Abstract
Objectives
This study aimed to investigate vancomycin therapeutic drug monitoring (TDM) in patients on continuous renal replacement therapy (CRRT) and explore the risk factors for exceeding the target concentration.
Methods
This retrospective study enrolled patients aged ≥18 years who were admitted to the intensive care unit and treated with ≥3 intravenous vancomycin doses during CRRT, and who underwent vancomycin TDM. Demographic and other information were collected. Multivariate logistic regression was used assess the risk factors for exceeding the target concentration.
Results
Sixty-nine patients were included, and 40.6% patients underwent TDM. Additionally, 14.5% of patients reached the optimal concentration, and 87.5% of patients who exceeded the target received a daily dose adjustment. The cumulative dose of vancomycin and serum albumin were risk factors for exceeding the target concentration in patients on CRRT.
Conclusions
Patients on CRRT did not meet the optimal vancomycin management; <50% of the patients routinely received vancomycin TDM, and <15% achieved the optimal concentration. Fewer patients in the subtherapeutic group received a daily dose adjustment than those who exceeded the target concentration. Cumulative vancomycin and serum albumin doses before TDM were the risk factors for exceeding the target concentration in CRRT patients.
Keywords
Introduction
The Infectious Diseases Society of America (IDSA) indicates that the clinical efficacy of severe methicillin-resistant Staphylococcus aureus (MRSA) infection is closely related to the vancomycin trough concentration. To confirm the efficacy and safety, a loading dose that is based on the actual body weight should be administered, and trough concentrations should be obtained before the fourth or fifth dose under steady-state conditions. The subsequent dosage adjustment should be administered on the basis of the actual trough concentration to achieve the optimal target of 15 to 20 mg/L. 1
When patients on continuous renal replacement therapy (CRRT) receive vancomycin for sepsis, vancomycin clearance becomes complex. CRRT parameters2–6 and patient conditions 7 may affect vancomycin pharmacokinetics (PK)/pharmacodynamics (PD). For example, continuous venovenous hemodiafiltration (CVVHDF) clearance of vancomycin was 1.3- to 7.2-times greater than that of other types of CRRT. 3 Additionally, severe hypoalbuminemia influenced the half-life of vancomycin in older patients. 7 Inappropriate therapeutic drug monitoring (TDM) time points and nonalternative daily doses may lead to a trough concentration that does not achieve the optimal target and contributes to adverse clinical outcomes. 8 Previous studies showed that the vancomycin trough concentration in CRRT patients has a wide variability.9–13
The purpose of this study was to investigate vancomycin TDM in patients on CRRT and explore the risk factors of vancomycin trough concentrations that exceeded the target.
Methods
Study design and data collection
This retrospective observational study was performed at the Department of Critical Care, a general intensive care unit (ICU) with 24 beds in a tertiary hospital in Beijing from 1 January 2014 to 31 December 2019. Because this was a retrospective study, the requirement for patient consent was waived by the ethics committee at Fuxing Hospital, Capital Medical University (approval notice number: 2013FXHEC-KY018).
The inclusion criteria were as follows: age ≥18 years; admitted to ICU; received CRRT; and treated with ≥3 doses intravenous vancomycin during CRRT. The exclusion criterion was patients who did not receive TDM.
Sex, age, body mass index (BMI), comorbidities, creatinine clearance (calculated using the Cockcroft–Gault formula), serum albumin level before TDM, Acute Physiology and Chronic Health Evaluation II (APACHE II), survival at day 28, infection sites, and etiology were collected. The following vancomycin parameters were recorded: daily dose, intermittent or continuous infusion, TDM time point (TDM before the fourth or fifth dose of vancomycin), trough concentration, and alternative therapy. The following CRRT parameters were recorded: mode (continuous venovenous hemofiltration [CVVH] or continuous venovenous hemodiafiltration [CVVHDF]), blood flow rate (mL/minute), dose (mL/kg/hour) in the CRRT effluent volume, duration (hours; including cumulative CRRT working hours, which is the CRRT machine run time from the first vancomycin dose administered for TDM from which the circuit priming time is subtracted), and the cumulative dose (L/kg; the product of duration and dose [duration was unchanged]).
The enrolled patients were divided into the following two groups on the basis of a trough concentration of 20 mg/L: nonexceeding target group and exceeding target group.
