Abstract
Background:
Panton-Valentine Leukocidin (PVL) is one of the major virulence factors known to be associated with invasive, life-threatening Staphylococcus aureus (S. aureus) soft tissue infections. Several studies have shown that methicillin-resistant S. aureus (MRSA) and methicillin-susceptible S. aureus (MSSA) are carriers of the lukSF-PV; however, data describing their prevalence and distribution in the Nigerian setting are sparse in the literature, and thus informed the need for the current study.
Objective:
We aimed to detect mecA and analysed the risk factors associated with lukSF-PV-producing S. aureus wound infections.
Design:
This was a single-centre hospital-based descriptive cross-sectional study conducted between March 2019 and September 2019 at the University of Calabar Teaching Hospital, Calabar, Nigeria.
Methods:
Aspirates from participants with soft tissue infections were cultured, and all isolates of S. aureus were tested for the presence of lukSF-PV using endpoint polymerase chain reaction. The mecA was also detected, and antibiotic susceptibility testing was performed.
Results:
Eighty S. aureus isolates were identified from 360 participants. Of the eighty, 47 (58.8%) were MRSA and 10 (12.5%) were lukSF-PV-producing S. aureus strains. Of the ten, six were MSSA and four were MRSA, but the difference was not statistically significant. A significant association was observed between lukSF-PV-producing S. aureus-infected wounds and recurrent skin infections (p = 0.024), as well as working in a day care nursery home (p = 0.0008). The majority of S. aureus isolates were susceptible to tigecycline (76%) and vancomycin (76%), followed by susceptibility to linezolid (72.5%), quinupristin/dalfopristin (67.2%), levofloxacin (38.6%) and erythromycin (11.7%).
Conclusion:
The prevalence of PVL-positive S. aureus strains causing soft tissue infections in our setting is seemingly high. There is a need for active surveillance of this gene in patients presenting with S. aureus soft tissue infections in our setting, ensure antibiotic susceptibility testing, evaluate the impact of these strains on clinical outcomes and prevent the spread of lukSF-PV-positive S. aureus strains.
Introduction
Staphylococcus aureus has emerged worldwide as an important causative agent of skin and soft tissue infection as well as a life-threatening pathogen, especially those containing the Panton-Valentine Leukocidin (PVL) gene. 1 The bacterium has been recognized as the most common isolate from wound infections, constituting a significant healthcare burden due to increased morbidity, mortality and cost of care. 2 Methicillin-resistant S. aureus (MRSA) strains have been of immense concern amongst healthcare providers because of increased involvement in causing multidrug-resistant infections. 3 MRSA strains cause both hospital-acquired (HA-MRSA) and community-associated (CA-MRSA) infections. 4 MRSA contains PVL determinants, a bicomponent beta-pore forming exotoxin that is composed of two water-soluble proteins LukF-PV and LukS-PV.5,6 These proteins exhibit high specific lytic activity against polymorphonuclear cells, monocytes and macrophages in humans by creating pores in their membranes, thereby leading to cell death and cytokine release.5,6 The frequent identification of S. aureus isolates that produce leukocidal toxins from patients with soft tissue infections, particularly furunculosis, cutaneous abscesses and severe necrotizing diseases, suggests that PVL is a virulence factor that plays a major role in the pathogenicity of S. aureus infections. The virulence potential of these toxins is further highlighted by the fact that they cause apoptosis and necrosis in human monocytes and polymorphonuclear cells by provoking inflammatory reactions.7,8 Besides MRSA, lukSF-PV has also been found to be expressed in methicillin-susceptible S. aureus (MSSA). 9
Globally, there are increased reports of infections caused by lukSF-PV-producing S. aureus from heightened surveillance and improved case recognition, and this has been of immense concern to experts. From Asia, a Pakistani study showed most of the S. aureus (60%) isolates identified from pus samples from children in a hospital setting had lukSF-PV in their genome, and a high prevalence was observed in MSSA (64%). 10 Similarly, a review on the epidemiology of S. aureus infection in Africa reported an exceptionally large number of MSSA isolates harbour lukSF-PV, thus portraying Africa as an endemic region for lukSF-PV-positive S. aureus. 11 Despite the elaborate studies performed on lukSF-PV-producing S. aureus across the globe, data on lukSF-PV-positive S. aureus in Nigeria are sparse, and this could negatively impact the management of patients with S. aureus soft tissue infections. We aimed at detecting lukSF-PV in S. aureus causing wound infections in Calabar, Nigeria, and the associated risk factors.
