Abstract
Background:
Intimate partner violence (IPV) is associated with suboptimal HIV treatment outcomes, but its distribution and risk factors among certain subpopulations of people living with HIV in resource-limited settings are not well known. We examined the prevalence, incidence, and recurrence of IPV and its association with adverse childhood experiences (ACEs) among pregnant/breastfeeding women living with HIV in Malawi.
Methods:
This study used longitudinal data for 455 pregnant women living with HIV continuously enrolled in the VITAL Start trial. IPV was assessed at baseline and months 1, 6, and 12 using the widely validated WHO IPV survey. Forms of IPV assessed were physical IPV, emotional IPV, and sexual IPV measured as prevalence, incidence, and recurrence. ACE histories were assessed using WHO’s ACE International Questionnaire (ACE-IQ) tool. Logistic and log-binomial regressions were used in multivariable analyses that controlled for factors such as depression and alcohol use.
Results:
Participants’ mean age was 27.6 ± 5.7 years. Forty-three percent (43%) reported IPV prevalence, 13% reported IPV incidence, and another 13% reported IPV recurrence, with emotional IPV being the most commonly reported IPV type. Over 96% reported experiencing ⩾1 ACE. In regression analysis, cumulative ACE scores were significantly associated with IPV prevalence and IPV recurrence and in both cases, the magnitude of association was greatest for sexual IPV compared with physical IPV and emotional IPV. ACE scores were not significantly associated with IPV incidence.
Conclusions:
IPV is highly prevalent among pregnant women living with HIV and continues to occur throughout the pregnancy and postpartum period; its graded relationship with ACEs is a concern in resource-limited settings where HIV/AIDS remains a public health concern. Strategies aimed to address the needs of pregnant/breastfeeding women living with HIV may benefit from the regular screening of this population for IPV and ACE, including in antenatal care clinics.
Keywords
Introduction
Intimate partner violence (IPV) is a major public health and human rights concern. IPV, which can occur in heterosexual or same-sex couples, includes any behavior by a current or former intimate partner that causes emotional, physical, or sexual harm. 1 IPV affects millions of people globally, but women and some regions are disproportionately affected.2–4 According to the World Health Organization, about 30% of women globally experience IPV, making IPV the most pervasive form of violence against women. 2 In sub-Saharan Africa (SSA), 33% to 44% of ever-partnered women report experiencing IPV.2,5 In Malawi, 40% of women aged 15 to 49 report experiencing IPV. 6 IPV and sexual violence against women violate the sexual and reproductive health and rights of women,7,8 and there is a responsibility to prevent, investigate, report, punish acts of gender-based violence, and support survivors and their families. 9
IPV is also a major public health issue because it has downstream health consequences1,2 and can exacerbate other public health issues such as the HIV pandemic.10,11 IPV is associated with the acquisition of HIV and/or other sexually transmitted infections, physical impairment, emotional trauma, and mental health problems such as depression and increased risk of suicide.2,7 IPV during pregnancy has been linked to poor maternal and neonatal outcomes, including substance use, suicide, depression, maternal death, and preterm birth. 12
In the context of the HIV pandemic, data suggest that the risk of IPV is greater among people living with HIV compared with those without HIV and that IPV is associated with suboptimal HIV treatment outcomes, including poor adherence to antiretroviral therapy (ART) and unsuppressed viral load.10,13,14 In SSA, community-based studies have shown that the risk of IPV is consistently higher among women living with HIV compared with those without HIV. 10 It has been suggested that women living with HIV are at high risk of experiencing IPV upon disclosure of HIV serostatus to their partners.10,11,15 Among pregnant women, both prevalence and incidence of IPV are higher among those with HIV compared with those without HIV.11,16 In SSA, IPV prevalence ranges from 20% to 60% among pregnant/postpartum women living with HIV.17–20 Considering global ambitions to eliminate vertical HIV transmissions by 2030, 21 the high rates of both prevalent and incident IPV among pregnant women living with HIV are particularly concerning. This is because pregnant women living with HIV reporting IPV are more likely not to adhere to ART thereby leading to unsuppressed viral load and subsequent elevated risk of vertical transmission.22–24
While global and regional data show that IPV is associated with poor HIV treatment outcomes, there is a paucity of research investigating the factors associated with IPV among pregnant/breastfeeding women living with HIV in resource-limited settings. This is particularly true about the association between IPV and other factors of similar public health importance such as adverse childhood experiences (ACE) 25 in these settings. 26 ACEs include any traumatic events such as abuse or neglect in the first 17 years of life.27,28 ACEs are widespread, with nearly one billion children exposed to at least one form of ACE worldwide annually. 29 In many countries, particularly those in SSA, children experience multiple adversities.30–39 In Malawi, ACEs are highly prevalent, with over 80% of adolescents and young adults reporting at least one ACE. 36 Because ACEs have been linked to poor health outcomes over the life course,27,28,40 and may also affect HIV treatment outcomes,41,42 understanding their association with IPV among pregnant women living with HIV can help inform the development of interventions aimed to address the needs of pregnant/breastfeeding women living with HIV. Data suggest a positive association between IPV and ACEs,43–45 but these data come from the general population and not specifically from pregnant/breastfeeding women living with HIV in resource-limited and HIV-endemic settings such as Malawi. Moreover, to date, much of the research on IPV, regardless of associated risk factors, has been conducted in resource-rich settings, with the studies mainly focusing on either the prevalence or incidence of IPV but not both. In terms of population subgroups, some studies have focused on IPV among pregnant women,46,47 but much less on pregnant women with HIV. Our previous work in Malawi showed that ACEs are highly prevalent (95%) among pregnant women living with HIV, 26 but we did not examine the association between ACEs and IPV. In all, gaps remain in our understanding of both the distribution of IPV and the association between IPV and ACEs among pregnant/breastfeeding women living with HIV in resource-limited settings.