Vancomycin hydrochloride injection was manufactured by of Eli Lilly Japan KK, Seishin (Kobe, Japan). Each bottle contained 0.5 g of vancomycin. The CVVH was performed using Prismaflex with the PRISMAFLEX ST150 SET filter (Deerfield, IL, USA). The CVVHDF was performed using a Multifiltrate, and the filter was Fresenius AV600S (Fresenius, Bad Homburg, Germany). An Abbott ARCHITECT i1000 immunoassay analyzer (Abbott, Abbott Park, IL, USA) with a quality control box and standard curve box administered fluorescence polarization immunoassay were used for the vancomycin TDM.
Statistical analysis
Continuous data were presented as the mean ± standard deviation (SD) or as the median and interquartile range (IQR). Categorical data were presented as the number and percentage. A Student’s t-test and the Mann–Whitney U test were used to compare continuous data. The Chi-square test and Fisher’s exact test were used to compare categorical data. Multivariate logistic regression was used to analyze the risk factors associated with a vancomycin trough concentration that exceeded the target. Additionally, a P value <0.05 was considered to be statistically significant. Statistical analyses were conducted using SPSS version 23.0 (IBM Corp., Armonk, NY, USA).
Results
Study population and characteristics
Four hundred eighty-seven patients who were admitted to the ICU and underwent CRRT were enrolled into our study. Among them, 119 patients were treated with vancomycin and 69 patients received TDM (Figure 1).

Flowchart of patient inclusion in the study.
Among these 69 patients who received TDM, the median age was 77.0 (56.0, 83.5) years, 35 (50.7%) were men, and the median BMI was 22.9 (20.6, 26.7) kg/m2. Most patients had comorbidities (63, 91.3%), and 42 (60.7%) patients had died at day 28 in the ICU.
Among all of the included patients, 43 (62.3%) patients had pneumonia, ten (14.5%) patients had a bloodstream infection, 13 (18.8%) patients had an abdominal infection, and three (4.3%) patients had a urinary tract infection. Among all the included patients, 27 (39.1%) patients had MRSA.
Vancomycin TDM and daily dose adjustment
All included patients received vancomycin by intermittent infusion, and each infusion time was greater than 1 hour.
More patients received vancomycin TDM in 2018 and 2019 than those in 2014 to 2017 (75.0% versus 48.0%, P = 0.004). There were five (7.2%) patients who received TDM before the third dose, and 28 (40.6%) patients who received TDM before the fourth or fifth dose. There were 36 (52.2%) patients who received TDM after the fifth dose. Fourteen (20.3%) patients were administered a loading dose.
Ten (14.5%) patients achieved the optimal concentration of 15 to 20 mg/L. A subtherapeutic concentration (<15 mg/L) was observed in 27 (39.1%) patients, and 32 (46.4%) patients had a concentration >20 mg/L.
Thirty-nine (66.1%) patients received a daily dose adjustment. Among them, 11 patients with a concentration <15 mg/L and 28 patients with a concentration >20 mg/L received alternative therapy for their daily dose (40.7% versus 87.5%, P < 0.001).
CRRT parameters
There were 50 (72.5%) patients who underwent CVVH and 29 (27.5%) patients who underwent CVVHDF. Among all included patients, the blood flow rate was 120 (120, 150) mL/minute. The therapeutic dose was 26.7 (25.0, 30.0) mL/kg/hour. The CRRT duration was 46.0 (34.0, 67.0) hours, and the cumulative dose was 1.17 (0.91, 1.74) L/kg.
Risk factors for a vancomycin concentration exceeding the optimal target in patients on CRRT
The exceeding target group seemed to have a lower BMI and received a higher cumulative dose of vancomycin before TDM compared with that of the nonexceeding group (Tables 1 and 2).
Demographics, baseline, and comorbidities.
