Abstract
Prompt treatment of candidemia, especially in immunocompromised hosts, is known to improve outcomes. We present a case of discordance among results of Gram stain, multiplex polymerase chain reaction (PCR)-based rapid diagnostic technology, and conventional cultures that subsequently resulted in delayed therapy and hospitalization. An immunocompromised patient presented to the outpatient oncology clinic with signs and symptoms of systemic infection. Blood cultures were obtained, and Gram stain showed gram-negative rods, while multiplex PCR results (BioFire® FilmArray® BCID 1) returned positive for both Enterobacter cloacae and Candida parapsilosis. Conventional cultures only grew E. cloacae. Because of the discordant results, the primary team elected to give ertapenem monotherapy and defer antifungal therapy. The patient’s symptoms progressed, and 11 days later, the patient was admitted with subsequent positive blood cultures for C. parapsilosis. The patient required a 9-day hospitalization due to complications associated with candidemia. This case highlights the value of understanding and interpretation of rapid diagnostics, shared decision-making in antimicrobial management of high-risk patients, and the important responsibility of antimicrobial stewardship teams across the continuum of care.
Introduction
Candida species are a frequent cause of bloodstream infection (BSI) in the United States with an associated mortality of up to 47%.1,2 Detection of Candida spp. with rapid diagnostic tests (RDTs) leads to earlier initiation of appropriate antifungal therapy, thus improving patient outcomes. 3 Multiplex polymerase chain reaction (PCR)-based RDTs, including BioFire® FilmArray® Blood Culture Identification Panel (BCID), report high sensitivity and specificity for Candida spp. (98.8% and 99.6%, respectively); however, limited data have demonstrated a higher degree of discordance among polymicrobial cultures.4–6 The BioFire® BCID 1 panel detects Candida albicans, Candida glabrata, Candida krusei, Candida parapsilosis, and Candida tropicalis, while the newer BioFire® BCID 2 panel additionally includes Candida auris. 4 Given the high sensitivity of the PCR-based test, it is not unusual to have a discordant result between the PCR-based test, Gram stain, and conventional cultures. Prior data at our institution have shown the discordance rate to be 27%, driven primarily by the detection of Candida spp. via BCID 1 and subsequent negative blood cultures. 6 Some institutions’ microbiology laboratories do not report discordant results (e.g. negative Gram stain and positive RDT); thus, antimicrobial stewardship or primary treatment teams may be unaware of their occurrence. To date, there are limited published data describing the clinical significance of discordant results. We present a case of discordant results between BioFire® BCID 1 and conventional blood culture in an immunocompromised patient with C. parapsilosis BSI, leading to delay in antifungal treatment and subsequent hospitalization.
Patient case
An elderly man in his eighth decade of life with a history of bladder and prostate cancer, as well as a prosthetic knee joint replacement, presented to the emergency department with mild abdominal pain 7 months prior to the index admission. A computed tomography (CT) scan revealed a bladder mass that was consistent with recurrence of bladder cancer and metastasis to the third and fourth thoracic vertebrae. The patient underwent a successful transureteroureterostomy and cutaneous ureterostomy and was referred for outpatient management. However, 3 months after this procedure, he was initiated on cisplatin and gemcitabine dual chemotherapy with the goal of decreasing the size of his tumor. This regimen was discontinued 2 months later due to the progression of malignancy, and he was switched to palliative pembrolizumab instead.
Twelve days prior to the index admission, the patient presented to the outpatient oncology clinic with a fever, and two sets of blood cultures were obtained. The Gram stain only showed gram-negative rods and the blood culture only grew Enterobacter cloacae; however, the BioFire® BCID 1 panel indicated that both C. parapsilosis and E. cloacae were present. The patient remained hemodynamically stable and was initiated on ertapenem 1 g intravenous (IV) daily as outpatient parenteral antibiotic therapy (OPAT) through a peripheral IV access at the infusion center. The Antimicrobial Stewardship and Support Team (ASST) at the affiliated institution was alerted to the positive BioFire® BCID 1 panel and recommended treatment with IV echinocandin or high-dose oral fluconazole, given the patient’s risk factors for candidemia (e.g. active cancer and recent chemotherapy). Because of the negative Gram stain for yeast and subsequent negative conventional culture, the decision was ultimately made by the primary oncology team to decline antifungal therapy at that time. The patient continued to receive outpatient ertapenem to complete a 14-day course for the E. cloacae BSI. A follow-up set of blood cultures were obtained on day 4 of ertapenem OPAT without bacterial or fungal growth; thus, the BioFire® BCID 1 panel was not performed.
