Abstract
Poxviridae have been successful pathogens throughout recorded history, infecting humans among a variety of other hosts. Although eradication of the notorious smallpox has been a globally successful healthcare phenomenon, the recent emergence of Monkeypox virus, also belonging to the Orthopoxvirus genus and causing human disease, albeit milder than smallpox, is a cause of significant public health concern. The ongoing outbreak of monkeypox, demonstrating human–human transmission, in previously nonendemic countries, calls for critical need into further research in the areas of viral biology, ecology, and epidemiology to better understand, prevent and treat human infections. In the wake of these recent events, it becomes important to revisit poxviral infections, their pathogenesis and ability to cause infection across multiple nonhuman hosts and leap to a human host. The poxviruses that cause human diseases include Monkeypox virus, Molluscum contagiosum virus, and Orf virus. In this review, we summarize the current understanding of various poxviruses causing human diseases, provide insights into their replication and pathogenicity, disease progression and symptoms, preventive and treatment options, and their importance in shaping modern medicine through application in gene therapy, oncolytic viral therapies for human cancers, or as poxvirus vectors for vaccines.
Introduction and viral taxonomy
Poxviruses are infrequently encountered in clinical practice but are well-established agents of human disease belonging to the family Poxviridae. Current viral taxonomy divides the family into subfamilies Chordopoxvirinae and Entomopoxvirinae. 1 The genera Orthopoxvirus, Parapoxvirus, Molluscipoxvirus, and Yatapoxvirus belong to the Chordopoxvirinae family and are best known for human diseases caused by Poxviridae. Major orthopoxviruses include Variola, Vaccinia, and Monkeypox virus species. Among Parapoxviruses, Orf virus and Molluscum contagiosum virus species of Molluscipoxvirus are well-described human pathogens. Finally, Yatapoxvirus species Tanapox virus and Yaba monkey tumor virus can affect human hosts.
Viral taxonomy of Poxviridae is detailed in Table 1. 1
Viral taxonomy of Poxviridae.

Structurally, these are large viruses (220–450 nm length) with an envelope that contains the viral genetic material, namely, double stranded deoxyribonucleic acid (dsDNA) arranged linearly with hairpin terminals forming continuous polynucleotide chains. 2 The virions are oval or brick shaped, which can differ between species. Poxviruses are unique in their replicative process which occurs in host cellular cytoplasm opposed to host-cell nuclei where most other DNA viruses are known to replicate. 3
In this narrative review, we discuss the epidemiology, pathogenesis, clinical manifestations, and management of the best known ‘human poxviral diseases’, namely, Smallpox, Monkeypox, Orf, Tanapox, and Molluscum among others; this is followed by a discussion of emerging therapeutics that employ Poxviruses with oncolytic potential.
Clinical diseases
Genus Orthopoxvirus
The Orthopoxvirus genus of the Poxviridae family has at least 12 species. 1 Multiple vertebrates including humans play host to this genus. In the evolutionary history of this genus multiple genes have been lost or truncated. The species in this genus include Abatino macacapox, Akhmeta, Camelpox, Cowpox, Ectromelia, Monkeypox, Raccoonpox, Skunkpox, Taterapox, Vaccinia, Variola, and Volepox. The cowpox strain seems to have the most conserved genes. 4 The orthopoxviruses, around 250 nm long and 220-450 nm wide, have linear dsDNA genomes, put together in a brick shaped geometry. 5 The genome, housed within the nucleoprotein complex, consists of about 170–250 kbp of dsDNA closed covalently into telomere-like hairpin bends. Each virion also contains a host of structural proteins and ribonucleic acid (RNA)-polymerase units that allow robust viral gene transcription, as seen in Figure 1. 5

Structure of the extracellular enveloped and intracellular mature orthopox virion.
The well-orchestrated life cycle of orthopoxvirus occurs in the host-cell cytoplasm. Upon fusion of viral proteins to cellular membrane glycosaminoglycans, the former releases its core into cytoplasm, while early expression of viral proteins, through RNA-polymerase, uncoats its genome to begin replication by viral DNA-polymerase. This process takes about 100 minutes post initial infection. Further, the proto-virion is assembled in host cytoplasmic viral factories, over the next 140 minutes to 48 hours post-infection. The mature virions are released via host-cell lysis or acquiring a double membrane from the golgi apparatus and budding as an external enveloped virion (EEV). The latter utilizes the cellular microtubule transport, fusing to the lipoprotein layer before releasing into the outer surface. 5 These mature virions, with enveloped or nonenveloped forms (IMV), attached to the outer cell surface each have distinct antigenic properties and are believed to have different attached sites on the cells. 5 Variability in antigenic expression from these virions modulates the host immune response and infectivity of progeny virions. 6 Figure 2 summarizes the replication cycle of orthopoxviruses in cytoplasm.

Schematic representation of the Orthopoxvirus replication cycle and release of mature virion in the host-cell cytoplasm.