Methods
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material S1). 12
Study design
We conducted a single-centre hospital-based descriptive cross-sectional study between March 2019 and September 2019.
Study setting
The study was conducted at the University of Calabar Teaching Hospital (UCTH), Calabar, Nigeria. Calabar is the capital of Cross River State, located in southern Nigeria. It has two local government areas: Calabar South and Calabar Municipality. The UCTH is located in Calabar municipality, a tertiary health facility with an annual turnover of over 100,000 patients and renders specialized healthcare services to Cross River State inhabitants and also receives patronage from bordering states of Akwa Ibom, Abia, Rivers and Benue as well as adjoining countries such as Cameroon and Equatorial Guinea.
Ethical considerations
Ethical clearance was obtained from the Health Research Ethics Committee, UCTH, Calabar (UCTH/HREC/33/622). A written informed consent was obtained from participants before the commencement of the study. Our study population comprised patients recruited from all wards as well as the specialized units and general outpatient departments of the hospital who presented with soft tissue infection.
Sample size estimation and eligibility criteria
The minimum sample size of 360 was calculated using the following formula, N = (Z2Pq)/d2. 13
Where N = minimum sample size, Z = the standard normal deviation corresponding to 95% level of significance (1.96), P = prevalence rate of S. aureus- (29.49%) associated wound infection in Nigeria, 14 q = (1 – p) and d, the degree of accuracy desired, set at 5% (0.05%).
Inclusion criteria
Participants with soft tissue infections aged ⩾ 18 years.
Consent to participate in the study.
Exclusion criteria
Patients with soft tissue infection who had commenced antimicrobial treatment.
Refusal of consent.
Data collection
The consecutive sampling method was used for case selection. A structured proforma was used to collect patient information. The proforma was divided into (1) socio-demographic characteristics such as gender, age, level of education, occupation and the location of participants; (2) medical and drug history and (3) possible risk factors including diabetes mellitus, sharing personal items, intravenous drug use, use of invasive devices, working in a day care nursery home and recurrent skin infections.
Specimen collection
Specimens collected were wound aspirates. The wound surface was cleaned with sterile gauze moistened with sterile normal saline to remove debris. Aspirates were collected with sterile syringes and needles into sterile specimen containers and labelled. The samples were cultured, and all isolates of S. aureus were tested for the presence of lukSF-PV using conventional polymerase chain reaction (PCR) at the molecular biology unit, Faculty of Basic Medical Sciences, Wilberforce Island, Niger Delta University.
Laboratory procedure
Standard laboratory safety precautions were observed throughout processing. All specimens were correctly labelled, registered with the patients’ study number and processed in a level 2 biosafety cabinet. The samples were inoculated on Mannitol salt agar and incubated aerobically at 37°C for 18 h. Discrete growths were then examined for morphological characteristics suggestive of S. aureus. Subcultures were made of these growths on nutrient agar to generate colonies on neutral media for biochemical testing, including catalase test and coagulase test, following standard protocols. 15 An initial Gram stain was done to determine the morphology of the isolate. Quality control was done using S. aureus ATCC 25923 as a positive control and Escherichia coli ATCC 25922 as a negative control. Susceptibility testing for S. aureus isolates was performed as per Clinical and Laboratory Standards Institute Standards, 28th edition. 16
DNA extraction
DNA extraction was performed using the Zymo bacterial/fungal DNA miniprep extraction kit (Zymo, California, USA) following the manufacturer’s instructions. The extracted DNA was quantified using the NanoDrop 1000 spectrophotometer.
