Abstract
Background:
Patients living with HIV (PLWH) with multi-drug resistance (MDR) and prior episodes of virologic failure have few therapeutic options remaining. These patients are often prescribed ‘salvage’ antiretroviral therapy (ART) regimens with high pill burdens, leading to potential decreased medication adherence and increased side effects and drug–drug interactions.
Materials & Methods:
In this retrospective, observational cohort study, we included adult patients with a diagnosis of HIV-1 who received care at our institution’s Ryan White Clinic and who received ‘salvage’ ART, defined as three of more antiretroviral agents from at least three different HIV drug classes. Patients were grouped into two cohorts, simplified ART cohort and non-simplified ART cohort, based on whether their ART regimen was reduced by at least one tablet daily. The primary outcome was the percentage of patients who had their viral load suppressed (HIV-1 RNA <50 copies/ml) at their most recent clinic visit. Secondary outcomes were virologic failure (HIV-1 RNA ⩾200 copies/ml), percentage of time patients were virologically suppressed over the past 2 years, and the emergence of new treatment-resistant mutations.
Results:
There were 50 patients included in the final analysis, 28 in the simplified ART cohort and 22 in the non-simplified ART cohort. The percentage of patients who had their HIV-1 viral load suppressed at their most recent clinic visit was n = 24 (86%) in the simplified ART cohort and n = 16 (73%) in the non-simplified ART cohort (p = 0.302). There were no statistically significant differences between the two cohorts in terms of the secondary outcomes.
Conclusion:
Our study found that simplification of ART regimens based on HIV genotype in PLWH with a history of MDR and prior virologic failures, regardless of the presence of HIV-1 viremia at the time of simplification, resulted in similar rates of virologic suppression and virologic failure as non-simplified ART regimens.
Introduction
Human immunodeficiency virus (HIV) has the ability to confer resistance to antiretroviral therapies (ARTs) if patients are non-adherent to their medications. Historically, patients living with HIV (PLWH) who experienced virologic failure and development of resistance had few therapeutic options remaining and were thus put on ‘salvage’ ART regimens. These salvage ART regimens subjected patients to high pill burdens and could potentially result in increased adverse effects and drug–drug interactions. Current Clinical Practice Guidelines for patients who experienced virologic failure recommend a treatment regimen with at least two fully active agents with at least one of these agents having a higher barrier to resistance, such as dolutegravir, bictegravir, or boosted darunavir. 1 A common salvage ART regimen used for patients at our institution includes boosted-darunavir, etravirine, and an integrase inhibitor, with or without additional agents. This regimen has been validated for PLWH and a history of treatment failure in retrospective and prospective clinical trials.2,3 Our institution has utilized this regimen in patients with known multi-drug resistance (MDR) based on cumulative genotype data or following virologic failure of multiple regimens. Recently, data have emerged to support the simplification of antiretroviral regimens, from modified instructions for medications such as darunavir to the approval of multiple single-tablet regimens and emerging data on two-drug therapy.4,5 Simplification of ART regimens is crucial as it can lead to increased adherence and, subsequently, increased virologic suppression. However, there are limited data on the clinical efficacy of ART treatment simplification in patients with a history of MDR and subsequent treatment failures.
Previously, our institution’s Ryan White Clinic and pharmacy department performed a review to determine the eligibility for salvage regimen simplification based upon genotypic test results and clinical review of ART appropriateness. 6 In a cohort of 53 patients, 72% were determined to be eligible for simplification of their ART regimen by at least one pill per day. Since this review was conducted, eligible patients have slowly transitioned to a simplified ART regimen during routine clinic visits; however, the virologic and clinical outcomes of this simplification strategy have not yet been formally assessed since regimen changes have been implemented. The purpose of this study was to determine whether simplification of salvage ART regimens in PLWH and a history of MDR and prior treatment failure led to sustained virologic suppression.
