Abstract

Introduction
The frequency of invasive fungal diseases (IFDs) continues to increase, producing significant morbidity and mortality in parallel with advances in medical care that tend to weaken host defense mechanisms.1,2 Unfortunately, the current episode of coronavirus 2019 (COVID-19) has just exposed the deficiencies of our health systems, particularly in Africa. Even though the African region was the least affected by this pandemic, the few serious cases confronted constituted a real challenge due to a lack of infrastructure and means of care. The health systems in Africa are generally fragile and are not prepared to face such epidemics. The number of qualified human resources, the availability of biomedical materials and drugs are the challenges of African countries. 3
Globally, we are witnessing the emergence of pathologies requiring prolonged treatment during which patients receive broad-spectrum antibiotic therapy and are subject to numerous invasive procedures such as the presence of a central venous catheter, which may promote direct vascular access of Candida spp. following previous colonization. 4 These conditions, even temporary, can provoke the development of IFDs. This is the case with AIDS, cancers, especially blood cancers such as leukemia, hemodialysis, etc. Indeed, fungi are responsible for more than a billion cutaneous infections, more than 100 million mucous infections, 10 million serious allergies, and more than a million deaths each year. Global mortality due to fungal diseases (FDs) is higher than for malaria and breast cancer and is comparable to that from tuberculosis (TB) and HIV. These statistics show that FDs are a threat to human health and a major burden on healthcare budgets around the world. 5
Antifungals – molecules used to treat FDs – fall into two categories. On the one hand, local antifungals or topicals are not absorbed orally, they are used locally for a local effect. On the other hand, systemic antifungals are absorbed orally or administered intravenously; they diffuse in the viscera. 