Abstract
Background and objectives:
Achieving vancomycin therapeutic levels is essential for antibacterial success and resistance prevention. Multiple studies have shown that most of the children fail to reach therapeutic trough levels (10–20 µg/mL). This study aims to determine the frequency of achieving therapeutic vancomycin initial trough levels in children, evaluate the effect of age on that achievement and the mean initial trough levels, and the frequency of supratherapeutic levels.
Methods:
Children aged 1 month to 12 years who received three or more vancomycin doses 15 mg/kg every 6 h while admitted at our hospital from February 2016 to January 2017, and had a level before the fourth dose were included. Cases with high baseline serum creatinine, acute kidney injury, and congenital heart disease were excluded.
Results:
Out of 75 included cases, one third, 28/75 (37.3%), achieved goal. The lowest frequency was 6/28 (21.4%) of the 2–5 years group, which were statistically less likely to achieve, and had significantly lower mean initial trough than the 1–23 months group (P = 0.026 and 0.013, respectively). Mean initial trough levels were 10.1, 7.3, and 8.2 µg/mL in the 1–23 months, 2–5 years, and 6–12 years groups, respectively (P = 0.014). No supratherapeutic levels were observed.
Conclusion:
Vancomycin dose of 60 mg/kg/day is insufficient to attain target levels for most of the children. Children aged 2–5 years are the least likely to achieve and have the lowest mean levels. More intensified doses are warranted to be studied prospectively to identify the most effective empiric dose for children.
Background
Vancomycin had been introduced to clinical use 60 years ago. 1 Its use has been markedly increasing over the past 40 years. 2 It is the drug of preference for the treatment of invasive gram-positive bacterial infections caused by methicillin-resistant Staphylococcus aureus (MRSA) and ampicillin resistant Enterococci. 3 It also has multiple empiric indications such as septic shock, meningitis, meningoencephalitis, central venous catheter infections, skin and soft tissue infections, endocarditis, severe pneumonia, and bone infections. 4
The leading pharmacokinetic/pharmacodynamic parameter that efficiently predicts vancomycin effectiveness against gram-positive organisms is the ratio of the 24-h area under the concentration-time curve over the minimum inhibitory concentration (AUC24/MIC). 4 One adult study reported better microbiological and clinical responses with vancomycin AUC24/MIC ⩾ 400 in adults with methicillin-sensitive Staphylococcus aureus (MSSA) and MRSA lower respiratory tract infections (P = 0.0046). 5
The Infectious Diseases Society of America (IDSA) 2011 guidelines for the treatment of MRSA recommended trough levels of 15–20 µg/mL in serious infections for adults and, by extrapolation, children.4,6 Nevertheless, this level does not seem to be necessary for children. Using Monte Carlo simulation, one study suggested that AUC24/MIC ⩾ 400, unlike adults, correlates with vancomycin trough levels of 8 and 9 µg/mL for children dosed 60 and 70 mg/kg/day, respectively. 7 Another study correlated much lower trough levels, 7 to 10 µg/mL, to AUC24/MIC ⩾ 400 in more than 90% of 5000 virtual pediatric subjects. 8 Kishk et al., 9 using the trapezoidal method of AUC calculation which was the most correlated method with trough levels (r2 = 0.59113), found that AUC24/MIC of 400 in children corresponded to a vancomycin trough level of 10.98 µg/mL. In practice, 10–20 µg/mL remains the usual target to achieve the desired IDSA-recommended AUC/MIC ratio,8,9 and was suggested for children by several studies.10–12 While the new IDSA 2020 guidelines recommend targeting AUC instead of trough levels, 13 that is logistically challenging as it requires software and/or more levels. It is likely that many centers will continue to use trough targets.
Multiple pediatric studies have shown that it is hard to reach the concentration of 10–20 µg/mL even with the higher dosing recommended by the 2011 IDSA guidelines 4 (15 mg/kg/dose IV every 6 h).14–17 In most of the studies, around one third to less than half of the children have been reported to reach the desired vancomycin trough levels.14–17 Vancomycin half-life and clearance differ with age, 3 and different levels were reported at different pediatric age groups.12,18,19 Explanation of the lower than expected vancomycin trough levels in children, specific age cut-points for different corresponding levels to the same doses, and the optimum pediatric initial dosing regimens remain as research gaps that need prospective studies to be investigated.
