Abstract
Febrile neutropenia (FN) is associated with mortality rates as high as 40%, highlighting the importance of appropriate clinical management in this patient population. The morbidity and mortality of FN can be attributed largely to infectious processes, with specific concern for infections caused by pathogens with antimicrobial resistance. Expeditious identification of responsible pathogens and subsequent initiation of empiric antimicrobial therapy is imperative. There are four commonly used guidelines, which have variable recommendations for empiric therapy in these populations. All agree that changes could be made once patients are stable and/or with an absolute neutrophil count (ANC) over 500 cells/mcL. Diagnostic advances have the potential to improve knowledge of pathogens responsible for FN and decrease time to results. In addition, more recent data show that rapid de-escalation or discontinuation of empiric therapy, regardless of ANC, may reduce days of therapy, adverse effects, and cost, without affecting clinical outcomes. Antimicrobial and diagnostic stewardship should be performed to identify, utilize, and respond to appropriate rapid diagnostic tests that will aid in the definitive management of this population.
Keywords
Introduction
In 1966, Bodey and colleagues described the clinical association between neutropenia (neutrophil count < 0.50 × 109 cells/L) and increased mortality in acute myeloid lymphoma (AML) patients.1–3 Five decades later, neutropenia in the setting of cancer remains a challenging clinical situation. Specifically, febrile neutropenia (FN) is of great interest due to both the associated economic burden and increased mortality.3,4 In the United States, neutropenic patients with cancer had hospital stays that amounted to nearly $6000 higher and 3 days longer than patients with cancer admitted for other reasons.5,6 In addition, FN is associated with mortality rates as high as 40% for invasive fungal infections, highlighting the importance of appropriate clinical management in this patient population. 7
The morbidity and mortality of FN can be attributed largely to infectious processes, resulting from defects in humoral immunity coupled with disruption of mucosal barriers, gastrointestinal colonization, and previous antimicrobial exposure.
8
While historically there has been dominance between Gram-positive and Gram-negative causative organisms, the etiology of infections in patients with neutropenia and fever continues to shift with changing prophylaxis regimens.
9
In particular, the global increase in incidence of Gram-negative resistance is of concern in FN. Extended-spectrum beta-lactamase-producing (ESBL) Enterobacterales are among the most frequently isolated pathogen in FN patients.
9
Carbapenem-resistant Enterobacterales (CRE) have been reported in this population as well, though frequency varies pending geographical region.
8
While
Because of the risk of these pathogens, expeditious identification of responsible pathogens through proper diagnostic techniques and rapid initiation of empiric therapy is imperative. Current FN guidelines agree on appropriately broad empiric antibiotics but differ with regard to discontinuation of therapy.4,10–12 In patients with identified or defined infections, these panels generally recommend following clinical practice guidelines or best practices for those individual infections. In patients with fever of unknown origin (FUO), there is a growing body of literature supporting shorter antibiotic courses.4,10–12 In addition, newer diagnostics have the potential to improve knowledge of pathogens responsible for FN and decrease time to results. Taken together, this information can help improve the work-up and management of patients with FN.
Methodology
A systematic literature search of the PubMed and Google Scholar databases was performed with the search terms FN, antibiotic duration with FN, treatment of FN, FN and discontinuing antibiotics, short antibiotic course with FN, and rapid diagnostic and FN. References of relevant articles were reviewed and added as appropriate. Guidelines identified using commonly referenced clinical sources were included in the Guideline Summary. English-language clinical trials evaluating therapy in patients with high risk of prolonged FN and published between 2016 (since the publication of the most recent guidelines) and September 2022 were included in the Updates section.
Guideline summary
There are several guidelines on the management of patients with FN, but the four guidelines discussed here are presented by the National Comprehensive Cancer Network® (NCCN), the European Conference on Infections in Leukemia (ECIL-4) the Infectious Diseases Society of America (IDSA), and the European Society of Medical Oncology (ESMO).4,10–12 According to the IDSA, FN is defined as ‘a one-time oral temperature of greater than 38.3°C or a sustained temperature of great than 38°C for ⩾ 1 h in a patient who has an absolute neutrophil count of less than 500 cells/µL within a 48-h period’.3,4 The guidelines agree that all patients presenting with FN should be initiated on empiric antibiotics. To determine which therapy should be initiated, clinicians should consider assessing the patient’s risk of infection; determining the local antimicrobial susceptibilities, the most common infecting organisms, and the potential sites of infection; ascertaining the need for therapy against Gram-positive or fungal pathogens; and assessing the patient’s clinical stability, recent antibiotic use, and presence of antibiotic allergies. 11 These guidelines are relatively similar in their recommendations for approaching FN patients; however, there are some subtle differences (Table 1).