6 The latter group is divided into four classes: polyenes (amphotericin), azoles (fluconazole, itraconazole, voriconazole, posaconazole, isavuconazole), echinocandins (caspofungin, anidulafungin, micafungin), and nucleoside analogs such as 5-fluorocytosine (flucytosine).7,8 For pharmacokinetic reasons, molecules such as griseofulvin (naturally occurring extract from Penicillium griseofulvum) or terbinafine (allylamines) are absorbed orally but are concentrated exclusively in the superficial skin layers. Thus, they are compared to topicals concerning therapeutic indications. 6
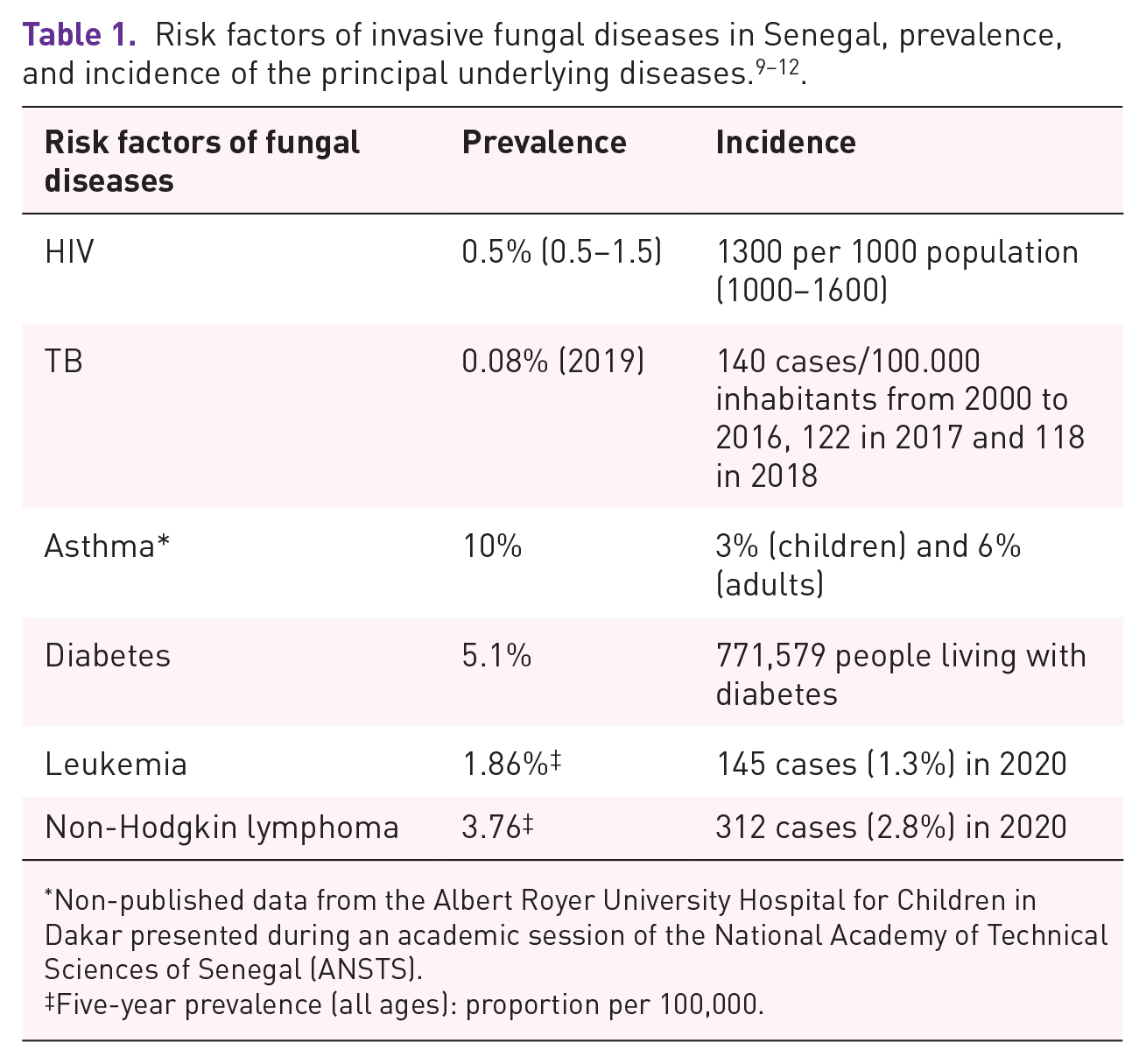
Senegal is hardly an exception to this situation. Our country is also going through a socio-economic transition responsible for a medical transition. Risk factors for IFDs are present across the country; for example, the number of patients with HIV was estimated at 42,000 in 2019 with 63% on antiretroviral treatment. 9 Other risk factors identified include leukemia, diabetes, asthma, and TB9–12 (Table 1). Thus, the Senegalese specialist will be increasingly confronted with the diagnosis and management of FDs. If the current antifungal arsenal in Senegal has molecules efficient for treating superficial FDs with molecules such as griseofulvin or fluconazole, 13 many deep fungal diseases (DFDs) do not yet benefit from effective treatment with the absence or unavailability of antifungal drugs.14,15 Consequently, based on data from Aristide Le Dantec and Fann University Hospital Centers in Dakar, which are the only public health establishments with a medical mycology laboratory, the objective of this communication was to update the table of the main FDs diagnosed in Senegal and the antifungals available for their therapeutic management to draw attention to these infections increasingly diagnosed with the emergence of new opportunistic fungi.
Non-published data from the Albert Royer University Hospital for Children in Dakar presented during an academic session of the National Academy of Technical Sciences of Senegal (ANSTS).
Five-year prevalence (all ages): proportion per 100,000.
