Abstract
Background:
Considering the therapeutic difficulties and mortality associated with bloodstream infection (BSI), it is essential to investigate other potential factors affecting mortality in critically ill patients with BSI and examine the utility of the quick Pitt bacteremia (qPitt) score to improve the survival rate.
Objectives:
To improve the predictive accuracy of the qPitt scoring system by evaluating the five current components of qPitt and including other potential factors influencing mortality in critically ill patients with BSI.
Design:
This was a retrospective cohort study.
Methods:
Medical information from the Medical Information Mart for Intensive Care IV database was used in this retrospective cohort study. The risk factors associated with mortality were examined using a multivariate logistic regression model. The area under the receiver operating characteristic curve (AUC) was used to assess the discriminatory capability of the prediction models.
Results:
In total, 1240 eligible critically ill patients with BSI were included. After adjustment for age, community-onset BSI, indwelling invasive lines, and Glasgow Coma Scale (GCS) ⩽ 8, acute kidney injury (AKI) was identified as a notable risk factor for 14-day mortality. Except for altered mental status, the four other main components of the original qPitt were significantly associated with 14-day mortality. Hence, we established a modified qPitt (m-qPitt) by adding AKI and replacing altered mental status with GCS ⩽ 8. The AUCs for m-qPitt and qPitt were 0.723 [95% confidence interval (CI): 0.683–0.759] and 0.708 (95% CI: 0.669–0.745) in predicting 14-day mortality, respectively. Moreover, m-qPitt also had acceptable performance and discrimination power [0.700 (95% CI: 0.666–0.732)] in predicting 28-day mortality.
Conclusion:
AKI significantly influenced the survival of critically ill patients with BSIs. Compared with the original qPitt, our new m-qPitt was proven to have a better predictive performance for mortality in critically ill patients with BSI. Further studies should be conducted to validate the practicality of m-qPitt.
Keywords
Introduction
In the last few decades, bloodstream infections (BSIs) have been proven to be one of the leading causes of morbidity and mortality in critically ill patients. The estimated death rate of patients with BSI ranges from approximately 17% to 60%, and varies across different organisms.1,2
To identify critically ill patients at high risk of death, several scoring systems have been designed and applied to evaluate the severity of BSI and other infections, such as the quick Sequential Organ Failure Assessment (qSOFA) and Systemic Inflammatory Response Syndrome (SIRS) scores.3,4 Known as a useful BSI prognosis prediction algorithm, the quick Pitt bacteremia (qPitt) score is widely used to predict mortality in patients with BSI because of its convenience and reliability. The qPitt score consists of five binary variables, with one point allocated for each, and a qPitt score of 2 is considered a reasonable cutoff value to assess the acute severity of illness. 5
However, various risk factors have not been included in the qPitt score but are associated with mortality in BSI. It is essential to further modify the qPitt score by evaluating suspected meaningful variables such as the occurrence of acute kidney injury (AKI). As the most common and threatening complication of severe infections, such as sepsis and BSI, AKI has been verified as an independent influencing factor associated with death in critically ill patients.6–10 AKI is strongly associated with poor clinical outcomes, including a higher probability of in-hospital death and longer duration of hospital stay, especially in critically ill patients. 11 Moreover, although there are several studies designed to elucidate the practicability of the qPitt score in predicting the prognosis of BSI caused by different organisms and even non-bacteremia carbapenem-resistant Enterobacteriaceae infections, there is still a lack of comprehensive large-scale clinical studies covering both Gram-negative and Gram-positive BSI.5,12,13
To this end, we carried out a retrospective cohort study in critically ill patients with BSI to validate the clinical utility of the qPitt score and identify any other potential variables affecting the prognosis of BSI to modify the criteria of the qPitt score based on the Medical Information Mart for Intensive Care IV (MIMIC-IV) database.
Methods
Study design and participants
This was a retrospective cohort study that used the MIMIC-IV (version 2.0) database. The MIMIC-IV database is a large, single-center, freely accessible database consisting of comprehensive information on more than 40,000 intensive care unit (ICU) patients across the United States from 2008 to 2019. 14 This study conformed to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 15 All consecutive hospitalized patients diagnosed with BSI in the MIMIC-IV database were enrolled in our study. Patients were included only once, when an initial episode of BSI was identified during hospitalization. The exclusion criteria were as follows: (1) age <18 years and (2) missing data.
Study objectives
The primary aim of the current study was to determine the risk factors affecting 14-day all-cause mortality in patients with BSI, especially to appraise the predictive ability of the qPitt score and modify the specific criteria of individual components from the qPitt score if necessary. Moreover, the 28-day all-cause mortality was examined as a secondary outcome.