This study seeks to assess the prevalence, incidence, and recurrence of IPV among pregnant/breastfeeding women living with HIV and examine the association between ACEs and IPV in this population. We hypothesized that the likelihood of experiencing IPV would increase as the number of adverse childhood events (ACEs) increased. Recognizing that various IPV forms may co-occur, 45 the study examined all three forms of IPV: emotional IPV, physical IPV, and sexual IPV.
Methods
Study design and setting
This study used longitudinal data from the VITAL Start (Video-based intervention to Inspire Treatment Adherence for Life) trial (ClinicalTrials.gov identifier: NCT03654898. Registered 31 August 2018, https://clinicaltrials.gov/ct2/show/NCT03654898)—a multisite randomized controlled trial in Malawi that started in 2015 and still ongoing. 48 Details of the VITAL Start trial, including the process for obtaining written informed consent, have been described elsewhere. 48 Briefly, VITAL Start is a brief facility-based video intervention, created with formative participatory research that included conducting focus group discussions with people living with HIV, applied theoretical frameworks, and evidence-informed message-framing techniques. VITAL Start was designed to help optimize retention and adherence to ART among pregnant/breastfeeding women in Malawi. 48 In Malawi, nearly 13% of the female adult population is living with HIV,49,50 and vertical HIV transmission remains a challenge. 51 Malawi is in Southeastern Africa and has one of the lowest GDPs per capita in the world. 50
Sample derivation
For the present analysis, two exclusion criteria were applied to a sample of pregnant women living with HIV enrolled in the VITAL Start study. First, women who were not married or did not have a steady partner were excluded. This ensured that we had a consistent sample of women to report on their IPV experiences both before and after enrollment; it would be challenging to fully capture IPV data from women without regular or steady partners. Second, women not continuously enrolled in the VITAL Start study for 12 months were also excluded. This ensured consistency in estimating IPV prevalence, incidence, and recurrence. Thus, our analytic sample was 455. Questions about IPV experiences were asked at four time points: baseline, month 1, month 6, and month 12.
Measures
IPV
This was assessed using responses to a tool developed by the WHO in 2006. The WHO developed the tool after it had assessed IPV prevalence across 15 countries. 52 This tool has since been used in Malawi and other settings.52–55 The tool consists of 13 behavior-specific questions which assess aggression or abuse in three dimensions: emotional (four questions), physical (six questions), and sexual aggression (three questions). All the questions are binary. This study distinguishes IPV prevalence, incidence, and recurrence:
IPV prevalence is any IPV that occurred before enrolment in the VITAL Start trial. 56 For IPV prevalence, respondents were asked at baseline if they had ‘ever’ experienced a specific act from their current or previous partner. Responses in the affirmative were coded as ‘1’ and ‘0’ otherwise.
IPV incidence is any first-time experience of IPV that occurred in the 12 months of the VITAL Start study (during the current pregnancy and part of the breastfeeding period) among participants without IPV experiences at baseline.56,57 For this, respondents were asked if they had experienced a specific act of IPV from their current or most recent partner in between surveys (between baseline and month 1, month 1 and month 6, and month 6 and month 12). Any responses in the affirmative meant the participant had experienced that specific act of IPV. Responses in the affirmative were coded as ‘1’ and ‘0’ otherwise.
IPV recurrence is defined as the repeated experience of IPV. This variable was constructed from responses across all the four-time points. So, recurrent IPV was coded ‘1’ if the sum of IPV experience across the four-time points was ⩾ 2 and ‘0’ otherwise. Thus, participants experienced recurrent IPV if they reported baseline IPV and experienced IPV during the VITAL Start trial at least once or had no baseline IPV and experienced IPV at least twice during the trial.
For IPV prevalence, incidence, and recurrence, separate variables were created for emotional IPV, physical IPV, sexual IPV, and any IPV. 57 Any IPV was coded ‘1’ if the participant experienced any of the three IPV forms (emotional, physical, or sexual).