Although Ectromelia and Camelpox are known to be highly host-specific, the four zoonotic orthopoxvirus species known to cause human diseases are Cowpox/Buffalopox, Monkeypox, Variola (smallpox), and Vaccinia (smallpox vaccine). Until eradication in the 1980s, Variola caused significant morbidity and mortality. Although Vaccinia continues to be utilized as a disease-prevention tool through immunization, there have been sporadic reports of disease in laboratory workers and vaccinated individuals. In addition, in Brazilian and Indian cow/buffalo cow herders, a Vaccinia-like virus has been reported to infect not just cows, rodents but also humans in contact.7,8 Cowpox infection in humans is commonly associated with occupational exposure in farmers. Occupational exposure to Cowpox virus in veterinarians is not only through cows but also other animals like cats, rats, and elephants.9,10 Interestingly, monkeypox was distinguished as a separate infection than smallpox only after the 1970s, causing disease mainly in humans and monkeys, although maintained in nature in rodent hosts. Most clinical symptoms are similar to the ordinary smallpox infection although milder. 11
In humans infected with orthopoxviruses initial infection symptoms are constitutional including fever, headache, myalgia, malaise, vomiting, and lymphadenopathy. Dermatological manifestations include a skin rash comprising either vesicles or pustules or both. Although skin lesions alone are not diagnostic of Orthopoxvirus infections, individual lesions are very often seen progressing to eventually form a dry crust. In severe cases, this may progress to general erythema and edema and involve the nervous system causing encephalitis or myelitis. 8 Diagnosis in infected individuals is clinical, eventually established by a confirmatory molecular test; the Centers for Disease Control and Prevention (CDC) recommends use of the diagnostic algorithm to aid the process. 8 Management in most cases includes supportive care (nutritional supplementation, hydration, covering of lesions to prevent spread) and preventing secondary infections with rigorous infection prevention and control practices, 8 allowing the course of infection to subside combated by the host immune response. Although human disease caused by orthopoxviruses have common traits, it is important to understand their individual epidemiology, pathogenesis, transmission, and disease progression pathways to treat them robustly.
Variola (smallpox), Cowpox, and Vaccinia
History and Epidemiology
The Variola virus species is the most prominent member of the Orthopoxvirus genus. In nature causing infection primarily in humans, it has been historically reported in both Indian and Chinese medical texts from 1500 to 1100 BCE, originally speculated to have been spread from Egypt by traders. 12 Over the middle ages, smallpox had become significantly prevalent across Europe and spread further into the new world through increasing colonization, with estimates suggesting over 30% mortality. 13 By the 18th century smallpox was a leading cause of death in Europe and killed an estimated 400,000 people each year, 14 and although remaining largely endemic to separate regions in the world, it was still a significant burden to human health.
Virology and Pathogenesis
Variola is a large, linear dsDNA virus – 186 kbp in size with telomere-like hairpin loops in the end. 15 The brick-like outer structure housing the genome is around 330 nm × 250 nm in size (Figure 1), presenting 2 viral strains–Variola major and Variola minor. 16 Fascinating in their replication, as with the other species in the genus, Variola replicate primarily in host-cell cytoplasm and utilize a robust machinery of special proteins, specifically the DNA-dependent RNA polymerase. Both the enveloped or nonenveloped forms of the mature virion emerging at the cell surface are highly infectious. Smallpox was highly contagious and transmission was predominantly through the inhalation route of airborne viral agents, from an infected individual’s oral, oropharyngeal, and nasopharyngeal regions, over sustained contact. Infection transmission through fomites, contaminated sheets, clothes, and so on, was also possible. Although transmission could occur throughout the illness, viral shedding is high past the prodromal stage, particularly with rash or lesion development in the oropharyngeal regions. Although the peak of infectivity wanes after 7 to 10 days, as the scabs cover lesions, transmission is possible until all lesions heal completely.17,18 Infection with one strain often conferred immunity against the other. 19 Upon inhalation, the virus could gain entry into the mucous membrane of the nasal, oral, pharyngeal, or respiratory epithelium of the host, before invading the lymph nodes. During the course of disease, an increasing viral load may cause widespread host-cell lysis and may be detectable in the bloodstream causing viremia. This could further potentiate and subsequently exacerbate the disease by furthering viral spread into other organs such as the bone marrow and spleen. 19
Clinical Manifestations and Diagnosis
Initial infection is often associated with fever (at least 101°F), fatigue, malaise, myalgia and headache. Generalized symptoms may also include nausea and vomiting. After the prodromal and incubation phase the initial visible red lesions appear by days 12 to 19, over mucosal membrane in the mouth, tongue, palate, and pharynx, while the body temperature may lower to normal. Soon these lesions enlarge and rupture, increasing the salivary viral load dramatically. 19 The striking feature of smallpox is the appearance of rashes, 24 to 48 hours after mucosal lesions develop, in different regions of the body starting from the forehead, face, and proximal extremities. These lesions, seated deeper epidermally; 0.5 to 1 cm in size, pass through the stages of macules, papules, and vesicles before developing scabs during week 2 to 3, resulting in the classical smallpox pockmarks. 19 Although the skin lesions make smallpox recognition distinct, differential diagnosis considered include cowpox infection, molluscum contagiosum, severe chickenpox, disseminated varicella zoster, herpes simplex infection, and secondary syphilis. 20 The CDC has designated a streamlined diagnostic algorithm to aid patient evaluation. 21
Microscopic diagnosis of variola depicts the characteristic cytoplasmic inclusion bodies – guarnieri bodies, appearing as strong pink areas in the hematoxylin and eosin (H&E) staining of skin biopsies. These may appear in other poxviral diseases too, and their absence does not rule out smallpox.19,22 In addition, viral agents in the scabs or pustular fluid can be identified using electron microscopy, identifying the brick-shaped poxvirus and eliminating other infectious agents. A definite diagnostic method that was used involved culturing the virus over the chorioallantoic membrane of chicken eggs to observe development of the classic smallpox pock lesions. Critically, real-time polymerase chain reaction (RT-PCR) assessment generates a specific and sensitive characterization of the infecting viral strain and species.23–25 Point-of-care antigen detection assays are also of high value to help initiate rapid countermeasures to disease management. 25