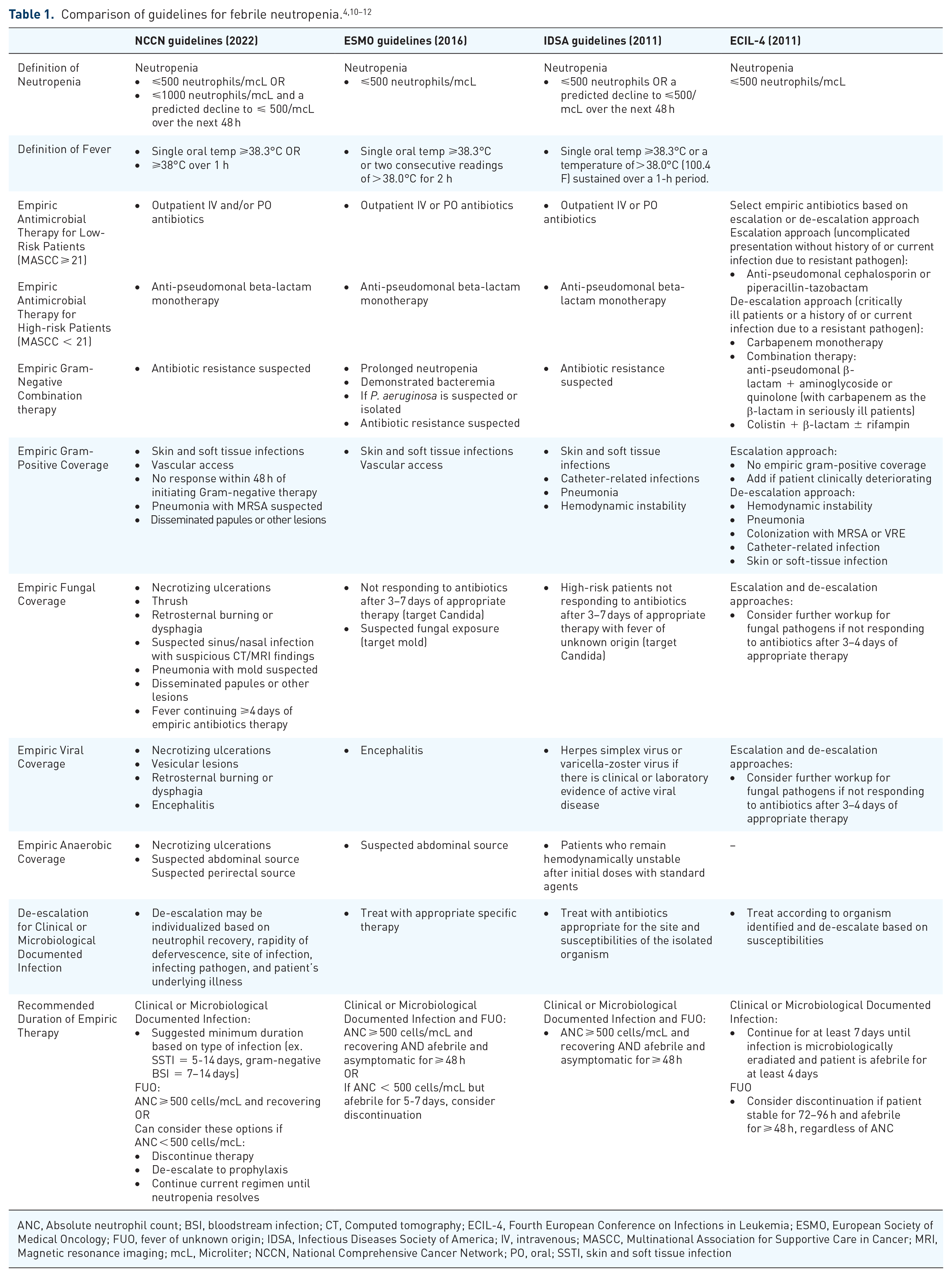
ANC, Absolute neutrophil count; BSI, bloodstream infection; CT, Computed tomography; ECIL-4, Fourth European Conference on Infections in Leukemia; ESMO, European Society of Medical Oncology; FUO, fever of unknown origin; IDSA, Infectious Diseases Society of America; IV, intravenous; MASCC, Multinational Association for Supportive Care in Cancer; MRI, Magnetic resonance imaging; mcL, Microliter; NCCN, National Comprehensive Cancer Network; PO, oral; SSTI, skin and soft tissue infection
Three guidelines (IDSA, ESMO, NCCN) divide their treatment recommendations into low-risk versus high-risk patients based on the Multinational Association for Supportive Care in Cancer (MASCC) score. 13 High-risk patients are identified by a MASCC score less than 21 and are at risk for complications if they acquire an infection. The ECIL-4 guidelines differ from the other major guidelines as the focus is on empiric antibiotic selection and the treatment duration for FN caused by FUO. 10
Duration of therapy
The duration of empiric therapy for FN is variable between guidelines (Table 1) and is currently one of the most controversial aspects of managing patients with FN. Due to the number of studies related to this topic since the latest guidelines, this review will focus on this area. All guidelines indicate that therapy can be discontinued when the absolute neutrophil count (ANC) is ⩽500 cells/mcL and is showing a trend toward recovery, but the ESMO and IDSA guidelines recommend also waiting until the patient has been afebrile and asymptomatic for at least 48 h with negative blood cultures.4,12
The NCCN, ESMO, and ECIL-4 guidelines also provide some guidance for patients who become afebrile, but in whom the ANC is still <500 cells/mcL. The NCCN suggests options ranging from discontinuing therapy to de-escalating to prophylaxis or continuing treatment until neutropenia resolves. 