Amplification of lukSF-PV
The lukSF-PV was amplified using the PVLF: 5′ 5′-GCTGGACAAAACTTCTTGGAATAT-3′ and PVLR: 5′-GATAGGACACCAATAAATTCTGGATTG-3′ primers, 17 on an ABI 9700 Applied Biosystems thermal cycler at a final volume of 30 microlitres for 35 cycles. The PCR mix included: the X2 Dream Taq Master mix supplied by Inqaba Biotec, Pretoria, South Africa (Taq polymerase, DNTPs, MgCl), the primers at a concentration of 0.5 µM and the extracted DNA as template. The PCR conditions were as follows: Initial denaturation, 95°C for 5 min; denaturation, 95°C for 30 s; annealing, 58°C for 30 s; extension, 72°C for 30 s for 35 cycles and final extension, 72°C for 5 min. The product was resolved on a 1% agarose gel at 130 V for 30 min and visualized on a blue light transilluminator.
Amplification of mecA
The mecA fragments were amplified using the mecA F: 5′ GTAGAAATACT GAACGT CCGATAA-3′ and mecA R: 5′-CCAATTCCACATTGTTTCGGTCTAA-3′ primers, 17 on an ABI 9700 Applied Biosystems thermal cycler at a final volume of 30 microlitres for 35 cycles. The PCR mix included: the X2 Dream Taq Master mix supplied by Inqaba Biotec, Pretoria, South Africa (Taq polymerase, DNTPs, MgCl), the primers at a concentration of 0.5 µM and the extracted DNA as template. The PCR conditions were as follows: Initial denaturation, 95°C for 5 min; denaturation, 95°C for 30 s; annealing, 56°C for 30 s; extension, 72°C for 30 s for 35 cycles and final extension, 72°C for 5 min. The product was resolved on a 1% agarose gel at 130 V for 30 min and visualized on a blue light transilluminator.
Statistical analysis
Analysis was done using SPSS version 21 (SPSS Inc, Chicago, Illinois). Descriptive analyses were used to summarize the data. The mean and standard deviation were described for continuous variables, whereas categorical variables were analysed using the χ2 test. A p value of <0.05 was considered statistically significant. A multiple logistic regression analysis was performed to identify the independent risk factors for lukSF-PV-producing S. aureus infections among participants.
Results
Socio-demographic characteristics of study participants
A total of 360 participants were recruited in the study, of which 188 (52.2%) were males and 172 (47%) were females. The mean age of study participants was 41.0 ± 17.4 years. The predominant age group was 30–39 years (26.1%), 45.9% had secondary education, while 54.7% were residents in urban areas. The participant’s major occupation was business (n = 89, 24.7%), followed by public service (n = 69, 19.2%; Table 1). The proportion of study participants recruited from different departments is represented in Figure 1, the highest being from the general outpatient department (GOPD; n = 97, 26.9%), and least from the paediatric medical ward (PMW; n = 5, 1.4%).
Socio-demographic characteristics of study participants (N = 360).


Departmental distribution of study participants.
Distribution of the PVL gene among S. aureus isolates
Of the 360 participants recruited, 80 had S. aureus soft tissue infections, which were all considered to be community-acquired. Of the 80, 47 (58.8%) were MRSA, while 33 (41.2%) were MSSA. A total of 10 lukSF-PV-producing S. aureus strains were identified. Of the ten, six were MSSA and four were MRSA, but the difference was not statistically significant (Table 2). The amplified mecA and lukSF-PV are shown in Figures 2 and 3 in the Supplemental Material S2 and the uncropped gels as Supplemental Material S3.
Distribution of the PVL gene among MSSA and MRSA isolates.

MRSA, methicillin-resistant S. aureus; MSSA, methicillin-susceptible S. aureus; PVL, Panton-Valentine Leukocidin.
Possible risk factors for PVL-producing S. aureus infection
The risk factors considered in this study were diabetes mellitus, sharing personal items, intravenous drug use, use of invasive devices, day care nursery home and recurrent skin infection. A significant association was observed between lukSF-PV-producing S. aureus-infected wounds and recurrent skin infections (p = 0.024), as well as working in a day care nursery home (p = 0.008; Table 3). However, only recurrent skin infection (odds ratio (OR): 9.2, 95% confidence interval (CI): 1.995–43.138; p = 0.05) was significant as an independent risk factors for the acquisition of lukSF-PV-producing S. aureus compared with working in a day care (OR: 0.8, 95% CI: 0.725–3.455; p = 0.999) which was not an independent risk factor of acquiring PVL-positive S. aureus (Table 4). Regarding socio-demographic characteristics, there was no significant association between lukSF-PV-producing S. aureus infection and age, sex, level of education, location of participants or occupation (Table 5).
