Abstract
Background:
Resistance to antibiotics is steadily increasing. Ceftaroline has a broad spectrum of activity against clinically relevant gram-positive strains including methicillin-resistant Staphylococcus aureus.
Objectives:
This systematic review was conducted to evaluate whether ceftaroline is effective and safe, leading to a lower rate of treatment failures than comparators.
Material and methods:
Studies were included if they were comparing the efficacy and safety of ceftaroline with other antibiotics.
Data sources:
Using the search terms ‘ceftaroline’ or ‘ceftaroline fosamil’, a search strategy was developed. The efficacy endpoint was the rate of treatment failure, while the safety endpoint was the incidence of adverse events. Heterogeneity bias was estimated using the Q-test, and publication bias was estimated using Egger’s test. Null hypothesis was rejected if p value was less than 0.05.
Results:
Only 10 studies were included.
Synthesis of results:
The risk of treatment failure was significantly lower for ceftaroline than for comparators, and cumulative meta-analysis showed that the effect size was relevant and precise. Pooled risk ratio was 0.79 (95% confidence interval = 0.65–0.95). The rates of adverse events were similar among the studies, and there were no statistically significant differences between groups. For this endpoint, there was a significant heterogeneity among studies (p = 0.03). Pooled risk ratio for adverse events was 0.98 (95% confidence interval = 0.87–1.10), without a statistical difference.
Discussion:
The risk of treatment failure was significantly lower for ceftaroline than comparators, while the rate of adverse events was similar. To the best of our knowledge, this is the first systematic review on the efficacy and safety of ceftaroline including children and adults. A limitation is that no randomized controlled trials were found in non-complicated skin- and soft-tissue infection and non-community-acquired pneumonia infections; only few cases with methicillin-resistant Staphylococcus aureus isolations and no patients admitted to the intensive care unit were evaluated.
Interpretation:
Ceftaroline may be an option of treatment in complicated skin- and soft-tissue infection and community-acquired pneumonia.
Introduction
Resistance to antibiotics has been steadily increasing, posing a growing worldwide health problem.
Methicillin-resistant Staphylococcus aureus (MRSA) has emerged as a common cause of complicated skin infections and pneumonia, among others, leading to the need for new effective and safe therapies.
Vancomycin remains the first option in the management of patients with invasive MRSA infections; however, renal toxicity, the narrow spectrum, low concentration in tissues, and an increase in resistance have warranted new treatment alternatives.
Ceftaroline fosamil is a cephalosporin antimicrobial that has generated much interest as a potential treatment option. However, detailed descriptions of its use remain limited. 1 As it is the case of other cephalosporins, the antibacterial activity of ceftaroline is the result of binding to essential penicillin-binding proteins (PBPs) inhibiting bacterial cell wall synthesis.1,2
Ceftaroline has a broad spectrum of activity against clinically relevant gram-positive, strains including MRSA and resistant Streptococcus pneumoniae strains, as well as some gram-negative pathogens involved in complicated skin- and soft-tissue infections (cSSTI) and community-acquired pneumonia (CAP).3,4 Currently, the drug has been approved by the US Food and Drug Administration (FDA) to be used in adults and children (from 2 months of age) with cSSTI caused by methicillin-sensitive and methicillin-resistant strains of Staphylococcus aureus, Streptococcus pyogenes, Streptococcus agalactiae, Escherichia coli, Klebsiella pneumoniae, and Klebsiella oxytoca.1–3 In addition, the drug has been approved by the FDA for CAP caused by Streptococcus pneumoniae, methicillin-sensitive, Staphylococcus aureus (non-MRSA), Escherichia coli, Haemophilus influenzae, Klebsiella pneumonia, and Klebsiella oxytoca.1,3–5 Nevertheless, data on the clinical use of ceftaroline are scarce, especially in the pediatric population. 5
Objective: This systematic review was conducted to evaluate efficacy and safety of ceftaroline and to assess if the drug is associated with a lower rate of treatment failures compared to antibiotic comparators. The secondary aim was to assess effectiveness of the drug in infections in which MRSA was isolated.