ACEs
Maternal histories of ACEs were reviewed using the World Health Organization’s Adverse Childhood Experiences International Questionnaire (ACE-IQ) tool. 25 This tool has been validated in multiple settings and used in Malawi.36,58,59 For the VITAL Start study, the tool was culturally adapted and administered in Chichewa – Malawi’s local language. The tool assesses childhood adversity across 13 categories covering childhood abuse, childhood neglect, household dysfunction, and community dysfunction. While the original tool combines parental death and divorce/separation into one category, 25 this study separated them. Both were pervasive, and based on attachment and social learning theories, their impact on the child may not be the same. 60 To score ACE reports, we used the WHO’s ‘frequency’ method whereby exposure to specific childhood adversity was a ‘1’ (Yes response) and ‘0’ otherwise. 25 For operationalization, we created an ACE score which was a summation of the number of ACEs reported,30,35,38 with 14 as the maximum possible score.
Other variables
We also included confounders such as sociodemographic and behavioral variables and depression. Among the sociodemographic variables were the age of the mother, gestational age, education, literacy, income, employment, whether the current pregnancy was intended, the number of sexual partners in the last year, cohabitation with a partner, partner’s age, partner’s HIV status, amount of time (years) in the relationship, and time since HIV diagnosis. Behavioral variables included maternal alcohol use and drug use. Alcohol use was assessed using the AUDIT (Alcohol Use Disorders Identification Test) tool – a 10-item tool with a maximum possible score of 40. 61 From the scores and as recommended, 61 an alcohol use variable tool was created with three categories (nonusers (total score: 0), moderate alcohol use (total score: 1 to 7), and heavy alcohol use (total score: 8 and above). For this study, however, alcohol use was reduced to a binary variable (0 or 1) because of the small number of cells in the third category (heavy users). Drug use was assessed using the DUDIT (drug use disorders identification test) tool – an 11-item tool. 62 From the scores, a binary variable indicating users and nonusers of drugs was created. We also included depression which was assessed using a 20-item WHO’s self-reporting questionnaire (SRQ-20). 63 The SRQ-20 can reliably identify probable cases of depression even among pregnant women.64,65 Participants scoring at least 8 (out of 20) were considered depressed.66,67 As part of the VITAL Start procedures, participants with IPV experiences, harmful alcohol use, drug use, and probable cases of depression were referred for nonstudy-related activities such as psychosocial counseling.
Data analysis
Data were analyzed using both univariable and multivariable approaches. In univariable analyses, frequencies and proportions summarized categorical variables while means and standard deviations summarized continuous variables. Chi-square analysis was used to test associations between IPV (prevalence, incidence, and recurrence) and the covariates (all control variables were categorical). In multivariable analyses, the association between IPV prevalence and ACEs was modeled using logistic regressions. Log binomial regressions, with robust error variances, were used to model the association between IPV incidence and ACEs as the relative risks were of interest as a measure of association. Finally, logistic regressions were also used to model the association between IPV recurrence and ACEs. To determine the final models in all cases, we included the primary exposure (ACE), important behavioral and biological variables in the model regardless of significance, and then used the change in estimate rule to select potential confounders. A variable was a confounder if its addition to a regression model led to a change in the effect size by at least 10%. The models were checked for specification errors (using the linktest) and goodness-of-fit (using the Hosmer–Lemeshow test). In addition, we used the LOWESS (locally weighted scattered plot smoothing) to check or confirm that the logits of the outcome variable and ACE scores had a linear relationship. Because of multiple testing on the outcome variable, the traditional alpha level of 0.05 for determining statistical significance was adjusted using the Bonferroni correction. 68 A p-value < 0.0125 was, therefore, determined to be statistically significant. All tests were two-sided. Results from logistic regressions are presented as odds ratios and those from log-binomial regressions as risk ratios (RRs). Predicted probabilities of the outcome are presented where necessary. All analyses were performed in Stata 14.
Sensitivity analysis
In sensitivity analysis, we removed one exclusion criterion in deriving the sample for this study. Unlike in the main analysis where only women with continuous enrollment for all 12 months were included (n = 455), all women (married or with a steady partner) enrolled in the VITAL Start study regardless of enrollment length or missing a study visit were now included in the sensitivity analysis (n = 733). While having this exclusion helps us to achieve internal consistency in estimating IPV incidence, removing it helps to make our findings more generalizable as women continuously enrolled in the VITAL Start may be inherently different from those that exited the study or missed some study visits.
Results
Baseline sample characteristics
This analysis included 455 women who had partners and were continuously enrolled for 12 months in the VITAL Start study. All participants had complete data both at baseline and follow-up. Participant ages ranged from 17 to 49 years, with a mean age of 27.6 ± 5.7 years. The majority of the participants (>50%) were literate, self-employed, had incomes not exceeding 50,000 Malawi Kwacha per month (Table 1). Alcohol and drug use were not pervasive but two in five participants were probable cases of depression. ACEs were highly prevalent, with 96% and 65% reporting at least one form and three forms of childhood adversity, respectively. The mean number of ACEs reported by each participant was 3.7 ± 2.3.
Demographic characteristics, adverse childhood experiences, and intimate partner violence among women continuously enrolled in the VS study for 12 months (n = 455).

