Abstract
Lobomycosis or lacaziosis is a chronic subcutaneous fungal infection, caused by the fungus Lacazia loboi, which is phylogenetically related to Coccidioides, Blastomyces, Histoplasma, and Paracoccidioides. The disease was first recognized in 1931 by Jorge Lobo, who found the disease to be a keloidal blastomycosis and named it Jorge Lobo’s disease. This case was perplexing initially as this fungal infection is very uncommon in the USA. However, with the ever-increasing frequency of international travel, many more cases of lobomycosis have been diagnosed in areas of nonendemicity, such as the USA, Europe, and South Africa. The clinical histories of such imported fungal infections often illustrate their long latency periods. In lobomycosis, the onset of the disease is usually insidious and often difficult to document. We describe a case of a New York resident who presented with multiple skin nodules over both his arms and forearms, and was subsequently diagnosed with Jorge Lobo’s disease. The case, diagnosis, histopathologic findings, complication, and management of this rare clinical disease are discussed.
Introduction
Cutaneous lobomycosis is a chronic cutaneous or subcutaneous fungal infection, caused by the fungus Lacazia loboi, which is phylogenetically related to Coccidioides, Blastomyces, Histoplasma, and Paracoccidioides [Al-Daraji, 2008]. The disease was first recognized in 1931 by Jorge Lobo, who identified the organism as resembling Paracoccidioides brasiliensis, the agent that causes South American blastomycosis, and thus named it keloidal blastomycosis or Lobo’s disease [Lupi et al. 2005; Al-Daraji, 2008]. L. loboi can be differentiated from P. brasiliensis by its intracellular obligate nature [Burns et al. 2000].
The natural reservoir of L. loboi remains unknown, but it is believed to have an association with soil, vegetation, and aquatic environments [Lupi et al. 2005; Bermudez et al. 2009]. The majority of cases involve transmission through trauma, insect bite, or inoculation, but there have also been cases in which no trauma is reported [Bermudez et al. 2009]. One such example involved a traveler to Venezuela who walked beneath Angel Falls and transmission of the disease is believed to be due to his exposure to high water pressure [Burns et al. 2000]. The incidence of lobomycosis is higher in men, aged 21–40 years, but this is thought to be due to their closer association with the environment rather than hormonal or genetic factors [Elsayed et al. 2004; Xavier et al. 2008; Bermudez et al. 2009]. In Amazonian tribes, the female role is to cultivate the land, and in these societies, more females are infected with mycoses than men [Xavier et al. 2008].
The disease is endemic in the Amazon (i.e. Brazil, Venezuela, Ecuador, Guyana, Suriname, Bolivia, Colombia, and Peru), but rare cases have occurred in distant parts of the world in people who have never traveled to these endemic areas [Al-Daraji, 2008; Bermudez et al. 2009]. Endemic areas are tropical continental regions that are 200–250 m above sea level, with dense vegetation, annual rainfall <2000 mm, mean temperature of 24℃, and mean relative humidity >75% [Baruzzi et al. 1989]. The occurrence and documentation of the disease in different species of dolphins have expanded the geographic range associated with lobomycosis [Bermudez et al. 2009]. Several cases have been reported of infection in bottlenose dolphins (Tursiops truncatus) and Guiana dolphins (Sotalia fluviatilis) in the waters of places as far as Florida and Texas in the USA, South Brazil, Spain, France, and in the Suriname river estuary [Baruzzi et al. 1989; Burns et al. 2000]. The signs of disease and pathology are similar in both humans and dolphins, and using serology it was found that both species are affected by similar strains of L. loboi [Baruzzi et al. 1989]. To date, only one case of dolphin-to-human transmission has been documented in PubMed [Baruzzi et al. 1989].