Management and Prevention
Initial treatment is often supportive in nature and involves fluid resuscitation, wound care, infection control and secondary bacterial infection prevention. Importantly, vaccination, even 3 days post-exposure, significantly prevents and decreases disease severity. 26 Immunization through ‘variolation’ was common practice in the early history of smallpox in India and China, introducing pustular material from infected individuals into infected naive individuals. 25 Although this immunization was successful, smallpox associated mortality was still significant. The discovery of Cowpox virus, infection in milkmaids and the inoculation of its infectious material into infection naive individuals led to a new era of vaccines, by Edward Jenner, with the same level of protection as variolation but lower adverse effects. 27 Subsequently, vaccines were mass produced under animal skins and the chorioallantoic membranes of embryonated eggs. 25 This led to the development of Vaccinia virus, used for vaccination as ‘calf-lymph’, although its actual history remains undetermined. Recent studies show that Horsepox may be closely related to Vaccinia virus (99.7%). 28 20th- and 21st-century vaccines were developed in calves (Dryvax – freeze-dried live-virus smallpox vaccine prepared from calf lymph and used for smallpox eradication efforts) or cell lines (ACAM2000 – live Vaccinia, cloned from the same strain as Dryvax, cultured in vero cell lines).25,29,30 A third vaccine – MVA-BN, is a live, nonreplicating vaccine for smallpox and monkeypox, derived from Modified Vaccinia Ankara, an attenuated strain of the Vaccinia. 31
Therapeutics have also been developed to counter smallpox, marking a significant achievement in drug development. ‘Tecovirimat’, the first drug approved by the United States Food and Drug Administration (FDA) for smallpox treatment, acts by targeting a key viral phospholipase thus inhibiting viral exit from host cell. It also blocks formation of enveloped virions, thereby effectively inhibiting virus dissemination both in vitro and in vivo. 32 More recently a second drug – brincidofovir, a prodrug of cidofovir, has been approved for smallpox therapy caused by Variola. 33 It acts by increasing the intracellular concentration of cidofovir thus effectively acting against viral replication as a nucleoside analogue that selectively inhibits the viral DNA polymerase. 34 Development of therapeutics and vaccines in the post-smallpox eradication era has been given special attention, due to the scare of potential biothreats and biorisks, from intentional misuse of the in-store Variola and Vaccinia. Routine smallpox vaccination was stopped in 1972 after the disease was eradicated in the United States. The World Health Organization (WHO) declared smallpox an eradicated disease in 1980. 35
Monkeypox
History and Epidemiology
Monkeypox virus is a species of the Orthopoxvirus genus, an emerging zoonotic infectious disease, that has gained prominence in the post-smallpox eradication era. Human infection presents very similarly to smallpox–prodromal stage followed by skin lesions. 36 It was first described in 1958 in an outbreak in cynomolgus monkeys, used for polio vaccine research, at Statens Serum Institut, Denmark. 37 Subsequently other cases were reported in monkeys in Europe and the United States. 38 The first human case was reported in 1970 in the Republic of Congo. 39 Since then cases have been recorded in over 11 countries including western and central Africa. 40 Once considered a rare zoonotic disease, new cases have increased and spread to multiple regions, highlighting its emergence as an important global disease.
Virology, Pathogenesis and Clinical Manifestations
Interestingly, Monkeypox virus is similar to Variola. Monkeypox virus is a large brick like virus, often even discernable by light microscope, ranging in size from 220 to 450 nm in length and 140 to 260 nm in width. The inner dsDNA is covered in a core complex, surrounded by the lateral bodies and wrapped in a lipoprotein outer cover. The monkeypox genome, one of the largest of all viruses, is about 197 kbp. There is significant homology of the genes in the central region, across orthopoxviruses, and variability in the terminal ends, leading to the different virulence across species in the genus. 41 Similar to other orthopoxviruses, each virion has a complex system of proteins and enzymes that hack host cellular mechanisms to aid stimulation of its replication, assembly and exit, responsible for further transmission. 42 Two distinct genetic clades of the virus have been identified leading to infection – the west African and the central African clade. 43
The virus can spread from animals to humans by either animal bite or contact with infectious bodily fluids. In human-to-human contact, the virus is spread through inhalation of oral, nasal, and oro-pharyngeal droplets, or fomite contact from infected individuals. The incubation period remains under speculation but thought to be commonly between 7 and 14 days and can range from 5 to 21 days. 44 It is proposed that incubation may vary by the route of transmission. 45 As with smallpox infections, the prodromal stage in monkeypox presents with symptoms such as fever, myalgia, malaise, fatigue, headache, sore throat, chills/sweats, and lymphadenopathy.46,47 Lymphadenopathy is a distinguishing feature of Monkeypox and differentiates it from smallpox. Patients can also experience gastrointestinal symptoms such as nausea, vomiting, loss of appetite, and diarrhea. The second stage to infection – rash phase manifests from 24 to 72 hours after onset of fever and lymphadenopathy. Lesions have been described as well circumscribed, deep seated, often with umbilication and may affect palms and soles. 44 The lesions appear simultaneously across the body in severe disease, more in the periphery in milder form, progress from papules, vesicles, pustules, and finally crusts, over 2–3 weeks duration. Importantly, individuals immunized against smallpox with Vaccinia develop significantly less lesions.48,49 Ocular and respiratory involvement during the course of infection can occur and may lead to complications such as loss of vision or bronchopneumonia. Encephalitis, secondary bacterial infections, and sepsis are other critical complications. Mortality associated with monkeypox is 1–11% and higher rates are noted in the central African clade than west African. 48 Monkeypox lesions can easily be mistaken for other conditions such as varicella, secondary syphilis, measles, and even smallpox.
