Abstract
Monkeypox is an emerging zoonosis that has been declared a public health threat due to its spread outside of endemic areas. We present two cases of monkeypox: one involving a 39-year-old male patient living with HIV on antiretroviral therapy with detectable viraemia and a low CD4 count, and another involving a 44-year-old male, negative for HIV, who visited our Diagnostic and Consultative Unit with a new-onset skin rash. Both individuals identify as men who have sex with men and had travelled abroad before the disease onset. One patient was hospitalised, while the other was treated as an outpatient with follow-up care. Both cases were managed conservatively due to the unavailability of tecovirimat in Bulgaria. We emphasise the importance of recognising and diagnosing monkeypox and the significance of HIV screening, as monkeypox could be considered an AIDS-defining condition in people living with HIV. Maintaining a high level of suspicion for new cases is essential, as instances of monkeypox continue to be reported in non-endemic countries.
Introduction
Monkeypox (mpox), caused by the monkeypox virus (MPXV), an emerging zoonosis initially endemic in Africa, has spread to non-endemic regions since 2022 and was declared a global public health threat.1,2 Between 23 June 2022 and 26 June 2023, a total of six cases of human mpox were confirmed in Bulgaria. 3 Based on strong clinical suspicion, two of these patients sought medical attention at our Diagnostic and Consultative Unit (DCU) and were confirmed positive for MPXV.
Case 1
A 39-year-old male was newly diagnosed with HIV in March 2022 and was admitted to the Acquired Immune Deficiency Unit at the time. Initially, he was diagnosed during an HIV screening before an elective cosmetic procedure. Two consecutive fourth-generation HIV tests were performed and were reactive. A confirmatory Western blot was obtained to support the diagnosis. The patient had a medical history of syphilis for which he had received treatment with depot penicillin G. He identified as a man who has sex with men. There was no family history of note, and he was not on any regular medication, nor did he use recreational drugs. He was diagnosed with AIDS stage C3, presenting with oropharyngeal candidiasis, HIV-wasting syndrome and HIV-associated thrombocytopenia and anaemia. Laboratory workup revealed a CD4+ cell count of 118 cells per mm3 and CD8+ count of 752 cells per mm3 with a CD4:CD8 ratio of 0.17. HIV viral load was 196,449 copies/µl. The comprehensive metabolic panel and liver function tests were unremarkable. Hepatitis B and C assays tested negative.
During his stay at the clinic, he remained afebrile, with an adequate response to the antifungal therapy, with no other HIV-related conditions. Antiretroviral treatment with doravirine/lamivudine/tenofovir disoproxil fumarate was initiated on 30 March 2022, resulting in good immunological and virological responses, as evidenced by an increase in CD4+ cell count and a sharp drop in viral load before the patient’s discharge (Figure 1). The patient was subject to monthly outpatient follow-up, during which he admitted to therapy non-adherence due to work-related travel and social activities, which is reflected in subsequent immunological and virological test results (Figure 1).

Cell counts prior to and after antiretroviral therapy (ART) initiation. A logarithmic scale of the viral load is superimposed with the respective absolute values (copies per μl). The dashed line and the red mark designate the approximate time of MPXV infection.
Five months following admission, on 9 September 2022, he attended our DCU with a new-onset rash he initially noticed on his palms. Lesions later appeared on the trunk, face, genitals, perianal area and lower extremities (Figure 2). Further history revealed recent travel abroad, where he engaged in unprotected sexual intercourse on several occasions but had no knowledge of contact with symptomatic individuals. He denied feeling ill, having a fever or experiencing malaise at any given moment but was bothered by pruritus.
