Abstract
Background:
Acute uncomplicated cystitis is common among outpatients and frequently leads to antibiotic prescriptions, making urinary tract infections (UTIs) an important area for antimicrobial stewardship initiatives. Infectious Disease Society of America (IDSA) guidelines promote alternative agents in place of fluoroquinolones for acute uncomplicated cystitis. Despite IDSA guidance, adherence to the guideline remains low in the United States (US). Several studies have described interventions to improve guideline-concordant prescribing for UTIs. However, the long-term sustainability and impact of fluoroquinolone (FLQ)-sparing strategies on community antimicrobial resistance and treatment outcomes are unknown. The objectives of this study were to characterize current antibiotic prescribing patterns, treatment failures and Escherichia coli resistance rates in a setting which instituted FLQ sparing strategies for UTIs in 2007.
Methods:
Retrospective cohort study of women aged ⩾ 18 diagnosed with acute uncomplicated cystitis based on International Classification of Diseases, 10th Revision (ICD-10) codes were included. Data were abstracted for ambulatory visits over a 6-month period, January 2018 to June 2018, at a large urban health care system. Treatment decisions were made by individual providers, and data were analyzed retrospectively. Nitrofurantoin (NFT) resistance was obtained from the institutional antibiogram and patient-level data. Treatment failure was defined as the need for a different antibiotic for UTI within 28 days of the original prescription.
Results:
NFT was the most frequently prescribed antibiotic (n = 386, 71.6%) of empiric antibiotic prescriptions for UTIs. FLQs comprised 4.6% of all antibiotic prescriptions (n = 25). Treatment failure rate was 2.3% in patients treated with NFT. Urine culture was ordered for only 26.8% of patients. Among the small group of patients with cultures ordered, E. coli remained 98.5% susceptible to NFT.
Conclusions:
This study is the first to report significantly low rates (4.6%) of FLQ prescribing for acute uncomplicated cystitis. Treatment failure rate was low with empiric NFT. Increased NFT resistance among E. coli was not observed at the institution or among the subset of patients with E. coli positive urine cultures. These findings support current IDSA treatment guidance for uncomplicated cystitis.
Keywords
Background
Acute uncomplicated cystitis, otherwise known as urinary tract infection (UTI), is a common reason for antibiotic prescription in the outpatient setting. 1 This likely stems from the fact that UTIs are one of the most common bacterial infections overall, affecting 50–60% of adult women at least once in their lifetime. 2 It has been estimated that there are 10.5 million office visits for UTI symptoms and 2–3 million emergency department visits annually in the United States (US).3–5 Given the high prevalence and patient burden of UTIs, it is essential to optimize antibiotic use for this condition. In addition, The Joint Commission (TJC) recently implemented new standards for outpatient antibiotic stewardship (AS) which will bring renewed focus on prescribing practices for UTIs. 6
Current Infectious Disease Society of America (IDSA) guidelines recommend nitrofurantoin (NFT), fosfomycin, and trimethoprim-sulfamethoxazole (in areas with <20% resistance rates) as first line agents for UTIs. Pivmecillinam is also recommended, where available. 7 Despite these guidelines, evidence to date shows that fluoroquinolones (FLQs) are often prescribed for UTIs.8–12 For example, a study in the United States from 2002 to 2011 illustrated that 49% of antibiotics prescribed for UTIs in the outpatient setting were FLQs. 8 In addition, a more recent nationwide analysis of outpatient prescription activity for adult ambulatory care visits showed that FLQs were prescribed for 40.3% of uncomplicated UTIs. In this study by Kabbani et al., 9 genitourinary conditions were the most common diagnosis associated with FLQ prescribing, comprising 24.5% of total FLQ prescriptions equating to over four million fluoroquinolone prescriptions over a one-year period. Antibiotic prescribing practices differ in countries outside the United States, with some reporting much higher level of guideline adherence. In Sweden, for example, 84.7% of antibiotic prescriptions for uncomplicated UTI were deemed appropriate. 13 FLQs were prescribed in only 13.8%, and the authors note this is still inadequately high. A large population-based cohort study in the Netherlands showed that nitrofurantoin has been the most frequently prescribed antibiotic for UTIs since 1999. 14 In countries such as Italy, Belgium, Russia, and Brazil, fosfomycin has been found to be the first-choice antibiotic for UTIs. The use of FLQs was still widespread among these countries, with rates ranging from 9.6% to 24.6%. 15 High levels of FLQ prescriptions for treatment of UTIs have correlated with increased incidence of FLQ resistance among enteric organisms responsible for UTIs. 16