The pathogenesis model of monkeypox in humans has been best mimicked and studied in black-tailed prairie dogs. During initial infection, the virus is largely local to the site and associated with acute inflammatory host response – phagocytosis, cell necrosis, and vasculitis. This is accompanied with local replication of the virus, followed by spread to the regional lymph nodes and lymphatics eventually resulting in primary viremia. This further propels viral spread systemically to other organs–bone marrow, liver and spleen resulting in secondary viremia, causing disseminated lesions.50,51 Morphological features observed in H&E staining include the classic guarnieri bodies type b inclusion bodies and type a inclusion bodies. Importantly, there are pathological differences among the two clades of monkeypox, with the central African clade exhibiting a more rapid spread and accumulation in tissues causing increased disease burden. 51
Diagnosis, Management, and Prevention
Although historical diagnostic methods using confirmation of viral pocks on a chicken embryo chorioallantoic membrane and electron microscopy can confirm infection by orthopoxvirus, contemporary confirmatory testing is the RT-PCR. The variable terminal end of the genome makes for ideal targets for detection. Other genes targeted for the confirmatory diagnostic test are hemagglutinin, envelope protein, inclusion body gene, crmB gene, B7R gene, and so on.52–54 As confirmatory tests are time-consuming, point-of-care tests can play a critical role in early detection, even as confirmatory tests are awaited. Two tests have been developed in this area: Antibody Immuno Column for Analytical Processes (ABICAP) immunofiltration system and Tetracore Orthopox BioThreat.55,56
Once diagnosed, treatment for monkeypox is largely supportive and symptomatic. There are no approved therapies for monkeypox, but animal studies suggest cidofovir, tecovirimat, and brincidofovir may have a role in treatment or lowering disease burden. 57 Adler et al. have reported successful clinical treatment of human Monkeypox with oral Brincidofovir in three cases – the dosing regimen was 200 mg as a single dose in one case and two doses in the other two cases. The major clinical complication associated with Brincidofovir use was reportedly transaminitis.
They also report clinical success treating a case of Monkeypox with oral Tecovirimat, 600 mg twice daily for 2 weeks. 58
Vaccinia immune globulin (VIGIV) is another therapy that could be utilized in monkeypox complication treatment. Antibodies are derived and purified from individuals inoculated with smallpox vaccine and administered intravenously to manage the serious side effects caused in some individuals after being dosed with the smallpox vaccine. Although no data are available on its effectiveness in treating severe monkeypox, VIC and cidofovir may be used to control monkeypox outbreaks. 59
The spread of monkeypox, once a rare zoonotic disease, raises concerns in an era when there may be lowered smallpox herd immunity owing to cessation of immunization posteradication. Monkeypox can pose a threat if transmission becomes widespread, combined with changes in virulence. 60 It continues to be treated as a biosafety level 3 infectious agent. 61 In this context, prevention becomes an important factor. Although prior vaccinia immunization lowers disease burden in monkeypox, there is a need for newer vaccines targeting monkeypox. Significantly, the modified vaccinia Ankara – JYNNEOSTM has been approved by the FDA for prevention of both smallpox and monkeypox. 62 JYNNEOSTM can be administered subcutaneously and the regimen consists of two doses given 28 days apart. Although its approval has been based on survival studies in nonhuman primates, results on effectiveness, immunogenicity, and safety tests on human subjects are awaited. 63
Monkeypox outbreak 2022 – current state of affairs
Monkeypox is considered to be endemic within the countries of Cameroon, Central African Republic, Democratic Republic of Congo (DRC), Nigeria, and Republic of Congo. Among endemic countries, highest case counts were reported out of DRC – a total of 1284 cases were captured via Integrated Disease Surveillance and Response (IDSR) between January 1 and May 8, 2022. Recently, cases have surfaced among countries that are not considered endemic for monkeypox. As of this writing, the WHO has reported a total of 61,282 confirmed cases of Monkeypox in 97 nonendemic countries, as of September 16, 2022. 64 Most cases have been reported to occur among males aged 20 to 50 years. 65 The first case was reported out of the United Kingdom on May 7, 2022, in a returning traveler from Nigeria. 66
Per preliminary reports, the vast majority of cases were not associated with travel while epidemiological investigations remain ongoing. Zumla et al. underscore an important aspect of this outbreak – a speculation as to whether silent transmission of Monkeypox may have preceded the current cases. Although there has been much flutter around whether a change in viral genetics could be a contributor to transmission beyond endemic regions, the authors note that the Monkeypox virus has a large genome thus conferring stability and efficiency at repairing genetic mutations.67–70 Per the CDC and European Centers for Disease Prevention and Control (ECDC), many cases of the current Monkeypox outbreak were diagnosed among men who self identify as gay, bisexual or men who have sex with men (MSM). There is speculation that disease transmission may have occurred during sexual intercourse and this is a stark feature of the current outbreak. The likelihood of spread among MSMs with multiple sexual partners is considered to be high thus highlighting the need for robust contact tracing programs. 71 Some cases were also reported among people residing in the same household as the infected person. 67 Fortunately, at the time of this publication, no cases of severe disease or death amid the current Monkeypox outbreak have been reported. A summary of monkeypox transmission, disease progression and symptoms is depicted in Figure 3.