Case presentation
A 65-year-old man, residing in New York, USA with a past medical history of hypertension and type II diabetes mellitus presented with chronic skin nodules over his arms and forearms that had been gradually increasing in size for years. The nodules initially appeared a few months after his return to New York from a hunting trip to the Amazon forest in 2007. These lesions had slowly grown into a large keloid-like lesion over his elbow with smaller lesions nearby. The patient could not recall any history of previous trauma, skin injuries, or an insect bite during his trip to Brazil. He did not report any unusual rashes or fever. On initial physical examination, there was a large nodular keloid-like lesion over the extensor surface of the right elbow measuring 3.0 × 2.8 cm. The lesion was nontender. An excisional biopsy of this skin nodule was performed and diagnosis was made as keloidal blastomycosis. Systemic antifungals were started, but the patient reported no improvement of this lesion during a follow-up visit, as well as no other systemic complaints. About 2 years later, the patient developed multiple similar, but smaller nodules, 2–3 cm, on the bilateral forearm with surface discoloration. The patient was sent for surgical excision of the skin nodule and the diagnosis was reconfirmed as keloidal blastomycosis.
The specimen was received in formalin and labeled ‘left arm skin lesion’. It consisted of a skin ellipse measuring 2 × 1.5 × 1.2 cm. The skin surface showed an ill-defined, grayish lesion measuring 1.5 × 1.0 cm. The cut surface was firm and rubbery.
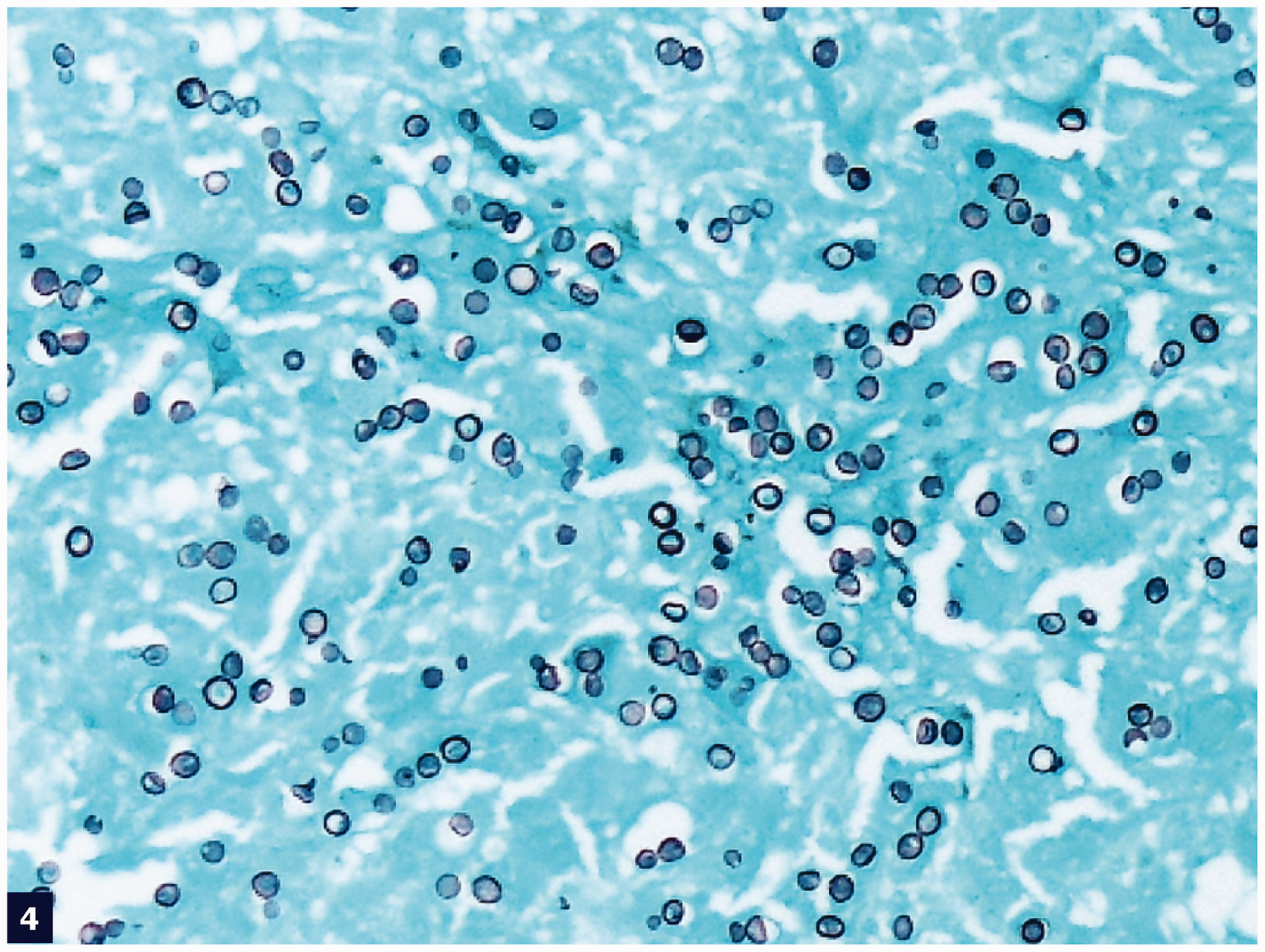
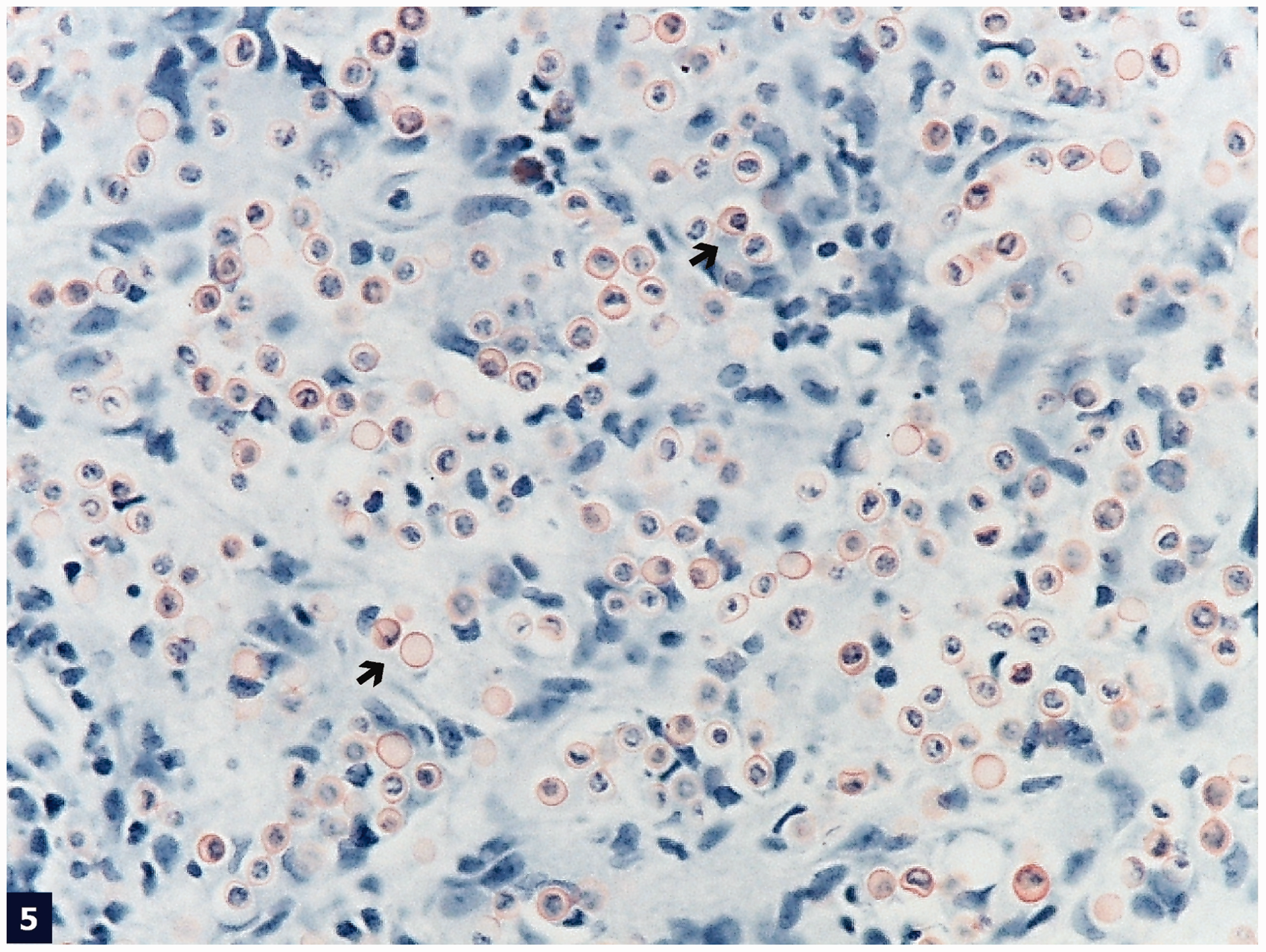
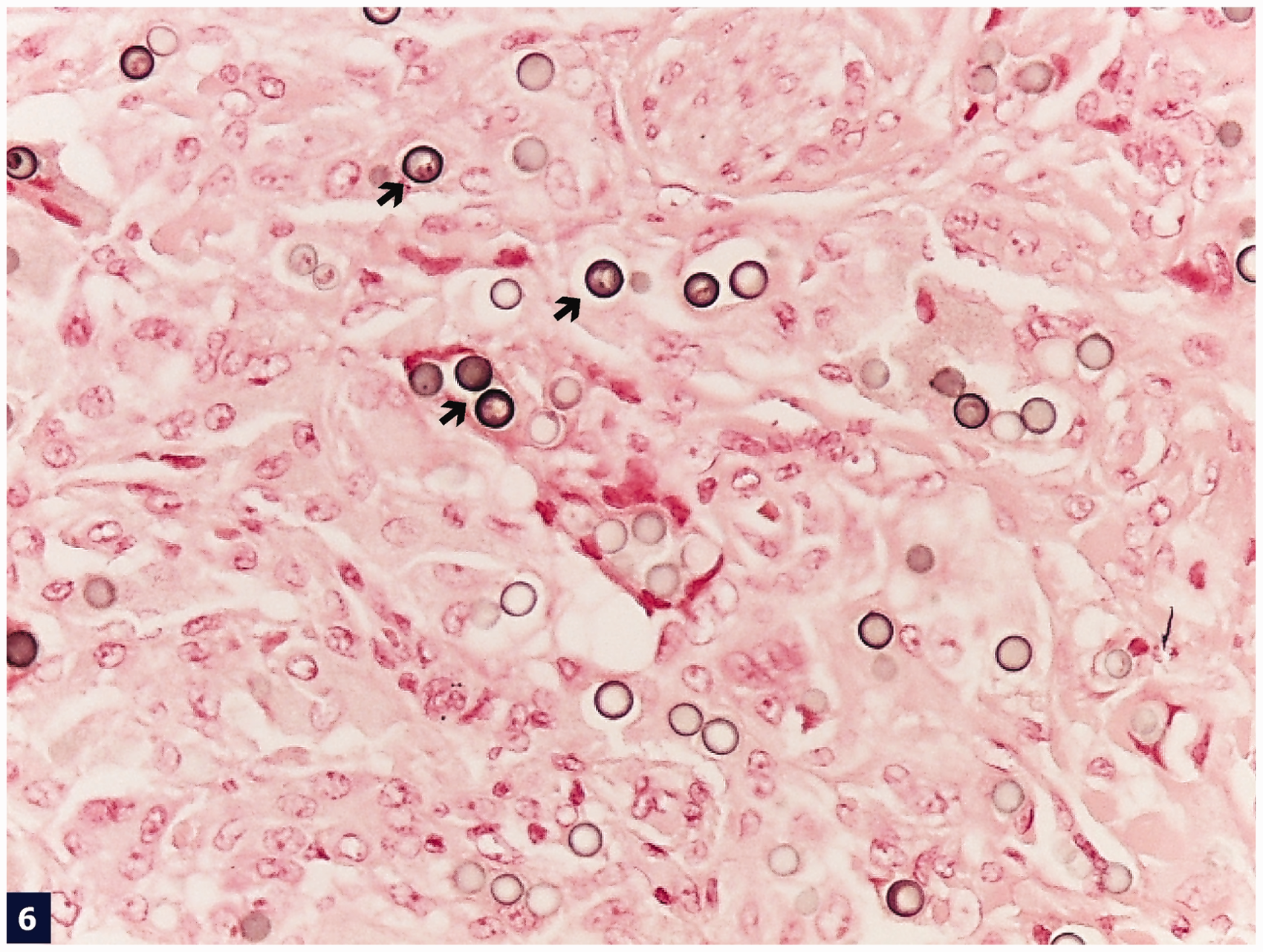
Histological sections from this skin nodule stained with hematoxylin and eosin showed a predominantly atrophic epidermis with areas of pseudoepitheliomatous hyperplasia. A diffusely infiltrative process in the subcutaneous area appeared to be the main lesion of this nodule (Figure 1). Upon further observation, the superficial and deep dermis revealed a proliferation of many cells with a thick and refractile cell membrane, interspersed by an abundant proliferation of fibrous bands. In medium magnification, numerous histiocytes and giant cells admixed with many inflammatory cells were noted in between the fibrous areas. At higher magnification, organisms were seen in the cytoplasm of these histiocytes, as