Evaluation of risk factors for PVL-positive S. aureus infection.
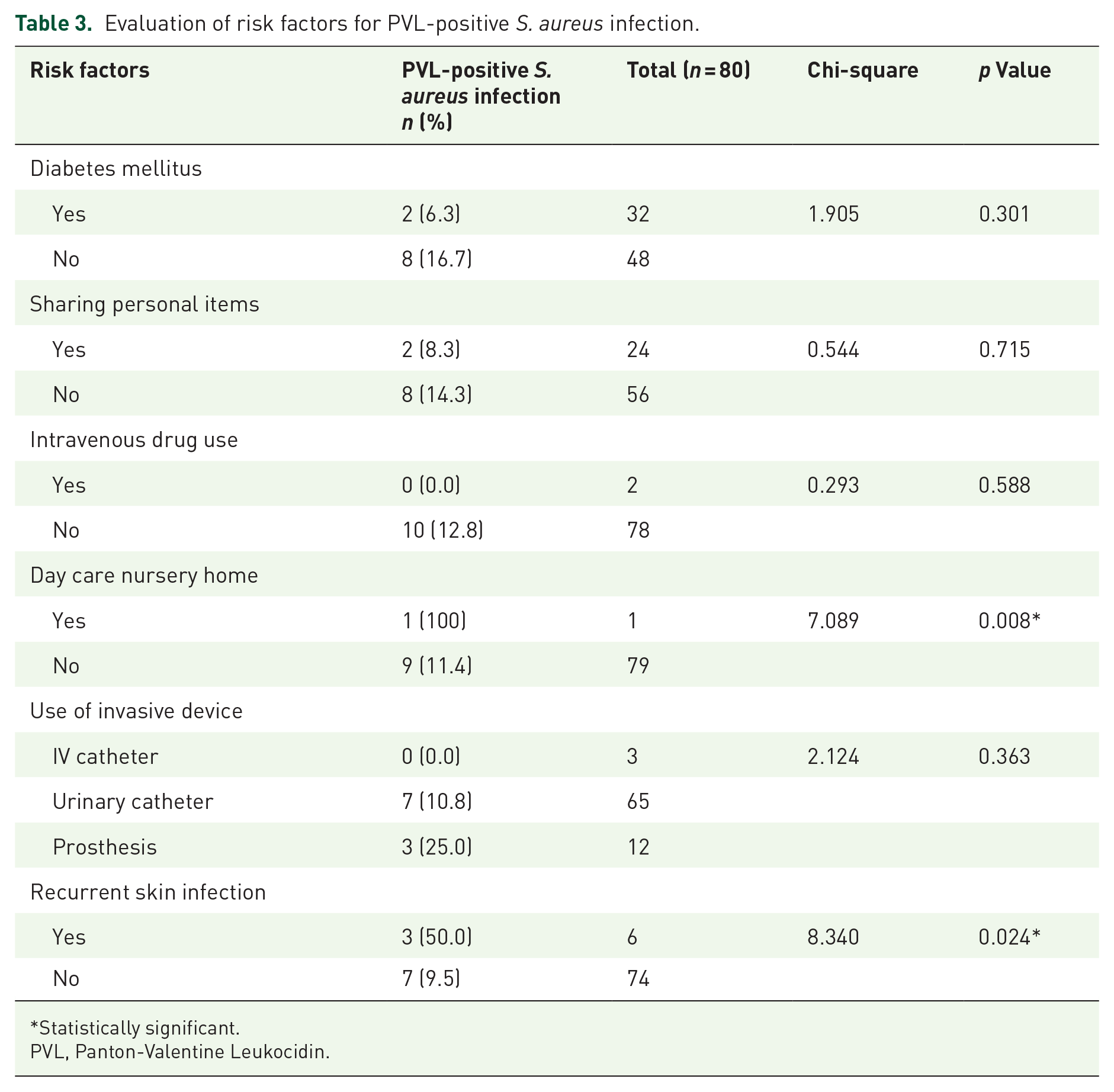
Statistically significant.
PVL, Panton-Valentine Leukocidin.
Multivariate binary logistics regression analysis for independent risk factors for acquiring PVL-positive S. aureus.
