhan et al. 27 reported a median duration of hospital stay of 2.5 days in 26 patients with standard catheterisation. The catheterisation and hospital stay may be prolonged if cysto-biliary communication or cystic infection occurs.35–38
A modification to PAIR, the MoCat, has been utilised for uncomplicated CE2 and CE3b cysts and cysts larger than 10 cm in diameter. As with standard catheterisation technique, MoCat, results in a longer hospital stay. The MoCat approach uses a drainage catheter placed via the Seldinger technique, wherein the cyst contents and membranes can be aspirated. At the end of the procedure, the catheter can be left in place for any ongoing fluid drainage.3,13,20,28,33,39
The first step in the standard PAIR technique is to puncture and aspirate the cyst under ultrasound guidance. A scolicidal agent is then injected into the cystic cavity to sterilise the cyst of any viable protoscoleces and destroy the germinal layer. Various scolicidal agents have been adopted in the use of CE. Either 20% hypertonic saline or 95% alcohol is recommended with the PAIR technique. The fluid is re-aspirated approximately 15–20 minutes later (Figure 4).1,3,15,23 If bile is aspirated, initially after the first puncture, it is suggestive of cysto-biliary communication. Scolicidal injection into the cyst may result in a devastating risk of chemical sclerosing cholangitis and subsequent biliary stricturing and thus should be avoided.18,23 There are limited recommendations regarding the management of cysto-biliary communication noted during PAIR. Khuroo 34 recommends aborting PAIR if ‘yellow cyst fluid’ or ‘positive bilirubin dipstick’ on fluid aspiration occurs. Öztürk et al., 28 Turkish consensus report recommends catheterisation after percutaneous therapy can be done for cysts larger than 10 cm and those with biliary content. Catheterisation should be left in place for roughly 1 week, with removal only once the fistula output is less than 10 cc. If there is an ongoing or increasing biliary leak, endoscopic retrograde cholangiopancreatography (ERCP) is recommended. 28

Management approaches for liver cystic echinococcosis vary from minimally invasive percutaneous approaches (PAIR) to more invasive surgical treatment options, which include partial cystectomy, total cystectomy and hepatic resection.
Surgery
Surgical options for liver CE can be divided into conservative and radical approaches (Figure 4). 40 The radical approach, which includes total cystectomy and hepatic resection, has been criticised as an aggressive form of therapy associated with significant morbidity for a relatively benign disease process.9,40 It is also a technically demanding surgery and usually requires patients to be operated on in a specialised hepatobiliary unit.10,18 For these reasons, the conservative approach with partial cystectomy is more widely adopted. This involves the removal of the cyst content and sterilisation of the residual cavity, together with partial cyst resection, under albendazole cover.3,11,25
The partial cystectomy approach uses an appropriate incision to gain access to and expose the liver cyst(s). The surgical field is protected by abdominal swabs soaked in a scolicidal agent (20% hypertonic saline). This helps to prevent contamination in the event of spillage during cyst-content evacuation. The cyst is then punctured, aspirated and instilled with a chosen scolicidal agent. As with PAIR, if bile is aspirated within the cyst, biliary communication is present and the scolicidal agent is avoided. After 15 minutes, the contents are re-aspirated, and the cyst is opened to evacuate the endocyst contents with suction (Figure 5). The cyst is then unroofed by excising the cyst wall outside the liver parenchyma (Figure 6). If a cysto-biliary fistula is noted intra-operatively, it can be managed by suture placement. Once the cystic cavity is evacuated of its contents, the cavity needs to be obliterated either with capitonnage or omentoplasty.4,23,25

Intra-operative image showing bile-stained floating membrane evacuated during a partial cystectomy (Original image).