Materials and methods
Search strategies: A literature search was conducted to identify all clinical trials comparing safety and efficacy of ceftaroline versus any or none comparator using the strategy described in Table 1. Only articles published in English, Spanish, or French published up to 4 December 2017 were reviewed. Efficacy endpoint was the treatment failure rate because that is the main concern at the moment of antibiotic prescription. The safety endpoint was the incidence of adverse events.
Search strategy.

Study selection: Data extraction and qualitative assessment were performed by two reviewers (M.T.R. and N.S.) who independently appraised the literature and considered only randomized controlled trials (RCTs) for further assessment. In case of disagreement, a third reviewer (R.L.) analyzed the data and managed the scientific discussion until consensus was reached.
A study qualified if (a) it was a RCT and (b) it compared the efficacy and safety of ceftaroline with other antibacterial agents or placebo. Both blinded and open-label trials were included. The methodological quality of the included studies was assessed using the Jadad scale. 6 The Jadad scale is a five-point questionnaire (Table 2) in which each question is to be answered with either a yes or a no. Each yes scores a single point and each no, zero points. Trials scoring ⩾2 were considered for evaluation.
Jadad score template.

Data analysis and statistical methods: Efficacy endpoint incidence was based on intention-to-treat (ITT) populations of each study, and relative risks were determined based on this measure. Pooled risk ratio (RR) and 95% confidence intervals (CIs) were calculated for failure and safety outcomes using the random-effects model (DerSimonian–Laird), in order to be more conservative. Calculations were carried out using the meta-analysis calculator by EpiData software version 3.1 (WHO). Heterogeneity bias was estimated using the Q-test. Potential publication bias was estimated using Egger’s test. The null hypothesis was rejected if p value was less than 0.05. Systematic review was carried out using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement (Table 3).
PRISMA checklist.
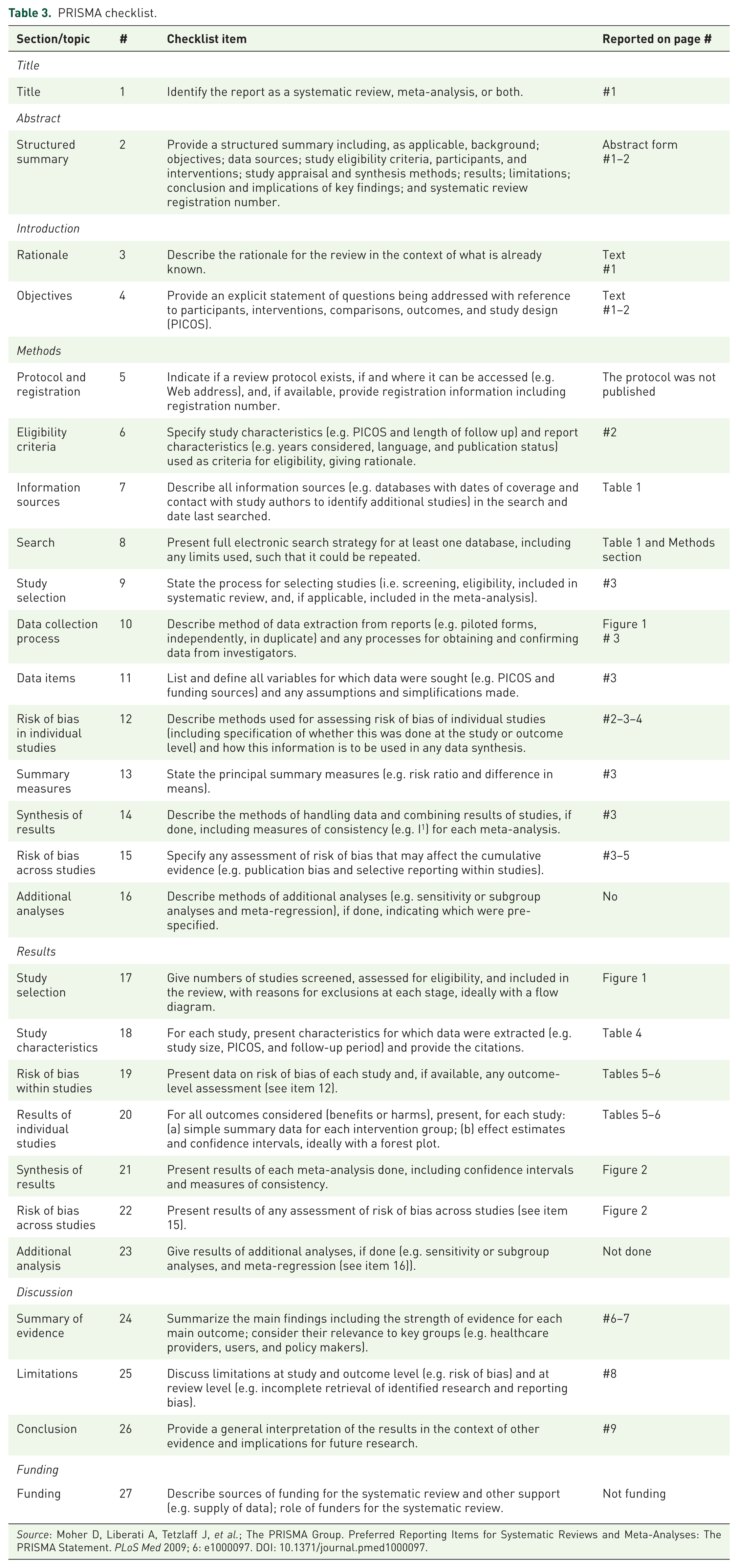
Source: Moher D, Liberati A, Tetzlaff J, et al.; The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med 2009; 6: e1000097. DOI: 10.1371/journal.pmed1000097.

Flow diagram of the process of identification and selection of the articles included.
Main characteristics of the included studies.
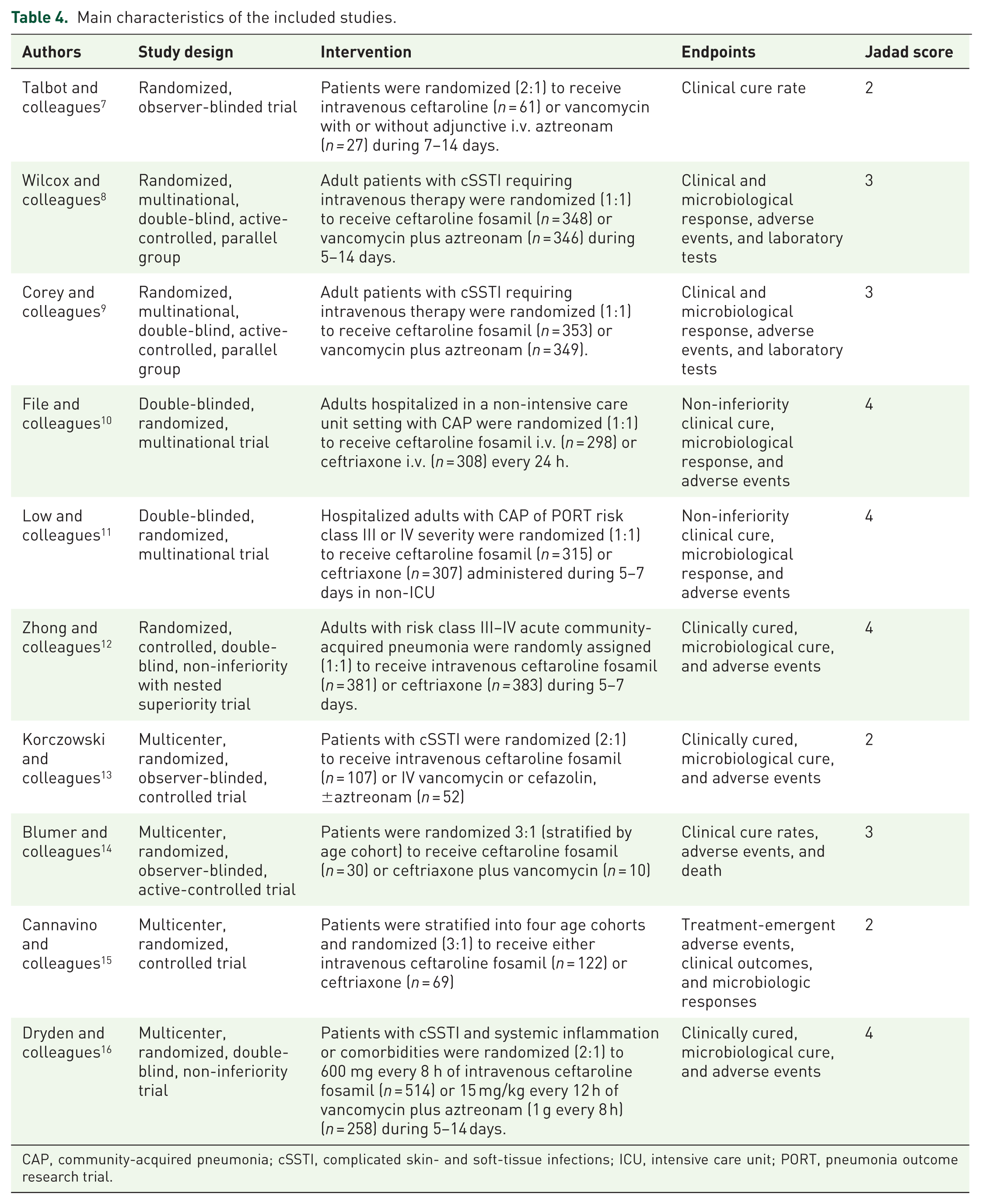
CAP, community-acquired pneumonia; cSSTI, complicated skin- and soft-tissue infections; ICU, intensive care unit; PORT, pneumonia outcome research trial.
Efficacy of the studies included: Therapeutic failure rate (intention-to-treat analyses).

CI, confidence interval; RR, risk ratio.
Safety of the studies included*.

CI, confidence interval; RR, risk ratio.
At least one adverse event.

Risk of therapeutic failure: cumulative meta-analysis.
Results
Included studies and their main characteristics: The literature search identified a total of 1021 potentially relevant abstracts. Screening by title and abstract, 30 RCTs were selected for initial evaluation. A total of 20 RCTs were excluded as they did not meet inclusion criteria or were duplications or new analysis of previously published studies. Finally, 10 full-text articles were selected to be potentially included in this review7–16 (Figure 1).
