Abstract
Clostridium difficile infections have a high recurrence rate following acute treatment. Extended duration vancomycin (EDV) is a mainstay for the treatment of recurrent Clostridium difficile infections (rCDI). Clinical disease guidelines recommend a variety of different vancomycin treatment regimens though based on weak, low-quality evidence. Patients typically receive an initial vancomycin treatment course of 7–14 days for the acute infection, followed by an extended duration vancomycin course. Multiple publications on the utility of EDV regimens have been published but few include reported effectiveness outcomes associated with a prescribed treatment regimen. The purpose of this review is to evaluate the safety and efficacy data on extended duration vancomycin regimens used in recurrent clostridium treatment. Five articles, three case series and two randomized open-label clinical trials, were identified which included both elements. Outcomes were evaluable in 174 patients, 31 from randomized trials, with prior average recurrent episodes ranging from 3 to 4. Vancomycin dose ranged from 3500 to >6800 mg with therapy durations extending from 21 days to over 60 days. Follow-up duration ranged from 10 weeks to 12 months. Case series reported success rates for EDV in rCDI from 61% to 100%, while randomized trials found lower success rates from 26% to 58%. Taper and pulse regimens reported superior outcomes compared to pulse-only regimens, 58–100% versus 26–81%, respectively. Comparative EDV data is limited. Current available data supports an EDV regimen which includes both a daily dosing taper followed by an every 48 or 72 h pulse.
Introduction
Clostridium difficile infection (CDI) is the most common nosocomial infection in the United States and a leading cause of morbidity and mortality. Based on data collected in 2011, an estimated incidence of approximately 453,000 infections in the United States was caused by C. difficile. The incidence rates are higher among females, Whites, and persons over 65 years of age. The majority of cases (65.8%) were deemed to be health care-associated, with only a 24.2% onset during hospitalization. Most initial infections are successfully treated, although on average 20% of patients may experience recurrent CDI (rCDI). 1 When possible, prompt discontinuation of offending antibiotics may hasten recovery and reduce the risk of recurrence. The health care costs associated with rCDI are estimated to top US$13,000 per relapse. 2
Clostridium difficile is a gram-positive, spore-forming anaerobic organism capable of producing toxins, leading to clinical manifestations ranging from mild diarrhea to pseudomembranous colitis (PMC). 3 The spore-forming nature of the organism makes it challenging to eradicate the infection. Recurrence is secondary to C. difficile spores germinating and allowing the organism to regrow before the normal colonic flora has time to adequately repopulate and suppress future infection. Recurrence rates following subsequent infections are even higher, reportedly near 45%. Recurrent CDI can be due to either the persistence of the initial strain or acquirement of a new strain. 4
Recommendations for initial CDI treatment are based on strong, high-quality evidence, whereas regimens for recurrence are based on weak, low-quality evidence. 5 Oral metronidazole has been widely used for acute treatment of CDI but prolonged courses are associated with an increased risk of neurotoxicity. 6 Oral vancomycin has been the most common guideline recommended therapy for rCDI.5–8 It is a glycopeptide antibiotic with a gram-positive bacterial spectrum and minimal systemic absorption even in patients with C. difficile-induced PMC. Oral vancomycin formulations include capsules, suspensions, and solutions. However, injectable vancomycin can be used to compound an oral formulation when commercial products are unavailable or to reduce cost. 9
Guidelines across the United States and the world consistently recommend a vancomycin taper and pulse regimen therapy for rCDI. Although recommended regimens vary, all include a similar initial vancomycin treatment course (see Table 1).5–8 An extended duration vancomycin (EDV) regimen may involve a slow dose taper over several weeks and / or a pulsed dose every 48–72 h. Multiple review articles on CDI treatment have promoted use of alternative EDV regimens.10–12 EDV regimen effectiveness has been estimated to be 69% at treating rCDI. 13 This review will summarize the published literature on the safety and efficacy of EDV regimens for the treatment of multiple rCDI.