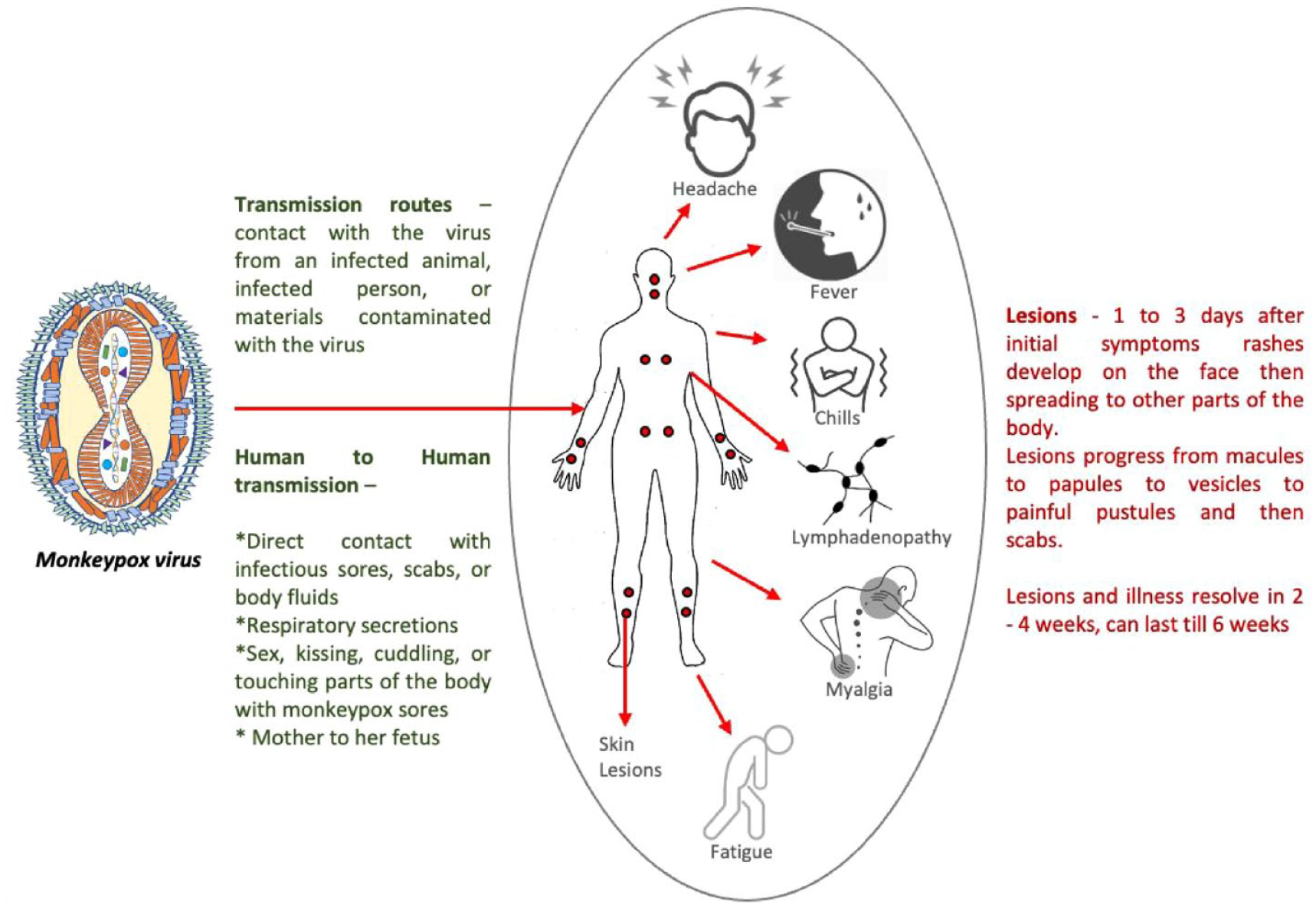
A diagrammatic summary of the transmission, symptoms, and disease progression in monkeypox. 72
Although the risk of spread to the general public has currently been deemed low, the CDC has issued a level 2 travel health notice urging persons traveling to countries where cases have been reported to practice enhanced precautions which includes avoiding contact with ill individuals especially those with dermatological or genital lesions and their belongings, avoiding contact with dead or alive wild animals and avoiding eating or preparing bushmeat. 67 Outbreak containment strategies include clinical education and re-education, early clinical recognition, speedy clinical and laboratory assessment, contact tracing, along with practicing infection control measures. For suspected and confirmed cases of Monkeypox, the CDC recommends standard, contact, droplet, and airborne precautions with patients placed in a negative pressure room if possible. The duration of isolation precautions would vary depending upon the length of time required for all lesions to crust and thereafter fully re-epithelize. Once newly formed skin is noted, isolation precautions may be discontinued. 73 Furthermore, disease containment would also require exposed healthcare workers to be identified and offered protection by immunization. The Advisory Committee on Immunization Practices (ACIP) published recommendations on the use of ACAM2000 and JYNNEOSTM for preexposure prophylaxis among those at risk of occupational exposure and this includes laboratory personnel handling Orthopoxviruses, designated response team members at the discretion of public health authorities and healthcare workers who care for patients with Orthopoxviral diseases and/or administer ACAM2000, the live Vaccinia vaccination. Boosters are recommended for those who have a continuing risk of occupational exposure. The recommended booster frequency for those working with more virulent Orthopoxvirus, that is, Variola virus and Monkeypox virus is every 2 years if using the JYNNEOSTM or every 3 years if using ACAM2000. Those working with less virulent Orthopoxviruses such as Vaccinia virus or Cowpox virus should seek boosters at least every 10 years. Notably, ACAM2000 is contraindicated among immunocompromised persons, patients with atopic dermatitis or another exfoliative dermatological condition, persons with an allergy to a vaccination component, pregnant or breastfeeding women and in those with underlying cardiac conditions such as coronary artery disease or cardiomyopathy. 74
Genus Parapoxvirus
Genus Parapox comprises species Orf virus, Pseudocowpox virus, Bovine papular stomatitis virus, Red deerpox virus, and Gray sealpox virus. Parapoxviruses can result in zoonotic skin infections among humans while the primary host is usually sheep, goat, cattle, camels, reindeer, caribou, chamois, musk-ox, and red deer among terrestrial animals; and seals or sea lions among animals with a predominantly aquatic habitat. Human infections are most often an occupational hazard among animal handlers including shepherds, farmers, slaughterhouse workers and veterinarians but can also occur as a recreational health hazard among hunters or after petting infected animals.75,76 Structurally, parapoxviruses are oblong, ovoid, or round in shape with lengths between 220 and 300 nm and widths reported between 140 and 170 nm. Their genome has been described to be one of the smallest among Poxviridae and is about 140 kbp of linear dsDNA.76,77 They have also been described to have a ‘ball-of-yarn’ appearance on electron microscopy. 78 Their replicative life cycle is similar to Orthopoxviruses and occurs within cellular cytoplasm.