Out of the 10 studies, three studies13–15 were conducted in pediatric population (two for treatment of CAP14,15 and one for cSSTI13); seven were carried out in adult patients7–12 (four for cSSTI and three for the treatment of CAP).
Table 4 summarizes the main characteristics of the articles included, and Table 5 shows the effect size, the proportional weight, and the pooled RR (95% CI) for the risk of failure of each study. Even when no heterogeneity was detected, pooled RR was calculated using the random-effects model (DerSimonian–Laird).
In individual studies, ceftaroline performance in CAP and cSSTI was not notably better than comparators regardless of the microbiological features (Table 6). However, cumulative meta-analysis revealed a lower risk of therapeutic failure for ceftaroline and the effect size became more relevant and precise when sample size increased, showing a sustained trend as in Figure 2.
Rates of adverse events were similar among the studies and there were no statistically significant differences between groups; however, significant heterogeneity among studies (p = 0.03) was found for this endpoint. Pooled RR for adverse events was 0.98 (95% CI = 0.87–1.10). The most commonly reported adverse drug reactions for ceftaroline were rash, fever, and gastrointestinal symptoms.7–16
In the majority of studies, the rate of direct Coombs test seroconversion was higher in the ceftaroline group than in the comparator groups. Nevertheless, no cases of hemolytic anemia were reported.