In the pediatric units of Hamad General Hospital (HGH), Doha, Qatar, it has been noticed that most of the vancomycin initial trough levels in children are subtherapeutic. Our current practice is to use the dosing of 60 mg/kg/day divided every 6 h and to collect trough levels 30–60 min before the scheduled time of the fourth dose. The aim of this study is to determine the frequency of therapeutic vancomycin initial trough level attainment with the current dosing strategy among the hospitalized pediatric patients in HGH, and to detect the age-related differences in those levels. The primary objective is to determine the overall percentage of admitted pediatric patients at HGH during the study period who achieved therapeutic vancomycin initial trough levels. Secondary objectives are to evaluate the influence of age on the frequency of achieving therapeutic vancomycin initial trough levels in children, to investigate whether there is a meaningful difference in the mean vancomycin initial trough levels among different pediatric age groups, and to assess the overall frequency of supratherapeutic vancomycin initial trough levels and their distribution among different pediatric age groups.
Methods
A 1-year retrospective study of the hospital profiles of children received vancomycin in Hamad General Hospital. Laboratory data, such as vancomycin levels, serum creatinine and blood cultures, patients weights, radiographic diagnostics, and clinical notes were extracted from each patient’s hospital profile. A data collection form was designed and used for the study which included demographic data, baseline renal function and serum creatinine, concomitant nephrotoxic medications, indication for vancomycin, vancomycin dose/interval, appropriateness of initial trough level collection time, and the value of vancomycin level in µg/mL. A therapeutic vancomycin initial trough level was defined as 10 to 20 µg/mL.10,12,15
Pediatric inpatients aged 1 month to 12 years at the time of starting vancomycin which was received during the period from 1 February 2016 to 31 January 2017 were included. They must have received at least three vancomycin doses of 15 mg/kg/dose every 6 h (intended total daily dose of 60 mg/kg/day divided every 6 h). The vancomycin initial trough levels must have been appropriately drawn within 1 h before the scheduled time of administration of the fourth dose. That was confirmed through reviewing each case’s electronic profile for specimen collection times and the doses administration times in the medication administration record. Children with congenital heart diseases (CHD), post cardiothoracic surgery (CTS), on extracorporeal membrane oxygenation (ECMO), on peritoneal dialysis, had a sign of acute kidney injury (AKI) before starting vancomycin, defined based on the Acute Kidney Injury Network (AKIN) criteria 20 or (a high baseline serum creatinine for age) 21 were excluded.
Eligible patients were then categorized into three age groups for the secondary outcomes: Group (1): ⩾1 month to 23 months, group (2): 2–5.99 years, and group (3): 6–12.99 years.10,12 For convenience, ages 2–5.99 and 6–12.99 years will be expressed all through as 2–5 and 6–12 years, respectively.
Ethical approval for this retrospective study was provided by the Medical Research Committee (MRC) of Hamad Medical Corporation (HMC).
Statistical analyses
Descriptive statistics were used to summarize demographics, indications for vancomycin, and other baseline characteristics. For the primary outcome, the frequency of patients achieving the required vancomycin level was expressed as a percentage (%). For the secondary outcomes, the categorical data of achieving therapeutic vancomycin initial trough levels were expressed as percentage (%) in each age group. Whether there is a statistically significant difference among the three age groups and between each two groups was tested using chi-square (χ2) test. A two-tailed P value < 0.05 was considered to be statistically significant. Mean (SD) was used to express the continuous data of vancomycin levels in each of the three groups. One-way analysis of variance (ANOVA) with Bonferroni multiple comparison test was utilized to analyze the association of age group with the mean initial vancomycin trough levels. The categorical data of the relative frequency of supratherapeutic vancomycin initial trough levels were expressed as a percentage. A two-tailed P value < 0.05 was considered to be statistically significant. All statistical analyses were done using statistical packages SPSS 22.0 (SPSS Inc. Chicago, IL, USA).
Results
From 1 February 2016 to 31 January 2017, a total of 75 pediatric patients aged from 1 month to 12 years received vancomycin and fulfilled the study inclusion criteria. Out of these, 28 patients (37.3%) were in the 1–23 months age group, 28 (37.3%) in the 2–5 years group, and 19 (25.3%) in the 6–12 years group. There were 42 males (56%) and 33 females (44%). The most common location of admission was the general pediatrics units with 44 cases. The rest of the cases were almost equally distributed between pediatric hematology oncology unit (15 cases) and pediatric intensive care unit (PICU) (16 cases). The most frequent indication of vancomycin was the suspected or confirmed central nervous system (CNS) infection (28 cases) followed by pneumonia (14 cases). A summary of the baseline characteristics and patients demographics is presented in (Table 1).