Given the concordance with IDSA guidelines is overall low in the United States, several studies have described interventions to improve guideline-concordant prescribing for UTIs. This includes a study at our institution, Denver Health (DH), which is an urban, integrated public healthcare system. In 2007, the institutional guidelines at DH were revised to advise providers to use NFT for acute uncomplicated cystitis and to reserve FLQs for complicated UTIs. Resistance of E. coli to trimethoprim-sulfamethoxazole in the DH system is >20%. The change in guidance involved revision and dissemination of an existing local clinical care guideline. The dissemination was accomplished via an e-mail to providers regarding the recommended change and posting of the guideline on the hospital intranet. After the change in prescribing guidance, there was an immediate 26% (95% CI [20, 32]) decrease in FLQ use (p < 0.001), and a nonsignificant 6% (95% CI [−2, 15]) increase in nitrofurantoin use (p = 0.12). 17 Details of the AS intervention and results directly following the intervention were published previously. More recently, Funaro et al. described an AS intervention involving education and feedback for UTIs. The study was a two-phase, prospective, quasi-experimental study to estimate the effect of an outpatient AS intervention on guideline-concordant antibiotic prescribing in a primary care and urgent care setting. Follow-up period was 3 months after the intervention was completed. 18
Despite these prior studies, the sustainability of AS interventions for outpatient antibiotic UTI prescribing has not been determined. In addition, the long-term impact of FLQ-sparing strategies for acute uncomplicated cystitis on community antimicrobial resistance and treatment outcomes are unknown. In this study, we sought to provide an updated assessment of UTI prescribing practices at DH. This study was a follow up to the prior AS intervention, with a goal to provide a current snapshot of empiric treatment patterns for acute uncomplicated cystitis and determine if the previously noticed change was sustained. The primary aims were to characterize empiric antibiotic prescribing patterns and treatment failures associated with UTIs in ambulatory patients. The secondary aim was to evaluate the effect of FLQ-sparing strategies on NFT resistance rates among E. coli.
Methods
Study design
In this retrospective cohort study, diagnostic workup and treatment were not standardized. Management decisions were made by treating providers based on their individual clinical judgment. Data from patients’ charts were abstracted electronically and analyzed from the time of their initial UTI diagnosis and through the end of the study period.
Study population
The study population consisted of ambulatory patients aged > 18 years, diagnosed with an uncomplicated UTI at DH during the study period. Patients who were seen at an outpatient clinic, at an urgent care or emergency department (ED) were included. Patients seen in the ED setting were discharged directly from the ED and not admitted to the hospital. The ED visits consist of patients seen at an adult and pediatric ED which occasionally sees patients 18 years and older. Men, pregnant women, patients age > 65 years, patients with diabetes (DM), and patients with pyelonephritis (ICD 10 code N10) were excluded as they would qualify as a complicated UTI. The study population was chosen to correlate with the focus of the IDSA guidelines on premenopausal, nonpregnant women with no known urological abnormalities or comorbidities.
Case identification
Cases were identified from the DH data warehouse. The DH data warehouse is a secure repository of accumulated data from DH’s electronic medical record system. Cases were included for analysis when a visit was coded with the International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10) codes for acute cystitis. Three ICD-10 codes were included: Cystitis NOS (N39.0), Acute cystitis with (N30.01), and Acute cystitis without hematuria (N30.00). Patients diagnosed with UTI based on these ICD 10 codes were included. Data including their demographics, clinical data, antibiotic prescriptions, and diagnostic testing were abstracted from the DH data warehouse.
Study period
Data were abstracted for a 6-month period, from January 2018 to June 2018. For patients with multiple visits, only the first instance was included. Radiology, laboratory, and operating room visits were excluded.
Data validation
Manual chart review was conducted on a random sample of 10% of patients to confirm diagnosis and treatment information. The random sample was chosen by reviewing every 10th patient in the data set.