Guideline recommendations for extended duration vancomycin therapy.

ACG, American College of Gastroenterology; ASID, Australasian Society of Infectious Diseases; BID, twice daily; ESCMID, European Society of Clinical Microbiology and Infectious Diseases; IDSA, Infectious Disease Society of America; SHEA, Society for Healthcare Epidemiology of America.
Literature search
The literature search included PubMed (1980–February 2018), Web of Science (1998–February 2018), and OVID MEDLINE (1946–February 2018). Selected search terms included pulse, vancomycin, Clostridium difficile, recurrent, and taper. Only articles available in the English language were considered. The bibliographies of all pertinent articles were reviewed to assess for additional references. To be included in the analysis, patients had to receive an initial vancomycin treatment course (7–14 days) for the acute infection followed by a specified EDV regimen. Only articles which reported response rates were included in the analysis. See Figure 1 for the search results. Five articles met inclusion criteria: three case series and two randomized open-label clinical trials (see Table 2).14–18

Summary of Study Selection.
Outcomes with extended duration vancomycin in recurrent Clostridium difficile.
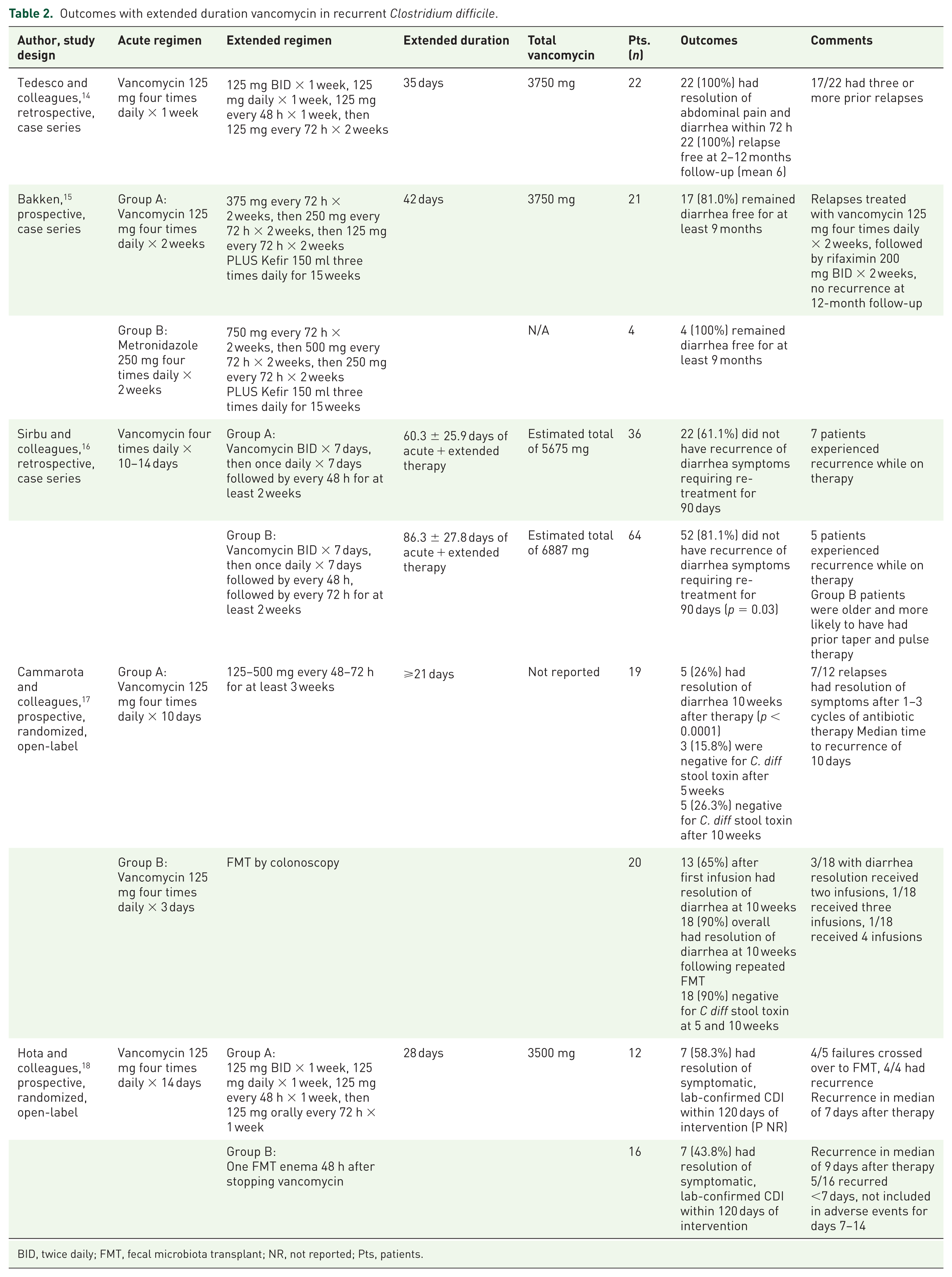
BID, twice daily; FMT, fecal microbiota transplant; NR, not reported; Pts, patients.
EDV
Case series
Tedesco and colleagues 14 were the first to report outcomes following a vancomycin taper in 22 patients (14 female) with 2–5 relapses of antibiotic-associated pseudomembranous colitis (AAPMC) or CDI. The mean age among the patients was 59 years and diverticulitis was noted in 82% of patients. Patients experienced multiple relapses with the last relapse confirmed by endoscopy. The regimen for relapses included an initial vancomycin taper and concluded with pulse dosing at two different intervals. Diarrhea and abdominal pain resolved within 72 h in all patients without AAPMC recurrence during a follow-up of 2–12 months. The positive results in this early report likely contributed to the taper regimen noted in the US CDI guidelines. 