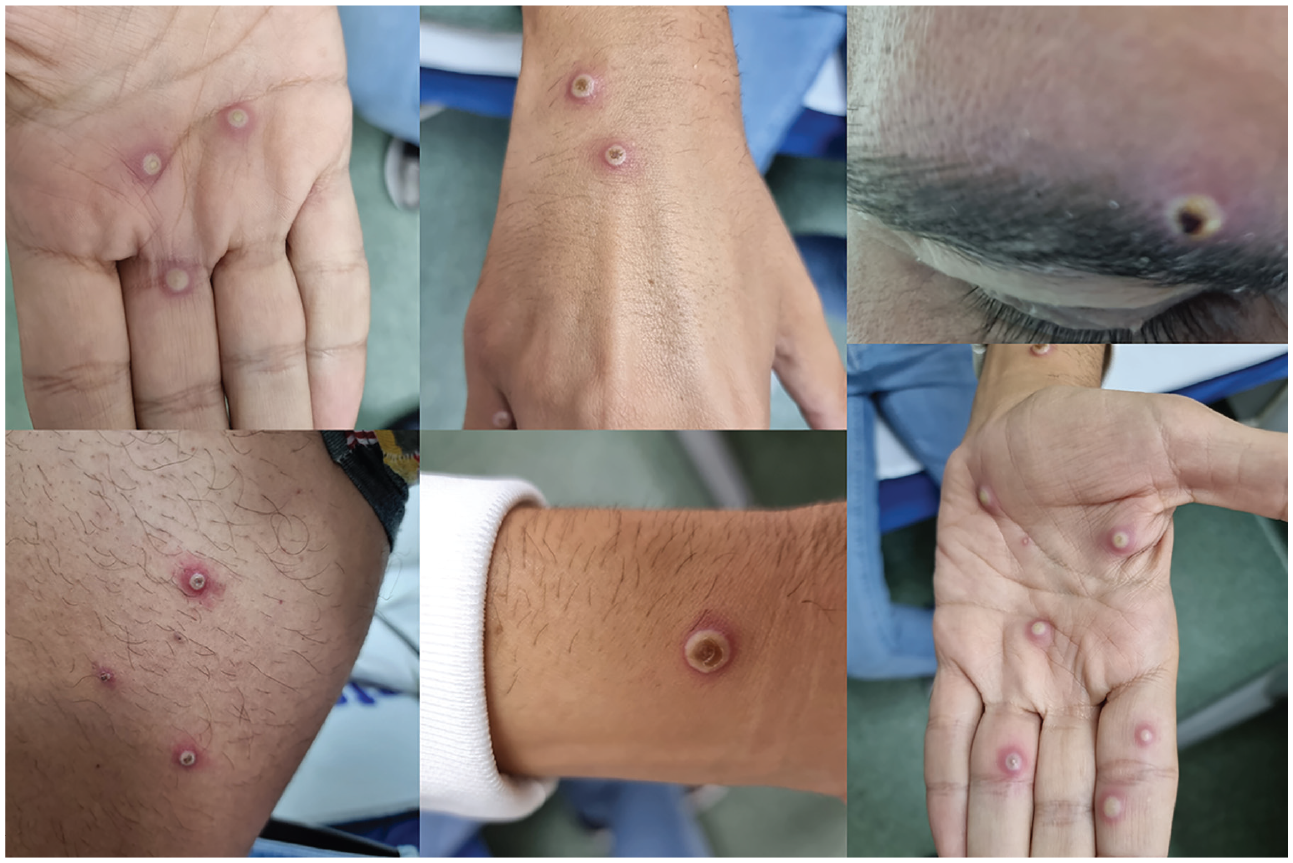
Multiple well-circumscribed pustules are visible on the patient’s face, upper and lower extremities. Some of the pustules appear dented in the centre.
Upon examination, the patient appeared non-toxic, well-oriented and apyrexial with notable firm, well-circumscribed umbilicated pustular skin lesions measuring 0.5–1 cm in diameter, with minimal anogenital involvement. The total number was slightly over 25 lesions. Submandibular, cervical, occipital and inguinal lymphadenopathy was also noteworthy. The rest of the general physical examination revealed no abnormal findings or signs of an impending health emergency.
Nasopharyngeal, oropharyngeal and lesion swabs were obtained for virological assays. The diagnosis was confirmed by qPCR targeting the rpo18 gene and the G2R (TNFR) gene, with all samples testing positive. The patient was managed as an outpatient with symptomatic treatment with loratadine and topical cream for pruritus relief and recovered fully within 2 weeks. Treatment with tecovirimat was considered in view of the sustained virological non-suppression and poor CD4 count recovery, but the antiviral was not available in Bulgaria.
Case 2
A 44-year-old man with unknown HIV serostatus, who identified as a man who has sex with men and had a history of sexually transmitted infections, was admitted to the hospital with an acute febrile illness and skin vesicles. Symptoms of fever (40.0°C), fatigue, and myalgia began 4 days before hospitalisation. The onset of the flu-like symptoms was followed by a pruritic vesicular rash on the head and genitalia, along with swollen lymph nodes. The patient reported having unprotected sexual intercourse during a 1-month stay in the United Kingdom.
The patient was conscious, oriented, febrile, pale and dehydrated upon examination. He presented with fewer than 10 eruptions on the face, scalp and inguinal area, mainly in the stage of vesicles, with some crusted lesions and a single painful, large, crusted eruption with a haemorrhagic centre on the chin (Figure 3). Papules or pustules were absent. Bilateral cervical lymphadenopathy was observed. The tongue and buccal mucosa were normal, besides the signs of dehydration. Abdominal examination revealed slight tenderness in the upper quadrants, with no masses or organomegaly. No abnormalities were noted during the examination of the respiratory, cardiovascular or neurological systems. Laboratory workups showed no significant abnormalities. Considering a potential epidemiological link to cases in the United Kingdom, nasopharyngeal, oropharyngeal and vesicle swabs were sent for analysis and detection of MPXV DNA. Screening tests for HIV and hepatitis B were negative. He received intravenous isotonic fluids, antipyretics, analgesics, antihistamines and a brief course of acyclovir, which was discontinued after a positive PCR confirmed mpox diagnosis. No new eruptions developed during the hospital stay and all other lesions became crusted. After clinical improvement, the patient was discharged for isolation at home until complete recovery.