ACE, adverse childhood experiences; IPV, intimate partner violence; VS, VITAL Start.
The percentages may not add up to 100% due to rounding.
IPV at baseline and follow-up
Many participants reported IPV both at baseline and follow-up although baseline proportions were consistently higher across the three forms of IPV. The proportion reporting any IPV was nearly half (196/455 or 43%) at baseline but decreased to about 10% at each of the follow-up time points (month 1, month 6, and month 12), Table 2. At all-time points, emotional IPV was the leading form of IPV with 31% and 6% reporting this IPV at baseline and each of the follow-up time points. IPV prevalence was at least 20% for each of the three IPV forms (emotional, physical, and sexual) while incident and recurrent IPV ranged from 4% to 9% (Table 3).
IPV reports at each study time point (N = 455).

IPV, intimate partner violence.
IPV prevalence, incidence, and recurrence.
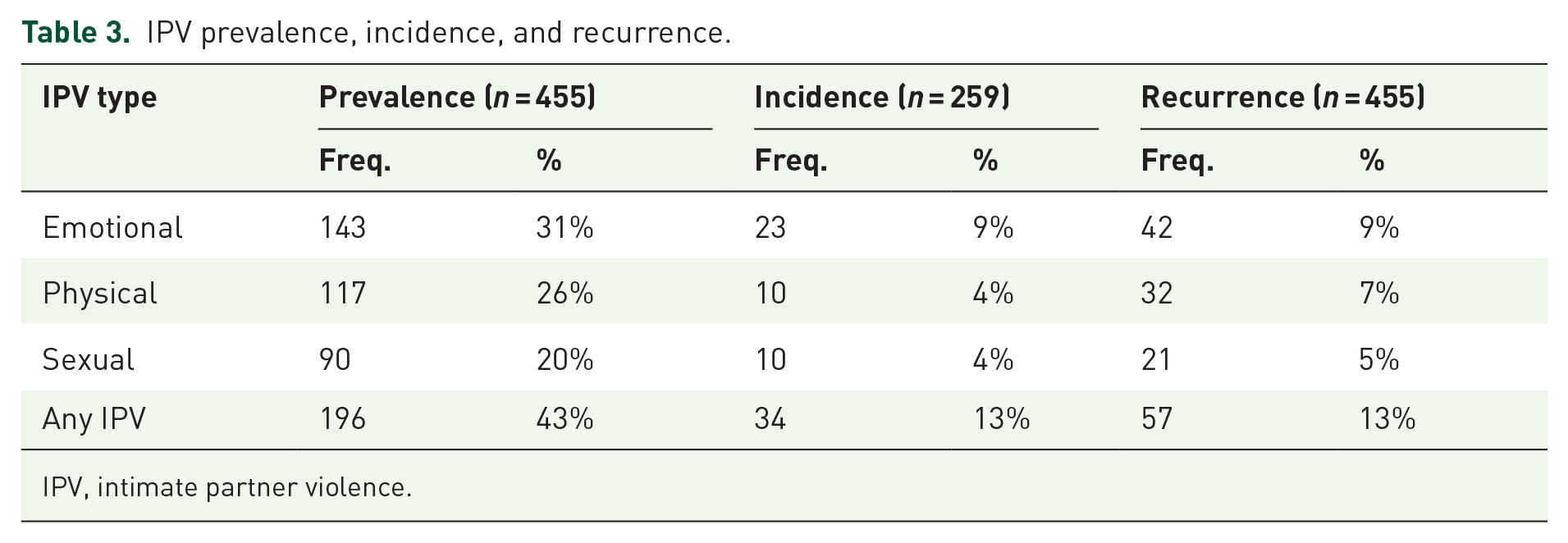
IPV, intimate partner violence.
ACE exposure and IPV (bivariable analysis)
In unadjusted bivariable analyses, ACE scores were significantly associated with IPV prevalence and IPV recurrence, but not with IPV incidence. On average, there was at least a 1.5 ACE score difference between participants reporting any IPV prevalence and those not reporting any IPV prevalence (mean ACE scores 4.6 ± 2.4 versus 3.1 ± 2.0). Among the three forms of IPV, sexual IPV prevalence had the largest difference in mean ACE scores between those reporting this form of IPV (mean ACE score = 5.3 ± 2.5) and those not reporting it (mean ace score = 3.4 ± 2.1). The difference in ACE scores was the lowest among those reporting and not reporting physical IPV prevalence, but it was nonetheless significant. While similar results were observed when we examined the association between ACEs scores and IPV recurrence, the differences in mean ACE scores among those reporting a particular IPV form and those not reporting that form of IPV were now larger for all IPV forms. For example, participants reporting recurrent sexual IPV had a mean ACE score of 6.2 ± 2.4 compared with a mean ACE score of 3.7 ± 2.2 among those not reporting recurrent sexual IPV (p < 0.001). Notably, as well, ACE scores were not significantly associated with IPV incidence. Also, those with prevalent IPV were more likely to have recurrent IPV; there was no association between incident IPV and recurrent IPV.