11 The ESMO guidelines recommend that therapy can be discontinued in patients with an ANC ⩽ 500 cells/mcL if the patients is afebrile for 5–7 days and has suffered no complications except in certain high-risk cases with acute leukemia when therapy can be extended up to 10 days. 12 The ECIL-4 guidelines recommend considering de-escalating therapy when the origin of fever is unknown once the patient has been stable for at least 72–96 h and afebrile for at least 48 h regardless of the ANC. 10 IDSA guidelines are the outlier with no recommendations for discontinuation prior to neutrophil recovery but allow for resumption of fluoroquinolone prophylaxis after an appropriate treatment course has been completed and signs and symptoms of infection have resolved. 4
Updates
Although these guidelines have been used clinically for many years, they are dated (6–18 years old). Over the past decade, several new studies have examined patients with fever and neutropenia.14–24 In order to best manage patients today, practitioners should be familiar with the most updated diagnostic and treatment information when considering best practices.
Updates in diagnosis
A key principle of the approach to a patient with FN is the importance of accurate diagnosis. Possible infectious agents that may cause fevers in a neutropenic host are too broad to accurately cover empirically in many circumstances, and only a small percentage of patients presenting with FN have a confirmed pathogen. 4 This necessitates rapid and definitive measures to obtain appropriate specimens to promptly make a diagnosis, which can ultimately impact decisions regarding escalation or de-escalation of antimicrobials in this patient population. Newer technologies like syndromic polymerase chain reaction (PCR) panels and other rapid diagnostic tests (RDT) improve the ability to identify a pathogen and decrease time to identification. By leveraging these technologies, clinicians may be able to provide targeted therapy in a timelier manner in microbiologically confirmed infections and determine if changes to antimicrobial therapy are appropriate if alternate pathogens or no etiology is identified.
