Abstract
Background:
A vancomycin AUC/MIC (area under the curve/minimum inhibitory concentration) of 400–600 mg•h/L is associated with improved clinical outcomes for the treatment of methicillin resistant Staphylococcus aureus (MRSA) infections. Currently, there are still limited studies evaluating the relationship between vancomycin trough and AUC.
Objectives:
To evaluate the correlation between vancomycin trough and AUC/MIC and determine if trough-guided monitoring is an adequate predictor of AUC/MIC in the Urban Health population at St Paul’s Hospital.
Methods:
This was a retrospective chart review of 29 patients from November 2019 to February 2021. Patient demographics and laboratory data were collected from electronic medical records. The two-level equation-based approach was used to calculate AUC/MIC. The proportion of AUC/MIC values within target (400–600 mg•h/L) despite subtherapeutic troughs, and the proportion of AUC/MIC values supratherapeutic when trough is within target (15–20 mg/L) were the primary endpoints.
Main Results:
Fifty-seven sets of levels were collected and 75% of vancomycin troughs and AUC24 were found to be discordant. When trough was 10–14.9 mg/L, AUC24 was > 400 mg•h/L in 94% of cases and when trough was 15–20 mg/L, AUC24 was > 600 mg•h/L in 69% of cases. There was a moderate correlation between vancomycin trough and AUC24h (R2 = 0.57; p < 0.001).
Conclusion:
A vancomycin trough between 15 and 20 mg/L may result in an AUC/MIC greater than necessary for clinical efficacy. Considering these findings and the practical concerns of AUC-guided monitoring, a modest reduction in target troughs to prevent vancomycin toxicity warrants clinical consideration and further evaluation.
Introduction
Vancomycin is a glycopeptide antibiotic that is commonly used for the treatment of infections caused by methicillin-resistant Staphylococcus aureus (MRSA). 1 The pharmacodynamic/pharmacokinetic (PK/PD) target associated with vancomycin effectiveness against MRSA is the ratio of the area under the area (AUC) over 24 h to the minimum inhibitory concentration (MIC; AUC/MIC). 2 For invasive MRSA infections, a target AUC/MIC of 400–600 mg•h/L is recommended when MIC is determined to be ⩽1 mg/L by broth microdilution.2,3 Vancomycin requires the use of therapeutic drug monitoring (TDM) for delivery of optimal therapy outcomes.3,4 Despite more than 60 years of use in clinical practice, knowledge gaps regarding the most appropriate method for optimizing vancomycin therapy and minimizing toxicity still exist.
The previous consensus guidelines on vancomycin TDM recommend targeting trough concentrations of 15–20 mg/L as a surrogate marker of AUC/MIC ⩾400 mg•h/L to simplify the management of therapy. 5 However, the clinical benefits of obtaining trough concentrations of 15–20 mg/L for invasive MRSA infections are not well established. Since the implementation of these recommendations, there has been evidence of increased dose-related adverse effects, most notably nephrotoxicity.6,7
Trough concentrations of 15–20 mg/L have been shown to result in AUC values well above 400 mg/L*h. 8 This is of concern due to the direct relationship between vancomycin exposure and toxicity.9,10 Several previous studies have found a positive correlation between vancomycin trough concentration and AUC in various populations, including geriatric, critically ill, and pediatric patients.11–13 However, there is a high degree of interindividual variability between a measured trough level and the actual AUC/MIC.8,14 The therapeutic discordance between AUC and trough may lead to poor treatment outcomes due to supratherapeutic or subtherapeutic AUC/MIC despite trough level being within target range, or unnecessary vancomycin dose escalations due to seemingly subtherapeutic trough levels despite therapeutic AUC/MIC values. Recent PK/PD studies have demonstrated a significant decrease in total vancomycin exposure and nephrotoxicity rates without compromising outcomes when AUC/MIC monitoring was utilized instead of trough-only level monitoring.8,15 Recent guidelines on vancomycin TDM also suggest that AUC/MIC-guided dosing and monitoring is the most accurate and safest method in patients with suspected or definitive serious MRSA infections. 3 Despite these new recommendations, trough-only level monitoring is still widely practiced, 16 and logistical considerations may limit the feasibility of performing AUC/MIC monitoring where two time-sensitive levels, as opposed to one, must be drawn. 17