Methods
Study design and study population
This observational, retrospective, single-center, cohort study included adult patients with a diagnosis of HIV-1 who received care at our institution’s Ryan White Clinic, a non-urban clinic in the United States, and who received ‘salvage’ ART between July 2016 and July 2021. Salvage ART was defined as three or more ART agents from at least three separate drug classes. A report was generated through a local database to identify all patients within our clinic who had received salvage ART since 2016. Patients were excluded if they transferred care or were deceased prior to simplification eligibility review. Clinical data were gathered from the electronic medical record, including demographics (age, sex, date of HIV diagnosis, race), laboratory data (HIV-1 viral load, CD4 cell count, HBV serology), prior and current antiretroviral regimens, and HIV-1 resistance genotype profiles. To assess resistance, all available patient genotypes were then entered into the Stanford Database to analyze cumulative resistance data. 7 Categories of high, intermediate, low-level, and potential low-level resistance were defined based on qualitative interpretation from the Stanford Database for each of the respective antiretroviral categories. 7 For nucleoside reverse transcriptase inhibitors (NRTIs), clinically relevant agents included abacavir, emtricitabine, lamivudine, tenofovir, and zidovudine. For non-nucleoside reverse transcriptase inhibitors (NNRTIs), clinically relevant agents included doravirine, efavirenz, etravirine, and rilpivirine. Clinically relevant protease inhibitors included atazanavir, darunavir, and lopinavir. All integrase strand transferase inhibitors (INSTIs) were considered clinically relevant. Genotypic sensitivity score (GSS) was calculated for the current regimen and new simplified regimen, if applicable, as follows: 1 point to each drug categorized as susceptible or potential low-level resistance, 0.5 points to low-level resistance or intermediate resistance, 0 points to high-level resistance, with total GSS of the regimen representing the sum of each component. 8 Patients were placed into two study cohorts based on whether they had their salvage regimen simplified between 2016 and 2021, defined as a reduction in ART pill burden by at least one pill daily. Patients were placed into the simplified cohort if they underwent a reduction in pill burden. Patients were placed into the non-simplified cohort if their regimen remained unchanged during this time. Regimen simplification occurred during routine clinic visits. HIV-1 RNA levels and CD4 T-cell counts were performed at initial screening and at all subsequent Ryan White Clinic visits. Per our institution’s policy, HIV-1 RNA levels are assessed 4–6 weeks after a regimen change and then every 3–6 months based on the patient’s clinical status. Genotypic testing for treatment-emergent resistance was performed if breakthrough HIV-1 viremia occurred while on an ART regimen and HIV-1 RNA ⩾1000 copies/ml, which is the lowest level at which our laboratory could perform this test. Adherence assessments and evaluation of missed doses were also performed at each clinic visit. A clinical pharmacist was utilized to review dispense and refill history and also pill box fills in a small cohort of the patients. The protocol was approved by the University of Virginia Institutional Review Board (IRB # 17871), and patients were exempt from requiring informed consent given the retrospective nature of the study.
Variables of interest and outcomes
The primary endpoint of the study was the percentage of patients with HIV-1 RNA less than 50 copies/ml at their most recent clinic visit. The secondary endpoints were virologic failure, defined as an HIV-1 viral load of ⩾200 copies/ml, percentage of time patients were virologically suppressed over the past 2 years, and the emergence of new treatment-resistant mutations.
Statistical analysis
Categorical values were reported as counts and percentage of the population and continuous values were reported as median (interquartile range). The Fisher’s Exact Test was used to analyze categorical values and endpoints. The Mann–Whitney U test was used to analyze continuous values and endpoints. A p value of less than 0.05 was considered to be statistically significant. Statistical analyses were performed using SPSS Statistics 28 (Armonk, NY, USA).
Results
Patient characteristics
Fifty-one patients received salvage ART between 2016 and 2021. One patient was excluded from the study due to loss of follow-up and transfer of care in 2016, prior to simplification eligibility review, resulting in a total of 50 patients in the final analysis. Twenty-eight patients had their regimens simplified between 2016 and 2021 and were included in the simplified cohort. Twenty-two patients did not undergo regimen simplification and were included in the non-simplified cohort. Patient characteristics are listed in Table 1. The cohorts were similar to each other in baseline characteristics except for the greater number of patients with darunavir-specific mutations in the non-simplified cohort. Prior to simplification, the median ART pill burden in both groups was eight pills daily. Patients in the simplified cohort had their ART regimen reduced by a median of six pills daily to a median total of two pills daily. Importantly, not all patients in the simplified ART cohort had their HIV-1 viral load suppressed prior to simplification of their ART regimen (n = 23; 82%).
Patient characteristics.

ART, antiretroviral therapy; CD4, cluster of differentiation 4; DRV/r, darunavir/ritonavir; ETR, etravirine; HIV, human immunodeficiency virus; INSTI, integrase strand transfer inhibitor; IQR, interquartile range; NNRTI, non-nucleotide reverse transcriptase inhibitor; NRTI, nucleotide reverse transcriptase inhibitor; PI, protease inhibitor.
V11I, V32I, L33F, I47V, I50V, I54L/M, G73S, L76V, I84V, and L89V.
