Abstract
Background:
Evidence suggests statins may improve survival in patients with bloodstream infections. However, there is no consensus on optimal timing and duration of exposure.
Objectives:
To quantify statin therapy duration associated with decreased mortality in bacteremic statin users.
Methods:
We conducted a case-control study using OptumClinformatics™ with matched Premier hospital data (1 October 2009–31 March 2013). Cases who died during the hospitalization were matched 1:1 to survivors on disease risk scores (DRSs). Post-admission statin therapy duration was evaluated in patients with at least 90 days of pre-admission continuous statin use. Classification and regression tree (CART) analysis was conducted to identify the optimal duration of statin continuation which provided the lowest inpatient mortality. Logistic regression was used to calculate the odds of mortality.
Results:
We included 58 DRS matched pairs of cases and controls: 47 patients (41%) continued statin therapy during the hospital admission, 15 (32%) cases and 32 (68%) controls. The CART analysis partitioned the continuation of statin therapy at ⩾2 days, representing lower mortality for patients who continued statins for 2 days or more and higher mortality for patients who did not continue or remained on statins for only 1 day. Inpatient mortality was 76% lower among those with at least 2 days of continued statin use (odds ratio 0.24, 95% confidence interval 0.11–0.55).
Conclusion:
Among matched cases and controls with at least 90 days of baseline statin use prior to the admission, the continuation of statins for at least 2 days after admission demonstrated a survival benefit among bacteremic patients.
Introduction
Inpatient mortality among patients with bloodstream infections remains high (16.3%). 1 Evidence from observational research suggests that statins may improve survival in patients with bacteremia2–5 and sepsis,6–8 including 14 day, 5 15 day, 2 31–180 day, 9 and all-cause hospital mortality.3,5,7 While several studies have reported reduced mortality with statins in bacteremic patients, statin duration and measurement of outcomes differ across these studies.3–5 As a result, rates of survival vary, particularly as statin exposure varies.3,9 In addition, several of these studies have identified an increase in mortality after cessation of statin therapy.3,10 Since the length of statin treatment time varies between studies, there is no consensus as to whether statin therapy should be continued among patients presenting to the hospital with bacteremia, and if so, what duration of statin continuation would provide the maximum advantage in terms of clinical outcomes.
While several meta-analyses11,12 and observational3–5,9 studies observed protective effects with statins in bacteremia, one meta-analysis 13 did not observe improvements in clinical outcomes with statin use. However, this meta-analysis was conducted among critically ill patients with severe sepsis, and some of the included studies only had short durations of statin use.3,4,9 Other studies with shorter statin durations also did not demonstrate a statistically significant association between statin use and mortality.9,13,14 A recent randomized controlled trial (RCT) evaluating the potential benefits of continued statin therapy on inflammatory parameters and sepsis among patients with pre-existing statin use 15 did not find clinical benefits with continuation. As such, there is a lack of consistent evidence regarding the appropriate exposure duration needed for statins to provide the greatest protective effects in bacteremic patients. The main objective of this study was to identify a duration of statin therapy continuation which minimized inpatient mortality among bacteremic patients.
Methods
A case-control study design was used to estimate a time breakpoint in statin continuation at which the highest clinical benefit would be seen in terms of survival (i.e. lowest inpatient mortality). This study was conducted using de-identified OptumClinformatics™ database (OptumInsight, Eden Prairie, MN, USA) with matched Premier hospital data (1 October 2009 to 31 March 2013) among adult (⩾18 years) patients with a primary diagnosis of bacteremia during a hospital admission.