Baseline characteristics and demographics of children who received vancomycin.
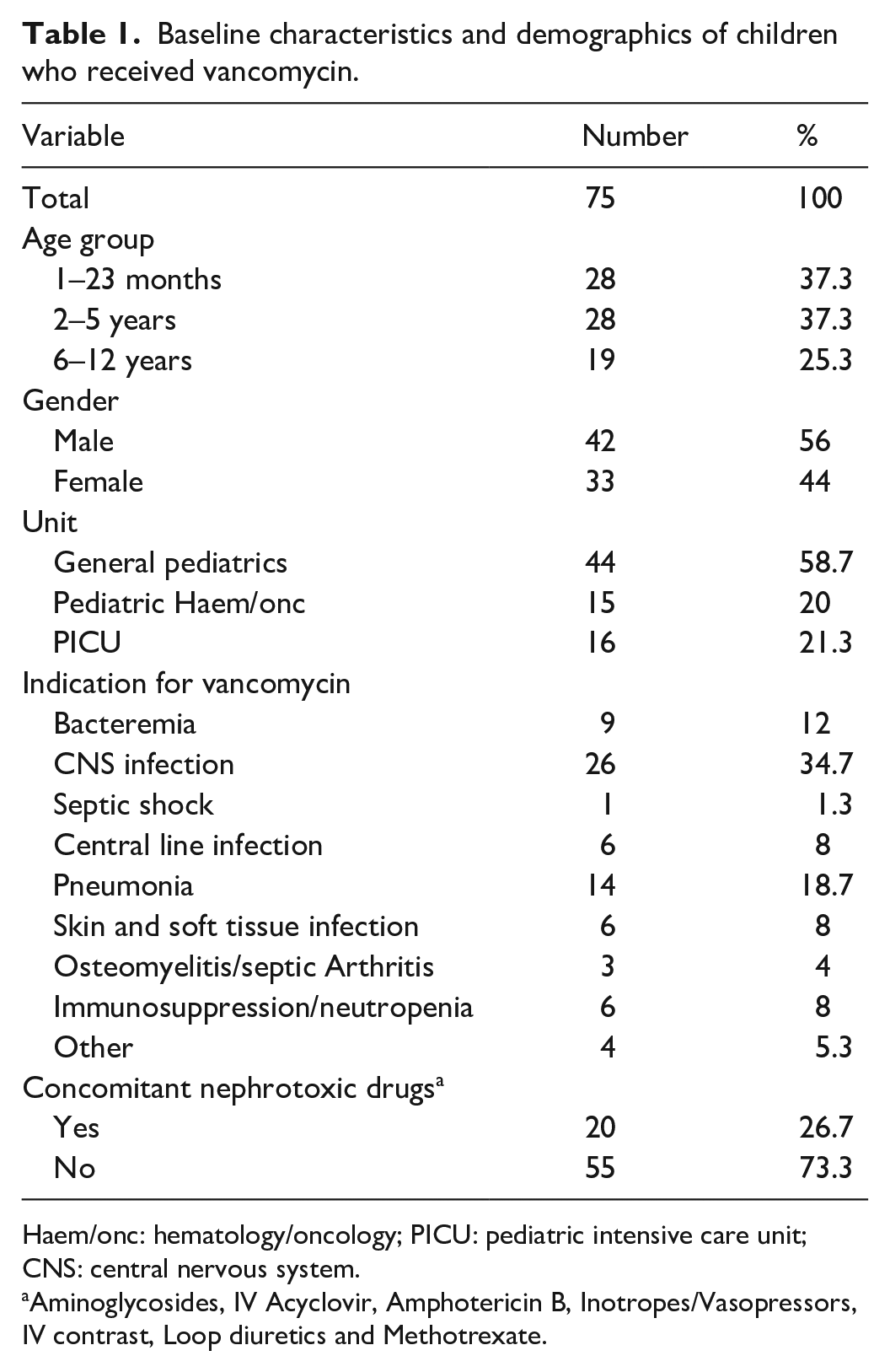
Haem/onc: hematology/oncology; PICU: pediatric intensive care unit; CNS: central nervous system.
Aminoglycosides, IV Acyclovir, Amphotericin B, Inotropes/Vasopressors, IV contrast, Loop diuretics and Methotrexate.