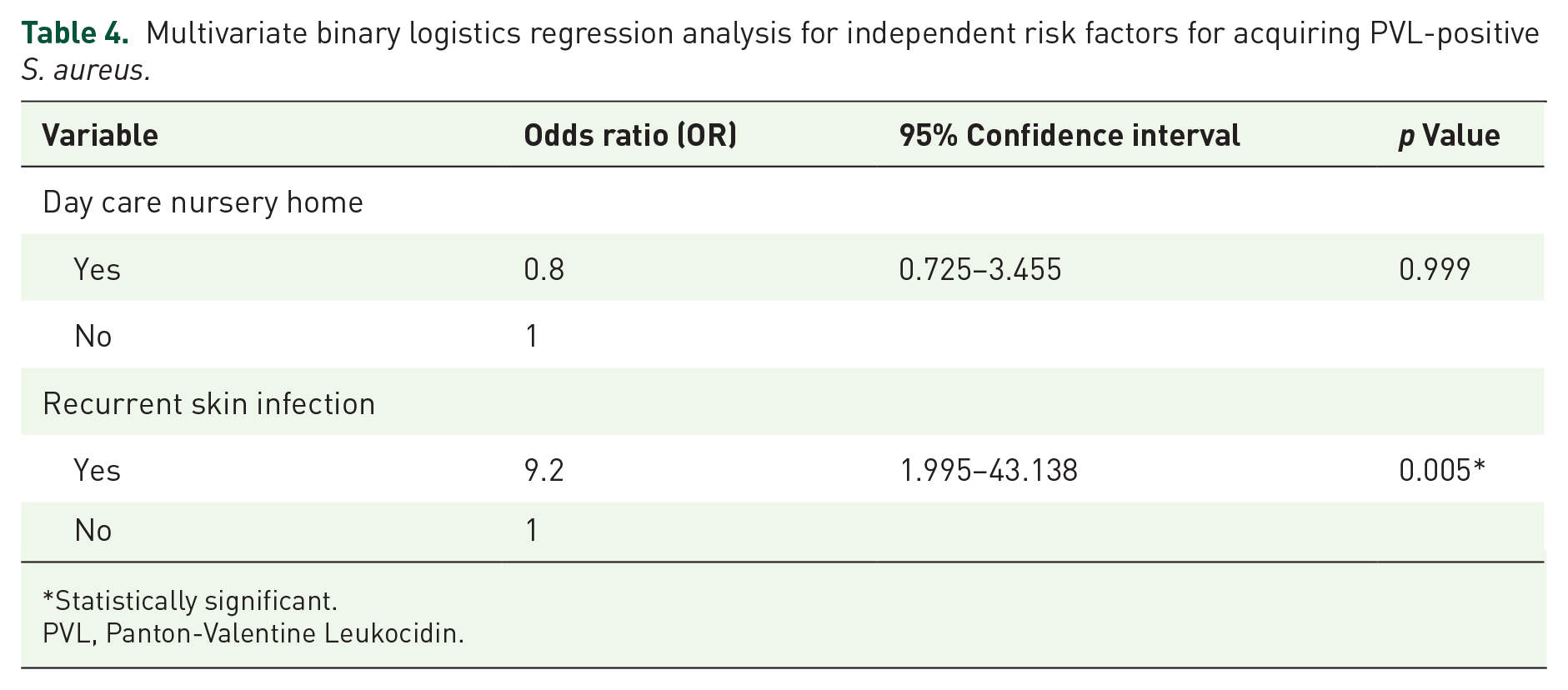
Statistically significant.
PVL, Panton-Valentine Leukocidin.
Association between socio-demographic characteristics and risk of acquiring PVL-positive S. aureus infection among participants.

PVL, Panton-Valentine Leukocidin.
Antibiotic susceptibility profile of S. aureus isolates among study participants
The majority of S. aureus isolates were susceptible to tigecycline (76%) and vancomycin (76%), followed by susceptibility to linezolid (72.5%), quinupristin/dalfopristin (67.2%), cefoxitin (41.2%), levofloxacin (38.6%) and erythromycin (11.7%). The least susceptibility was to clindamycin (6.1%). All the S. aureus isolates were resistant to penicillin.