Adult patients with continuous enrollment for at least 6 months in the commercial health plan prior to hospital admission were included. Patients were included if they were hospitalized between 1 April 2010 and 31 March 2013 with a primary diagnosis of bacteremia or septicemia [International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM) codes 003.1, 020.2, 022.3, 036.2, 038.0, 038.1, 038.10–038.12, 038.19, 038.2, 038.3, 038.40–038.44, 038.49, 038.8, 038.9, 054.5, 449, 771.81, 995.91, 995.92, 790.7] 16 by any causative organism. We excluded patients who, on the first 3 days after hospital admission, did not receive a minimum of two successive days of at least one antibiotic therapy that would be used to treat bacteremia.17–20 The index date was defined as the date of the first hospital admission during the study period, and subsequent hospital admissions were not considered for the analysis. From this cohort, only patients with a minimum of 90 days of continuous statin use in the 90 days prior to admission were selected for inclusion to capture prevalent and adherent statin users (Figure 1). The Charlson comorbidity index and chronic comorbidities were captured from ICD-9-CM codes in the 6 months prior to admission and during the index admission. 21

Case-control study design.
Cases included those who died during the admission. Controls were selected from survivors of the same cohort of adult patients who had a primary diagnosis of bacteremia on hospital admission and received antibiotic therapy. Controls were matched to cases on disease risk scores (DRSs). 22 DRS is a confounder summary method, which can be used in case-control studies to control for confounding by calculating the predicated probability of an outcome in the absence of exposure.23,24 The stratified DRS is a retrospective balancing score and therefore, it works in a similar manner in case-control studies as the propensity score works in cohort studies. While propensity score models predict the probability of exposure, DRSs predict the probability of the outcome, which in our study was mortality. 24 DRSs were calculated using logistic regression. The c-statistic for the final DRS model was 0.91. The full DRS model equation can be found in the footnote of Figure 2. Using nearest neighbor matching within a caliper of 0.25 distance, a single control without replacement was selected for each case. 25 We checked DRS balance between cases and controls using graphical displays (Figure 2).

Disease risk scores (DRS) distribution among cases and controls.
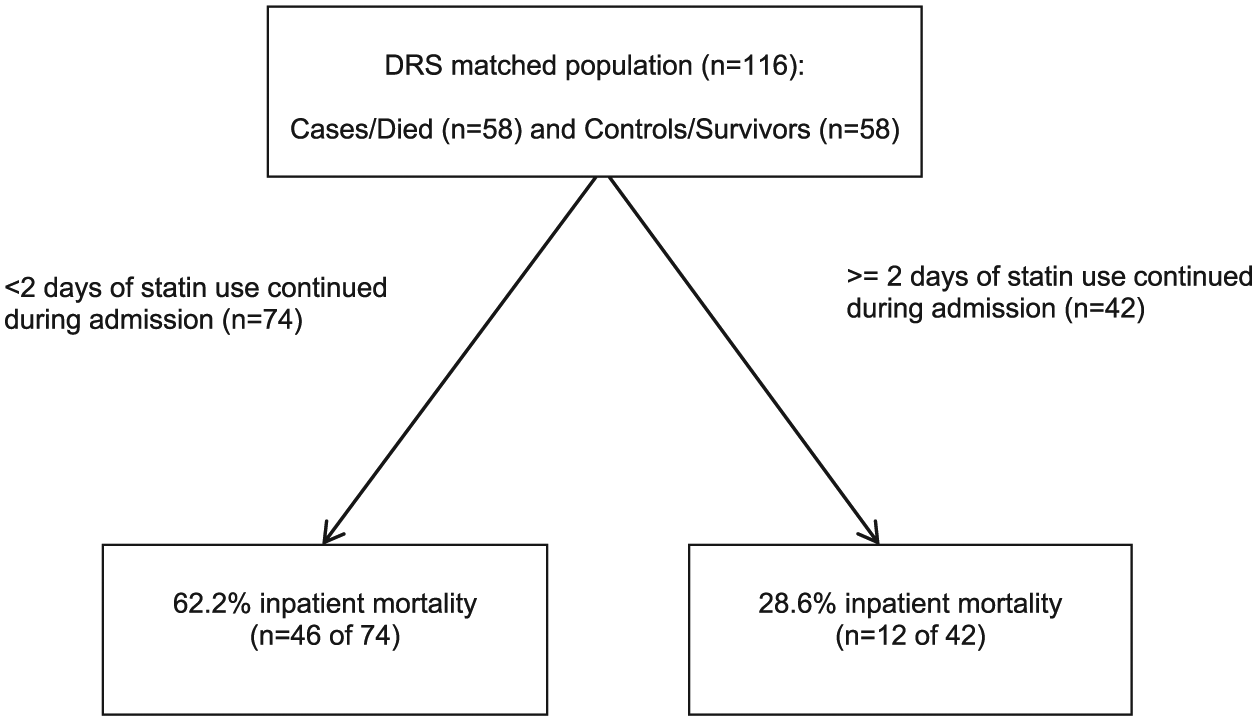