For the primary outcome, out of the 75 cases included in the study, 28 cases (37.3%) achieved therapeutic vancomycin initial trough levels of 10–20 µg/mL. Subtherapeutic levels below 10 µg/mL were observed in 47 cases (62.7%).
For the secondary outcomes, therapeutic vancomycin initial trough levels of 10–20 µg/mL were achieved by 14 out of 28 patients in the 1–23 months group (50%), 6 out of 28 patients in the 2–5 years group (21.4%), and 8 out of 19 patients in the 6–12 years group (42.1%). Although the percentage of patients who achieved therapeutic trough level in the 2–5 years group was the smallest, an overall statistically significant difference could not be detected among the three groups (P = 0.077). Multiple comparisons revealed that the percentage of patients who achieved vancomycin level of 10–20 µg/mL in the 1–23 months group was significantly larger than the 2–5 years group with a relative risk (RR) of 2.33 (95% confidence interval [CI] 1.05, 5.19) (P = 0.025) (Table 2).
Multiple comparisons of % patients with trough 10–20 µg/mL.

RR: relative risk; CI: confidence interval.
Group (1): 1–23 months; group (2); 2–5 years; group (3): 6–12 years.
Mean (SD) initial trough level was the lowest for children aged 2–5 years, 7.3 (3.2) µg/mL, followed by 8.2 (3.5) µg/mL for the 6–12 years group, then 10.1 (3.8) µg/mL for the 1–23 months group. An overall comparison among the three groups showed a statistically significant difference (P = 0.014). Multiple comparisons showed that the mean initial trough level was significantly lower in children aged 2–5 years than children aged 1–23 months (P = 0.013) (Table 3).
Multiple comparisons of means of vancomycin initial trough levels.
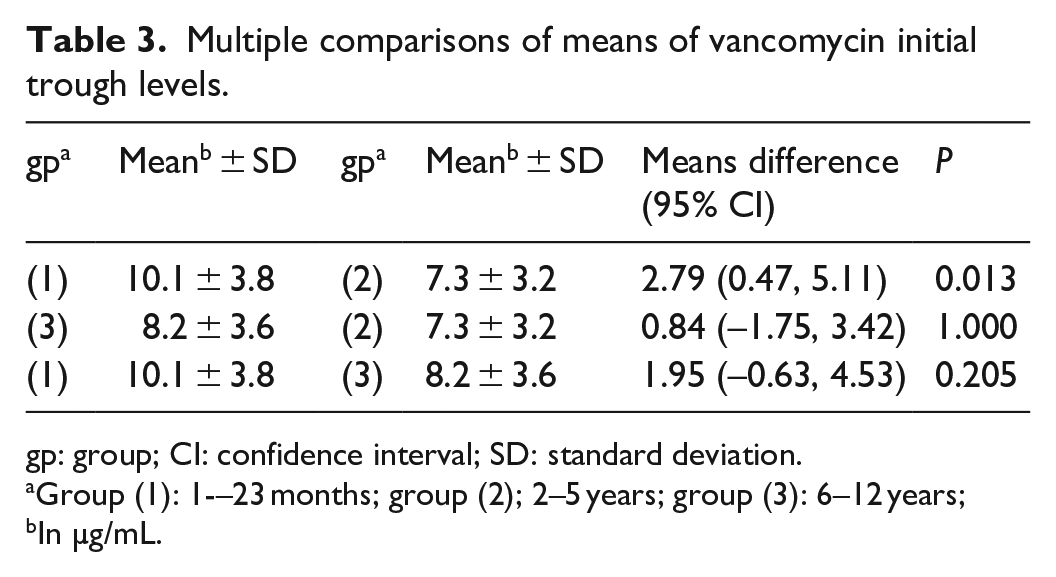
gp: group; CI: confidence interval; SD: standard deviation.
Group (1): 1-–23 months; group (2); 2–5 years; group (3): 6–12 years;
In µg/mL.
None of the patients (0%) was found to have a supratherapeutic vancomycin initial trough level.
Discussion
This study showed that only 37.3% of patients, aged 1 month to 12 years, have reached vancomycin initial trough of 10–20 µg/mL with the dose of 60 mg/kg/day divided every 6 h. Children aged 2–5 years had the lowest frequency of achieving the desired trough, 21.4%, and the lowest mean level, 7.3 µg/mL. Statistical significance could only be detected between them and the 1–23 months group but not the 6–12 years group. That might be explained by the limited numbers of patients, particularly in the later group.