Antibiotic prescriptions
Antibiotics were chosen at the discretion of the provider given this was a retrospective chart review. Antibiotics prescribed at the time of the UTI visit were included in the analysis. This included antibiotics prescribed with an indication for UTI at time of visit and up to 10 days after the visit. Antifungals or metronidazole were not included. Encounters without antibiotics prescribed within 10 days were excluded from data analysis for antibiotic prescribing. Antibiotic percentages were expressed as the number of prescriptions of a single antibiotic or antibiotic class over the total number of antibiotic prescriptions in either the ED/urgent care, outpatient category, or total study population, depending on the description provided.
Clinical outcomes
As in previous studies, we used the need for a second course of antibiotics as a proxy measure of treatment effectiveness.18–20 For this study, we defined treatment failure as need for a second course of a different antibiotic within 28 days after the date of the initial antibiotic prescription. Treatment failure rate was expressed as the number of treatment failures divided by the total number of prescriptions of that specific antibiotic or antibiotic class. ED and outpatient data were combined to calculate failure rate due to low number of failure rates overall. Antibiotics prescribed in response to urine culture results were not considered treatment failures as this is consistent with routine care. In addition, an extension of the primary antibiotic was not considered a failure, as duration of therapy was not evaluated in this study.
Urine cultures and nitrofurantoin resistance
Patients in the study population who had a urine culture ordered at their provider’s discretion during the study period were reviewed from the abstracted data. Data on the urine culture results, including organism and susceptibility data, were included. In addition, E. coli susceptibility information was obtained from the DH antibiogram to capture the institution wide trend. The DH antibiogram is generated from organization-wide microbiology data, which includes hospital wards, the intensive care units (ICUs), ED, urgent care, and outpatient clinics. The microbiology lab follows the Clinical and Laboratory Standards Institute (CLSI) guidelines for E. coli isolation and susceptibility testing. The Microscan Walkaway plus system (Beckman Coulter) is utilized for susceptibility testing, in accordance with CLSI M100-S25, published in 2015. 21 Kirby Bauer testing is used for confirmation when indicated. Nitrofurantoin MIC thresholds did not change during the study period.
Analysis
Excel (2016) Microsoft, Redmond, WA and Tableu (2021.1) Salesforce, Seattle, WA, were used in data analysis. Chi-square test was used to calculate p-value for comparisons.
Results
Cohort description
Of 891 visits selected based on ICD-10 diagnosis codes, there were 772 unique patients seen for acute cystitis in the 6-month period from January to June 2018. Patients with DM (n = 117), Radiology (n = 29), OR (n = 1), and Lab (n = 22) visits were excluded. Therefore, 623 patient visits for acute cystitis were eligible for analysis. A manual chart review of 63 (10%) patients was performed to validate diagnosis, treatment, and outcome data electronically abstracted from the electronic medical record (EMR) abstraction. See Figure 1. There were no discrepancies found in the data between the chart review and EMR abstraction data. Baseline characteristics are shown in Table 1.

Study flowchart depicting the number of patients included in electronic medical record abstraction, chart review, and analysis.
Demographic and clinical characteristics.

Baseline characteristics of patients diagnosed with acute cystitis in the ED, urgent care, and outpatient setting. ED, emergency department; ICD, International Classification of Diseases; SD, Standard deviation; UTI, urinary tract infection.
Antibiotic prescribing
A total of 539 visits resulted in empiric antibiotic prescription for UTI at the time of visit. 364 of 380 (96%) patients in the ED or urgent care were prescribed an antibiotic for UTI, compared with 175 of 243 (72%) patients in the outpatient setting who were prescribed an antibiotic for UTI at the time of visit. Of encounters with antibiotics prescribed, NFT was the most frequently prescribed antibiotic among both outpatient and ED/urgent care visits (n = 386, 72% of total empiric antibiotic prescriptions for UTIs). NFT was prescribed for 74.3% of outpatients versus 70.3% of ED/urgent care patients. FLQs (levofloxacin and ciprofloxacin) comprised of 4.6% of total antibiotic prescriptions (n = 25). See Figure 2. Comparison of ED/urgent care and outpatient prescribing patterns can be seen in Table 2.