The Urban Health unit at St. Paul’s Hospital (SPH) is a clinical area that has adopted the use of AUC/MIC vancomycin dosing and monitoring since its recommendation by the Infectious Diseases Society of America (IDSA) in early 2020. An AUC between 400 and 600 mg•h/L is targeted for MRSA isolates found to have an MIC of less than 2 mg/L based on E-test. When MIC was not reported (i.e., for MRSA isolates in body fluids other than blood), the MIC was assumed to be 1 mg/L. The Urban Health service provides care to adult patients 19 years of age and over who need hospitalization for issues related to human immunodeficiency virus (HIV)/acquired immunodeficiency syndrome (AIDS), or require concurrent management of an infection and addiction. Patients under the Urban Health service are a unique population with altered PK parameters compared to other hospitalized patients. These patients are typically young in age, have low weight with muscle atrophy and have rapid renal clearance. Vancomycin dose adjustments based on trough level alone may result in supratherapeutic or subtherapeutic AUC/MIC values, leading to increased risk for toxicity or treatment failure. Characterizing patients with AUC/MIC-trough discordance may help predict which patients would generally benefit from AUC/MIC monitoring, and which patients can continue to be safely monitored with trough levels alone.
Clinical studies examining the relationship between vancomycin trough concentration and AUC are still limited. As appropriate vancomycin dosing and monitoring are essential to improving patient outcomes, this study aims to determine if there is correlation between trough and AUC/MIC, as well as whether trough-guided monitoring is an adequate predictor of AUC/MIC in the Urban Health population.
Methods
This retrospective chart review was conducted at St. Paul’s Hospital (a 550-bed tertiary care center) in Vancouver, British Columbia from November 16, 2019 to February 27, 2021. Ethical approval of the study was granted by the University of British Columbia – Providence Health Care Research Ethics Board. This study was deemed as minimal risk by the University of British Columbia – Providence Health Care Research Ethics Board, and informed consent of participants was not required.
This study included patients under the Urban Health service at SPH who were 18 years of age or older, received three or more doses of intravenous vancomycin and had one or more sets of steady state peak and trough concentrations. Steady state was defined as any time after the third consecutive vancomycin dose, in accordance with the 2020 IDSA vancomycin TDM guidelines. 3 Patients who were undergoing renal replacement therapy, were pregnant, or grew MRSA with an MIC ⩾ 2 mg/L were excluded. For the study patients, the following data were collected: age, sex, weight, height, serum creatinine levels, vancomycin levels, medication regimens, concurrent nephrotoxins or medications known to increase serum creatinine, comorbidities, type of infection, and microbiology results. Cockcroft and Gault equation utilizing total body weight was used to calculate an estimated creatinine clearance.
The primary objectives were to identify the proportion of AUC/MIC values within target (400–600 mg•h/L) despite supratherapeutic or subtherapeutic troughs, as well as the proportion of AUC/MIC values that are supratherapeutic or subtherapeutic when trough level is within target (15-20 mg/L). Secondary objectives were to determine the differences in vancomycin total daily doses between AUC/MIC monitoring (AUC/MIC target of 400–600 mg•h/L) compared to trough-guided monitoring (target trough of 15–20 mg/L), possible predictors of AUC/MIC, and the frequency of nephrotoxicity.