A ruptured vesicular rash, with some lesions showing ulceration.
Discussion
Since the declaration of mpox as a global health emergency, our understanding of the disease has been challenged, especially concerning transmission and clinical features. Sexual transmission has not been definitively confirmed, but it seems likely. In Bulgaria, MPXV DNA was detected in two seminal fluid samples, supporting the possibility of sexual transmission. 4 Nonetheless, cases with no known sexual or other established exposure have been reported. 5 Unlike previous monkeypox outbreaks, current cases may present with no prodrome, atypical symptoms and aberrant infections.5–7 Notably, one of our patients presented with no viral prodrome or respiratory symptoms despite detecting MPXV DNA in the nasopharyngeal and oropharyngeal swabs, while the other presented with a vesicular rash requiring а broader differential diagnosis. Interestingly, findings from large global case series postulate that severe mpox, such as fulminant or necrotising forms, should be considered an AIDS-defining condition in people living with HIV with CD4 count less than 200 cells per mm3. 11 In contrast, the simultaneous acquisition of acute HIV infection and mpox has already been documented on a few occasions.8–10 It has been well-established that local genital factors, such as inflammation and the loss of protective anatomical barriers, can facilitate HIV transmission.8,9 Therefore, it is reasonable to expect that MPXV lesions may also facilitate HIV transmission. 8 This further emphasises the significance of HIV testing in mpox cases.
A high pretreatment HIV viral load may preclude viral load suppression and lead to virological failure. In our case, a baseline drug resistance test with susceptibility confirmation was performed before therapy initiation. Moreover, a substantial decrease in the viral load was achieved in the first month, accompanied by a CD4+ cell count rise. Presumably, non-adherence, resulting in uncontrolled viraemia, contributed to the development of a more disseminated rash compared to the patient who was negative for HIV. A CD4 cell count below 100 per mm3 and a higher viral load are associated with a greater risk of complications, severe disease course and a fatal outcome. 11
Therefore, local and global management protocols for risk assessment and aetiological treatment have been implemented.12,13 Tecovirimat is the only antiviral approved for mpox treatment by the European Medicines Agency. 14 Despite concerns over the safety of coadministration of ART with tecovirimat and other antivirals known to have activity against poxviruses, current recommendations do not advise against it if therapy is indicated, given the unlikely occurrence of clinically significant adverse interactions.12,13,15 When there is a clinical suspicion of mpox in people living with HIV, treatment with tecovirimat is actively encouraged, especially for those with low CD4 counts or uncontrolled viraemia.12,16 However, it should be noted that tecovirimat was unavailable in Bulgaria, so we opted for conservative medical treatment with close follow-up.
Even though WHO declared that mpox no longer represents a public health emergency of international concern, new cases continue to be reported in non-endemic countries.3,17 Bulgaria has not reported the highest MPXV rates, yet a high index of clinical suspicion among patients in risk groups should be maintained for imported cases in Southeast Europe.
Conclusions
Mpox has rapidly and unprecedentedly spread to non-endemic countries worldwide. The few mpox cases we observed exhibited a relatively mild disease progression, including in a person living with HIV with unsatisfactory immunological reconstitution. However, HIV infection may contribute to a more pronounced clinical presentation and adverse outcomes. Furthermore, mpox should be in the differential diagnosis of vesicular exanthems in non-endemic countries. Testing for HIV in mpox cases is crucial as simultaneous transmission is possible and mpox could also be considered an AIDS-defining condition when accompanied by a low CD4 count. Finally, our data indicate that while Bulgaria has had a low incidence of mpox cases, the possibility of new cases cannot be dismissed, highlighting the importance of raising awareness among medical professionals in the country, Southeast Europe and other non-endemic countries with low incidence.
Footnotes
Acknowledgements
The authors would like to express their gratitude to associate professors Stefka Krumova and Lyubomira Glomb at the Department of Virology, National Centre of Infectious and Parasitic Diseases, as well as to the institute’s Director, professor Iva Christova, for their invaluable assistance in the virological diagnosis.
Author contributions
D.I. and N.Y. acquired and synthesised information about the clinical cases and the discussion part; D.I. wrote the original draft and edited the article; N.Y. and T.V. were involved in reviewing and finalizing the article; D.I. designed and edited the graphical content.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval and informed consent
This report did not require an ethical board approval because it did not contain any human or animal trials. Written informed consent was obtained from the patients for the publication of these case reports and the accompanying images.