well as in the extracellular spaces that show the characteristic tubular connections between yeast cells with thick refractile cell walls (Figures 2 and 3). Interestingly, there was an abundant proliferation of band-like collagen fibers interspersed throughout the lesion (Figure 2). Proliferation of these fibrous bands was morphologically similar to that of a typical keloidal lesion in the skin and subcutaneous area. Both acute and chronic inflammatory cells, although few in number in some places, were seen admixed with the organisms. The overall impression was of a diffuse dermal infiltrate by fungal organisms in the background of keloidal-type fibrosis. Staining with Gomori methenamine silver and periodic acid–Schiff showed many yeast-like organisms of uniform size, ranging from spherical to oval in shape (Figure 4). Staining for Congo red stained the fungal cell wall red to pink (Figure 5). For further confirmation, a Fontana–Masson staining was also performed and the fungal cell wall appeared black (Figure 6). The microbiologic culture result was negative for any bacterial organisms. No fungal culture was obtained in this case.
Diffuse infiltration of the dermis by fungal organisms (→), interspersed by chronic inflammatory and giant cells (▸) separated by pink collagenous stroma (hematoxylin and eosin stain, low [40×] magnification). Many intra- and extracellular fungal organisms (→) separated by prominent collagenous stroma (▸) (hematoxylin and eosin stain, medium [200×] magnification). Some fungal structures are connected to each other by ill-defined tubule formation (→) (hematoxylin and eosin stain, medium [20×] magnification). Numerous intra- and extracellular fungal organisms (Gomori methenamine silver stain, medium [200×] magnification). Cell wall of fungal organisms (→) are stained red by Congo red stain (medium [200×] magnification). Cell wall of fungal organisms (→) produces melanin pigment highlighted by Fontana–Mason stain (medium [200×] magnification).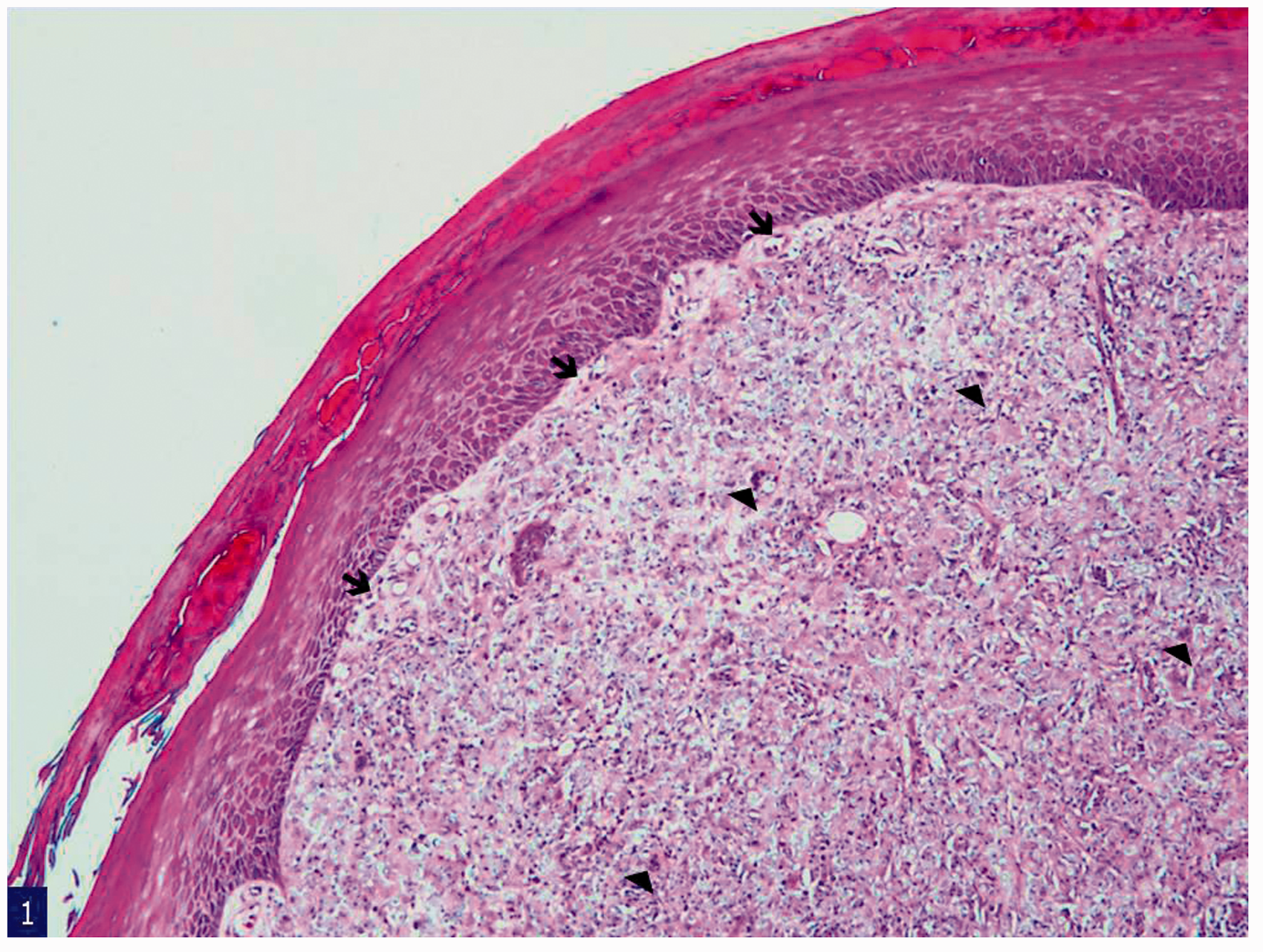


Discussion
The patient had travelled to Brazil, an endemic area of L. loboi, for a hunting trip and presented with the lesion shortly after. The lesion first presented on his elbow, and over several years came to involve a large area with many satellite lesions nearby. This presentation is consistent with how lobomycosis has been documented in the literature.
Cutaneous lobomycosis is a disease of slow insidious onset, progressing over 40–50 years with no reported deaths [Burns et al. 2000; Lupi et al. 2005]. Patients present with nodules, papules, or plaques that are variable in size, and can vary from smooth, to verrucose, to ulcerated [Burns et al. 2000]. The organism invades both the cutaneous and subcutaneous tissue, causing painless lesions that may have associated pruritus and a tingling sensation [Al-Daraji, 2008; Bermudez et al. 2009]. The lesions tend to occur on exposed, cooler areas of the body, with 50% on the pinna of the ear, 29% on the lower limbs, and a small percentage on the upper limbs [Elsayed et al. 2004; Lupi et al. 2005]. They do not involve the mucus membranes and internal organs [Elsayed et al. 2004]. The early lesions have been described as wart-like, and as the disease progresses they become more keloidal in appearance [Lupi et al. 2005]. The lesions may remain localized, or they can be disseminated by the lymphatics to several areas of the body [Lupi et al. 2005; Xavier et al. 2008]. Superinfection can occur in ulcerated lesions. The transformation of chronic lesions into squamous cell carcinoma has also been reported [Baruzzi et al. 1989].