The day prior to the index admission, day 10 of ertapenem OPAT, blood cultures were drawn at the outpatient oncology clinic during his ertapenem infusion appointment. The next day, the patient was admitted to the hospital with fever of 100.7°F, chills, dizziness, and shortness of breath. Initial laboratory work-up revealed white blood cell (WBC) count of 27.3 K/µl with 91.6% neutrophils and serum creatinine of 5.73 mg/dl (baseline 1.20 mg/dl) with an estimated creatinine clearance of 10.9 ml/min. On admission, ertapenem was discontinued and cefepime 1 g IV daily was initiated. The blood culture did not become positive until the evening of hospital day (HD) 2; the Gram stain showed yeast and the BioFire® BCID 1 panel again detected C. parapsilosis. On HD 3, nearly 72 h since blood culture obtainment, conventional culture grew C. parapsilosis and the patient was initiated on caspofungin 70 mg IV loading dose, followed by a maintenance dose of 50 mg IV daily. Ophthalmology ruled out ocular dissemination and his prosthetic joint was deemed free of involvement, but he required placement of nephrostomy tube on the right to relieve a pelvic mass obstruction. Once speciation was finalized, the patient was then de-escalated to fluconazole 600 mg (9.2 mg/kg) given orally once daily on HD 4, as the local institution reports nearly 100% susceptibility among C. parapsilosis isolates to fluconazole, although susceptibilities were not obtained for this patient. The patient’s serum creatinine normalized to 1.1 mg/dl on HD 6 on IV fluids and antimicrobial therapy. Follow-up Gram stain and cultures on HD 6 were negative. On HD 7, oral fluconazole was increased to 800 mg (12.2 mg/kg) daily, which he received for a total of 3 weeks. The decision was made to continue suppressive oral fluconazole 200 mg daily indefinitely given the obstructive uropathy caused by the pelvic mass. Nearly 3 months post discharge, the patient had no recurrence of candidemia, but was transferred to hospice due to the progression of cancer (Figure 1).
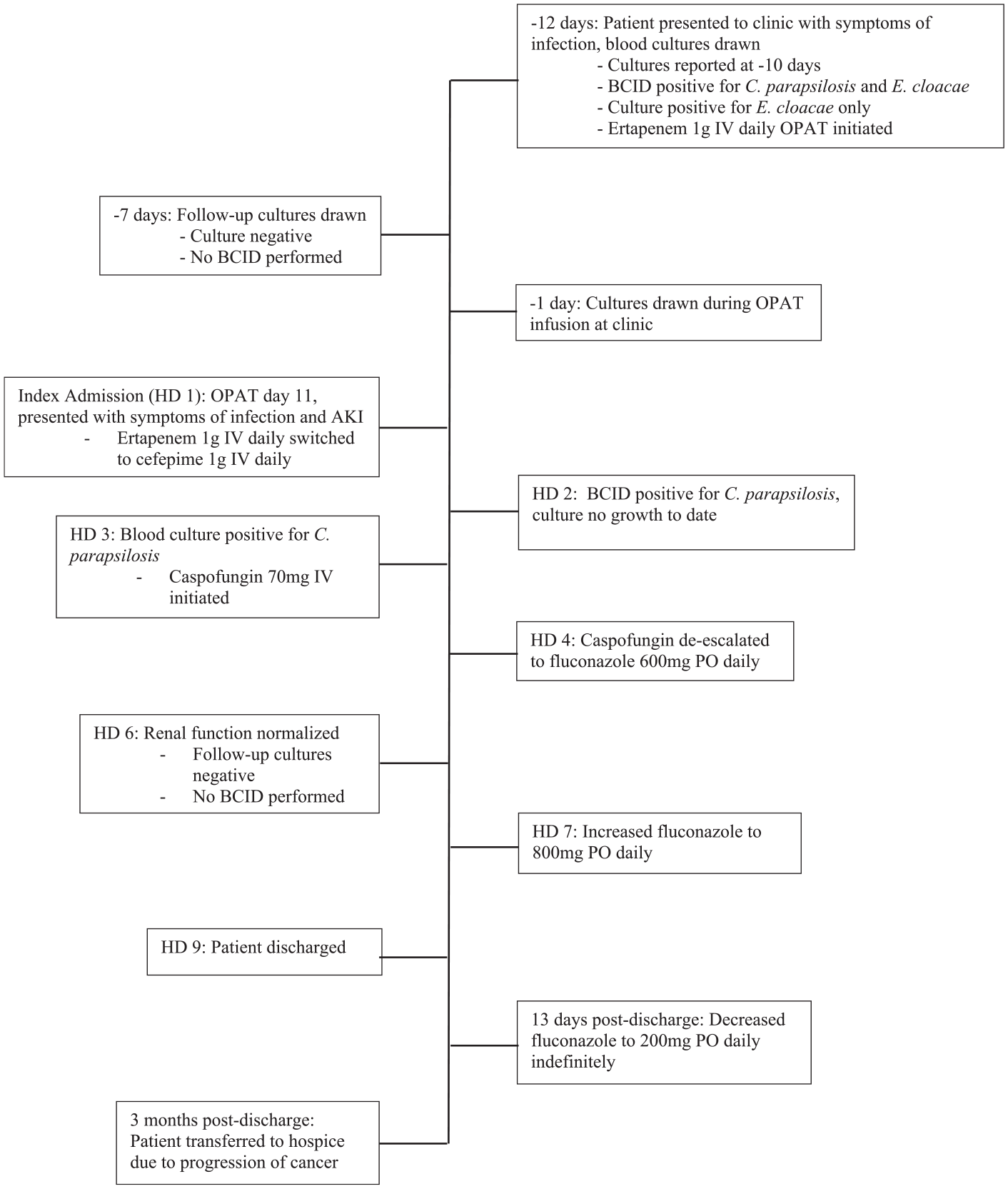
Timeline of events.