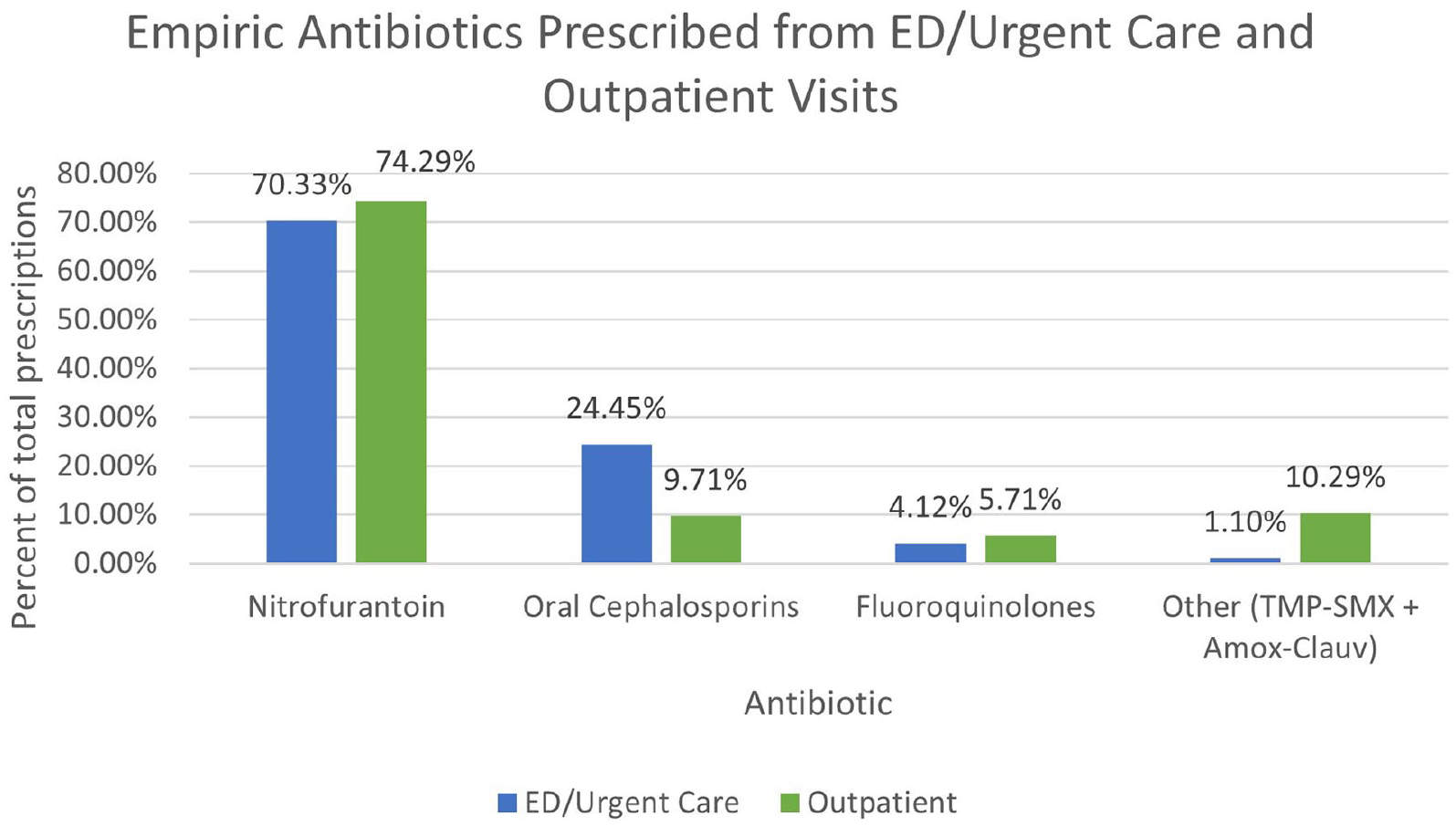
Percent antibiotics prescribed for UTIs for both ED/Urgent Care and Outpatient visits.
Antibiotics prescribed for UTIs for both ED/urgent care and outpatient visits with p-value, calculated by Chi-square test. Trimethoprim–sulfamethoxazole (TMP-SMX). Amoxicillin–clavulanate (AMOX-CLAV). Oral cephalosporins included cefdinir and cephalexin. FLQ included levofloxacin and ciprofloxacin.

ED, emergency department; FLQ, fluoroquinolone; UTI, urinary tract infection.