First-order PK equations (Sawchuk-Zaske method) and the trapezoidal model were used to calculate AUC (Appendix 1). Two steady state levels must have been available, with one level drawn following the distribution phase of vancomycin (at least 1–2 h after the end of the infusion) and another level just prior (within 30 min to 1 h) to the next scheduled dose. Ideally, these were drawn within the same dosing interval, but adjacent dosing intervals at steady state were acceptable due to logistical challenges in practice. These levels were used to calculate a patient’s PK parameters by clinical pharmacists, which were then used in the linear trapezoidal and logarithmic trapezoidal formulas to calculate the AUC for a single dose. 24-h AUC was calculated based on the number of daily doses administered. The two-level equation based approach has been validated to have similar accuracy and bias compared to Bayesian modeling, 17 which is not available at SPH. Because the method used to determine MIC at SPH, E-test, overcalls MIC by 0.5 to 2 dilutions, 3 the MIC was assumed to be 1 mg/L for all patients included.
We determined differences in total daily dose by first comparing calculated AUC and trough and assessing whether these values were concordant. These values were considered concordant if trough was < 15 mg/L and AUC was < 400 mg•h/L, or if trough was between 15 and 20 mg/L and AUC was between 400 and 600 mg•h/L, or if trough was > 20 mg/L and AUC was > 600 mg•h/L. If values were deemed discordant and the vancomycin dose was not changed (i.e., AUC within range but trough concentration was subtherapeutic), we determined the theoretical daily vancomycin dose required to achieve a trough of 15–20 mg/L using proportional dosing based on first order PK principles.
Nephrotoxicity was defined as an absolute increase in serum creatinine ⩾ 26.4 μmol/L, or a 50% increase from baseline within a 48-h period. This definition was developed by the Acute Kidney Injury Network to assist in early identification of drug-induced kidney damage to improve clinical outcomes.18,19
Statistical analysis
Baseline characteristics were summarized using means (standard deviations) for normally distributed continuous variables and medians (interquartile ranges) for non-normally distributed continuous variables. Number of patients (percentages) were used for categorical variables. Correlation analysis of AUC/MIC and trough concentration was performed using Pearson’s correlation coefficient. Univariate analysis was also performed with the outcome variable as AUC and dependent variables being trough along with other pre-specified covariates hypothesized to affect patient pharmacokinetics. These included age, weight, height, baseline serum creatinine and the presence of a concurrent nephrotoxin or medication known to increase serum creatinine. Results from the univariate analysis were used to identify variables for the multivariate model. For inclusion in the logistic regression model, the threshold p-value was 0.2. Regression analysis was used to identify factors confounding the correlation between vancomycin AUC/MIC and trough concentration. Statistical analyses were performed using R (version 4.2.2, R Foundation for Statistical Computing, Vienna, Austria).
Results
A total of 155 patients were screened based on the inclusion and exclusion criteria to reach a sample size of 29 patients (Figure 1). The average age of the cohort was 42 years, and 76% of patients were male (Table 1). The average weight was 65 kg. The mean serum creatinine level at baseline was 60 μmol/L, with a calculated creatinine clearance of > 120 mL/min. 41% of patients were on a nephrotoxin or medication that could affect their serum creatinine (Table 1). The most common infection being treated was bacteremia and the most common causative organism was MRSA. The average duration of vancomycin therapy was 28 days and patients achieved a median trough level of 13.5 mg/L with a median AUC of 572 mg•h/L.

Flow diagram of inclusions.
Patient demographics, clinical characteristics and vancomycin PK/PD parameters.

AUC24h, 24-h area under the curve; COPD, chronic obstructive pulmonary disease; HIV, human immunodeficiency virus; MRSA, methicillin resistant Staphylococcus aureus; MSSA, methicillin sensitive Staphylococcus aureus; PK/PD, pharmacodynamic/pharmacokinetic; SCr, serum creatinine; SD, standard deviation.