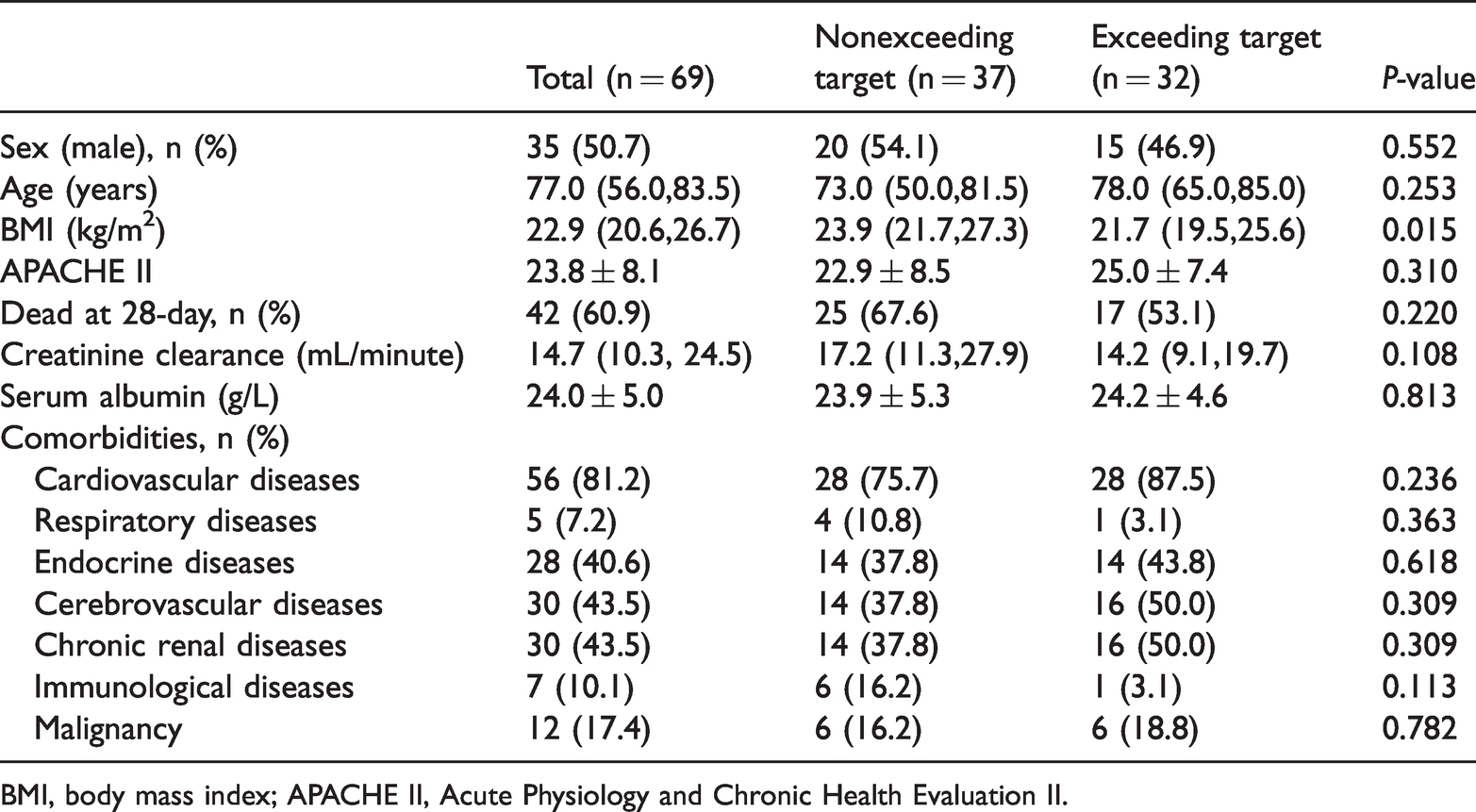
BMI, body mass index; APACHE II, Acute Physiology and Chronic Health Evaluation II.
Vancomycin therapy and CRRT parameters.

CRRT, continuous renal replacement therapy; TDM, therapeutic drug monitoring; CVVH, continuous venovenous hemofiltration; CVVHDF, continuous venovenous hemodiafiltration.
The cumulative dose of vancomycin (odds ratio [OR]: 1.505, 95% confidence interval [CI]: 1.066–2.125) and serum albumin (OR: 1.204, 95%CI: 1.001–1.449) were the risk factors for a concentration exceeding the optimal target. The cumulative CRRT dose (OR: 0.264, 95%CI: 0.091–0.767) and BMI (OR: 0.816, 95%CI: 0.683–0.976) (Table 3) could reduce the risk of the concentration exceeding the optimal target.