5 Adverse events were not reported. Strengths include endoscopic and tissue culture assays for C. difficile toxin and using a defined vancomycin dose and duration. Limitations with this report include retrospective reporting, uncontrolled and open-label design, single center, and small sample size.
Bakken 15 examined the efficacy of tapering pulse regimens of metronidazole and vancomycin in conjunction with regular intake of kefir, a milk-based product containing active probiotic cultures. A total of 25 patients (21 female), with a mean age of 68 years, were evaluated during this 8-year prospective case series. Vancomycin was used as the taper therapy in 17 patients. The mean number of rCDI was 4 (range, 1–9) and the median days of diarrhea at presentation were 135. The rCDI was verified by ⩾1 positive stool sample. Common comorbidities predisposing to recurrent infection included ulcerative colitis, diabetes, previous bowel surgery, and malignant illness. Patients were given an option to receive fecal microbiota transplantation (FMT), but all declined. The pulse regimen was initiated once bowel movements normalized and were maintained for at least 7 days. No additional antibiotics were given during this treatment course, although four patients took immunosuppressive therapy and eight were on histamine-2 blocking agents. Patients were interviewed about rCDI via telephone or mail at 90 days and quarterly intervals for up to 1 year. Diarrhea resolved in all but four patients after 9 months, an 84% treatment success rate. Adverse events were not reported. Strengths include being prospective with predefined treatment dose, duration, and detailed patient demographics. Limitations include single investigator, small number of cases, open-label, and uncontrolled design.