Outcomes
Of the 386 patients empirically prescribed NFT for UTI, the treatment failure rate was 2.3% (n = 9), that is, nine patients were prescribed a second antibiotic for UTI in the subsequent 28 days. The most common second empiric antibiotic prescribed was cefdinir. Only one of these nine patients had a urine culture performed as part of their initial routine care, which grew E. coli (susceptible to nitrofurantoin). Therefore, the reason for a second antibiotic was based on clinical characteristics, not on urine culture results in these nine cases. The treatment failure rate for oral cephalosporins, trimethoprim–sulfamethoxazole, and fluoroquinolones was 1%, 3.85%, and 0%, respectively. Amoxicillin–clavulanate failure rate was not analyzed due to low numbers of prescriptions (n = 1).
Urine culture results and nitrofurantoin resistance among E. coli
A urine culture was ordered among 167 patients as part of their routine care (26.8%). This included 88 (23.2%) ED/urgent care patients and 79 outpatients (32.5%). Urine culture changed management in 19 patients (3.0%). E. coli was the most common isolate among both groups. There were 95 E. coli isolates total in the study population, of which 93 were susceptible to NFT (98%). The remaining two isolates were intermediate to NFT. During the calendar year that encompassed the study period, E. coli remained 98% susceptible to nitrofurantoin at DH (Figure 3).
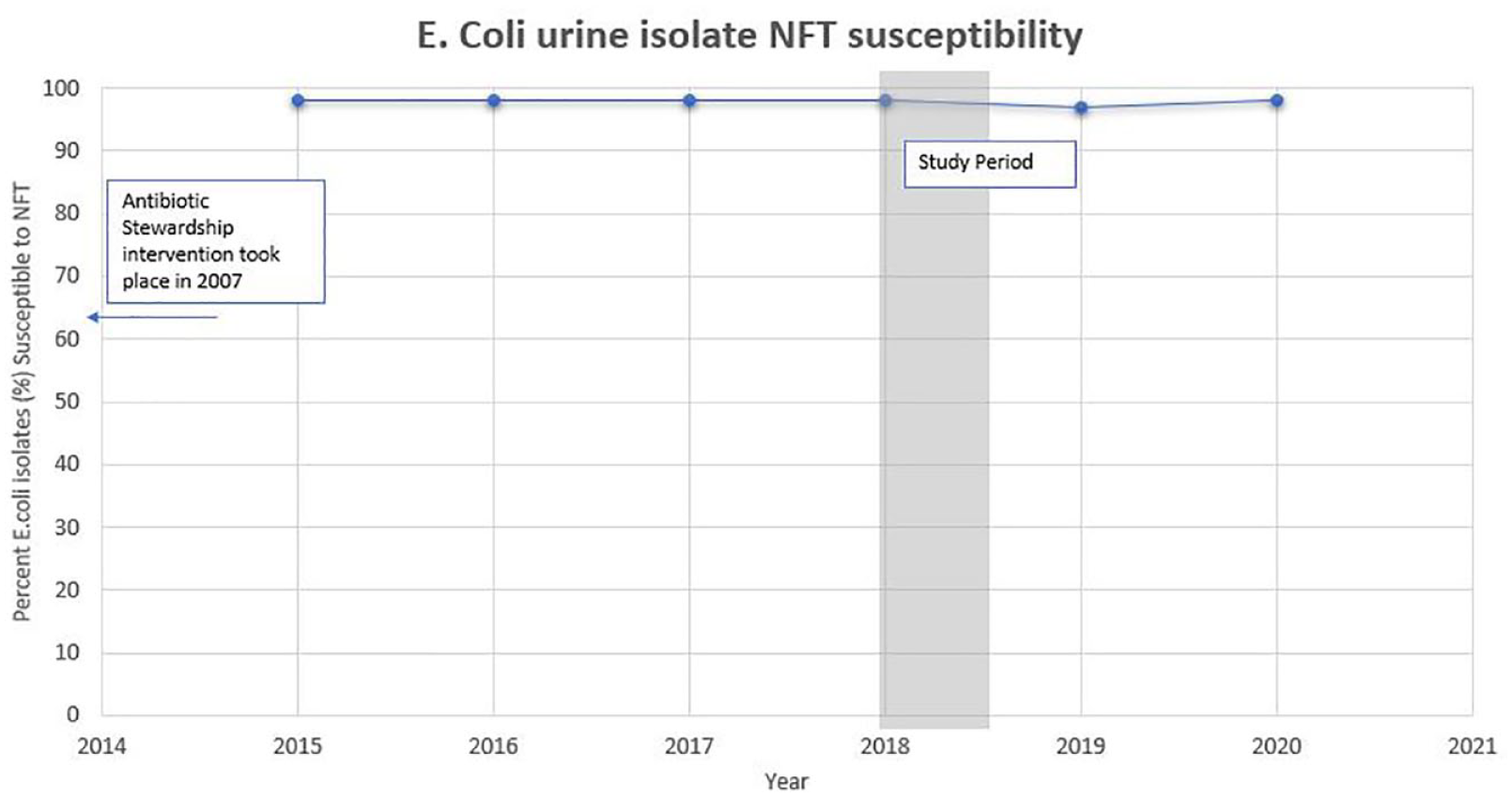
Percent susceptible E. coli over the last 5 years based on the institutional antibiogram. Only urine isolates were included for analysis in the antibiogram.