In order to analyze risk of treatment failure specifically in infections due to MRSA, a secondary analysis was performed including the six studies (357 patients) reporting these data.7–9,12,13,16 Pooled RR was 0.71 (95% CI = 0.37–1.35).
No significant publication bias was detected (p > 0.05) at any stage (efficacy or safety analysis) of the meta-analysis, although its probability is close to the boundary of significance. Figure 3 shows the corresponding funnel plot for efficacy analysis (Q-test: 16.46; p = 0.058).

Risk of publication bias in efficacy analysis.
In other infections, such as endocarditis, osteoarticular infections, and bacteremia, only case series showing good results with ceftaroline fosamil were found;17–21 however, these studies were not analyzed in this study.
Discussion
This systematic review was conducted to evaluate the risk of therapeutic failure and safety of ceftaroline in children and adults in order to assess the comparative effectiveness and safety of the drug as monotherapy against available comparators.
This meta-analysis suggests that ceftaroline was effective and well tolerated, consistent with the good safety profile of the cephalosporins. 22 This finding, which does not arise from the observation of the individual studies, is probably the result of the increasing sample size.
It is worth pointing out that the risk of therapeutic failure of ceftaroline was found to be significantly lower for both types of infection, and a sustained trend was seen in the cumulative meta-analysis.
Concerns arose, when all the included RCT studies were observed to be conducted only in patients with CAP or cSSTI infections, in patients who were not admitted to intensive care units (ICUs) or were treated with different doses having different follow-up periods, or were using antibiotic comparators that are not typically indicated in the clinics for the treatment of these infections, among others’ characteristics.
In CAP, ceftriaxone is the only cephalosporin that has been demonstrated superiority to penicillin in Streptococcus pneumoniae, even in penicillin-resistant strains, and the drug may be an option in these cases. 8 Ceftaroline may be useful against gram-positive organisms and in areas with a high incidence of MRSA infections.
For complicated pneumonia and patients in the ICU, antimicrobial therapy must be broadened to cover pathogens such as MRSA.
The study by Zhong and colleagues 12 was the only report that found non-inferiority, even superiority, of ceftaroline versus ceftriaxone for the management of CAP in Asian patients not admitted to the ICU. However, the conclusions of this study are of limited validity because of the observed risk of bias regarding the timing of assessment of clinical cure between groups, doses, and conflict of interest, among others’ concerns. 23
In other infections such as endocarditis, osteoarticular infections, and bacteremia, only case series showing good results with ceftaroline fosamil were found,17–21 but these reports were not analyzed in this study.
The strengths of our study are that, to the best of our knowledge, this is the first systematic review and meta-analysis on the efficacy and safety of ceftaroline including children and adults, only RCTs were included, and that the quality of the studies included was assessed in detail.
Limitations are that no RCTs including other non-cSSTI and non-CAP infections were found, cases in which MRSA was isolated were few, and none of the patients was admitted to the ICU. Quality of evidence of studies carried out in other types of infection or in patients admitted to the ICU was limited. In addition, very few studies were conducted in children.
Inherent to systematic reviews, publication bias is always a potential problem, and although the comprehensive search strategy may overcome this issue and the funnel plot showed no relevant evidence of publication bias, this possibility can never be completely excluded.
Clinical implications: The superior efficacy of ceftaroline, its safety profile, and the possibility of its use as monotherapy decrease the need for combined antibiotic treatments, making the drug an attractive option in clinical practice.
Ceftaroline may be considered, particularly in patients with CAP and cSSTI that are intolerant or refractory to other antibiotics used as first-line treatment.
It is remarkable that none of the patients was studied in an ICU setting; however, given the effectiveness of ceftaroline, it may be speculated that even in patients admitted to the ICU, ceftaroline could be useful.
Future research: Further randomized and controlled studies are needed to better understand the role of ceftaroline in other non-CAP and non-cSSTI infections in ICU settings and in children.
Footnotes
Acknowledgements
The authors thank Leticia Cuellar Pompa, Janneke Deurloo, and David Bes for the revision of manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
