Abstract
Background:
Pulmonary mycoses are important diseases of the respiratory tract caused by pulmonary fungal pathogens. These pathogens are responsible for significant morbidity and mortality rates worldwide; however, less attention has been paid to them. In this study we determined the prevalence of pulmonary fungal pathogens among individuals with clinical features of pulmonary tuberculosis at Mbarara Regional Referral Hospital.
Method:
This was a hospital based cross sectional survey. Sputum samples were collected from each study participant. For each sample, the following tests were performed: Sabouraud dextrose agar for fungal culture, GeneXpert for
Results:
Out of 113 study participants, 80 (70.7%) had pulmonary fungal pathogens whilst those with pulmonary tuberculosis numbered five (4.4%).
Conclusion:
Our findings highlight the diversity of neglected pulmonary fungal pathogens whose known medical importance in causing pulmonary mycoses cannot be overemphasised. Therefore this presents a need for routine diagnosis for pulmonary mycoses among TB suspects and set-up of antimicrobial profile for pulmonary fungal isolates to support clinical management of these cases.
Keywords
Introduction
Pulmonary tuberculosis (PTB) remains a fundamental cause of sub-acute and chronic pulmonary diseases and thus a major cause of mortality attributed mainly to
Materials and methods
Study design
This was a hospital based cross sectional study. The study samples were processed from the Mycology Laboratory, Department of Microbiology, Mbarara University of Science and Technology (MUST), whose clinical Mycology Laboratory serves Mbarara Regional Referral Hospital (MRRH) patients.
Study duration
The study was carried out from November 2019 to June 2020.
Sample size
Sputum samples were collected from 113 clinically PTB suspected patients referred to the TB clinic of MRRH.
Inclusion criteria of study participants
Patients with signs and symptoms of PTB who visited the TB unit who required MTB GeneXpert diagnostic test were included.
The study included TB suspects with a negative TB diagnostic history and those who were not on antifungals.
Signed informed consent was sought from each study participant prior to sputum sample collection. As for children, written informed assent was obtained from their care givers.
Exclusion criteria
TB suspects who failed to provide sputum samples and whose request forms lacked demographics were excluded from this study.
Specimen collection
First morning sputum sample for MTB GeneXpert and fungal culture was collected aseptically from study participants in a sterile dry wide-necked, leak-proof universal container from each of study participant suspected for PTB.
Transport and storage of samples
Samples were transported in courier boxes from point of collection to the laboratory. Samples which were not processed within the same day of collection were stored in a refrigerator at 4–8°C awaiting processing
Specimen processing
Collected sputum samples were processed at the TB and Mycology Laboratory Unit at Microbiology Department, Faculty of Medicine, MUST. Demographic data and HIV status of each study participant were recorded.
GeneXpert MTB/ Rifampicin (RIF) assay
Sample reagent was added to sputum sample in a ratio of 2:1 ml and the sputum sample was allowed to liquefy for 15 min. A sterile dropper was used to transfer 2 ml of mixture into the GeneXpert cartridge, which was loaded into the GeneXpert machine for MTB detection for 2 h. The results were reported as ‘MTB detected’ or ‘MTB not detected’.
Potassium hydroxide (KOH) mounts
A drop of 10% KOH was placed on a clean glass slide using Pasteur pipette. A small portion of sputum was added into the KOH drop using a sterile wire loop and mixed well. The coverslip was placed on top of this mixture and the preparation was placed in a moist chamber and kept at room temperature for 30 min. The preparation was examined under low power microscope objectives for the presence or absence of fungal elements.
Gram stain
Gram stain smear was made from the sputum sample to test for the Gram reaction of fungi and also the size, shape and arrangement of fungal elements. For Gram positive yeast-like cells, mucopurulent absence or presence of pseudo hyphae was recorded.
Fungal culture
Sabouraud dextrose agar (SDA) containing antibiotic chloramphenicol and gentamicin was used to culture sputum samples. The specimens were streaked onto the medium in the Universal bottles with a sterile inoculating loop in order to obtain isolated colonies. The preparations were then incubated at 25–30°C in an inverted position (agar side up). Cultures were examined at least weekly for fungal growth and held for 4 weeks before being reported as negative. After sufficient incubation, considering colony morphology, texture, rate of growth, surface of the colony and pigmentation on the surface and reverse of the colony on SDA tubes were recorded. The significant fungal isolates on culture were identified to the species level, using standard mycological procedures.
Lactophenol cotton blue staining
Lactophenol cotton blue was used for microscopic identification and characterisation of fruiting bodies such AS conidia, sporangia, rhizoids and hypha or mycelia of cultivated fungi on SDA. A drop of lactophenol cotton blue stain was placed on a clean grease-free glass slide. A small fragment of cottony, woolly or powdery colony was picked from the midpoint of the culture using a sterile straight wire and placed on a clean glass slide for the staining process. A clean coverslip was applied avoiding air bubbles. Excess stain was removed with blotting paper and the preparation examined using ×10 and ×40 objectives of the microscope. Fungal element features such as microconidia, macroconidia, chlamydospores and hyphae with spiral, pertinate and antler-like structures were investigated. These features seen on the stained slide were compared with established characteristic fungal features using mycology atlases.