Among patients with at least 90 days of statin therapy in the 90 days prior to admission (proportion of days covered 100% for all patients), the primary exposure of interest was the number of days of continued statin use during admission. The statins included were atorvastatin, fluvastatin, lovastatin, pravastatin, rosuvastatin, and simvastatin. A 1 day gap in therapy was allowed, but the gap was not counted in the calculation of the statin use period. To partition statin continuation days associated with the greatest survival benefit, we conducted a classification and regression tree analysis (CART).26,27 The CART analysis, which includes an optimal tree selection based on pruning and cross-validation, identified subsets of patients at lowest risk of death based on days of statin continuation. CART models are useful because of their non-parametric, non-linear structure. 27 The trees were automatically developed to forecast inpatient mortality by considering every possible cut-point on statin continuation duration at every node in the classification tree. Based on the split provided by the CART analysis, conditional logistic regression was conducted to calculate the odds of mortality. Statistical analyses were conducted using SAS version 9.4 (SAS Institute, Cary, NC, USA) and R software version 3.2.0 (The R Foundation for Statistical Computing) with a recursive partitioning technique “rpart” package that was developed for Splus (Insightful Corporation, Seattle, WA, USA). 28 This study was reviewed and approved as exempt by the University of Rhode Island’s Institutional Review Board.
Results
Among our study population of prevalent and adherent statin users in the 90 days prior to admission, 61 (6.9%) patients died and 821 (93.1%) survived their hospital stay. Using DRS matching, controls were identified for 58 cases. Due to matching, baseline characteristics were similar between cases and controls, including age (median 68 versus 67 years, p = 0.8520; Table 1), gender (39.7% versus 43.1% females, p = 0.7992), race (20.7% versus 15.5% non-whites, p = 0.7637), and Charlson comorbidity score during the admission (median 4 versus 4, p = 0.8239) and in the 6 months prior to admission (median 4 versus 4, p = 0.4959). The length of hospital stay was significantly longer among controls compared to cases (median = 9 versus 5 days, p = 0.0005). Of the 47 (41%) patients who continued statin use during the hospital admission, 32% (n = 15) were cases and 68% (n = 32) were controls. The average statin therapy duration during admission among cases and controls was 1.5 ± 3.7 versus 4.5 ± 7.5 days, respectively.
Demographic and hospitalization-related characteristics in cases and controls.