From the 29 patients included, we collected 57 sets of steady state peak and trough concentrations. Nineteen patients had 1 set each collected, 3 patients had 2 sets each collected, 3 patients had 3 sets each collected, 2 patients had 4 sets each collected, 1 patient had 5 sets collected, and 1 patient had 10 sets collected. The majority of steady state peak and trough sets (n = 54) corresponded to an AUC/MIC > 400 mg•h/L including 94% of troughs between 10 and 15 mg/L (Table 2). Of the 31 trough concentrations between 10 and 14.9 mg/L, 2 (7%) achieved an AUC of < 400 mg•h/L, 21 (68%) achieved a target AUC of 400–600 mg•h/L and 8 (26%) achieved a supratherapeutic AUC of > 600 mg•h/L (Table 2). Of the 13 trough concentrations within therapeutic trough concentration range of 15–20 mg/L, 4 (30%) achieved target AUC of 400–600 mg•h/L and 9 (70%) achieved a supratherapeutic AUC above 600 mg•h/L. Based on the predefined definition of concordance for this study, 43 (75%) AUC values were discordant with trough concentrations and 14 (25%) were concordant. Concordance was higher when a vancomycin trough of 10–15 mg/L was assumed to correspond to an AUC between 400 and 600 mg•hour/L. Based on this definition, 19 (33%) AUC values were discordant and 38 (67%) were concordant. Linear regression analysis produced a Pearson correlation coefficient of 0.57, indicating a moderately positive relationship between AUC and trough concentrations (Figure 2). The corresponding regression equation predicts AUC based on trough concentration [AUC24 (mg•h/L) = [trough concentration (mg/L) + 1.4936]/0.0274].
Comparison of 24-h AUC and trough concentrations.

AUC24h, 24-h area under the curve (mg•h/L).

Relationship between vancomycin AUC24h/MIC and trough.
Univariate analysis revealed that weight (p = 0.11), baseline serum creatinine (p = 0.02) and trough concentration (p ⩽ 0.001) were significantly associated with AUC (Table 3). A patient’s age, height and if they were on a concurrent nephrotoxin or agent that may alter serum creatinine did not show a statistically significant association. Multivariate regression using variables with p value < 0.2 on univariate analysis found that only trough concentration was an independent predictor of AUC (p ⩽ 0.001). The analysis showed that a 1 mg/L increase in trough concentration reflects an increase of 21.69 mg•h/L in the AUC (Table 4). The p value for the Hosmer-Lemeshow goodness-of-fit statistical test for the regression model was 0.58, which indicated a moderate association between AUC and trough concentration.