Analysis of the burden of fungal diseases in Senegal
Over the past two decades, the main publications mostly found on Pubmed and Google Scholar databases with ‘mycoses’ and ‘Senegal’ as keywords show the principal part of the epidemiology of FDs diagnosed in Dakar (capital of Senegal) which concentrates the only medical mycology laboratories in the country. These publications (Table 2) mainly concern superficial FDs such as tinea capitis,16–18 onychomycoses,19–21 intertrigos,22–24 among others,25–31 and subcutaneous FDs such as mycetomas32–35 and exceptionally chromomycosis. 36 These superficial and subcutaneous FDs generally have an arsenal of antifungals that permit their management even if the latter treatment sometimes remains probabilistic. 37 For DFDs, the reported publications essentially concern histoplasmosis,38,39 especially the African form often treated with disappointment, 15 and cryptococcosis with which alternative treatment regimens have been adopted with a success rate estimated at 36.7%. 40 Rare cases of pneumocystosis 41 and aspergillosis/aspergilloma have been reported, treated by surgery without any adjunction of antifungal for the aspergilloses’ management, possibly due to antifungals unavailability.42–44
Principal fungal diseases (mycoses) diagnosed in Senegal.

This relatively low frequency of DFDs aside from cryptococcosis,45,46 which represented more than half (50.8%) of the requests for diagnosis of DFDs in Fann University Hospital, 47 would be mostly related to the weakness of the technical material and use of little sensitive biological tools of diagnosis. 41 These diagnostic problems are corroborated by data from the infectious diseases department of this same institution. Indeed, according to a study carried out in this latter service on non-TB lower respiratory infections (LRI) in HIV patients between 2011 and 2015, out of the 255 patients with non-TB LRI, only three fungal etiologies (1.2%) were noted, including two cases due to unidentified yeasts. In addition, in 70.9% of cases, no etiology was found, which therefore motivated the administration of an antifungal (fluconazole) in 57.6% of patients and cotrimoxazole at a curative dose in 30.9% 48 for preventing a probable pneumocystosis. These probabilistic treatments could contribute to the emergence of antimicrobial resistance given the already unavailability of antifungals. All the more so since, out of the 477 requests for the diagnosis of pneumocystosis received between 2005 and 2017 by the parasitology and mycology laboratory in Fann University Hospital, which hosts one of the few pneumology services in the public in Senegal, only 28 were positive corresponding to a hospital prevalence of 5.9%. 47 Furthermore, in our microbiology laboratories in general and medical mycology laboratories in particular, the diagnosis is essentially based on conventional methods including direct microscopic examination and culture on Sabouraud dextrose agar (SDA), with identification of causative agents predominantly based on macroscopic and microscopic morphological characteristics. The only one matrix-associated laser desorption and ionization time of flight mass spectrometry (MALDI-TOF MS)-based device accessible in the country is installed in ‘Hôpital Principal’ in Dakar, a hospital that does not specially perform mycological analyses, and accordingly, the database of this device does not include fungal species aside from the classic species of the genus Candida. The same is true for most private laboratories that have the Vitek 2 instrument.
Another problem that could explain this situation is the unequal distribution of the health system in Senegal. Indeed, as human resources, most of the infrastructure of the health system is based in Dakar. Except for health huts and level 2 hospitals, Dakar is the region with the best health infrastructure. Senegal has 11 level-3 hospitals, among them, 10 are in the Dakar region and the other in Touba, Diourbel region (185 km from Dakar). 49 Therefore, to compensate, populaces in remote areas devote themselves to traditional medicine by attending traditional healers which sometimes delays treatment and leads to disastrous consequences. This is the case with patients with mycetoma. These patients are often received at an advanced stage of the disease with surgical excisions or amputation as the only treatment options. This is evidenced by the recent case of pulmonary metastasis of a knee mycetoma observed in Aristide Le Dantec University Hospital. The patient wasted a lot of time having their lesions treated by traditional practitioners at least for 10 years. 50
The low knowledge of IFDs among physicians may also explain this situation. In fact, according to our mycological laboratory experience, almost only dermatologists systematically send requests for mycological analyzes. Apart from them, the other specialists only resort to the mycology laboratory in no-way-out-situations when they have explored all other possibilities. Moreover, a cross-sectional multicenter survey across seven tertiary hospitals in five geopolitical zones of Nigeria between June 2013 and March 2015 showed that only 0.002% (2/1046) of the respondents had a good level of awareness of IFDs. 51
Evidence for unavailability of systemic antifungals in Senegal
In Senegal, there are only three pharmaceutical industries that meet around 10–15% of needs and none of them produce systemic antifungals. 52 According to the distribution circuit of drugs in Senegal (Figure 1), 13 populations can only obtain drugs from two sources: health organizations and pharmacies.