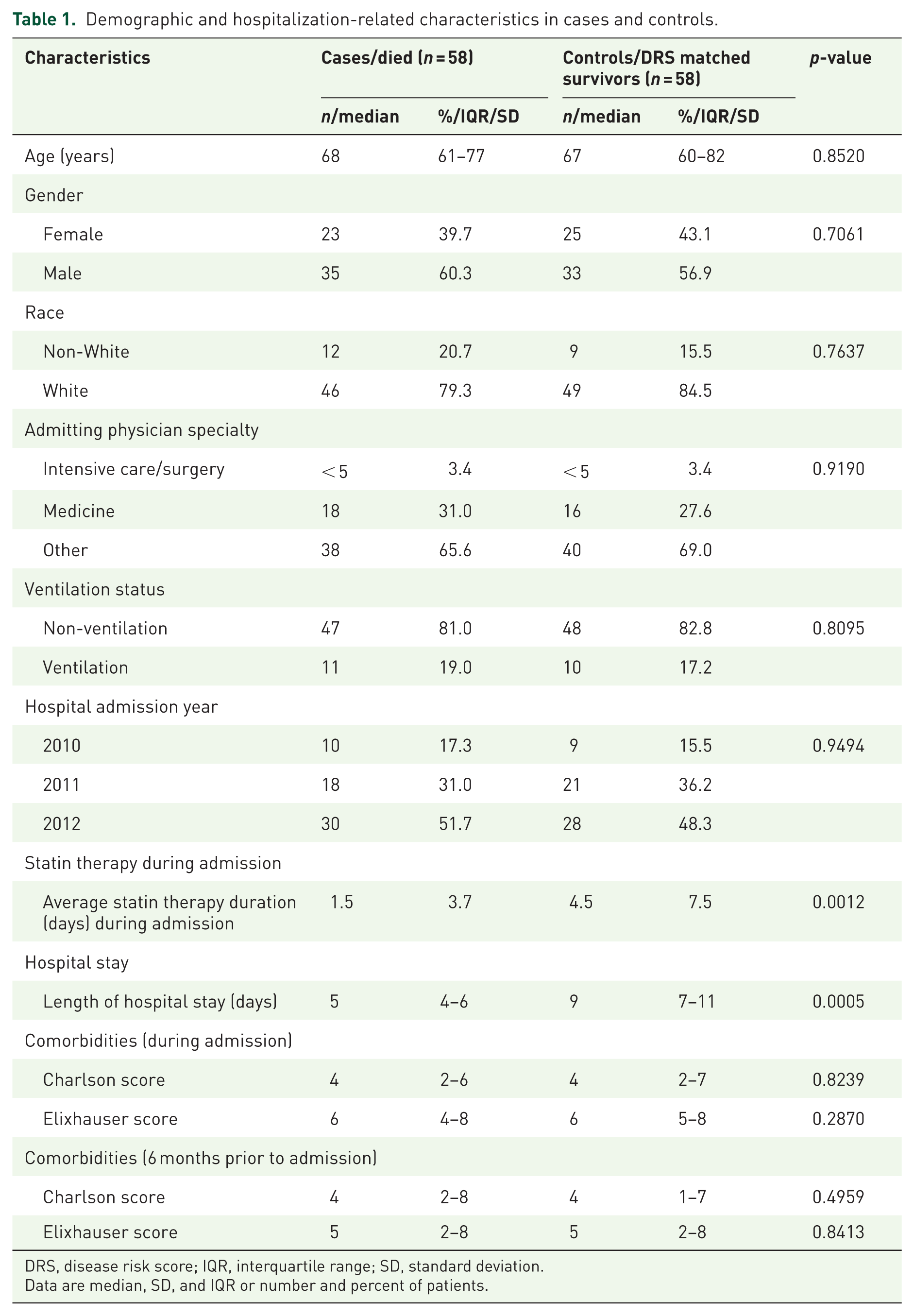
DRS, disease risk score; IQR, interquartile range; SD, standard deviation.
Data are median, SD, and IQR or number and percent of patients.
The study included an equal number (n = 58) of cases and controls, producing a 50% survival rate at the root node. The CART analysis partitioned the dependent variable of statin therapy duration at ⩾2 days. Those continuing statin therapy for at least 2 days had a survival probability of 71.4%, while those not continuing or only continuing for 1 day had a survival probability of 37.8% (Figure 3). The odds of inpatient mortality was 76% lower among those continuing statin therapy for at least 2 days (odds ratio (OR) 0.24, 95% confidence interval (CI) 0.11–0.55).
Partitioning from CART analysis.