The disease agent, L. loboi, is an obligate intracellular pathogen that resides predominantly in macrophage vacuoles. It is a very slow-growing fungus and has been difficult to culture, making it difficult to determine the natural reservoir [Burns et al. 2000; Rosa et al. 2009]. Inoculation of the footpads of BALB/c mice has shown some success and may become an animal model for the disease [Lupi et al. 2005]. Also, transmission of the disease agent to some mammals has been successfully demonstrated including armadillos, tortoises, and mice [Al-Daraji, 2008]. Many cases in humans have demonstrated a history of trauma, insect bite, cut from a sharp object, or exposure to an aquatic environment; often in people from rural areas that live or work in close contact with vegetation and water sources [Burns et al. 2000]. Disease transmission related to aquatic environments has been implicated due to multiple cases of Lobo’s disease found in dolphins [Burns et al. 2000]. These include two species of dolphins, the marine Tursiops truncatus, and marine–freshwater dolphins Sotalia fluviatilis [Burns et al. 2000]. However, only one case of direct contact from dolphins has been reported and that was in a dolphin trainer in Florida [Paniz-Mondolfi et al. 2007; Rosa et al. 2009]. Furthermore, transmission from person to person has not yet been documented [Rosa et al. 2009].
Our patient was on a hunting trip and spent a great deal of time in outdoor environments where he could have been exposed to many different environmental propagules. He does not recall any cuts or insect bites that occurred during this time. He did not develop a fever or rash during the hunting expedition. Although the majority of cases involved inoculation of the skin, there have also been cases where no trauma was reported, which is consistent with our case [Elsayed et al. 2004]. Our patient noticed the lesion shortly after the trip and after 7 years it had slowly grown into a large keloid-like lesion over his elbow with smaller lesions nearby.
The incubation time from inoculation to development of disease varies from 3 months to 4 years [Rosa et al. 2009]. A case of a French aquarium caretaker who handled an infected dolphin reported an inoculation time of 3 months, whereas another case of a man traveling through Venezuela reported a time of 2.5 years [Rosa et al. 2009]. Most specifically of all, a volunteer inoculated L. loboi into his skin in order to document the exact progression over time. At 4 years, the lesion had spread to cover 33 mm of skin [Rosa et al. 2009]. In our patient, the lesion gradually increased over 7 years.
The histopathologic analysis of lobomycosis specimens, stained with hematoxylin and eosin, showed a granulomatous reaction with large numbers of multinucleated Langhans-type giant cells, as well as many isolated thick-walled yeast-like cells that could be seen singly or in chains (Figures 1 and 2) [Bermudez et al. 2009; Rosa et al. 2009]. The rounded organisms were equal in size, and measured 6–12 µm in diameter as described in other case reports [Elsayed et al. 2004; Al-Daraji, 2008]. The cytoplasmic contents were clear, and with the thick walls, together created a ‘sieve-like’ pattern to the granulomatous inflammation [Elsayed et al. 2004; Rosa et al. 2009]. In addition, many of the cells were connected by a thin tube-like connection that is the hallmark of L. loboi [Elsayed et al. 2004]. The lesion of our patient when stained with hematoxylin and eosin showed the characteristic tubular connection between the yeast cells that contained thick refractile cell walls (Figure 3). This is consistent with the classic findings of L. loboi granulomatous inflammation. Axelson and colleagues described the Congo red staining as an important test for diagnosis of blastomycosis [Axelson et al. 2008]. The Congo red staining in our patient was also positive suggesting its importance as a diagnostic aid along with other staining methods (Figure 5). Taborda and colleagues described the cell wall of L. loboi as containing melanin, which can easily be detected by using the Fontana–Masson stain [Taborda et al. 1999] (Figure 6). This melanin-containing cell wall resists digestion by macrophages and may have a central role in the chronicity of the infection [Bhawan et al. 1976].