Intra-operative image of a partial cystectomy for liver hydatid cyst with biliary communication. After the surgical field is surrounded by abdominal swabs soaked in scolicidal agent solution, the cyst wall is deroofed and the cyst contents are evacuated (Original image).
Although this conservative technique is deemed a safer peri-operative approach than radical surgery, it is not without complications. It is associated with a higher risk of recurrence (20–25%) compared with radical surgery (0.6–4%). 23 The increased risk may be due to intra-abdominal spillage during cyst-content evacuation and incomplete removal of the cyst’s entire residual germinal layer. Another less common mechanism whereby cyst recurrence may occur is through exogenous vesiculation. With cyst enlargement, the germinal layer and hydatid fluid crosses through the pericyst and result in the growth of the daughter cyst outside the pericyst. Partial cystectomy is also associated with an increased risk of post-operative cystic complications, including biliary fistula and cystic cavity infection.9,23 Most post-operative biliary fistulas may be managed conservatively with percutaneous tube drainage. However, ERCP and biliary stent placement for biliary drainage are indicated for a post-operative biliary fistula with ongoing duration (> 10 days) or for a continuous, large output fistula.4,9,19,23
Laparoscopic or robotic surgery has revolutionised surgical techniques in most fields. The most notable advantages are shorter hospital stays, reduced wound complications and less post-operative pain. If surgical expertise allows, these minimally invasive approaches should be considered for accessible cysts in segments II, III, IVB, V and VI. However, there is concern regarding the handling of the cyst and the possible increased risk of seeding the cystic contents. 23
Khuroo et al. 41 conducted a study where 25 patients were assigned to percutaneous drainage with albendazole therapy. The other 25 patients underwent simple cystectomy to treat uncomplicated liver hydatid cysts. Results showed that the final cyst size and cyst disappearance on follow-up did not differ significantly between the two groups. However, significant outcomes were noted concerning procedure-related complications, which were less in the percutaneous drainage group (32%) as compared with the surgical group (84%) as well as hospital stay being shorter in the percutaneous group (4.2 +/– 1.5 days) as compared with the surgical group (12.7 +/–6.5 days). Percutaneous therapy combined with albendazole has shown to be a safe and effective modality for the treatment of uncomplicated liver CE cysts with fewer operative complications and shorter hospital stay outcomes as compared with surgical treatment.4,15,34,41,42
Preventive strategies
Eradication of CE is possible through effective dog-deworming programmes and legislative implementation of safe slaughtering practices, as seen in Tasmania and New Zealand. Other countries, such as Argentina, Chile and Uruguay saw a similar reduction in CE with dog deworming programmes. The programmes utilised supervised dog-deworming with praziquantel at intervals between 4 and 8 times yearly.2,43
A newer modality to reduce transmission of CE is through the vaccination of sheep with the EG95 vaccine. It is currently registered for use in China and Argentina as an additional intervention to reduce CE transmission. The high cost of the vaccination precludes this intervention for poorer countries where CE is usually endemic. Attempts at dog vaccinations have been described but have not shown proven measurable outcomes in CE transmission reduction.3,43
Preventive measures to assist in the reduction of CE transmission are possible. Public health strategies, such as regular dog-deworming programmes, strict safe slaughtering practices strengthened by local legislation and public education about the disease process may assist with transmission reduction in endemic populations. 18
Conclusion
Due to a paucity of epidemiological data in endemic areas, the true global burden of CE is grossly underestimated. Humans are accidental intermediate hosts in this disease process, and hepatic infection accounts for over two-thirds of all cases. Signs and symptoms are largely non-specific, especially in early disease. Therefore, clinicians should have a low threshold for considering the diagnosis in patients with positive serology and suggestive radiological findings, especially in endemic regions. The standard management for liver CE depends on the patient’s symptoms, the radiological stage, the size and location of the cyst, the presence of complications and the clinicians’ expertise. Implementation of well-established public health protocols developed by the WHO is paramount to mitigate the significant annual costs associated with CE. While the disease is largely seen in the developing world, increasing migration and travel, require clinicians to consider CE in their differentials for any suspicious cystic lesion of the liver.