PCR-based tests
Multiplex PCR panels
Approximately 10% to 25% of patients with FN have bloodstream infections (BSI), and several blood culture PCR panels (BC-PCR) have been evaluated in cancer patients.4,6,25–28 These studies demonstrated implementation of BC-PCR led to significantly shorter time to pathogen identification, but had limited impact on clinical outcomes, including length of stay, mortality, or readmissions.25–28 Clinicians are often hesitant to de-escalate therapy in patients with FN, so it is unsurprising that institutions were unable to show major differences in antibiotic coverage or duration of therapy with these tests. One unique aspect of cancer patients is that they are exposed to more antimicrobials than the general population, and rates of antibiotic-resistant pathogens are rising in this vulnerable group of patients.29–31 Many of the available BC-PCR panels can detect important Gram-positive and Gram-negative resistance patterns, including methicillin resistance, vancomycin resistance, and several beta-lactamases (KPC, IMP, OXA, NDM, and VIM carbapenemases and CTX-M-type ESBLs). Institutions with high rates of resistance may see greater benefit from the BC-PCR panels due to the ability to escalate or add antimicrobials sooner.
Many patients presenting with FN have respiratory viral illnesses, and previously reported rates likely underestimate the incidence due to lack of rapid testing. 32 Respiratory viruses are important in neutropenic cancer patients due to the possibility of progressing from upper to lower respiratory tract infection, increasing risk of mortality. 33 In patients with cancer, several international guidelines recommend respiratory viral testing based on local epidemiology and prevalence (one recommending for all cancer patients and the other two for hematologic and HSCT patients only).33–35 One study has assessed outcomes associated with implementation of a respiratory PCR panel in adult cancer patients. A retrospective evaluation in oncology patients found no difference in clinical outcomes (mortality, escalation to the intensive care unit [ICU], or 30-day readmission) between patients who had antibiotic therapy de-escalated at 72 h or continued based on the presence of a respiratory viral pathogen on the Luminex respiratory pathogen panel (Luminex Corporation, Austin, Texas). 36 Improving identification of respiratory viruses may allow for safe antibiotic discontinuation based on the limited data available. 36
Etiologies of diarrhea in cancer patients are numerous and include infectious and non-infectious causes. Stool culture and other conventional tests only diagnose a small portion of GI illnesses.37,38 A few studies have evaluated the use of the GI PCR panels in cancer patients.29,39–41 Results from the studies comparing the GI PCR panels to conventional testing in this population demonstrated significantly improved pathogen identification.29,40,41 It is unclear how many patients were presenting with FN or how the GI PCR panels influenced clinical outcomes in these studies, but they highlight the ability of these tests to improve diagnosis of infectious diarrhea in cancer patients. Notably,
There are limited data on rapid diagnostics for CNS infections in patients with hematologic disorders.
44
Incidence of meningitis and encephalitis (ME) is low in this population but is more common in those with allogeneic HSCT.
45
The Biofire FilmArray ME PCR panel (FA/ME) (BioFire Diagnostics, Salt Lake City, Utah) is widely used and can diagnose bacterial and viral pathogens along with
MRSA PCR
Neutropenic patients are at an increased risk for drug-resistant Gram-positive pathogens including methicillin-resistant
Given these indications, a significant number of patients will receive empiric vancomycin and would benefit from tools to assist with de-escalation. Assessment for nasal carriage of MRSA has been used as a screening tool for MRSA infection in a number of practice settings. Its utility for ruling out MRSA in patients with bacterial pneumonia has been well established and MRSA PCR is now recommended for routine use in patients with community acquired pneumonia.50,51 Neutropenic patients have generally not been well represented in these studies, but there are now three published studies evaluating the predictive value of PCR-based MRSA nasal carriage assessment and MRSA infection in neutropenic patients.52–54 All three studies found a low prevalence of MRSA colonization and infection. The MRSA PCR demonstrated a negative predictive value of 97.5% to 99%, consistent with studies in non-neutropenic patients. Emerging data support the utilization of MRSA screening to aid in de-escalation of vancomycin therapy and clinicians should work to incorporate them into clinical protocols and practice, particularly if local prevalence of MRSA infection is low.52–54
Fungal diagnostics
Neutropenic patients are at a significant risk for invasive fungal diseases (IFD) including invasive aspergillus (IA) and systemic candidiasis and candidemia. Current biomarkers recommended by guidelines include aspergillus galactomannan (GM) and 1-3-β-D-glucan.4,10–12 The GM assay is specific for invasive aspergillosis, whereas the beta-glucan assay identifies any pathogens that have 1-3-β-D-glucan as a component of the cell wall, including
Aspergillus galactomannan
A large meta-analysis found the GM assay had a sensitivity of 70% among patients with hematologic malignancies, but another study found a sensitivity of only 43% in patients on prophylactic therapy.