Germ tube test
Germ tube test is a simple, reliable procedure for the identification of
Safety and environment
All biological specimens, including used cartridges, capable of transmitting infectious agents were treated with universal precautions.
All laboratory procedures were done in a level 2 TB laboratory.
Personal protective equipment such as disposable gloves, laboratory coats were used when handling specimens and reagents.
Washing of hands was done thoroughly after handling specimens and test reagents.
Disposing of used Xpert MTB/RIF cartridges was done according to the country’s safety guidelines for hazardous material.
Quality control measures
Well collected sputum samples were accepted for analysis.
Known standard fungal element morphologies and known fungal atlases were used to confirm established characteristic fungal features. 12
Laboratory reference cultured fungal elements grown in the laboratory were also used.
Results
Demographic distribution, PTB, pulmonary fungal pathogens (PFPs) and PTB–fungal co-existence profiles
Of 113 sputum samples collected, 4.4% (
Demographic distribution, pulmonary tuberculosis (PTB), pulmonary fungal pathogens and PTB–fungal co-existence.

KOH, potassium hydroxide; PFP, pulmonary fungal pathogen.
Prevalence of PTB and PFP by HIV status
Following the HIV status of our study participants obtained through their medical history, the majority of the participants were HIV positive [70.8% (
Prevalence of pulmonary tuberculosis and pulmonary fungal pathogens by HIV status.

KOH, potassium hydroxide; PFP, pulmonary fungal pathogen; PTB, pulmonary tuberculosis.
Aetiological profile of pulmonary fungal infections and PTB–PFP co-existence
Categorically, fungal pathogens isolated were dominated by filamentous fungi [58.8% (
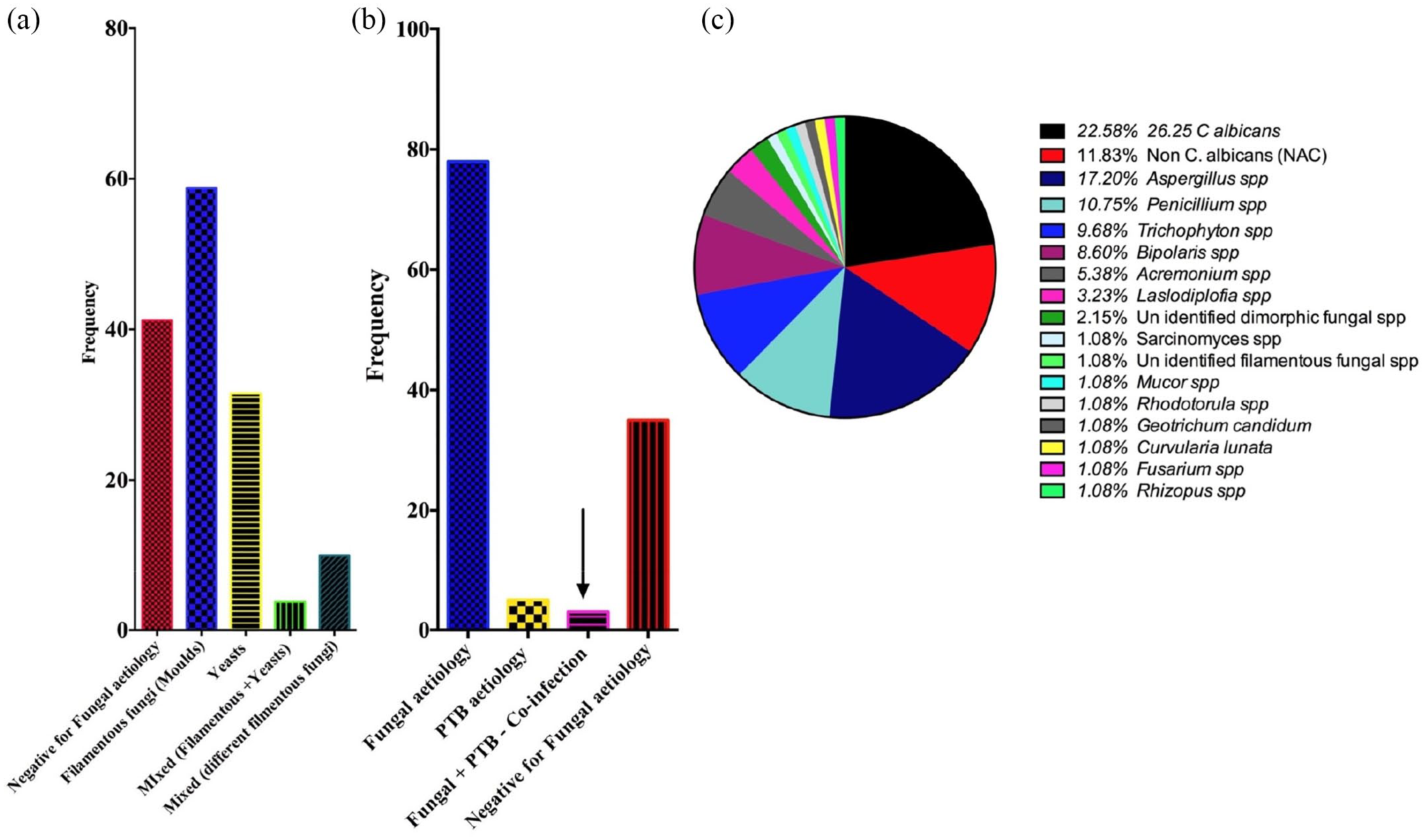
Aetiological profile of pulmonary fungal infections and pulmonary tuberculosis–pulmonary fungal pathogen co-infections.