Our results are comparable to data from several previous studies. Using the same dose, Madigan et al. 12 reported that 36.1% achieved initial trough goal. Also, Hoang et al. 19 reported 39% attainment, Geerlof and Boucher 10 reported 31.7%, and Rainkie et al. 22 29.5%. One study reported a comparatively larger percentage of achieving vancomycin initial trough 10–20 µg/mL, 50% of 74 children with complicated infections. 18 Rajon et al. 23 reported that out of 52 children, nine cases (17.3%) reached the initial target. Recently, Maloni et al. 24 reported only 22%–23% of children in the PICU who received 51 mg/kg/day or above attained 10–20 µg/mL.
Previous studies that evaluated the age effect on target vancomycin trough achievement repeatedly showed that children aged 2–5 years have the smallest likelihood to accomplish that goal.10,12,25 Consistent with our findings, Madigan et al. 12 reported that the patients aged 2–5 years had the lowest proportion to achieve goal trough (16.7%) and the lowest mean level, 7.5 µg/mL. Geerlof and Boucher 10 showed the same trend of goal attainment (16% in the 2–5 years, 20% in the 2–23 months, and 38.1% in the 6–12 years group) with mean levels of 7.3, 10.3, and 9.6 µg/mL, respectively. Another study observed that, particularly, children aged 1 to 5.9 years mostly do not achieve goal levels. 6 Rainkie et al. 22 found that only 6% of patients 1–6 years old achieved initial vancomycin trough of 10–20 µg/mL with the dose of 60 mg/kg/day, while the same level was reached by 28% and 48% of cases aged 1 month to 1 year and 6–13 years, respectively. 25
One explanation to the age-related differences is that in full-term neonates, vancomycin clearance is (30 mL/min) with a half-life of about 7 h, and then it is doubled (50 mL/min) around 3 months of age with reduced half-life down to 4 h. Vancomycin clearance rate continues to increase with age reaching 130–160 mL/min, and half-life continues to diminish from 4 to 8 years old (2–3 h), and they approach adults values around 12 years old. 3
None of the patients included in this study had a supratherapeutic vancomycin initial trough level (>20 µg/mL). Previous studies that used the same initial dose, excluded abnormal renal function, reported no initial supratherapeutic levels as well.10,12,18
Vancomycin trough level was the only supported monitoring parameter in the 2011 IDSA guidance, with a recommended pediatric daily dose of 60 mg/kg to reach a target of 15–20 µg/mL for both adults and children. 4 Alternatively, the 2020 IDSA guidelines suggest an AUC/MIC target of 400–600 through Bayesian estimation. 13 Still, they consider the same empiric dose yet stretch the range up to 80 mg/kg for children age 3 months to below 12 years. 13 Our study, aligned with multiple other studies,10,12,19,22–24 demonstrated that 60 mg/kg/day is not sufficient for most children. Hence, we can suggest 80 mg/kg/day as the starting dose provided normal renal function and no CHD.
Limitations of the study include the retrospective design, the limited number of patients, and the short data collection period (1 year). We did not initially calculate the sample size; the inclusion/exclusion criteria determined the total number of subjects. This study did not take account of other variables that might have affected vancomycin levels, such as weight, creatinine clearance, and malignancies.
Despite these limitations, this study introduced a valuable overview of therapeutic vancomycin initial trough attainment in children and age-related vancomycin trough concentration pattern. To the best of our knowledge, this is the first study to assess this subject among the pediatric patients in Qatar. Our findings are consistent with most of the previous literature and can serve as a starting point for future larger studies with intensified doses down the road.
Conclusions
Vancomycin daily dosing of 60 mg/kg is insufficient to attain the current target levels for most of the children up to 12 years of age. The dose is unlikely to produce supratherapeutic levels for children with normal renal function and cardiac structure. The least likely to achieve the goal are the patients aged 2 to 5 years. Prospective studies of empiric doses of 80 mg/kg/day are warranted to identify the most effective dosing tailored to pediatric age groups to achieve the desired AUC/MIC and clinical response.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this retrospective study was provided by the Medical Research Committee (MRC) of Hamad Medical Corporation (HMC) (#17089/17).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The publication of this article was funded by Qatar National library (QNL).
Informed consent
Informed consent was not sought for the present study because of the retrospective design of the study (IRB waived the need for informed consent).
Presentations
The abstract of this research study was presented at the American Academy of Pediatrics National Conference & Exhibition in Orlando, Florida (2018). Our study complies with Declaration of Helsinki.