Herein we discuss a clinically noteworthy Parapoxviral infectious disease; namely Orf.
Orf
Pathogenesis, Transmission, and Clinical Manifestations
Orf virus is a Parapoxvirus and can result in a zoonosis called ‘Orf’, also known as contagious ecthyma or contagious pustular dermatitis. Sheep and goats are natural hosts and suffer lesions in their mouth, around the lips or muzzle. Human transmission may occur via direct inoculation when broken or ulcerated skin, the portal of entry, comes in contact with an infected animal harboring the virus or via contaminated equipment. 79
Human pursuits that may place one at risk include, rearing sheep and goats (including bottle feeding and grooming), petting infected animals and having contact with contaminated material such as an animal harness. Animal bites and contact with recently vaccinated animals could also result in Orf since the most widely used veterinary Orf virus vaccines contain live, attenuated virus.80,81
Human to human transmission is not known to occur. Clinical disease may begin after an incubation period of 3 to 5 days as an erythematous macule progressing to form papules which may have a target-like appearance. The lesions further progress to form nodular or vesicular lesions which often ulcerate around the 2- to 3-week mark, also known as ‘acute stage’. This is usually followed by the appearance of a regenerative papilloma and lesion regression with self resolution, the entire process taking about 4 to 6 weeks of time. Orf lesions heal without any scarring. Stages of Orf were best described by Leavell et al. 82 and are summarized in Table 2. Rarely, immunocompromised hosts may develop substantial sized lesions, also called ‘giant orf’. Constitutional symptoms such as malaise and lymphadenopathy are uncommon as is disseminated disease. Veterinary Orf lesions are considered infectious even after crust formation and a crust that falls off may have potential to transmit the virus. The virus is believed to be resistant to heat and drying and is reported to persist on environmental surfaces for extended periods of time thereby contributing to potential downstream transmission. 82 Lesions are considered noninfectious after the epithelium has completely healed and regenerated itself.
Clinical stages of Orf, originally described by Leavell et al.

Diagnosis and Management
Diagnosis is largely clinical based on physical examination along with a suggestive exposure history. Diagnostic technologies that may be employed are polymerase chain reaction (PCR), negative stain transmission microscopy, viral culture and Parapoxviral serologies; of these, PCR and serology are the most widely used. Samples often need to be sent to reference or public health laboratories requesting Orf virus or Parapoxvirus detection. Most lesions self heal over 4 to 6 weeks’ time and do not need pharmacological therapies. Topical cidofovir has shown success when used for Orf treatment in an immunocompromised host, a renal transplant recipient. The regimen described by Geerinck et al. 83 encompassed 1% cidofovir cream administered topically to the site of the lesion over five cycles of 5 days of cream application followed by 5 days of no treatment. Notably, there was recurrence two months after lesion resolution which was successfully managed with two more courses of topical cidofovir in this case.