Predictors of AUC/MIC by univariate analysis.
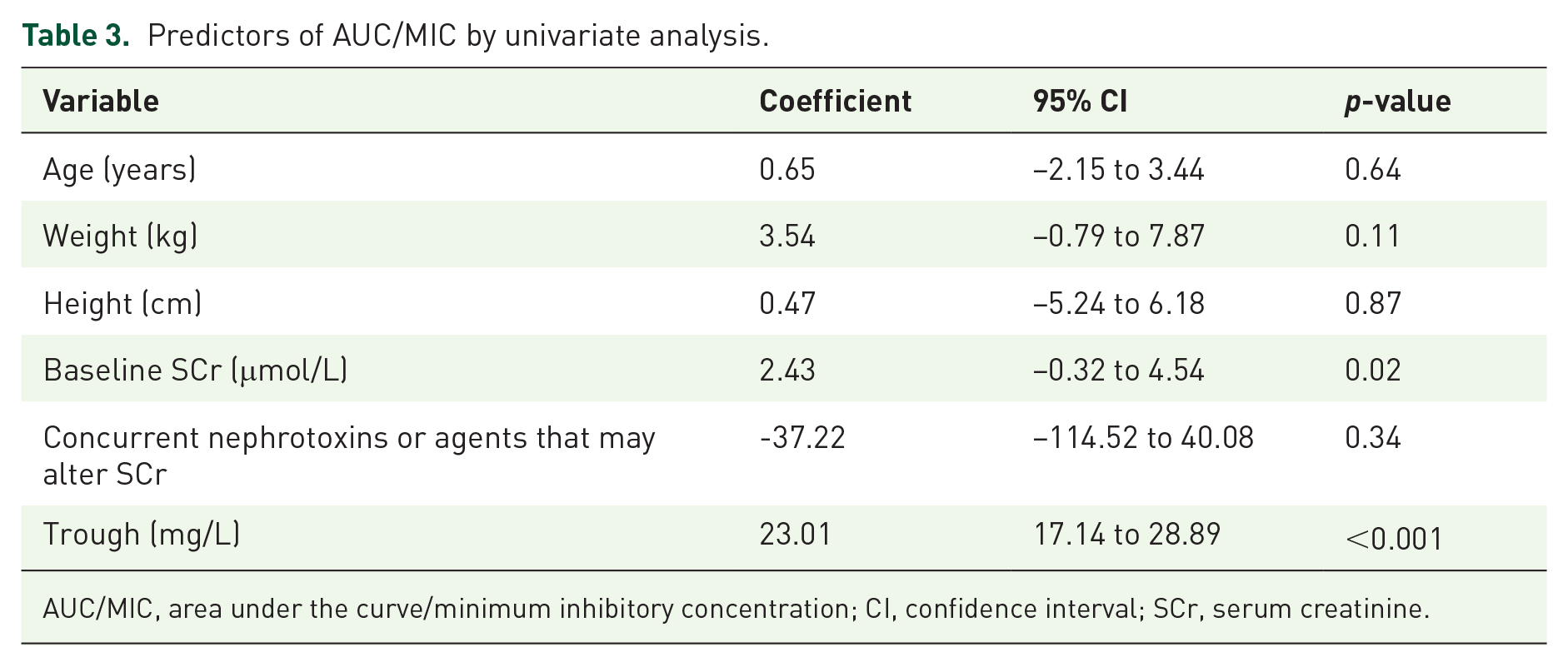
AUC/MIC, area under the curve/minimum inhibitory concentration; CI, confidence interval; SCr, serum creatinine.
Predictors of AUC/MIC by Multivariate Analysis.

AUC/MIC, area under the curve/minimum inhibitory concentration; CI, confidence interval; SCr, serum creatinine.
The average daily vancomycin dose of the population was 2089 mg with AUC-guided monitoring. The theoretical daily vancomycin dose with only trough-guided monitoring (target trough of 15–20 mg/L) was calculated to be 2726 mg. If target trough was 10–15 mg/L, the calculated theoretical daily vancomycin dose was 2180 mg. On average, the total daily dose of vancomycin was 637 mg lower and did not exceed 4 grams when AUC was available (Figure 3). It is estimated that the total daily dose of vancomycin was reduced by 30% when AUC-guided monitoring was performed.

Difference in vancomycin total daily dose.
Nephrotoxicity occurred in 5 (17%) of the 29 patients. The mean serum creatinine recorded in these patients at the time of the acute kidney injury was 116 μmol/L, representing a 55% increase above baseline. The mean glomerular filtration rate was 57.0 ± 26.0 mL/minute/1.73 m2. The mean total daily vancomycin dose was 2400 ± 978 mg and the mean vancomycin trough was 33.0 ± 10.4 mg/L during the acute kidney injury. Serum peak levels were not collected at the timeframe of nephrotoxicity and therefore, AUC was not available for comparison in all cases. In all five patients, renal function resolved before discharge and no patient required initiation of renal replacement therapy. Vancomycin was implicated in three of the five cases. Four out of five patients were on at least one other nephrotoxin or agent that may affect serum creatinine. Two of these patients were considered to have vancomycin-associated nephrotoxicity. One patient was concurrently on cobicistat, tenofovir disoproxil fumarate and furosemide, and the second was on sulfamethoxazole-trimethoprim. Nephrotoxicity was associated with dehydration and septic shock in the other two cases. One patient was on dolutegravir and the other was on cobicistat and tenofovir disoproxil fumarate. Vancomycin was not discontinued in any patient who experienced an acute kidney injury; however, the dose was adjusted in four patients.