Discussion
We report a lukSF-PV-producing S. aureus prevalence of 2.8% (10/360) amongst participants with wound infections and 12.5% (10/80) prevalence amongst S. aureus isolates, which suggests one out of every eight isolates of S. aureus in our setting is lukSF-PV producing. This is quite concerning as the presence of PVL secretion by S. aureus is often associated with increased antimicrobial resistance and worsening morbidity.18,19 However, compared with previous studies conducted in Nigeria, the prevalence of this index study was lower. A prevalence of 24.1% (28/116) was reported from South-Western Nigeria 20 and 31.3% (67/214) from North Central Nigeria. 9 The seeming low prevalence from this index study may be because our study was focused on S. aureus isolates detected from wound specimens only, whereas the studies mentioned above included S. aureus isolates from varied sources including wound swabs, ear swabs, urethral swabs, throat swabs, aspirates, blood, cerebrospinal fluid, urine, amongst others. By contrast, a 10.7% (6/56) prevalence was reported in a study from South–South Nigeria. 21 The above variations suggest more studies are needed to understand the epidemiology of PVL-positive S. aureus in our setting.
Regarding the distribution of the lukSF-PV among S. aureus isolates, a slightly higher proportion was found among the MSSA isolates compared with the MRSA group. This agrees with findings from the few studies conducted in Nigeria on the detection of lukSF-PV amongst S. aureus isolates identified from wound samples,9,20 compared with findings from other similar studies around the globe. For instance, a study conducted in Northern Cyprus showed that 27.7% of MRSA isolates were PVL-positive. 22 Similarly, a Cameroonian study reported that 53.3% of MRSA isolates were PVL gene-producing. 18 In Algeria, the frequency of MRSA strains containing PVL genes was 36%. 23 The coexistence of the lukSF-PV and mecA poses a serious challenge in the treatment of infections caused by such S. aureus isolates. While lukSF-PV is associated with severe skin infections and necrotizing pneumonia, mecA confers resistance to methicillin and other beta-lactam antibiotics, thus resulting in high virulence and difficult-to-treat infections. By contrast, as seen in this study, some factors may account for a higher frequency of lukSF-PV in MSSA, including genetic factors, epidemiological trends, selective pressure or virulence. The distribution of mobile genetic elements carrying lukSF-PV may vary between MSSA and MRSA and, as such, accounts for a higher frequency of lukSF-PV in either of the two. 24 On the other hand, selective pressure can occur following the use of antibiotics, thus influencing the prevalence of PVL-positive strains. 25 Also, as per the environments of these pathogens, the lukSF-PV may confer a fitness advantage, influencing its prevalence in MSSA compared with MRSA. 26
Amongst the possible risk factors considered in this study, recurrent skin infections were seen to be significantly linked with PVL-positive S. aureus infection. Several studies around the globe affirmed similar findings. In a German study, PVL-producing strains of S. aureus were detected in 61.3% of 411 patients with recurrent skin infections. Of those isolates, 80.6% were MSSA, while 19.4% were MRSA. 27 In another German study, amongst a cohort of 155 patients with recurrent skin infections, PVL-positive MSSA and PVL-positive MRSA were detected in 66% and 19% of cases, respectively. The remainder comprised symptomatic patients with close contact with PVL-producing S. aureus index patients but without detection of lukSF-PV. 28 PVL-producing S. aureus is often associated with recurrent necrotizing skin and soft tissue infections and can lead to more severe and invasive presentations, including necrotizing haemorrhagic pneumonia, necrotizing fasciitis and purpura fulminans. 8
Regarding susceptibility profiling, most S. aureus isolates were highly resistant to antibiotics commonly prescribed for wound or soft tissue infections, including clindamycin and levofloxacin. This may result in a delayed initiation of therapy with appropriate antibiotics and invariably worsen clinical outcomes, especially when the desired medications are seemingly not available. Particularly concerning is the fact that this index study was a single-centre study, suggesting there could be more cases of multidrug-resistant PVL-producing S. aureus in our setting going undetected and hence poorly treated with perhaps eventual fatal outcomes.
Limitations
Besides PVL, S. aureus also produces other toxins such as the exfoliative toxins, enterotoxins and toxic shock syndrome toxin 1, known to be associated with severe skin and soft tissue infections. However, this study only investigated PVL due to cost considerations. We recommend subsequent studies of this nature to investigate other S. aureus toxins as highlighted, explore their role in S. aureus soft tissue infections in our setting and the associated risk factors.