Microscopically there are other fungi that can resemble L. loboi, and these include: P. brasiliensis, Blastomyces dermatitidis, and Histoplasma capsulatum var duboisii [Elsayed et al. 2004]. Of these fungi, only L. loboi forms chains of fungal cells of uniform size, 6–12 µm in diameter, connected by thin tube-like connections [Elsayed et al. 2004]. The walls of these cells contain melanin and thus L. loboi can be detected using the Fontana–Masson stain [Burns et al. 2000]. P. brasiliensis shows multiple buds arrayed around a larger, thicker-walled mother cell; often referred to as a ‘ship’s wheel’ appearance [Elsayed et al. 2004; Al-Daraji, 2008]. The cell walls do not contain melanin and thus the Fontana–Masson stain will not detect P. brasiliensis [Burns et al. 2000]. B. dermatitidis is known for its broad-based buds [Elsayed et al. 2004]. H. capsulatum is much smaller in size than L. loboi, mostly intracellular, and the disease tends to affect the lungs and skin [Al-Daraji, 2008]. Lastly, L. loboi is the only one that is unable to grow in vitro on routine mycologic media [Elsayed et al. 2004].
An important feature of lobomycosis is the high expression of the cytokine transforming growth factor beta (TGF-β), which acts to suppress macrophage activity, as measured by a decrease in the marker CD68 [Quaresma et al. 2008; Xavier et al. 2008]. It has also been found in high quantities in other acute and chronic disease processes, such as chronic hepatitis, leprosy, and yellow fever [Xavier et al. 2008]. The high expression of TGF-β causing macrophage suppression may in part be responsible for the difficult clearance of the disease once it infiltrates the skin [Xavier et al. 2008]. In addition, due to the ability of this cytokine to modulate extracellular gene expression, the associated fibrosis and abundance of collagen bands leads to the typical keloidal appearance [Xavier et al. 2008]. Lobomycosis in humans is associated with only a decrease in cell-mediated immunity, whereas in dolphins it involves both cell-mediated and humoral immunity [Bermudez et al. 2009]. Patients present with little to no local inflammatory response and no systemic symptoms at all [Elsayed et al. 2004]. If the lesions are removed at an early stage the excision may be curative, but if the lesions are removed late in the progression they are likely to remain for life [Elsayed et al. 2004]. Also, it is interesting to note that as of yet there has not been a study reporting worsening of the lesions or widespread dissemination of the disease in cases in severely immunocompromised individuals, such as those with HIV/AIDS [Quaresma et al. 2008].
Treatment of lobomycosis is extremely difficult. As it currently stands, surgical excision of isolated lesions with wide margins is the most effective form of treatment and management [Burns et al. 2000; Paniz-Mondolfi et al. 2007; Al-Daraji, 2008]. If the disease has already disseminated, then with surgical excision it tends to recur [Al-Daraji, 2008]. There have also been cases where clofazimine and dapsone were used for their antimicrobial and anti-inflammatory effects, in combination with antifungals, such as itraconazole [Rosa et al. 2009]. Bustamante and colleagues described a case of disfiguring lobomycosis successfully treated with posaconazole and documented no recurrence after 5 years of follow up [Bustamante et al. 2013]. The treatment must be used for extended periods of time, usually 1–2 years, in order to prevent disease recurrence [Rosa et al. 2009]. The treatment is expensive and must be continued for a prolonged time [Quaresma et al. 2008]. Further research must be conducted on the possible treatment options for this disfiguring disease.
Conclusion
Lobomycosis is a slowly progressive fungal infection of the skin and subcutaneous tissue. It should be suspected in patients with single or multiple keloidal skin lesions if they have a history of travel to the Amazon, or a history of trauma related to marine activities [Burns et al. 2000; Bermudez et al. 2009]. The disease has a long latency period and insidious onset, but it is important to keep a high degree of suspicion in nonendemic areas due to the increasing amount of international travel [Burns et al. 2000]. If lobomycosis can be confirmed in its early stages, then surgical excision of local lesions may be curative for the patient [Elsayed et al. 2004]. Medical management remains difficult and lengthy at this time with limited success in treatment [Quaresma et al. 2008].
Footnotes
Acknowledgments
Author contributions: RA, acquisition of case history, preparing the histologic pictures with legends, drafting the case report; SA, critical review; MK, manuscript proofreading and drafting of the case report.
JPK, concept and design, acquisition of available literature, and drafting of the case report; SG, critical revision of the manuscript for important intellectual content.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