Discussion
The 2020 IDSA vancomycin TDM guidelines suggest that the optimal way to manage vancomycin dosing is through AUC-guided monitoring, as opposed to the traditional method of trough-guided monitoring. 3 The results of our study suggest that there is a moderate correlation between vancomycin trough concentration and AUC/MIC. The main finding was that the majority of vancomycin trough concentrations between 10–15 mg/L corresponded to AUC/MIC > 400 mg•h/L for MIC value of 1 mg/L, indicating that higher trough concentrations of 15–20 mg/L as previously recommended by the 2009 IDSA vancomycin TDM guidelines are likely unnecessary to achieve an AUC/MIC target of 400–600 mg•h/L. Furthermore, patients with vancomycin trough between 10–15 mg/L were more likely to have vancomycin levels within the therapeutic AUC/MIC range of 400–600 mg•h/L compared to a trough level of 15–20 mg/L. Our study contributes to the growing literature investigating AUC compared to trough-guided monitoring. Bel Kamel et al. observed a moderate correlation (R 2 = 0.51) between vancomycin AUC and trough concentration in 95 geriatric patients. 11 Through logistic regression, it was determined that a trough of 10.8 mg/L was predictive of an AUC > 400 mg/L•h. 11 A study of critically ill patients reported a correlation between AUC calculated by a trapezoidal method and trough (R 2 = 0.81); specifically, a trough of 13 mg/L was associated with an AUC of 445–568 mg•h/L; a trough of 15 mg/L associated with an AUC of 532–645 mg•h/L; and a trough of 19 mg/L was associated with an AUC of 541–752 mg•h/L. 20 Another study by Clark et al. of 34 primarily critically ill patients demonstrated that vancomycin trough greater than 9 mg/L achieved AUC/MIC values greater than 400 mg•h/L, with the majority being greater than 500 mg•h/L. 12 There was a direct correlation between vancomycin trough concentration and AUC/MIC (R 2 = 0.73). 12 We observed a similar, if not weaker correlation, between vancomycin trough concentration and 24-h AUC in our study compared to previous reports investigating the same relationship in varied patient populations. Similar to previous literature, we used two serum levels to calculate the AUC, which is more accurate than AUC calculated with only a trough concentration.15,17
Another key finding from our study was that the majority of trough concentrations between 15 and 20 mg/L achieved AUC/MIC > 600 mg•h/L This suggests that targeting previously recommended trough concentrations of 15–20 mg/L can lead to increased vancomycin exposure that may not be necessary for successful treatment of invasive MRSA infections and may increase the risk of vancomycin-associated nephrotoxicity.3,15 We were unable to calculate AUC in patients who experienced nephrotoxicity since peak levels were not available at the time of insult, but these patients all had elevated trough levels. Given the correlation between trough and AUC, it is highly likely that AUC would also have been above target. Vancomycin-associated nephrotoxicity is related to increased exposure to vancomycin, as measured by trough or cumulative daily doses.6,7,21 Hale et al. 21 demonstrated that in 100 patients with a positive MRSA culture that troughs of 15–20 mg/L did not increase the rate of achievement of AUC/MIC ⩾ 400 mg•h/L compared to troughs of 10–14.9 mg/L, and the average trough was higher in patients who developed nephrotoxicity. Neely et al. observed lower vancomycin trough concentrations in patients monitored using the Bayesian approach to AUC calculation, targeting AUC/MIC of 400–800 mg•h/L. 8 Compared to trough-guided dosing, AUC-guided monitoring was associated with decreased nephrotoxicity and shorter courses of vancomycin, without reducing efficacy. 8 In our study, most patients who continued receiving vancomycin following an AUC calculation subsequently remained on current vancomycin dose when AUC was within target despite a seemingly low trough concentration, or had their vancomycin dose reduced when AUC was above target despite trough concentration being within 15–20 mg/L.