Regression results
A similar pattern of results was also observed in multivariable regressions, with ACE scores significantly associated with IPV prevalence and IPV recurrence but not IPV incidence.
IPV prevalence
IPV prevalence was significantly associated with several baseline variables, including ACE scores, although the strength and magnitude of association varied by IPV form (Table 4). While all IPV forms were significantly associated with ACE scores, a unit increase in ACE scores had the greatest effect on the odds of experiencing sexual IPV followed by emotional IPV. For example, a one-unit increase in ACE score increased the odds of sexual IPV by a factor of 1.34 (99% CI: 1.15, 1.55) compared with 1.24 (99% CI: 1.09, 1.1.41) for emotional IPV. Notably, depression was significantly associated with all three IPV forms, with the magnitude of the association greatest for sexual IPV. In addition, women who reported using alcohol were significantly more likely to report experiencing physical IPV, but not the other two forms of IPV. And, women who were pregnant at least once before and those in the third trimester of the current pregnancy were significantly more likely to report emotional IPV, but not the other two forms of IPV.
The association between ACEs and IPV prevalence among pregnant women living with HIV (N = 455).

ACE, adverse childhood experiences; CI, confidence interval; HIV, human immunodeficiency virus; IPV, intimate partner violence.
Exponentiated coefficients; 99% confidence intervals in brackets.
p < 0.05, **p < 0.01, ***p < 0.001.
Consistent with the results reported in Table 4, the predicted probability of IPV prevalence increased as total ACE scores increased (Figure 1). The rapid rise of the sexual IPV curve, especially after a total ACE score of four, suggests that the effect of an additional ACE is smaller among those with fewer ACEs but the effect increases more rapidly among those with at least four ACEs. For example, a unitary increase in total ACE scores changes the probability of sexual IPV by + 3 percentage points from three to four ACEs, +4 percentage points from five to six ACEs, and +6 percentage points from nine to 10 ACEs. However, the steady rise in the emotional IPV curve suggests a more uniform effect of a unitary increase in total ACEs. And the flatness of the physical IPV curve suggests that the effect of a unitary increase in total ACEs is lower compared with sexual IPV or emotional IPV.

Average predicted probability of IPV prevalence at various levels of ACE scores. This figure shows that the average predicted probability of prevalent IPV (emotional, physical, sexual, or any IPV) increases with accumulating childhood adversity. The various shapes of the curves suggest differential effects of a unitary increase in total ACE score on the predicted probability of IPV prevalence across the various types of IPV.
IPV incidence
While trends suggested a higher risk of IPV incidence among those with higher ACEs scores, the association was not significant even at an alpha of 0.05. Similar results were observed with other variables in the model, except for depression. Even then, the risk was only significantly higher for sexual IPV among women who were depressed (RR = 5.75; 99% CI: 1.12, 29.61).
IPV recurrence
Similar to IPV prevalence, the odds of IPV recurrence were significantly higher among women reporting experiencing one or more forms of childhood adversity. A one-unit increase in ACE score was associated with higher odds of experiencing IPV recurrence as follows: emotional IPV (OR = 1.30; 99% CI: 1.07, 1.57); physical IPV (OR = 1.31; 99% CI: 1.05, 1.64); and sexual IPV (OR = 1.48; 99% CI: 1.13, 1.93). In addition, all three forms of IPV were significantly associated with being depressed while only physical abuse was associated with alcohol use.
We also examined the effect of a one-unit increase in ACE score on average predicted probabilities of IPV recurrence (Figure 2). Across the three forms of IPV recurrence (emotional, physical, and sexual), the average predicted probability of experiencing any of the three forms increases with accumulating childhood adversity. For example, among women reporting four ACEs (the average ACE score in our sample), the average probabilities are 0.08 for emotional IPV recurrence, 0.06 for physical IPV recurrence, and 0.03 for sexual IPV recurrence. However, doubling the ACE scores has a disproportionately higher effect on the average predicted probabilities. For example, an ACE score of eight is associated with average predicted probabilities of 0.19 for emotional IPV recurrence, 0.15 for physical IPV recurrence, and 0.13 for sexual IPV recurrence. With the concave shapes of the curves, the average probabilities of IPV recurrence get even larger with further accumulation of childhood adversity.