The majority of patients in both groups had high-level resistance to agents in the NRTI class and NNRTIs class, but a much lower percentage of these patients had high-level resistance to protease inhibitors (PIs) and INSTIs. INSTI resistance was low overall and only present in the non-simplified cohort. One patient in the non-simplified cohort had an H51Y mutation resulting in low-level resistance to elvitegravir (EVG) and raltegravir (RAL) and potential low-level resistance to bictegravir (BIC) and dolutegravir (DTG). Three patients in the non-simplified cohort had high-level INSTI resistance: one with Q148H and G140S mutations, resulting in high-level resistance to EVG and RAL and intermediate resistance to BIC and DTG; one patient with N155H mutation, resulting in high-level resistance to EVG and RAL and potential low-level resistance to BIC and DTG; and one with T66A mutation, resulting in high-level resistance to EVG and low-level resistance to RAL. Median baseline GSS was 3 in the simplified cohort and 2.5 in the non-simplified cohort. The majority of patients were simplified to either a single-tablet regimen (n = 8) or a two-tablet regimen (n = 15). Two patients had the ETR component of the salvage regimen eliminated due to high-level resistance (n = 2). Figure 1 summarizes the ART simplification regimens.
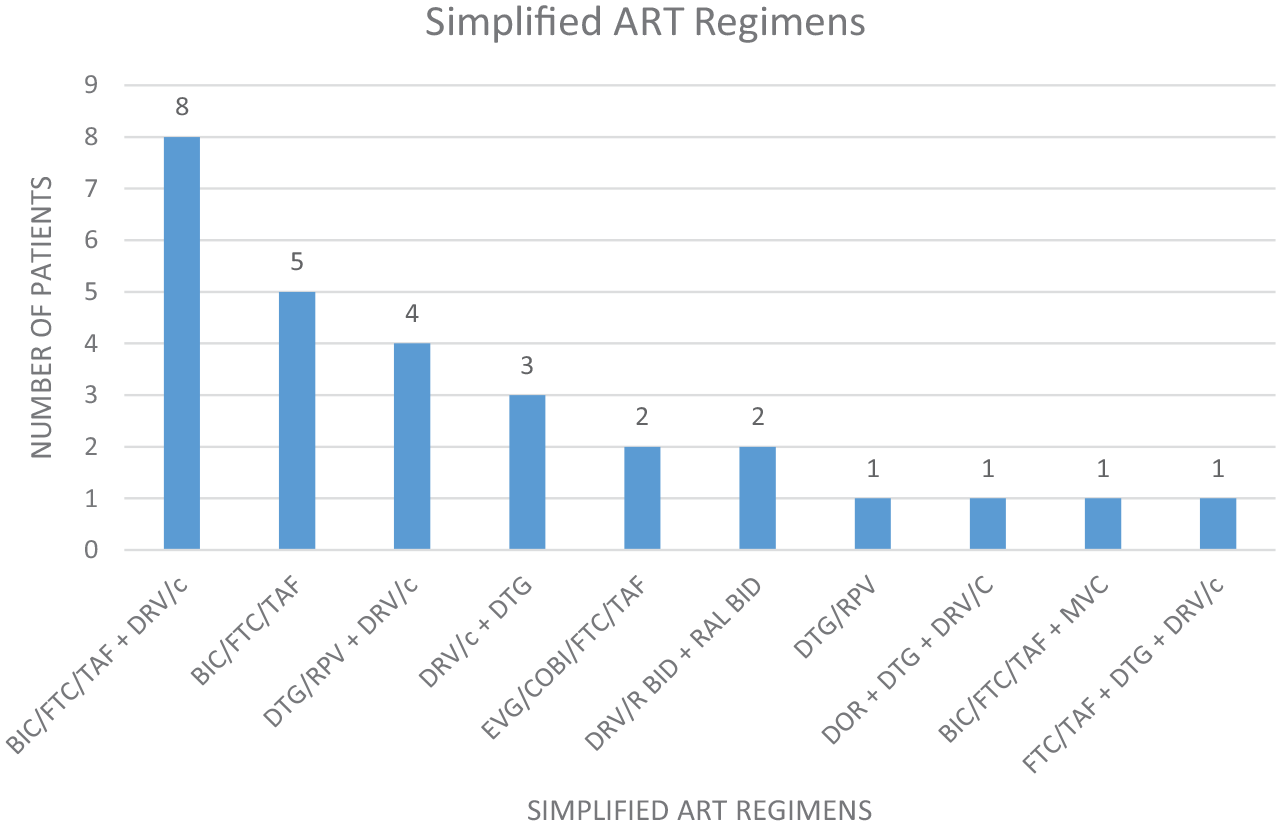
Simplified ART regimens.