The risk factors for the vancomycin concentration exceeding the target in patients on CRRT.
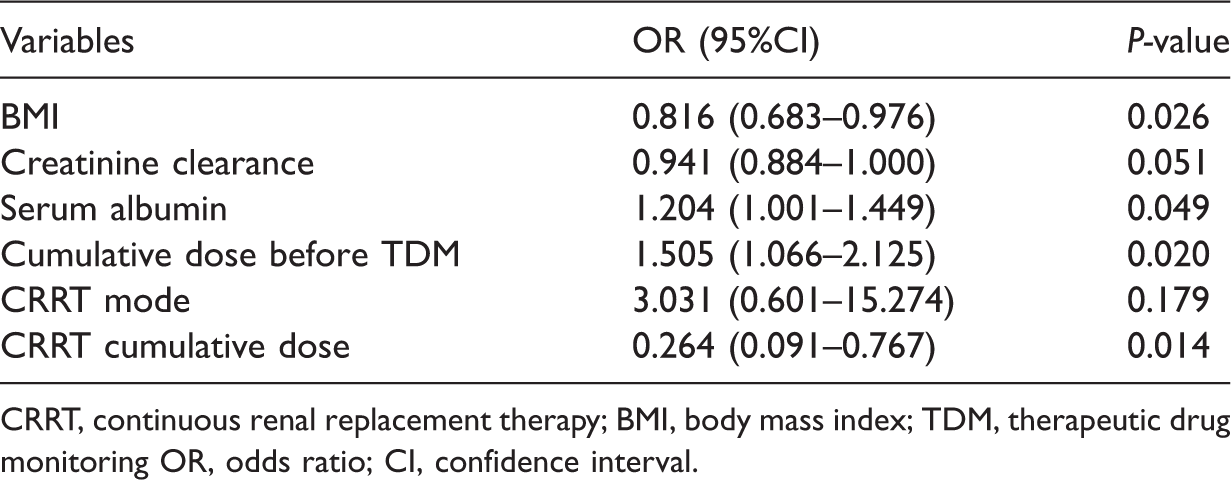
CRRT, continuous renal replacement therapy; BMI, body mass index; TDM, therapeutic drug monitoring OR, odds ratio; CI, confidence interval.
Discussion
In our study, patients on CRRT did not achieve optimal vancomycin management. Fewer than half of the patients on CRRT routinely received vancomycin TDM, and less than 15% of these patients achieved the optimal concentration. On the basis of the trough concentration, fewer patients in the subtherapeutic group (which included patients whose vancomycin trough concentration was less than 15 mg/L) received an daily dose adjustment than those in the exceeding target group. The cumulative dose of vancomycin and serum albumin before TDM were risk factors for the concentration exceeding the optimal target in patients on CRRT.
Vancomycin clinical efficacy is closely related to its trough concentration. To confirm the efficacy and safety, a dose regimen on the basis of a trough concentration target of 15 to 20 mg/L is recommended by IDSA. Providing a loading dose on the basis of the actual body weight rather than on the basis of the empirical dose is recommended to attain the optimal concentration as soon as possible. When obese patients receive vancomycin for infection, the loading dose should be increased, 14 while in our study, fewer patients received a loading dose.
The previous dose regimen was based on vancomycin TDM. The study performed by Ye et al. showed that nearly 50% of patients received TDM and that the TDM group had a significantly higher rate of clinical efficacy and a significantly decreased risk of nephrotoxicity compared with those without TDM. 15 Some studies16–19 with a small sample size showed that 10% to 60% of patients received vancomycin TDM, which was a large variation. Vancomycin clearance is complex in patients on CRRT, which means that physician should monitor vancomycin trough concentration particularly in CRRT patients.
Vancomycin will reach a stable trough concentration after the third or fourth dose, so TDM should be performed before the fourth or fifth dose. Additionally, the daily dose should be adjusted on the basis of the measured trough concentration. Van Der Heggen et al. showed that delayed sampling occurred in 16% of adults, 20 while over half of the patients received vancomycin TDM later than recommended in our study. In many tertiary hospitals in China, vancomycin TDM is only available on weekdays during working hours, and thus, TDM and the daily dose adjustment may be delayed, which may lead to a concentration that exceeds the optimal target. Therefore, balancing the supply and demand in the pharmacology and clinical departments seems to be important. Moreover, the clinical department should provide supervision for vancomycin TDM and improve its awareness to prevent a missed test resulting from human-related issues.