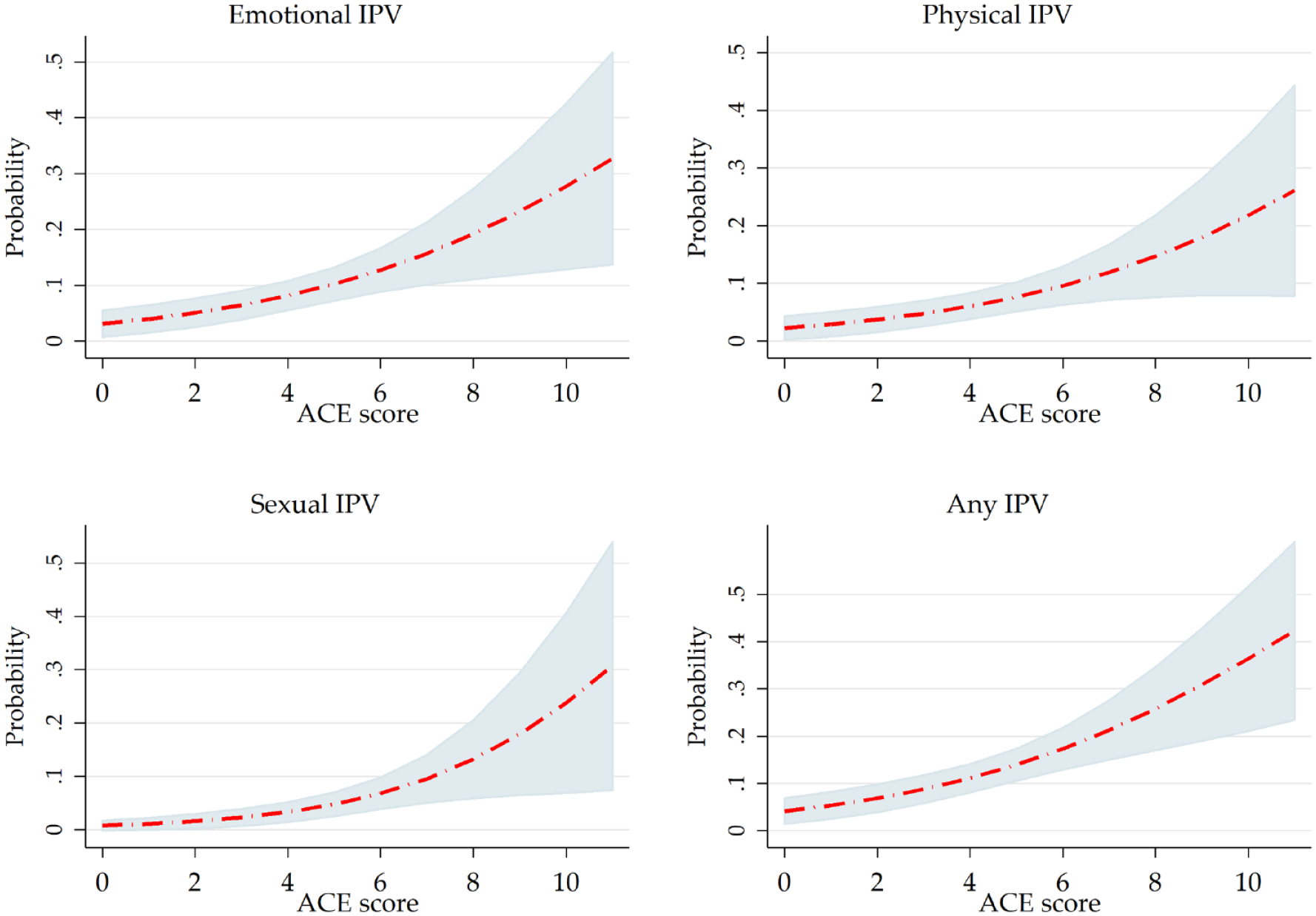
Average predicted probability of IPV recurrence at various levels of ACE score. The average predicted probability of experiencing recurrent IPV (emotional, physical, sexual, or any IPV) increases with accumulating childhood adversity. The concave shapes of the curves suggest that the average probabilities of IPV recurrence get even larger with further accumulation of childhood adversity.
Sensitivity analysis results
In sensitivity analysis, our findings remained robust to a change in the definition of the sample to include all women with partners regardless of enrollment length (Table 5). Results from both samples are similar in terms of both magnitude and strength of association between IPV and ACE scores, except for physical IPV (both prevalent and recurrent) which has a much stronger association with ACE scores in the sensitivity analysis compared with the main analysis. This suggests that women continuously enrolled in the VITAL Start are more likely to have reported physical IPV across the distribution of ACE scores compared with those not continuously enrolled in the VITAL Start trial. Nonetheless, as with other forms of IPV, the differences do not materially change our findings or conclusions.
The association between ACE scores and IPV in a sensitivity analysis.

ACE, adverse childhood experiences; CI, confidence interval; IPV, intimate partner violence; OR, odds ratio; RR, risk ratios.
In the main analysis, the sample size was 259 which increased to 267 in the sensitivity analysis.
p < 0.01, **p < 0.001.
Discussion
This study sought to examine the prevalence, incidence, and recurrence of IPV and its association with adverse childhood experiences among pregnant/breastfeeding women living with HIV in Malawi – a typical resource-limited setting where HIV is endemic. To our knowledge, this is the first study in this setting to report IPV prevalence, incidence, and recurrence across all three forms of IPV (physical, emotional, and sexual) in pregnant/breastfeeding women living with HIV. Multiple measures of IPV allowed us to differentiate women who had experienced IPV before study enrollment (IPV prevalence) from those who experienced it for the first time but during this pregnancy and postnatal period (IPV incidence) or experienced it on either side of study enrollment (IPV recurrence). We were able to estimate IPV prevalence at baseline and IPV incidence during follow-up, which other studies from resource-limited settings have been unable to achieve. In this study, IPV prevalence was reported by nearly half (43%) of the participants, followed by both IPV incidence and recurrence at 13%. ACEs were significantly associated with both IPV prevalence and recurrence but were not associated with IPV incidence. In sensitivity analysis, the association between ACEs and IPV was robust to a change in the definition of the sample to include all women with partners regardless of enrollment duration.