Discussion
Conventional blood cultures, which exhibit lower sensitivity and longer turnaround times than available RDTs, are the current gold standard for diagnosing candidemia in the United States. 7 The sensitivities of multiplex PCR-based assays, such as the BioFire® BCID 1 panel presented in this case and the magnetic resonance–based T2Candida® system, are superior to conventional blood culture techniques.4,8–10 The BioFire® BCID panels and the T2Candida® system, which are specific for the same Candida spp., have turnaround times of less than 2 and 5 h, respectively. It is recommended to use RDTs in conjunction with conventional blood culture methods because PCR and magnetic resonance–based assays are insufficient to test antifungal susceptibilities. 7 Earlier identification of the causative organism allows for prompt initiation of empirical antifungal therapy, preferentially echinocandins. 12 Because candidemia is associated with significant morbidity and mortality, RDTs may improve patient outcomes. 1
Patient populations at increased risk for developing candidemia include those with critical illness, receipt of parenteral nutrition, central venous catheter access, recent abdominal surgery or trauma, and solid organ malignancies consistent with our patient case. 1 It is important to note that a single positive Candida spp. among negative cultures may be considered a contaminant, in the absence of central line access and other risk factors. In contrast, a single positive Candida culture is considered to be a true infection if the patient has central line access at the time of culture obtainment.13,14 Risk factors for mortality in cancer patients with candidemia include the presence of a solid tumor, insufficient response to chemotherapy, advanced age, previous surgery, neutropenia, and previous antibiotic use. 15 The patient in the present case had multiple risk factors for candidemia and associated mortality, which prompted the local ASST to recommend initial antifungal therapy at the time of discordant results. Initiation of appropriate antifungal therapy was delayed by 14 days in this patient, which ultimately resulted in a 9-day hospital admission for the management of sepsis and acute renal failure. Several studies have demonstrated that delayed initiation of appropriate antifungal therapy results in increased mortality with differences in mortality becoming apparent with a 12- to 24-h delay and increasing with further delay thereafter.3,16 A meta-analysis of patients with candidemia concluded that infectious diseases specialist consultation should be the standard of care for all patients with candidemia, due to the statistically significant decrease in mortality that was associated with such consultations. Antimicrobial stewardship programs or ID specialists should be alerted to positive Candida spp. results from RDTs, Gram stains, and/or conventional cultures for prompt evaluation and potential treatment. 17 Conversely, overuse of empirical antifungals without indication can lead to increased resistance and unnecessary adverse effects.7
Previously, a statistically significant discordance between BioFire® BCID 1 and conventional blood cultures for Candida spp. was observed at this institution. 6 The discordance appears to be common in polymicrobial cultures, such as occurred in the present case. This institution releases all Gram stain, BioFire® FilmArray® BCID tests, and conventional culture results, regardless of concordance, to provide clinicians with all information required to make treatment decisions. While the reporting habits of other microbiology laboratories are unknown, there is a study presently underway to determine these practices. Table 1 presents the current reporting recommendations for discordant results provided in select RDT technology manuals.
Manufacturer recommendations for reporting of discordant results.

Because of the significant rate of discordance between diagnostic tests for C. parapsilosis observed at this institution and the overall sensitivity of 98.8% of the BioFire® BCID 2 for all targets, compared with less than 71% sensitivity of conventional blood cultures, we strongly recommend antimicrobial stewardship or infectious disease involvement in the decision-making for patients with discordant results, particularly those with immune compromising conditions or other significant risk factors for candidemia, making the diagnosis more likely.4–7 In addition, clinicians and stewardship teams should be aware of their microbiology laboratory’s reporting practices for discordant results at their institutions. Although knowledge of RDTs can be difficult to maintain, even among stewards, it is important for patient safety. 20
This case highlights the potential advantages of RDTs over conventional blood culture methods in early identification of candidemia in high-risk patients. It also emphasizes the role of antimicrobial stewardship programs in assessing patients across the continuum of care to optimize antimicrobial therapy, when needed, based on RDT and other test results. This case report along with the results of a previous investigation presents a reasonable argument to microbiology laboratories to consider the implications of reporting practices for discordant results, which might not currently allow healthcare providers with the opportunity to act on these results to improve patients’ outcomes.
Footnotes
Acknowledgements
This case was presented in part as a poster at the American Society of Health-System Pharmacists Midyear Clinical Meeting in December 2021. The authors thank the Microbiology Department at Prisma Health Midlands for their commitment to rapid diagnostic technology for our patients.