56
Duarte
1-3-β-D-Glucan
Assessment of 1-3-β-D-Glucan is recommended by multiple expert guidelines for the diagnosis of IFD including candidemia other mycoses. 60 This test is most routinely used to assist in the diagnosis of invasive candidiasis (IC) and may be particularly helpful for patients with blood culture negative IC. 61 It has shown utility in the diagnosis of IFD in patients with AML and myelodysplastic syndrome where the test was positive a median of 10 days before clinical diagnosis 1-3-β-D-glucan may be specifically useful in diagnosis of non-IA IFD including fusariosis and pneumocystis.62–64 A meta-analysis including data from 594 patients with proven or probable IFD calculated a pooled sensitivity of 76.8% and specificity of 85.3%, but significant variability was noted between studies. 65
Emerging diagnostics
Additional rapid diagnostics have emerged in recent years, but there is limited guidance and evidence for implementation.66,67
T2 magnetic resonance (T2Candida)
This test has been used for the diagnosis of IC in medical patients. In a prospective trial, T2Candida demonstrated a 89% sensitivity for the diagnosis of IC, and was more effective than blood cultures for patients on antifungal therapy.66,67 However, a trial evaluating T2Candida accuracy using stored samples found that T2Candida was only positive in 17 of 25 samples from patients with candidemia and 6 of 22 patients with deep-seated candidiasis. Implementation of this test is further limited by significant cost of the instrument and interpretation of discordant results. 68
Next generation sequencing
Non-hypothesis driven next-generation sequencing (NGS)-based assays have been introduced over the last few years for diagnosis of infectious syndromes. These tests use advanced, non-invasive, rapid methods in order to identify viral or bacterial pathogens in blood or other samples from patients. 69 Utility in neutropenic fever has been assessed in small studies but further research needs to be done to clarify the role of these costly tests.70,71 In a cohort of 101 immunocompromised patients, iDTECT® Dx Blood (PathoQuest, Paris, France) was three times more likely than standard microbiological tests to identify a clinically relevant virus or bacteria. 71 The Karius test (Redwood City, CA, USA), a plasma cell-free DNA-based assay, is commercially available in the United States. In a cohort of 55 patients with neutropenic fever, Karius test was found to have a sensitivity of 85%. 70
Clinicians need to be aware of the limitations associated with NGS-based assays prior to incorporating them in routine management of patients with neutropenic fever. 72 NGS-based assays can be expensive, frequently detect organisms of unknown significance, be falsely negative in the setting of prolonged antibiotic exposure, and do not provide phenotypic antimicrobial resistance information. Furthermore, the ideal time and frequency of obtaining these assays in neutropenic patients remains to be determined. Obtaining an NGS-based test at the onset of every neutropenic fever will likely contribute to overutilization of healthcare resources while waiting to use these tests as last resort may render them falsely negative because of prolonged antibiotic exposure.
Update in management
Although all four of the main guidelines comment on when discontinuation of therapy may be considered in FN, no consensus has been reached for the duration of empiric antibiotics for clinically stable, afebrile patients at high risk for prolonged neutropenia.4,10,11 Subsequently, the duration of antibiotics in this patient population has been primarily driven by the resolution of clinical signs and symptoms of infection, achieving defervescence, origin of fever, obtaining source control, if warranted, and recovery of ANC to ⩾ 500 cell/mm3. However, clinicians involved with antimicrobial stewardship continue to advocate for shorter courses of optimal antibiotics with hopes of avoiding unnecessary use of broad-spectrum therapy and limiting antibacterial resistance in this patient population.19,73,74 With the growing concern for antibiotic resistance and limited antibiotic development (10 for 20 initiative), it is imperative to reevaluate dated clinical practices in hopes of modernizing the standard of care.74,75 Within the past 6 years since the ESMO guidelines were published, several studies have been published that shed light on appropriate paths forward for appropriate duration of empiric antibiotic therapy in patients presenting with FN (Table 2).14–24
Studies published on FN empiric therapy since 2016.

ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; ANC, absolute neutrophil count; BSI, blood stream infection; C, control; DDD, defined daily dose; DOT, days of therapy; EAT, end of antibiotic therapy; ECIL-4, Fourth European Conference on Infections in Leukemia; Fourth European Conference on Infections in Leukemia; FN, febrile neutropenia; FQ, fluoroquinolone; FUO, fever of unknown origin; HR, hazard ratio; HSCT, hematopoietic stem cell transplantation; I, intervention; ICU, intensive care unit; MDS, myelodysplastic syndrome; PI, Phase I; PII, Phase II; Pts, patients; SD, standard deviation; SMC, serious medical complication.
Presented as median ± (interquartile range) unless otherwise specified.
ECIL-4 recommendations: discontinuation of antibiotic therapy when patients were afebrile, had resolution of infectious signs and symptoms, and normal vital signs for at least 48–72 h.
Randomized control trials evaluating antibiotic durations in FN patients
Only one randomized control trial (RCT) has been performed comparing the relationship between short courses of therapy for FN and clinical outcomes in patients at a high risk for prolonged neutropenia (Table 2). The How Long study conducted by Aguilar-Guisado and colleagues examined clinical outcomes in 157 patients with hematologic malignancy and hematopoietic stem-cell transplant (HSCT) presenting with FN without a microbiologic diagnosis. Patients in the experimental group had empiric antimicrobial therapy (EAT) stopped once afebrile and hemodynamically stable for at least 72 h. All control patients were given the standard of care based on hospital-specific protocols. Patient demographics were similar between the experimental and control groups, including the median number of neutropenic days (14 versus 11 days). EAT-free days was significantly lower in the experimental group (
Clinical studies evaluating antibiotic durations in hematopoietic stem cell transplantation (HSCT) with FN
In 2018, Snyder and colleagues retrospectively compared allogeneic HSCT patients admitted with FN who were de-escalated from empiric broad-spectrum antibiotics (BSA) to prophylactic agents by day 5 (cohort 1) to patients who received empiric BSA until neutrophil engraftment (cohort 2). The primary outcome of recurrent fever within 72 h was experienced by 7 (15%) in cohort 1 vs 14 (19%) in cohort two (
Clinical studies evaluating antibiotic durations in hematological malignancies (HM) with FN
In 2016, Kroll and colleagues retrospectively evaluated 52 patients with acute myeloid leukemia (AML) and acute lymphocytic leukemia (ALL) who had empiric antibiotics de-escalated to prophylactic therapy after 14 days. Patients who had antibiotics de-escalated at day 14 were just as likely to remain afebrile as those who did not (80.7% vs 61.5%
In 2020, Fuller and colleagues compared the risk of recurrent fevers in AML patients who had their empiric antibiotics de-escalated before neutropenia resolved and those who continued or escalated antibiotics until recovery of neutropenia. Results from this study demonstrated a 46% relative risk reduction in recurrent fever in patients with antibiotic de-escalation (hazard ratio [HR]: 0.54; 95% confidence interval [CI]: 0.34–0.88;
In 2022, 3 studies examined the role of short courses of antibiotics in AML patients with FN. Metais and colleagues compared short (<7 days) and long courses (>7 days) of therapy for confirmed BSI in 71 AML patients with FN.
20
Of the 104 BSI, 42% were caused by enterobacterales, followed by coagulase-negative staphylococcus (21%), and streptococci (13%). Eight (7 patients; 7.6%) BSIs relapsed within 30 days of antibiotic discontinuation and no difference was found between patients receiving short and long courses. The median length of neutropenia was higher in relapsing vs non-relapsing episodes (45 days vs 22.5 days;
Alegria and colleagues performed a pre–post, quasi-experimental trial evaluating a novel de-escalation guideline for AML patients with FN.