Discussion
In this DRS matched case-control study, we identified a statin continuation duration threshold providing the maximum survival benefit among bacteremic patients. Our findings are consistent with existing literature3,4,9 evaluating this association, but we expanded these findings to identify an optimal statin duration of at least 2 days. Although other studies have observed similar protective effects with statin continuation,3,4,9 as we observed in our study, duration of pre-admission statin therapy, with and without continuation, not only varied between these studies but also within studies. In considering our findings with those from previous studies, the period of statin exposure is directly related to crucial inflammatory periods, including as the infection develops (pre-admission statin exposure) and the time period right after admission when antibiotics are begun (continued statin exposure for at least those first 2 days).29,30
A retrospective cohort study among bacteremic patients from a 300-bed acute care hospital in Ipswich, Australia found a reduced adjusted hospital mortality rate (OR: 0.39, 95% CI: 0.17–0.91, p = 0.029) in those taking statins prior to admission (n = 66), which decreased even further with the continuation of statins (n = 56) during the admission (OR: 0.06, 95% CI: 0.01–0.44, p = 0.0056) compared to patients not receiving statins (n = 372). 3 Pre-admission statin use was based on medication use reported at admission, and therefore, duration of prior statin use was not assessed. Similar effect estimates were observed when restricting the analysis to death attributable to bacteremia (statin use only before admission: OR: 0.29, 95% CI: 0.10–0.86, p = 0.025; continued during admission: OR: 0.09, 95% CI: 0.01–0.64, p = 0.016). 3 Another retrospective cohort study 4 conducted among bacteremic patients taking a statin at the time of admission and continuing throughout the hospitalization (n = 35) at a Veterans Affairs Medical Center in Washington, identified a therapeutic benefit with statin continuation (adjusted OR: 0.13, 95% CI: 0.02–0.99) compared with patients not taking statins (n = 353). Again, duration of pre-admission statin use was not assessed.
These results should be taken into consideration with clinical judgment with regard to safety as they can contribute to liver dysfunction and life-threatening rhabdomyolysis.31,32 Moreover, elevated statin levels have been observed in critical illness, possibly related to pharmacokinetic and pharmacodynamic changes during sepsis but also due to concomitantly prescribed medications with cytochrome P450 inhibition of statin metabolism.33,34 Among critically ill patients on continued statins, monitoring of liver function and creatine phosphokinase may be warranted.
Our study has a number of limitations. First, we were unable to assess statin drug or dose-dependent effects that might affect bacteremic mortality. Second, our study relied on a claims database, which is subject to misclassification due to coding selected for medical claims processing and reimbursement. Third, we could not study differences in mortality by causative pathogen. A previous study 24 observed greater protection with statins in Staphylococcus aureus bacteremia compared to bacteremia caused by Gram-negative bacilli, while also suggesting greater survival in nosocomial versus community-associated bacteremia. 24 Our study could not evaluate these differences. We also could not distinguish bacteremic severity or changes in oral intake; however, we incorporated potential causative pathogen proxies (identified using ICD-9 codes) in our DRS model, as well as sepsis and ventilation status proxies from diagnosis-related group codes. Moreover, the sample size of our study was small. While we included antibiotic treatment in the DRS mode, we could not evaluate the appropriateness of antibiotic therapy. The intensity of statin therapy was also not assessed. In addition, the limitations of CART analysis include an inability to fully describe the observed data due to uncertainty that remains in the prediction of the model and potential existence of multiple threshold values despite a single “optimal” split. 35 Finally, since statin use prior to admission was captured from pharmacy dispensing, misclassification due to non-adherence may have impacted our findings.
In conclusion, we found that continuation of statins for at least 2 days in prevalent, adherent statin users, significantly reduced hospital mortality in our DRS matched case-control study conducted in a real-world clinical population. To further understand the relationship between statin use and improved clinical outcomes among those with serious infections, future research should assess drug/dose-response, while accounting for duration-response. Although our findings indicate benefits with continuation of statins during admission, greater information is needed regarding the risks of continuation, in terms of adverse events, to enable a clear benefit–risk assessment.
Footnotes
Author’s Note
The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the U.S. Department of Veterans Affairs.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ajinkya M. Pawar has no conflicts to disclose. Tristan T. Timbrook, has received honoraria for speaking and/or consulting from BioFire Diagnostics, GenMark Diagnostics, and Roche Diagnostics. Kerry L. LaPlante, has received research funding or acted as a scientific advisor for Allergan, Bard, Merck (Cubist), Pfizer, and The Medicines Company. Aisling R. Caffrey, has received research funding from Pfizer Inc, Merck (Cubist), and The Medicines Company.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