Overall IPV prevalence among pregnant women living with HIV was high (43%). This finding is comparable with other IPV prevalence reports from WHO (37%), 69 SSA (44%), 5 and Malawi (39–41%).20,55 Our findings are also similar to studies in Ethiopia (41%) 46 and Nigeria (45%) 70 but higher compared with reports from Namibia (8%), 71 South Africa (21%), 17 and Tanzania (27%). 72 These differences could be attributed to differences in the study population, time period of assessing the outcome (IPV), and differences in methodology, including sample size and eligibility criteria. Similar to previous studies, we were able to distinguish specific forms of IPV and found that out of the overall prevalence, 31%, 26%, and 20% were emotional, physical, and sexual IPV, respectively. This appears to be consistent with previous research demonstrating that pregnant women living with HIV most commonly experience emotional IPV compared with physical and sexual IPV.46,71 This study has also demonstrated that ACEs were significantly associated with all forms of IPV with the highest odds of experiencing sexual IPV followed by emotional IPV. The odds got higher as the ACE score increased, which is consistent with similar studies in SSA and resource-rich settings.43,44 Similar to other studies, depression, alcohol abuse, and number of previous pregnancies were associated to IPV prevalence.73,74 While additional research focusing on the links between ACEs, IPV, depression, and alcohol abuse outcomes among pregnant/breastfeeding women living with HIV in this setting is warranted, the evidence from this study suggests that those experiencing forms of IPV and ACEs also have concomitant conditions such depression and alcohol abuse.
IPV incidence was 13%, with 9% incident emotional violence, 4% physical violence, and 4% sexual violence, which is higher than the IPV incidence reported in other studies conducted in SSA.75,76 Recent studies in South Africa and Ethiopia have reported IPV incidence of 7% to 8% during pregnancy.75,76 These differences, however, could be attributed to differences in study populations, with those studies not limiting their samples to pregnant women living with HIV. In multivariable analyses, ACEs, alongside all other demographic factors, were not associated with any form of incident IPV. This could be due to rare incident events or underreporting in the follow-up study period. However, it was noted that the risk of incident sexual IPV was higher among women who were probably depressed. Therefore, it will be important to continue to explore risk factors of IPV incidence in resource-limited settings and conduct further research to better understand the variations in IPV incidence among studies.
Reports of IPV recurrence (13%) among pregnant/breastfeeding women living with HIV during the vulnerable period of pregnancy and postpartum are a source of worry. Despite continued concerns about underreporting, 77 the rate of IPV recurrence suggests that screening for and attending to IPV should be continuous during pregnancy. Out of the overall IPV recurrence, 9% was emotional, 7% physical, and 5% sexual violence. Thus, a one-time clinical assessment of IPV (emotional violence, physical violence, and sexual violence) during pregnancy may not be sufficient in identifying the majority of women still experiencing IPV. The recurrence of IPV in a relationship is dangerous for the woman’s well-being. For example, it puts the pregnant woman at risk of preterm delivery. 78 In multivariable analyses, ACEs were significantly associated with all forms of IPV recurrence with the highest odds of experiencing sexual IPV followed by emotional and physical IPV. The odds of experiencing emotional, physical, and sexual IPV increased with accumulating ACEs scores indicating a graded relationship between the exposure and the outcome. ACEs and depression were found to be predictors of IPV recurrence and strategies to interrupt the recurrence of violence should be advocated and implemented during pregnancy and postpartum. A robust understanding of when and why IPV continues to occur is critical for developing appropriate screening recommendations and clinic-based interventions in resource-limited settings.
This study offers important insights into the distribution of IPV and its association with ACEs among pregnant/breastfeeding women living with HIV in a resource-limited setting. The likelihood of IPV increased as the total ACE score increased. Strategies to address the needs of pregnant/breastfeeding women living with HIV must include screening for IPV and ACE during antenatal or HIV services and tailored programs or interventions to address and/or prevent IPV. As ACEs are also highly prevalent in this setting, additional research to understand the causes of the high level of ACEs in our setting is warranted.
This study has several limitations. First, our analytic sample comprised women in relatively steady partnerships which may affect the generalizability of our findings given that women not in steady relationships or without regular partners such as those who engage in transactional sex may have different IPV experiences. A second limitation is that we relied on self-reports to collect data on ACEs and IPV which may introduce either recall bias or social desirability bias or both. Unfortunately, if the bias exists, we are unable to tell its direction. Notwithstanding this limitation, almost all prior research on ACEs or IPV has relied on self-reports.40,79 Importantly, widely validated instruments were used to estimate ACEs and IPV, strengthening the validity of the study results.52,80 A third limitation is that we might be missing insights from women living with HIV as they were not involved at every step of this particular research. Although women living with HIV were involved in creating the video for the VITAL Start intervention, they were neither involved in interpreting findings nor making recommendations in this particular research. All our future work will address this limitation.