Co-infection or co-existence by fungal pathogens
Through this study, we were able to establish what seemed like a co-infection or simply a co-existence by more than one organism in an infection niche in about 15.9% (
Aetiology of co-infections or co-existence by age, gender and HIV status.


Aetiology of mixed and co-infections.
Discussion
For some time now PTB has been established as a major opportunistic pulmonary disease, especially in the HIV/AIDS infected cohort, with about two million people dying and one million infected worldwide each year.
13
However, in many parts of the world endemic pulmonary mycoses, particularly the deep seated ones, are also prevalent and, just like TB, are also responsible for high rates of morbidity and mortality in an array of patient populations’.14,15–17 The challenge has always been that in addition to the fact that both entities present with similar symptoms, patients with a history of suffering from TB have proven prone to certain opportunistic fungal infections such as
This brief background highlights the medical importance of both PTB and pulmonary mycoses. Yet the role of opportunistic fungi as secondary invaders of lungs, kidneys and other organs of patients having underlying conditions such as HIV/AIDS and cancer is documented in the literature with a considerable variation of rate of incidence of 9–80%.
19
In Uganda, though, there is a general paucity of the burden of pulmonary mycoses. As a result, these infections have remained a silent challenge to public health due to the fact that they have been either neglected, ignored, missed or misdiagnosed.10,20,21 The spectrum of pulmonary fungal aetiology has been evolving over the years from the commonly isolated
In this study, the prevalence of fungal pathogens using culture on SDA as our diagnostic test was 80 (70.7%) which dominated the prevalence of MTB in patients only suspected for MTB [5 (4.4%)]. The low TB prevalence here is interesting to know and it would have been important to establish whether some of these patients that presented with fungal pathogens had a history of TB treatment. The prevalence of pulmonary fungal pathogens according to gender was higher in females at 62.0% (
In this study,
On the other hand it is not uncommon for fungi to co-exist with other microbes in the same infection niche. In this study, we also sought to establish the prevalence of PTB, PFP and fungal–PTB co-infections among the PTB suspects by HIV status. We established a prevalence of 57 (71.3%) for PFP, three (60.0%) for MTB in HIV positive patients and 18 (22.5%) for PFP in HIV negative patients and zero (0.0%) for MTB in HIV negative patients. On the other hand, two (100%) HIV positive patients were co-infected with both PFP and MTB (Table 2). These results prove the fact that HIV/AIDs is a major predisposing factor to both MTB and many of the saprophytic and environmental fungal opportunists. There is also existing evidence that individuals treated for PTB are prone to fungal infection. 18
In the wider environment, fungi co-exist and interact with similar or other microbes to form different kinds of relationships including the highly addictive mutualistic or endosymbiotic interactions, whilst others can be of antagonistic nature. However, the impact of such interactions in an infection niche on clinical outcomes remains unclear. Through this study, we were able to establish what seemed like a mixed, a co-infection or simply a co-existence by more than one organism in an infection niche in about 15.9% (
Already shown by prior studies, our findings continue to highlight the medical importance of pulmonary fungal pathogens among patients suspected for TB. Most importantly, the aetiological diversity determined in this study reveals other neglected pulmonary fungal pathogens. Therefore this presents a need for routine diagnosis for pulmonary mycoses among TB suspects and set-up of antimicrobial profile for pulmonary fungal isolates to support clinical management of these cases.
Footnotes
Acknowledgements
We are grateful to the study participants and staff Mbarara University Microbiology department for all the support given to us during data collection.
Author contributions
IKN, MP and AM contributed in study conception and design, KK and JM collected data and participated in laboratory analysis, JT and EN carried out data cleaning, LA and BM carried out data analysis, IKN, BA and HI wrote the first draft of the manuscript while JK and TK reviewed the manuscript and JB supervised the whole research process.
Availability of data and materials
Data and materials are readily available from the corresponding author upon request.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics statement
Ethical approval to conduct the study was obtained from Mbarara University of Science and Technology Research and Ethics Committee (REC) (Ref. MUREC 1/7). Upon ethical clearance, administrative clearance was sought from Mbarara Regional Referral Hospital to conduct the study from the hospital and written informed consent was sought from each participant. As for children, their written informed assent was obtained through their care givers. All participants who were found with fungal infections and/or pulmonary tuberculosis were referred to clinicians for further management.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