Erbağci et al. describe four cases of Orf treated successfully with topical imiquimod including one case of giant Orf. Imiquimod topical application two times a day resulted in clinical success among the described cases with duration of therapy varying from 5 to 18 days (average time to resolution ~ 8 days). 84 Randomized clinical trials for both pharmacological therapies for Orf, namely topical cidofovir and imiquimod are lacking. Prior literature has also described a case where surgical excision was performed for an Orf lesion with clinical success. 82
The exact duration of immunity derived from Orf virus infection among humans remains elusive, but there is literature to support a rapid cell mediated immune response followed by an antibody response after initial infection. 85 Notably, Yirrell et al. found that smallpox vaccination did not confer protection against Orf.
Genus Yatapoxviruses
Genus Yatapoxvirus comprises two viral species, namely, Tanapox virus and Yaba monkey tumor virus. Tanapox virus and Yaba like disease virus are considered different strains of species Tanapox virus (TPV). 86 Yatapoxviruses akin to other poxviridae are large, brick-shaped, enveloped dsDNA viruses and replicate in host cellular cytoplasm, while their genomes are among the smallest. Monkeys and other primates are believed to be natural hosts and may serve as a viral reservoir. 86 Yatapoxviral infections in humans are thought to be zoonoses that could spread during periods of ecological imbalance such as floods or via insect bites.
Tanapox was originally described by Downie et al. in 1972. It was noted among tribal people living in the Tana river valley in Kenya. The observations dated back to 1957 and 1962 and were reported as a short febrile illness along with other prodromal symptoms and a skin rash. 87 Since then, a few reports of Tanapox have emerged among returning travelers from the republic of Congo and Tanzania.88,89 A case series also describes numerous cases among residents of Zaire. 90
Yaba monkey tumor virus (YMTV) infections in humans proliferate slowly, involving mesodermal cells, from histiocytomas to large subcutaneous masses, comprised of polygonal mononuclear cells, considered benign tumors. The lesions became apparent as nodules after 7 to 10 days, while becoming raised tumors peaking in size after 3 to 6 weeks and regressing only after 2 to 3 months. 86 Whereas, TPV produces rapidly resolving dermal lesions, localized to the extremities, with little evidence of vesiculation. 86 Lesions begin as a papule and grow into raised, umbilicated vesicles that do not become pustular, while histopathological examination shows pronounced dermal hyperplasia and cytoplasmic eosinophilic inclusions, typical of poxviral infection. 86 The TPV lesions appear at 3 to 4 days and peak at 1 to 2 weeks, while the lesions regress after 4 to 6 weeks. TPV infection usually occurs as a single lesion and seldom more than 2, a characteristic quite different from Monkeypox or other poxviral infections even though the nodules appear similar.86,91 A widespread TPV illness observed in at least 50 individuals, across ages and sex, suggested the disease occurred during flooding and subsequently increased mosquito activity. In addition, the lesions were restricted to the exposed region of skin, confirming that mosquitoes may be the transmitting vector. 91 In subsequent studies, transmission of disease by viral inoculation was observed only in monkeys, while the viral strain could only be grown in human and monkey cell lines. These studies suggested that monkeys may be the reservoir species, 91 although the natural host of Yatapoxviruses are still uncertain. 86
Importantly, YMTV and TPV, while similar morphologically to other members of Poxviridae, are immunologically distinct. Vaccination or infection from vaccinia or variola or monkeypox does not confer immunity against infection from YMTV or TPV and is the same conversely too.86,91 Interestingly, YMTV and TPV cause different diseases but are immunologically related and have a similar host range. 86 Although natural YMTV infections have not been observed, TPV generally causes self limiting disease. 91 The Yatapoxviruses, an interesting genus, are not regarded as a major human health threat, and are a poorly researched group of viruses that require further investigation including prevention and therapeutic options. YMTV infection in humans can be controlled by following stringent biological safety protocols when handling monkeys, while TPV infection control is associated with mosquito control measures. 91 Another potentially valuable perspective to research Yatapoxviruses comes from the understanding of their slow replication cycle and sustained antigen expression. This can be helpful in developing recombinant vaccines or vectors for cancer gene therapy.86,92
Genus Molluscipoxvirus
Genus Molluscipoxvirus comprises a single viral species namely Molluscum contagiosum virus (MCV). MCV is oval shaped and contains linear dsDNA, approximately 190 kbp. 93 MCV replicates in cellular cytoplasm like its other poxviral counterparts. Human disease has been vastly reported, although some veterinary literature has described Molluscum contagiosum in horses.94,95
Though broadly regarded as a pediatric ailment, Molluscum contagiosum may affect adults. Those at risk include children between the ages of 1 to 10 years, persons with atopic dermatitis, immunocompromised persons, people residing in humid climates or crowded living circumstances. Transmission can occur by direct contact or via fomites such as shared clothing, linen, towels, or toys. Although a high frequency of swimming pool attendance was significantly more frequent among patients with Molluscum contagiosum, a definite causal association was not proven in the study by Castilla et al. 96 Per Choong et al., 97 only swimming in a school pool was significantly associated with Molluscum transmission along with sharing bath sponges and bath towels with an infected person. Autoinoculation is a distinct possibility due to itching or picking at skin lesions. Shaving may also result in the spread of lesions across the body.