Sirbu and colleagues 16 retrospectively evaluated 100 patients (64 female) with a mean age of 63.5 years who were referred for rCDI, mean 3.15 episodes, during a 6-year period. All patients received an initial 2-week vancomycin taper regimen. The vancomycin dose was not specified, but was reported as generally following Infectious Disease Society of America (IDSA) guidelines. The authors analyzed outcomes between groups receiving either an every 48 h (36 patients) or an every 48 h followed by an every 72 h (64 patients) pulse regimen. Patients were seen every month or more frequently if needed. All patients had a 90-day follow-up after completion of the vancomycin taper/pulse regimen. All subjects had at least one CDI confirmed by C. difficile stool assay. Recurrence of diarrhea was defined as ⩾2 CDI at the time of referral. The mean duration of the combined treatment and pulse therapy for both groups was 11 weeks. Patients receiving the 72 h pulse regimen averaged 26 more treatment days. Patients in the 72-h group were older and more likely to have had prior vancomycin pulse and taper therapy. A higher cure rate (81.1%) was seen in the group who received the extended 72-h pulse regimen (p = 0.03). The cure rate was higher in patients who had ⩽2 prior CDI compared to those with >2 prior episodes. The cure rate was not influenced by variables such as age, sex, concomitant antibiotic use, gastric acid suppressant use, and the total duration of vancomycin use. Adverse events were not reported. Strengths include a larger patient population. Limitations include variable vancomycin treatment durations and the single center, non-randomized, observational, and retrospective design.
Clinical trials
Cammarota and colleagues 17 randomized 39 patients (23 female), in an open-label design to a vancomycin pulse-only regimen (n = 19) for at least 3 weeks or FMT. Enrolled patients had an average age of 73 years, median of 3 rCDI, and 90% failed a prior vancomycin taper. The majority of CDI infections were considered to be hospital-acquired. The primary endpoint of this trial was the CDI resolution 10 weeks after the end of treatment. The study was concluded early as the 1-year interim analysis suggested that FMT showed higher efficacy than vancomycin. In the FMT group, 65% were cured after the first infusion; seven patients were diagnosed with PMC and received multiple FMTs. In the vancomycin group, 26% were cured. A total of 12 patients had recurrence at the end of treatment; the median time for recurrence was 10 days. These patients received repeated cycles of vancomycin and metronidazole therapy for the recurrences which resulted in symptom resolution in seven patients. All but one patient in the FMT experienced diarrhea and abdominal cramping immediately after infusion which resolved within 12 h; bloating and cramping were experienced by 60% of FMT patients. No vancomycin-related adverse effects were noted. Strengths include randomization and a comparator group. Limitations include lack of blinding, small sample size, single center design, and lack of standardization of vancomycin dose or pulse frequency.
Hota and colleagues 18 compared 14 days of oral vancomycin followed by a single FMT by enema versus a 6-week oral vancomycin taper/pulse in adult patients with rCDI. A total of 30 patients (19 female) with an average age of 72 years were randomized: 14 patients to vancomycin taper and 16 patients to FMT. Patients had at least two rCDI episodes (range, 2–9) confirmed by laboratory or pathology and received at least one cycle of oral vancomycin (500 mg/day for a minimum of 10 days). The primary endpoint was rCDI within 120 days. The study included two in-person and four telephone follow-up visits for evaluation of recurrence and adverse events. Two patients in the vancomycin arm did not receive treatment: one patient was dropped due to protocol violation and one patient received FMT at another center. Recurrent CDI was reported in nine FMT patients (56.2%) and five vancomycin patients (41.7%). The median days of recurrence were 9 days after FMT and 35 days after vancomycin tapering. Abdominal pain, tenderness, and bloating were equally reported in the early study period (⩽7 days) among both groups. Additional adverse effects such as mucoid and smelly stools were reported more frequently in the vancomycin group later in the study (⩾7 days). The trial was stopped early when it was determined further enrollment would fail to detect a difference between treatment groups. Strengths include randomization and a control group. Limitations include small sample size, single center, and open-label design.