Median duration of HIV diagnosis was over 25 years in both groups and median duration of salvage regimen was 8–9 years in both groups. The patients in the non-simplified cohort did not undergo simplification for various reasons. For five patients, there was no option to reduce pill burden and maintain a GSS of at least two active drugs. For three patients, simplification was discussed but declined by the patient due to inability to receive an entirely once-daily regimen. For four patients, the HIV provider did not elect to simplify due to history of virologic failure prior to salvage regimen initiation and distrust in a simplified regimen. One patient was not simplified due to persistent viremia and frequent gaps in care. For four patients, no genotype data were present to calculate GSS or identify specific mutations; however, due to failures of multiple previous regimens, these patients were empirically placed on salvage regimens and elected to continue in the presence of virologic suppression. For the remaining five patients in the non-simplified cohort, a reason was not disclosed for not discussing simplification.
Clinical outcomes
Patients in the simplified cohort were on their simplified salvage regimen for a median of 28 months prior to their most recent Ryan White Clinic visit where they were assessed for virologic suppression. The amount of patients who achieved virologic suppression at their most recent clinic visit was 24 (85.7%) in the simplified cohort and 16 patients (72.7%) in the non-simplified cohort (p = 0.302) (Table 2). Two of the four patients who did not achieve the primary outcome in the simplified cohort were those with HIV-1 viremia at the time of ART simplification. One patient who did not achieve the primary outcome was suppressed at the time of simplification, remained suppressed for 22 months following simplification, and subsequently stopped taking medication several months prior to most recent clinic visit. The final patient in the simplified cohort who did not achieve the primary outcome was suppressed at the time of simplification, remained suppressed for 17 months following simplification, and upon most recent clinic visit had low-level viremia (HIV-1 RNA 88 copies/ml). For the five patients in the simplified cohort who had viremia at baseline prior to simplification, three became suppressed after simplification. The number of patients in the simplified ART cohort who had their HIV-1 viral load suppressed ⩾80% of the time over the past 2 years was 23 (82.1%) and the number of patients in the simplified ART cohort who had their HIV-1 viral load suppressed ⩽50% of the time over the past 2 years was 4 (14.3%).
Clinical outcomes.

Regarding the non-simplified cohort, the number of patients who had their HIV-1 viral load suppressed ⩾80% of the time over the past 2 years was 16 (72.7%) and the number of patients who had their HIV-1 viral load suppressed ⩽50% of the time over the past 2 years was 4 (18.2%). Of the six patients in the non-simplified cohort who had detectable viral loads at the most recent clinic visit, four had been suppressed for ⩽50% of the time over the past 2 years. One patient had been suppressed for 75% of the past 2 years and had a newly elevated viral load following gaps in medication access. One patient had low level viremia (HIV-1 RNA 55 copies/ml) after over 3 years of virologic suppression, which was considered a likely virologic blip. Three patients in the simplified ART cohort (10.7%) and five patients in the non-simplified ART cohort (22.7%) experienced virologic failure (p = 0.277). Neither cohort experienced emergence of new treatment-resistant mutations (p = 1.000). No patients in the simplified cohort underwent re-escalation of regimen, and no further regimen changes were made in any patients in the non-simplified cohort.
Adherence was assessed informally at each clinic follow-up visit through patient interview as part of standard clinic workflow. In patients with viremia, barriers to adherence were assessed by the HIV medical provider while delivering laboratory results. As part of routine clinic workflow, all patients were counseled by the HIV medical provider about the importance of adherence at each visit. Three patients in the non-simplified cohort and one patient in the simplified cohort met with the clinic-based pharmacist weekly to monthly for pill box fills and adherence review. This adherence support began prior to this study and was maintained throughout the study period. These patients remained suppressed throughout the study period. One additional patient in the non-simplified cohort began meeting with the pharmacist monthly to have pill boxes filled upon presence of viremia during the study period. One additional patient in the non-simplified cohort had pill boxes filled weekly by his primary care provider (PCP), but remained viremic despite these efforts. The four patients in the simplified cohort who had detectable viral loads had refused additional adherence support at clinic visits.
Discussion
The literature surrounding the simplification of ART regimens in patients with MDR HIV and prior episodes of virologic failure is limited given that this patient population is typically excluded in clinical trials. This is the first study to assess the clinical outcomes of ART simplification in patients with MDR HIV and episodes of virologic failure, regardless of the presence of HIV-1 viremia prior to simplification. In addition, in contrast to prior studies, we looked at the percentage of viral suppression over the course of the past 2 years in addition to the most recent clinic visit. This allowed for a more robust evaluation of HIV-1 viral suppression throughout the patient’s clinical course versus a single time point.