Implications for policy and future research
The results of this study are crucial for policy and future research. The data demonstrate that IPV continues to be a significant serious public health problem for pregnant/breastfeeding women living with HIV in resource-limited settings. To start with, the Malawi government and its development partners should implement policies and programs designed to prevent, detect, and treat IPV. It would be easier for abused women to get help if healthcare workers routinely assessed for IPV and referred those requiring further attention. Unfortunately, in Malawi, screening for IPV among pregnant women in general or those living with HIV is not done as part of routine care, and there are no proper structures for effective referral of those requiring additional support. Developing a tool (a short questionnaire) to screen pregnant women for IPV on each ANC visit that captures several forms of violence and creating effective referral structures at the various levels of care can help in this regard. Alongside this tool, should be strategies (for example, promoting inclusion of peer mentor mothers as part of the healthcare workforce81,82) to help ensure that the tool is indeed administered among pregnant/breastfeeding women living with HIV given experiences of denial of care and discrimination based on HIV status when these women seek maternal health care. 83 Those reporting IPV can then receive additional care or support. While this tool/policy intervention can start in a few large sites, it can later be implemented in all health facilities dedicated to providing antenatal care, HIV testing, and ART services as evidence of its benefits becomes available. This recommendation can easily be extended to help address ACEs as noted in our work previously. 26
Furthermore, noting that IPV, particularly sexual violence, is not usually reported at the time it occurs, strategies to bring change at the community level must be quickly identified and introduced. Such strategies need to be multisectoral and could include proactively engaging communities and local leaders to ensure quick and safe reporting of any form of IPV, as well as the involvement of law enforcement and civil society agencies to ensure that both the women and community members who report violence are protected. As noted in research conducted by people living with HIV, enforcement of frameworks to protect the rights of women and young girls living with HIV in countries such as Malawi has been challenging. 9 Therefore, the involvement of law enforcement in these multisectoral strategies can enhance collaboration between communities and law enforcement agencies to ensure safe reporting of IPV and that perpetrators of IPV are prosecuted. In addition, awareness creation in the community about the availability of policies, programs, and interventions on IPV is paramount to encourage IPV reporting and uptake of services aimed to help victims of IPV. As part of an effort to reduce IPV in Malawi, in 2013 the Coalition of Women Living with HIV and AIDS (COWLHA) trained community paralegals in 12 districts (out of 28) to promote the rights of women living with HIV, address gender imbalances, and provide guidance/advice to women who have been abused in those districts. 84 The community paralegal initiative is worth supporting, but 10 years down the line its present and future statuses are unclear. Further, programs and interventions to address risk factors for IPV such as ACEs as well as concomitant conditions such as depression and alcohol abuse which should include screening, prevention, and treatment as part of antenatal or HIV care for women in general are needed. In summary, integration of IPV and ACE screening and psychosocial interventions into antenatal or primary care settings, maybe a simple and effective way to help improve care for pregnant/breastfeeding women living with HIV and for them to remain successfully engaged in HIV care for the long term. Future research should aim to assess the evidence to support the effectiveness of screening women for IPV and referral systems in the primary care setting in resource-limited settings. Such knowledge when obtained, will likely persuade governments and partners across the SSA region to make IPV assessment become part of the standard of care.
Conclusion
This study has demonstrated that IPV is highly prevalent among pregnant women living with HIV in resource-limited settings, and unfortunately, they continue to experience this violence throughout their pregnancy and the postpartum period. These findings should be of serious concern to policymakers and all stakeholders involved in the fight against the HIV pandemic since pregnant women living with HIV reporting IPV are more likely to not adhere to ART, thereby leading to unsuppressed viral load and subsequent elevated risk of vertical transmission. Our findings have also highlighted that ACEs have a graded relationship with IPV, suggesting that the problems affecting pregnant/breastfeeding women living with HIV in these settings are multifaceted and require concerted efforts. Routine screening of pregnant/breastfeeding women living with HIV for IPV and ACE and providing targeted support services need to be considered as part of the package of primary care for pregnant/breastfeeding women living with HIV.
Footnotes
Acknowledgements
We acknowledge and thank all study participants who took their valuable time to participate in this study. We also gratefully acknowledge the Malawi Ministry of Health, Tingathe Outreach Program Team, Baylor College of Medicine Children’s Foundation Malawi, Baylor International Pediatric AIDS Initiative, The U.S. President’s Emergency Plan for AIDS Relief (PEPFAR), and the U.S. Agency for International Development (USAID). We thank the participants and organizers of the INTEREST International Conference on HIV Treatment, Pathogenesis, and Prevention Research in Resource-Limited Settings where this work was initially presented. We also thank the anonymous reviewers of this article.