There were only 14.5% patients whose TDM met the optimal target range of 15 to 20 mg/L in our study. Al-Maqbali et al. showed that the vancomycin therapeutic level was not achieved in 16.8% of patients, and 47.5% of them had high vancomycin levels (>20 mg/L), 8 which was consistent with our study results. Subtherapeutic concentrations were measured by Van Der Heggen in 68% of adults in their study, showing that it is common for vancomycin TDM to not meet the optimal target. 20 When patients on CRRT receive vancomycin for sepsis, vancomycin clearance is complex and trough concentrations may be uncertain. Subtherapeutic concentrations may lead to inadequate drug efficacy and supertherapeutic concentrations may aggravate renal failure. Thus, vancomycin TDM should be carefully monitored in CRRT patients, but few studies have focused on them. The purpose of our study was to highlight the vancomycin regimen in CRRT patients.
Bakke et al. studied 245 patients who received vancomycin. When the concentration was not within the target concentration range, only 46.2% of patients received an adjustment. 21 Clinicians in our study were watching more carefully for supertherapeutic concentrations because more patients (87.5% versus 40.7%) in this group received a daily dose adjustment. When excessive serum levels were observed, clinicians decreased the daily dose and provided extra vancomycin if subtherapeutic concentrations occurred.
Hypoproteinemia may increase vancomycin clearance by CRRT. Vancomycin shows approximately 55% protein binding, and only free drug can be cleared by CRRT.20,22 When patients with hypoproteinemia receive vancomycin, the increasing amount of free drug can be cleared more easily by CRRT, leading to concentrations that do not meeting the target.
Different CRRT modes and doses will affect vancomycin clearance.23–25 The adequacy of CRRT can decrease the occurrence of excessive concentration.26–28 Some studies showed that a higher CRRT dose was closely related to subtherapeutic concentrations.13,29,30 In our study, a lower cumulative CRRT dose led to the supertherapeutic vancomycin concentrations.
Additionally, some studies showed that other factors influenced the vancomycin concentration. Lin et al. 31 and Covajes et al. 32 suggested that when patients are undergoing CRRT, it was easier to achieve and maintain the target concentration with continuous vancomycin infusion compared with intermittent infusion.
Our study results showed that patients on CRRT did not meet optimal vancomycin management. The physician should administer TDM at the correct time point, and the individualized drug regimen should be administrated on the basis of the trough concentration results.33–36
Our study had some limitations. This was a retrospective study at a single center, and although the observation time was extended, the number of patients who met the inclusion criteria was small. Thus, selection bias might exist. Additionally, we did not exclusively analyze medications such as exogenous albumin, diuretics, and citrate anticoagulation.
Conclusions
Patients on CRRT did not meet optimal vancomycin management requirements. Fewer than half of the patients routinely received vancomycin TDM, and less than 15% of them achieved the optimal concentration. On the basis of the trough concentration, fewer patients in the subtherapeutic target group received a daily dose adjustment compared with those in the group that exceeded the target. The cumulative dose of vancomycin and serum albumin before TDM were risk factors for a concentration exceeding the target in patients on CRRT.
Footnotes
Acknowledgements
The authors would like to thank the Department of Critical Care Medicine at Fuxing hospital and the Beijing Friendship hospital at Capital Medical University for their assistance with the study design and conduct.
Author contributions
YL and LJ designed the study. YL performed data collection. YL and LJ analyzed data and drafted the manuscript. YL and LJ were in charge of overall direction and planning and contributed to reviewing and editing the manuscript. All authors contributed to editing the manuscript for important intellectual content and approved the final manuscript.
Additional data
There are no additional data.
Declaration of conflicting interest
The author declares that there is no conflict of interest.
Ethics approval
The Ethics Committee at Fuxing Hospital, Capital Medical University approved the present study (Approval Notice Number: 2013FXHEC-KY018) and waived the requirement for patient consent because of its retrospective nature.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from the National Key Clinical Specialty Construction Project.
Research checklist
This paper is not associated with any research checklist.
Statement
Study participants or the public were not involved in the design, conduct, reporting, or dissemination plans of our research.