Clinical disease tends to be mild and self limiting in otherwise healthy hosts but can manifest as disseminated disease among immunocompromised persons. Molluscum is a dermal disease and manifests as small, raised, pearly white, pink or flesh-colored papules with central umbilication. These are the characteristic lesions of the disease also known as ‘Mollusca’ and can vary in size from 2 to 5 mm. Lesions may appear on any part of the body including face, trunk and extremities. Typically, lesions do not occur on palms and soles. Self resolution timelines vary between patients but most cases resolve in 6 to 12 months without scarring. For cases that require treatment, options include physical and chemical treatments. Physical treatment modalities that may be employed include cryotherapy, curettage and laser application. Chemical treatment could be oral therapy with cimetidine and may sometimes be preferred in pediatric patients who fear physical lesion excision. Although there are reports of clinical success with this regimen, conflicting reports cite the inefficacy of oral cimetidine for children with Molluscum who did not have atopic dermatitis.98–100
Topical treatments include 0.7% cantharidin application and 0.5% podophyllotoxin cream application twice daily for 3 days, which was reported to be more efficacious than the 0.3% strength.101,102 Other topical options include 5% Imiquimod cream, iodine and salicylic acid, potassium hydroxide and tretinoin. 103 Among those with human immunodeficiency virus (HIV) and Molluscum contagiosum, antiretroviral therapy and the consequent improvement of CD4 cell counts are highly encouraged in addition to physical remedies. Clinical success with the use of intralesional interferon injection among patients with HIV and Molluscum was reported by Nelson et al. 104 Interferon alpha intralesional injections were reported to have clinical success among two pediatric patients with combined immunodeficiency. 105
Application of poxviruses in emerging cancer therapies
Poxviruses possess strong virulence, harnessing the host-cell machinery to benefit its replication and transmission forward. These properties can be tuned for human benefit by utilizing them as oncolytic vectors. By definition, the FDA suggests an engineered recombinant therapeutic agent is one that – ‘mediate their effects by transcription and/or translation of transferred genetic material and/or by integrating into the host genome and that are administered as nucleic acids, viruses, or genetically engineered microorganisms’. 106 The poxviruses with large genomes have the capability to incorporate significant foreign DNA and deliver it to the host cell. 107 Zoonotic poxviruses like vaccinia have been studied extensively to manage disease, and it has been noted that during the replication phase (which happens completely in host cytoplasm) of the viral genome, it does not get incorporated into host DNA and no splicing of viral transcripts occurs. 108 Hence the vaccinia and myxoma viruses, among others, have been extensively studied to deliver therapeutic transgenes or manipulate the host cancer cell, as vectors.
Although initially these agents were envisioned and studied to selectively infect cancer cells and destroy them by cytolytic activity, more recently their use has pivoted toward remodeling host immune response toward cancer cells. They can also successfully alter the tumor microenvironment, particularly to facilitate the cytotoxic T-cell response over cancerous cells. 109 Synergistically, these poxviral vectors can also be complemented to transfer transgenes, prodrug activators, cytokines and chemokines, apoptosis inducers, and so on.110–112 Furthermore, the addition of luciferase, sodium iodide symporters113,114 have been beneficially delivered through these vectors. By far, the Vaccinia virus has been studied the most as an oncolytic vector candidate, and multiple candidates have been advanced into phase I/II clinical trials. 115 In the study by Cripe et al., the viral vector has been engineered to insert the human granulocyte-monocyte colony stimulating factor into the thymidine kinase (TK) gene. A second study uses modification in the vector for insertion of β-galactosidase into the viral TK gene, and the insertion of β-glucuronidase into the viral hemagglutinin open reading frame (ORF). 116 In addition the virotherapy may complement and benefit immunotherapy, chemotherapy and radiotherapy.117–119 More recently, the Myxoma virus, a Leporipoxvirus, has also gained prominence as an alternative to vaccinia vector therapy, in preclinical studies, of particular interest within aging populations where efficacy concerns with the Vaccinia arise. 120 Myxoma is antigenically similar to vaccinia and has high tropism for European rabbits. 121 In preclinical models, it has shown significant promise, particularly in difficult to treat cancers like pancreatic, medulloblastoma, and gliomas.119,122,123
Due to the high complexity of tumorigenesis, cancer therapy must target oncogene abnormalities, tumor suppressor gene abnormalities, immune evasion, tumor microenvironment, and much more. Poxviruses have been long studied, have individual species tropism, and have robust replication proficiency and progress with nonincorporation into the host genome, making them promising oncolytic virotherapy agents, with a strong potential to aid cancer therapy. 124
Conclusions
Poxviridae have been longstanding pathogens causing human disease. They have substantial potential in shaping the course of modern medicine. The current monkeypox outbreak in otherwise nonendemic regions illustrates human vulnerability owing to loss of cross-protective immunity derived from smallpox disease and vaccination in a posteradication era. Understanding poxviruses gives us critical insights toward their molecular mechanisms, replication, epidemiology, and ecology. This information can help us develop robust strategies to manage these viral diseases, prevent future outbreaks, and utilize certain poxviruses in oncolytic therapies, gene and vaccine delivery applications.