17
Pre-implementation patients received physician-directed durations and post-implementation patients’ empiric antibiotics were either de-escalated to fluoroquinolone prophylaxis or discontinued after 5 days of empiric therapy if the patient was afebrile and stable for 48 h. No difference was observed in suspected or documented infection after antibiotic de-escalation or discontinuation, and a decrease in the incidence of
Finally, Contejean and colleagues performed a quasi-experimental study evaluating pre- and post-intervention groups after an ECIL-4 based antibiotic de-escalation and discontinuation strategy was implemented in a hematology malignancy department in Paris, France. 273 hospital stays (164 patients) and 217 (148 patients) were analyzed in the pre and post groups, respectively. The primary composite endpoint was transfer to the ICU for more than 24-h or death. Using defined daily doses (DDD) per 1000 patients-days, it was determined that the post group had less raw consumption of carbapenems (pre-group 1187 DDD/1000 vs post-group 331 DDD/1000;
Eleven new studies, including one RCT, have evaluated early discontinuation or de-escalation of empiric antibiotics for patients presenting with FN. Four of the studies used the ECIL-4 recommendations and rates of mortality and other clinical outcomes were similar between patients with early discontinuation and those who continued antibiotics until neutrophil recovery.15,16,18,24 The remaining studies used similar strategies, but many de-escalated to antibiotic prophylaxis instead of discontinuing therapy as recommended in the ECIL-4 recommendations. Taken together, these results demonstrate cost-savings of shorter regimens without negative clinical outcomes. There are limitations to the studies discussed above, including most are retrospective with small patient populations. In addition, the findings cannot be applied to all patients with identified infection though it is reasonable to consider targeted antibiotic therapy for a duration appropriate for the disease state and organism. 10
Discussion
FN remains a significant clinical challenge marked by the challenge of identifying unusual pathogens and extended broad-spectrum antimicrobial therapy. The growing array of diagnostic tools have the potential to improve pathogen identification and lead to more rapid antimicrobial de-escalation and targeted therapy. Unfortunately, rapid diagnostics have not always had a clear positive impact on clinical outcomes and antibiotic use, leaving the optimal utilization of these tests unclear.76–81 Misuse and overuse of diagnostics have their own potential for untoward costs and outcomes. Some tests may reduce need for additional diagnostics, like imaging or endoscopic procedures; however, they also may have a negative financial impact if no action is taken based on the results or the tests are inappropriately ordered.76–81 In addition, PCR based tests cannot distinguish infection or disease from colonization; therefore, clinical context should be considered, and pre-test probability should be high before ordering these panels. Both fungal and syndromic tests should be targeted to appropriate clinical syndromes and broad ‘shotgun-like’ approaches should be avoided. Laboratories should consider creating multidisciplinary approved guidance, including order panels or interpretation criteria prior to implementation to encourage appropriate test utilization. For example, a GI PCR panel could be recommended first-line in patients with FN presenting with community-acquired diarrhea, but a two-step approach for hospital-acquired diarrhea that tests for
Antimicrobial de-escalation in FN without a clear diagnosis is more challenging but recent studies have demonstrated the safety of this approach.14–24 Across the studies, different methods and definitions for de-escalation and/or discontinuation were used, and the rate of de-escalation to antimicrobial prophylaxis varied. Despite this variance, there does appear to be a significant population of patients in which de-escalation of antimicrobial therapy is safe and appropriate. Ideal patients for de-escalation are those who have defervesced, are clinically stable, and who do not have a documented bacterial infection despite appropriate workup. To guide clinicians toward this goal, antimicrobial stewardship programs and health systems should establish guidance and processes that encourage de-escalation for these patients. A recently published survey of United States cancer centers found only 18 of 29 (62%) of surveyed hospitals had guidance for de-escalation of antibacterial therapy, highlighting this need. 82 Further studies are needed to evaluate the impact reduced antibiotic exposure has in patients with FN on drug resistance and cost and to identify favorable de-escalation strategies.
Conclusions
Historically, there have been many challenges for antimicrobial stewardship in patients with FN. There are four commonly used guidelines, which have variable recommendations for empiric therapy in these populations. All agree that changes could be made once patients are stable and/or with an ANC over 500 cells/mcL. Recent data show that rapid de-escalation or discontinuation of therapy, regardless of ANC, may reduce days of therapy, adverse effects, and cost, without impacting clinical outcomes. Antimicrobial and diagnostic stewardship should be performed to identify, utilize, and respond to appropriate rapid diagnostic tests that will aid in the definitive management of this population.