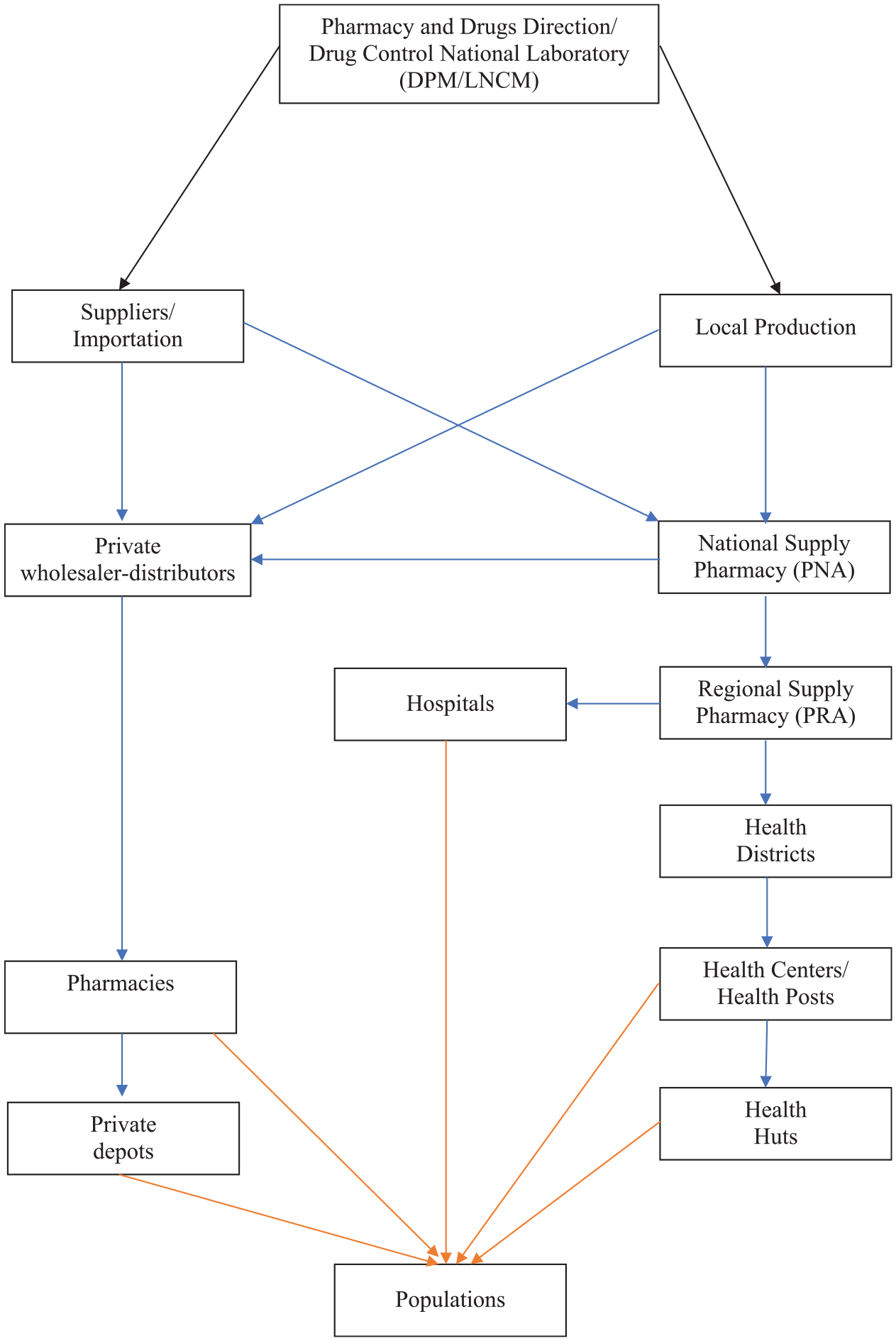
Diagram of the drug distribution circuit in Senegal.