Regarding the susceptibility profile of S. aureus strains to tested antibiotics, we advise caution in interpreting the high resistance rates of 24% detected for vancomycin and linezolid, as this was merely via phenotypic methods. A confirmation by molecular assay is needed, but we were somewhat limited in the conduct of this study due to a lack of funding.
From the epidemiological perspective, this was a single-centre study conducted at the UCTH, Calabar, Nigeria. The findings may not be extrapolated to ascertain the burden of PVL-producing S. aureus wound infections in Nigeria. A multicentre study capturing tertiary health facilities across the six geopolitical zones of Nigeria may produce more representative data on the occurrence of PVL-positive S. aureus in Nigeria and the associated risk factors, and generate policies on how to tackle the spread of such strains of S. aureus and develop antibiograms for effective antimicrobial treatment. However, considering the number of participants recruited, the inputs from this study can be used as a basis to drive similar studies in other healthcare institutions in Nigeria, as well as active surveillance, and invariably improve case finding and clinical outcomes.
Conclusion
We report a 12.5% prevalence of PVL-positive S. aureus isolates amongst S. aureus strains causing soft tissue infections in patients from Calabar, Nigeria. There is a need to scale up studies in our setting, evaluating the presence of PVL genes in S. aureus isolates cultured from wound or soft tissue infections. In addition, awareness of PVL-producing S. aureus infections should be done among prescribers with emphasis on the need to request antibiotic susceptibility testing on S. aureus isolates and adhere to antibiotic sensitivity test results in managing wound infections.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251357394 – Supplemental material for Molecular detection of mecA and lukSF-PV in patients with Staphylococcus aureus soft tissue infections in a tertiary hospital setting, Calabar, Nigeria: a cross-sectional study
Supplemental material, sj-docx-1-tai-10.1177_20499361251357394 for Molecular detection of mecA and lukSF-PV in patients with Staphylococcus aureus soft tissue infections in a tertiary hospital setting, Calabar, Nigeria: a cross-sectional study by Christian J. Ide, Godwin I. Ogban, Bassey E. Ekeng, Ubleni E. Emanghe, Asukwo E. Onukak, Anthony A. Iwuafor, Ubong A. Udoh, Stella T. Chukwuma, Emmanuel M. Jimmy, Ido E. Ukpeh, Tatfeng Y. Mirabeau and Daniel Z. Egah in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-2-tai-10.1177_20499361251357394 – Supplemental material for Molecular detection of mecA and lukSF-PV in patients with Staphylococcus aureus soft tissue infections in a tertiary hospital setting, Calabar, Nigeria: a cross-sectional study
Supplemental material, sj-docx-2-tai-10.1177_20499361251357394 for Molecular detection of mecA and lukSF-PV in patients with Staphylococcus aureus soft tissue infections in a tertiary hospital setting, Calabar, Nigeria: a cross-sectional study by Christian J. Ide, Godwin I. Ogban, Bassey E. Ekeng, Ubleni E. Emanghe, Asukwo E. Onukak, Anthony A. Iwuafor, Ubong A. Udoh, Stella T. Chukwuma, Emmanuel M. Jimmy, Ido E. Ukpeh, Tatfeng Y. Mirabeau and Daniel Z. Egah in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-3-tai-10.1177_20499361251357394 – Supplemental material for Molecular detection of mecA and lukSF-PV in patients with Staphylococcus aureus soft tissue infections in a tertiary hospital setting, Calabar, Nigeria: a cross-sectional study
Supplemental material, sj-docx-3-tai-10.1177_20499361251357394 for Molecular detection of mecA and lukSF-PV in patients with Staphylococcus aureus soft tissue infections in a tertiary hospital setting, Calabar, Nigeria: a cross-sectional study by Christian J. Ide, Godwin I. Ogban, Bassey E. Ekeng, Ubleni E. Emanghe, Asukwo E. Onukak, Anthony A. Iwuafor, Ubong A. Udoh, Stella T. Chukwuma, Emmanuel M. Jimmy, Ido E. Ukpeh, Tatfeng Y. Mirabeau and Daniel Z. Egah in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