Discussion
Reviewed literature identified three case series14–16 and two clinical trials17,18 encompassing a total of 174 patients treated with an evaluable EDV regimen, of these only 31 patients were included in randomized trials. The largest report was a case series covering 100 patients. 16 Baseline characteristics included a mean of 3–4 prior CDIs, were predominantly female, and with a mean age from the late 50s to early 70s. Each evaluation incorporated different vancomycin dosing, treatment duration, or taper/pulse regimens. The most frequently used regimens followed a taper and pulse combination similar to that recommended in the IDSA guidelines, which appears to be based on the original work of Tedesco and colleagues but referenced McFarland and colleagues.13,14,16,18 Response rates varied greatly with an initial published case series 14 reporting success rates of 100% to as low as 26% in a more recent randomized trial. 17 Duration of therapy ranged from 21 days 17 to exceeding 60 days. 16
Sirbu and colleagues 16 was the largest single report but also most difficult to evaluate. The authors reported using a regimen based on the IDSA vancomycin taper but did not include specific doses or report a target duration of therapy. This was the only analysis comparing two different vancomycin pulse strategies. Patients in this case series experienced the longest overall treatment duration and the standard deviation for an individual regimen varied by 3–4 weeks.
Two studies used pulse-only EDV regimens, with 19% of patients having a recurrence within 9 months in a reported case series 16 while 74% had a recurrence at 10 weeks in the randomized trial. 17 The trial with the fewest enrolled vancomycin patients (n = 12) was also the most well-designed evaluation of an EDV regimen. 18 The 120-day recurrence rate of 41.7% is similar to the 45% previously reported for multiple rCDI. 4 Follow-up duration ranged dramatically from 10 weeks to 12 months; however, this did not appear to impact efficacy outcomes. Longer follow-up would generally result in a greater risk of recurrence being identified. The study with the shortest follow-up time was also the study with the poorest outcomes. 17 Based on the mixture of regimens and durations in the reported literature, a clear association between duration and effectiveness cannot be made; but generally, regimens ⩾28 days reported better outcomes.
This review included two randomized, open-label, controlled trials comparing EDV to FMT.17,18 The response to vancomycin differed substantially between the studies. Cammarota and colleagues 17 had a 26% response to EDV while Hota and colleagues 18 a had 58.3% response rate when using a clearly reported regimen. Only one report by Bakken 15 incorporated a concomitant probiotic therapy, kefir, to re-establish normal gut flora preventing invasion by pathogens. 2 The influence of concurrent use of the probiotic product kefir or other probiotic formulations on EDV effectiveness is unclear.
Pulse-only regimens may have several potential advantages over taper/pulse EDV regimens. A consistent 48- or 72-h dosing schedule appears to be less complicated for patients. Pulse dosing may also result in lower total vancomycin requirements and subsequent associated cost. It is noteworthy that none of the literature reviewed included any discussion of patient compliance on regimen complexity. Total vancomycin doses were similar in most of the trials, ranging from an estimated 3500 to 3750 mg. One trial did not report enough data for an estimate to be made 17 and the largest case series used the most vancomycin with an estimated mean total dose exceeding 6800 mg. 16 Cumulative vancomycin dose and duration did not vary substantially among the majority of regimens; therefore, one cannot be favored due to potential cost.14–18 The least effective EDV regimen for rCDI, 74% recurrence rate, was seen in the lone randomized trial using a pulse-only regimen. 17 Findings from this review do not support the use of pulse-only EDV regimens as recommended by some guidelines.6,8
A lack of robust, randomized clinical trials limit the quality of evidence behind recommending a specific EDV regimen. Although the expert opinion is to initiate vancomycin in a taper and pulsed fashion for rCDI, no head-to-head comparisons have been studied. After an extensive literature search, only two randomized clinical trials have evaluated outcomes with an EDV regimen.17,18 A randomized controlled trial is currently recruiting patients to assess the optimal treatment for rCDI between vancomycin, a vancomycin taper/pulse regimen, and fidaxomicin (NCT 02667418), which will likely contribute to the available data once published. 19
Conclusion
Literature evaluating EDV regimens has demonstrated efficacy in small case series and small studies. Pulse-only regimens, as recommended in some guidelines, resulted in lower success rates. Current available data support an EDV regimen which includes both a daily dosing taper followed by an every 48- or 72-h pulse, similar to that recommended in the most recent guidelines.
Footnotes
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