Practical considerations and patient preferences do pose a clinical challenge to AUC-guided monitoring and affect the decision of how widely to apply this method, especially given the limited literature evaluating its impact on clinical outcomes. Training must also be provided to pharmacists, physicians, nurses, and laboratory technicians when implementing AUC-guided monitoring, in addition to the considerable cost and logistical challenges related to obtaining additional timely serum concentrations. Because of these concerns, it may be reasonable to initially target a vancomycin trough level of 10–15 mg/L in the Urban Health population as more than 90% of patients would be expected to have an AUC ⩾ 400 mg•h/L while reducing the risk for nephrotoxicity. AUC-guided monitoring for vancomycin may be considered to ensure appropriate drug exposure in certain patients who are on high doses of vancomycin (i.e., > 4 grams per day), at risk for nephrotoxicity, on prolonged therapy, not clinically improving, or have persistent bacteremia.
This study had several limitations. The small sample size of a unique population in a single center may limit the generalizability of our findings. However, we believe that our observations may be valuable to sites that provide care to similar patients, such as young adults with rapid renal clearance and invasive gram-positive infections, requiring prolonged treatment with vancomycin. The PK data used in this study were collected retrospectively and based on TDM conducted in a clinical setting. The timing of blood draws and doses, use of pre- or post-steady state levels and calculation errors could contribute to variability. Some vancomycin levels were drawn too early or late; extrapolating true peak or trough concentrations introduces additional error and potentially decreases the accuracy of these values. Our study uses a two-level equation based approach to calculate AUC (Appendix 1). This method is based on a 1-compartment model which does not capture the AUC of the initial alpha-phase (distribution) or the non-linear increase of concentration until the end of the infusion. We expect that our calculations may underestimate the true AUC values. However, the two-level equation based approach has been validated against the Bayesian approach and showed a median error of ⩽ 2%. 12 This is unlikely clinically significant in regard to dosing adjustments. Many patients presented challenges with intravenous access (e.g., complex infections requiring prolonged antibiotics; history of intravenous drug use resulting in fewer viable veins) and required peripherally inserted central catheters. Blood work drawn through these lines instead of peripherally could result in inaccurate levels if the lines were not adequately flushed after administering vancomycin, or if insufficient volume was removed from the lines beforehand. Some patients had multiple sets of peak and trough levels measured throughout the course of this study and would have had more representation in the discordance data. For differences in total daily dosing, each individual clinician could adjust vancomycin differently based on patient presentation and clinical judgment. Therefore, there may be variations in true differences in total daily vancomycin dose based on AUC-guided monitoring and trough-guided monitoring. For patients who experienced nephrotoxicity, peak and trough levels were not drawn immediately prior to the acute kidney injury event. As a result, it cannot be determined if high vancomycin concentrations led to the events or if acute kidney injury from other causes led to vancomycin accumulation. Finally, the limited sample size and absence of a comparative group precluded the ability to assess clinical effectiveness and safety.
In conclusion, we observed that AUC is moderately correlated with trough concentration in the Urban Health population. Most patients with trough concentrations above 10 mg/L achieved AUC/MIC values above 400 mg•h/L. This suggests that patients may be provided with unnecessarily high doses of vancomycin when traditional trough-guided monitoring to target 15–20 mg/L is used. The likelihood of subtherapeutic AUC/MIC was low in patients with vancomycin troughs between 10 and 15 mg/L, and so, this may be a reasonable target when AUC/MIC monitoring cannot be routinely performed. Further studies evaluating the clinical significance of vancomycin PK/PD parameters are needed to better define the optimal target level and approach to TDM.