To our knowledge, there is only one other study that assessed the clinical outcomes of salvage ART simplification in patients with MDR HIV and prior virologic failures. A prospective, randomized trial by Huhn et al. 9 included PLWH who were virologically suppressed (HIV-1 RNA ⩽50 copies/ml) with a history of at least two prior ART regimen failures and confirmed resistance to at least two different classes of antiretrovirals. However, patients were excluded if they had a history of integrase inhibitor resistance or if they had any darunavir-associated resistance mutations. The patients in this study were randomized to either stay on their current ART regimen or have their ART regimen simplified from a high pill-burden regimen to a two-pill ART regimen of elvitegravir, cobicistat, emtricitabine, tenofovir alafenamide, and darunavir once daily for a median reduction of three tablets daily. Patients who had their regimen simplified to the two-pill ART regimen consisting of an integrase inhibitor, two NRTIs, and a boosted PI maintained high rates of virologic suppression at 24 and 48 weeks (96.6%) and low rates of virologic failure (2%) with no episodes of treatment-emergent resistance.
The patient population in our simplified cohort experienced lower rates of virologic suppression (85.7% versus 96.6%) and greater rates of virologic failure (10.7% versus 2%) compared with the Huhn et al. 9 study. There are several differences between our study and the Huhn et al. 9 study that could explain these differences. First, although the Huhn et al. 9 study included patients with ART drug resistance, patients who had resistance to darunavir or INSTIs were excluded. Our study population had 18% of patients with high-level darunavir resistance and 6% of patients with high-level integrase inhibitor resistance. Our study also had a longer duration of time between ART simplification and the primary outcome, a median of 112 weeks in our study compared with 48 weeks in the Huhn et al. 9 study. This longer time period could allow for more opportunities for treatment adherence gaps and subsequent episodes of viremia. One of the biggest differences between the two studies was that we included patients with HIV-1 viremia prior to ART simplification, whereas these patients were excluded in the Huhn et al. 9 study. This allowed for the inclusion of patients with higher levels of drug resistance with few therapeutic treatment options remaining, indicating a more ‘real-world’ cohort of patients with MDR HIV.
The regimens for the patients in the simplified cohort were constructed based upon individual genotype results and GSSs, using combination products to lower the pill burden while maximizing the amount of active agents. Our most common simplification regimens included BIC/FTC/TAF with DRV/C. BIC/FTC/TAF is currently indicated as a complete regimen and is not recommended to be used with other antiretroviral products.1,10 This combination was often selected due to favorable tolerability profile of the BIC/FTC/TAF component, ability to retain partial NRTI activity in the patients for which it was prescribed, and due to the high barrier to resistance of bictegravir. 11 A pooled analysis of phase 3 clinical trial data by D’Antoni et al. 11 found that a small cohort of patients with primary INSTI resistance achieved or maintained virologic suppression through 48 weeks of BIC/FTC/TAF treatment. Pre-existing INSTI mutations were detected in 20 of the 1907 participants, and all but one of these 20 participants was virologically suppressed at baseline. Those suppressed at baseline maintained virologic suppression and the one treatment-naïve patient achieved a viral load of <50 by week 4 and maintained this through week 48. The detailed analysis of individual patient resistance data in this study showed that 12 out of the 20 patients with INSTI resistance also had NRTI, NNRI, or PI resistance as well. In our study, the majority of patients in both cohorts had high-level resistance to at least one NRTI or NNRTI, but low rates of INSTI resistance. This study by D’Antoni et al. 11 reinforced our confidence in utilizing BIC/FTC/TAF as part of our simplification strategies. While there is limited guidance regarding bictegravir use in treatment-experienced patients with drug resistance and in combination with other antiretroviral agents, we feel that our experience could help guide clinicians when multi-class combinations are needed with efforts to reduce pill burden. More studies are needed regarding this specific combination.
The limitations of this study include a small sample size and a retrospective study design. However, the addition of a non-simplified ART group increases the internal validity of our study. There are various reasons why patients in the control group did not undergo simplification, including patient familiarity with their current ART regimen with preference to continue, the presence of hepatitis B infection requiring additional ART agents added to regimen, and inability to simplify patients due to exhaustion of ART options because of extensive ART class resistance. Despite this, the extent of ART resistance among the simplified and non-simplified cohorts was similar except for greater darunavir resistance in the non-simplified cohort.
In conclusion, our study found that simplification of ART regimens based on HIV genotype in PLWH with a history of MDR and prior virologic failures, regardless of the presence of HIV-1 viremia at the time of simplification, resulted in similar rates of virologic suppression and virologic failure as non-simplified ART regimens.