This distribution circuit is also applicable to hospital pharmacies according to an investigation carried out in Aristide Le Dantec University Hospital and Fann University Hospital on the flow of antifungals situation. Indeed, these hospital pharmacies can only obtain antifungals through the national supply pharmacy (NSP). Consequently, in the case of medical prescription of non-available antifungals in health facilities, patients should look for them in pharmacies that may have them in situ or order them via their commercial connections, connections that are only within the reach of a small group.
By compiling the list of essential drugs in Senegal and the list of antifungals available from two of the four drugs wholesaler distributions to pharmacies, antifungals available in Senegal are summarized in Table 3 with their indications. 53
Antifungals available in Senegal with their medicinal forms and indications.

On the list of essential medicines in Senegal but not available from suppliers.
INN, international non-proprietary name.
Thus, the remark is that only fluconazole is available as a systemic antifungal in Senegal, whereas this molecule has no activity on a large number of the fungi responsible for DFDs encountered in Senegal. First, fluconazole does not work against infections caused by Aspergillus spp. 54 Then, regarding candidiasis, while Candida albicans is usually sensitive to antifungals comprising fluconazole, this is not the same for other non-albicans species. Candida krusei exhibits natural resistance to fluconazole while Candida glabrata has variable or even dose-dependent azole susceptibility. 55 As in general, our laboratories only performed the germ tub test to distinguish C. albicans from non-albicans species, 56 the probabilistic use of fluconazole could lead to therapeutic failure or selection of resistant strains, as already seen in immunosuppressed patients on chronic fluconazole prophylaxis. 57
So, our country is lagging far behind, particularly at a time when the country is witnessing the emergence of pathologies such as renal failure and cancer, but also the development of the means implemented for their management such as hemodialysis centers or chemotherapy centers and even the establishment of a national organ donation committee responsible for regulating kidney transplantation. 58 In our sub-region for example, four antifungal agents for IFDs (amphotericin B, fluconazole, itraconazole and voriconazole) were licensed in Nigeria. 51 Even better in north Africa, all classes of systemic antifungals (amphotericin B for polyenes, fluconazole, itraconazole, voriconazole for azoles, caspofungin, micafungin, anidulafungin for echinocandins and flucytosine for nucleoside analogs) are available in Morocco (http://dmp.sante.gov.ma/) and Tunisia (http://www.dpm.tn/).
Conclusions/recommendations
It appears from this analysis that FDs are regularly diagnosed in Senegal. However, DFDs are maybe underestimated probably related to the weakness of the technical platform and consequently the use of insensitive biological diagnostic tools. Also, the few cases of DFDs diagnosed do not benefit from antifungal molecules for specific effective treatment. Therefore, the most recent technical approaches and the most effective therapies must be adopted for rapid diagnosis and better management of DFDs. Likewise, molecular identification techniques should be added to conventional methods. For antifungal availability, a network based on the directory of African pharmacy and drug directorates must be set up and run in collaboration with the national laboratory directorates for harmonization of procedures to reduce morbidity and mortality related to IFDs, especially in Africa. Once again, this will depend at all decision-making levels, on the ability to provide to relevant actors the means to know, recognize, and appropriately treat FDs, especially for DFDs.
Footnotes
Acknowledgements
The authors are grateful to the following drug distribution companies: SODIPHARM and DUOPHARM in Senegal for providing them with their lists of marketed drugs. They are also thankful to hospital pharmacies staff in Aristide Le Dantec University Hospital and Fann University Hospital.
Authors’ contributions
Conception, design of the study, and drafting the article: Khadim Diongue; reviewing the article: Mamadou A Diallo, Mame C Seck, Mouhamadou Ndiaye, Aida S Badiane, Daouda Ndiaye D; final approval of the version to be submitted: all authors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Our study did not require an ethical board approval because it did not contain human or animal trials.
Data availability
Data used to support the findings of this study are included within the article.