Discussion
This study is the first to report patient outcomes and effect on NFT resistance in a US health system with high provider adherence to IDSA guidelines for treatment of UTIs. Eleven years after the AS committee changed the recommended treatment for acute uncomplicated cystitis from FLQs to NFT, our results show there has been a sustained increase in guideline-concordant antibiotic prescriptions for UTIs. Based on prior published rates and the current study results, over the past approximately 6 years, NFT use has increased from 28.1% to 70.2% among ambulatory patients at DH. Over that same period, FLQ prescribing has decreased from 34.9% to 4.6%. 17 In the interim period, the AS guidelines for UTI were not changed but were included in a web application made freely available to providers. This may account for the additional increase in guideline-concordant prescribing; however, prescription data in the interim period are not available. Treatment failure rates remain low, with only 2.3% of patients prescribed NFT requiring a second, different antibiotic within 28 days. This treatment failure rate is slightly lower than previously published studies, which has shown that between 4% and 16% of patients will return for further treatment within 28 days.19,20,22 The previous studies analyzed different patient populations which may account for the difference in findings. Our outcomes show that high empiric NFT use is not associated with need for frequent change in therapy. At the same time, there has been no significant change in the NFT resistance among E. coli isolates at the institutional level or among the subset of patients with urine culture ordered. Nitrofurantoin resistance rates remained low among E. coli (2%). There were no treatment failures among patients treated with FLQs, however this is likely due to small numbers in our study population (n = 33).
These results are biologically plausible, as previous in vitro studies have shown that E. coli does not easily acquire resistance to NFT use, as is the case with FLQs. This may be because NFT resistance confers a reduction in fitness in E. coli. In vitro studies have shown that even in the presence of therapeutic levels of NFT, resistant mutants do not grow adequately. 23
Overall, the majority (n = 73.2%) of UTIs were treated with empirical antibiotic prescriptions alone and urine culture was not ordered to guide therapy. The rate of patients with urine cultures ordered seems low; however, this likely reflects the study population of relatively young, healthy women. It is a common practice among providers to empirically treat women reporting typical UTI signs and symptoms without further testing. This is similar to findings by Pujades-Rodriguez et al., 22 which found that 85.7% of patients diagnosed with lower UTI were prescribed same day antibiotics with urine sampling undertaken in 25%. However, in the era of increasing antibiotic resistance, encouraging confirmatory testing could be an opportunity for further AS, as was previously done in the Netherlands. 24
It is also interesting that higher guideline-concordant prescribing has been reported in countries other than the United States. In the Netherlands, a large population-based study showed that NFT was the most frequently prescribed antibiotic for UTIs. They reported antibiotic use in prescriptions per 1000 person years (PY). In their study of women seen at primary care clinics, nitrofuran derivatives increased from 52 prescriptions per 1000 PY in 2008 to 98 prescriptions per 1000 PY in 2014. The Netherlands study did not report patient outcomes or resistance patterns. However, it is encouraging that high NFT use is sustainable in another setting. 14 In an assessment of UTI treatment in Switzerland, fosfomycin was prescribed in 44.7% of cases, with TMP/SMX in 25.8% and nitrofurantoin in 14.5%. This resulted in guideline conforming empiric antibiotic therapy in 84.7%. 13 Fosfomycin was also found to be the most frequently prescribed antibiotic for UTI in a multi-country study including Italy, Belgium, Russia, and Brazil (REWIND study). The second most common antibiotic choice differed between countries, with Belgium reporting the highest NFT and NFT derivative use, prescribed in 29.9% of patients. However, in the Italian cohort, NFT was rarely prescribed (<2% of patients treated with antibiotics). The REWIND study differs from the current study in that it did not focus on uncomplicated cystitis and had a more heterogeneous population. Differences in national and European guidelines, provider perceptions and age level may contribute to the differing prescribing practices between the United States, European, and Other countries.15,24
There was high guideline-concordant prescribing across all ambulatory settings in our study; however, each setting had slight variations in findings. Interestingly, there was no statistically significant difference in NFT prescribing rates between the ED/urgent care and outpatient setting [n = 256 (70.3%) versus 130 (74.3); p = 0.3402], indicating that adherence to IDSA guidelines is feasible even among more acute patients seen in ED/urgent care settings. There was a statistically significant difference in oral cephalosporin prescribing [n = 89 (24.5) in ED/Urgent Care versus 17 (9.7%) in outpatient settings (p ⩽ 0.0001)], indicating that providers are preferentially choosing cephalosporins more frequently in the ED/urgent care setting. Perhaps this is also due to the more acute patient population seen in the ED/urgent care setting. This is also consistent with IDSA guidelines, which suggests cephalosporins as a second-line treatment after NFT. However, these cases were not analyzed to see if the patient met criteria for NFT treatment. In contrast, TMP-SMX and AMOX-CLAV were more commonly prescribed in the outpatient setting [n = 4 (1.1) in ED/Urgent Care vs18 (10.3) in outpatients p < 0.0001], perhaps due to more provider variation in the outpatient setting.
It is important to note that fluoroquinolone use in general may have also been affected by new US Food and Drug Administration (FDA) guidance over the study period. In July 2016, the FDA advised that serious adverse effects have been associated with fluoroquinolone use and the risks typically outweigh the benefits for patients with uncomplicated UTIs who have alternative treatment options. 25 This warning took place 2 years prior to the study period and may have also impacted the low observed rate of fluoroquinolone use.
The preponderance of prior research in this area demonstrates that antibiotic prescribing practices for UTIs in the United States are not in concordance with IDSA guidelines, 26 and perhaps providers continue to remain wary of using narrow spectrum first-line agents for their patients. The new TJC AS requirements for ambulatory care settings that routinely prescribe antimicrobials will bring renewed focus on AS for ambulatory care. 6 UTIs represent an important target for outpatient AS programs. Our study is the first to describe high NFT use and sustained guideline-concordant prescribing for UTIs many years after an AS intervention within a large health care system. The results illustrate that policy has the ability to impact practice and adherence to available guidelines is achievable over the long term. It is our hope that this research bolsters confidence in, and adherence to, IDSA guidelines for treatment of acute cystitis among ambulatory patients. The results of this study support development of local antibiotic stewardship guidelines promoting FLQ-sparing strategies for treatment of UTIs.
Limitations
This was a retrospective study so is observational in nature. Cases were identified retrospectively using ICD-10 codes and coding may not always be accurate. The population was limited to diagnosis of acute cystitis and did not focus on pyelonephritis or catheter associated UTIs where resistance may be more common. The data may not have captured antibiotic prescriptions if the ICD-10 codes for acute cystitis were not used. Duration of therapy or dose was not studied. A urine culture was ordered in only 30% of the study population, thus the exact E. coli resistance rates for the whole study population is unknown necessitating the use of institutional antibiogram data. In addition, these data are from a single institution and may not be generalizable to other organizations.
Future directions
Future directions should focus on implementing antibiotic stewardship initiatives and improving adherence to IDSA guidelines among outpatients at other institutions. Duration of therapy was not evaluated in this study and may represent an additional target for antibiotic stewardship and future studies.
Footnotes
Acknowledgements
The authors thank David Kon for his assistance using Tableau software and Bryan Knepper with his help in data acquisition. We also thank Laura Triplett for her management of the clinical microbiology laboratory and consultation in preparing this manuscript.