Definitions
All definitions used in the study were ascertained prior to data collection. BSI was defined as the isolation of any Gram-positive or Gram-negative bacillus from an index blood culture. The site of BSI acquisition was categorized as community-onset if the index blood culture was collected within 48 h of admission; otherwise, it was categorized as hospital-onset. Multidrug-resistant organisms (MDROs) included the following five common drug-resistant pathogens for critically ill patients: carbapenem-resistant Acinetobacter baumannii (CRAB), carbapenem-resistant Pseudomonas aeruginosa, carbapenem-resistant Enterobacterales, vancomycin-resistant Enterococcus, and methicillin-resistant Staphylococcus aureus, in view of the World Health Organization global priority pathogens list. 16 Immunocompromised status was defined as the presence of at least one of the following: neutropenia (absolute neutrophil count < 500/μL), use of chemotherapy, diagnosis of Acquired Immune Deficiency Syndrome, prior organ or bone marrow transplantation, corticosteroid treatment (prednisone at a daily dose of 20 mg or higher for at least 7 days), or other immunosuppressive medications.17,18 AKI severity was staged according to the Kidney Disease Improving Global Outcomes (KDIGO) classification system. 19 The individual components of the qPitt criteria are summarized in Table 1. 5 Altered mental status was defined as a Glasgow Coma Scale (GCS) score of less than 15 points. The qSOFA and SIRS scores were graded according to their own criteria.3,4
The qPitt score criteria.

The worst (highest or lowest) variable is collected in the 24 h leading to and 24 h following collection of index blood culture.
qPitt, quick Pitt bacteremia; SBP, systolic blood pressure.
Data collection for potential predictors
In this study, we used the Navicat Premium software (PremiumSoft CyberTech Ltd., Hong Kong SAR, version 16.3.5) and Structure Query Language (SQL) with PostgresSQL (PostgresSQL Global Development Group, U.S., version 14.2) for data collection and management. Candidate prediction variables were all extracted from the MIMIC-IV database for further analysis, including patient demographics (age, gender, weight), BSI information (time to diagnosis of BSI after admission, Gram-positive or Gram-negative infection, and MDRO infection), comorbidities, Charlson Comorbidity Index (CCI) score, immunocompromised status, indwelling invasive lines, AKI variables [AKI stage and continuous renal replacement therapy (CRRT)], scoring system (GCS, qPitt, qSOFA, and SIRS), and qPitt variables (hypothermia, hypotension, respiratory failure, cardiac arrest, and altered mental status). All variables were recorded within 24 h prior to or 24 h following the collection of index blood cultures. If a variable was recorded more than once during the aforementioned period, only the value associated with the greatest illness severity was retained.
Statistical analysis
To establish a final multivariate logistic regression model to identify the risk factors associated with our primary outcome, all potential variables were assessed using univariate analysis. The distribution normality of each variable was verified using the Shapiro–Wilk test. For p value calculations, Pearson’s chi-square test or Fisher’s exact test was used to analyze categorical variables, whereas Student’s t-test or Mann–Whitney U-test was used to evaluate continuous variables. We separated the qPitt score into five individual components to determine the independent association between these components and 14-day mortality. Potential variables related to 14-day mortality in univariate analysis (p value < 0.05) were included in the multivariate analysis with forward stepwise selection, whereas only variables with p-values < 0.05 were retained in the final model. Odds ratios with 95% confidence intervals (CIs) were used to estimate each variable’s strength of association with 14-day mortality.
Subsequently, a derivative scoring system, the modified qPitt score (m-qPitt), was established based on qPitt individual components and other significant predictors of 14-day mortality. We then used the area under the receiver operating characteristic curve (AUC) and the Youden index (sensitivity + specificity − 1) to determine the most appropriate dichotomous cut-off value for m-qPitt.
To quantify the association between different scoring systems (m-qPitt, qPitt, qSOFA, and SIRS) and 14-day mortality, each scoring system was considered integrally as a continuous variable to develop logistic regression models with other independent risk factors for 14-day mortality from the previous model. AUC values were computed to assess and compare the discrimination power of the different scores. The AUC comparison was conducted using the Delong test. 20
As for the secondary outcome of our study, the AUC calculation process was utilized to analyze the discrimination ability of m-qPitt in predicting 28-day mortality as well. Finally, Kaplan–Meier survival curves stratified by the cutoff value of m-qPitt were produced to analyze 14-day and 28-day mortality, and the Bonferroni-based log-rank test was applied for comparison.
All statistical analyses were performed using JMP Pro (version 16.2.0; SAS Institute Inc., Cary, NC, USA). Statistical significance was set at p value < 0.05 (two-tailed).
Results
Demographics and clinical characteristics
After screening the data of 3215 patients with BSI, 1240 were eligible for our study (Figure 1). The median age of the patients was 64 years and 62.8% were male. A total of 514 (41.5%) patients had community-onset BSIs. The proportion of Gram-negative BSI was 35.6%, and nearly 40% of the BSIs were attributed to MDROs. Overall, congestive heart failure (32.7%), renal disease (27.7%), and diabetes without chronic complications (265%) were the three main comorbidities. The median CCI score was 6 (interquartile range, 3–8). Approximately 15% of the patients were immunocompromised when diagnosed with BSI. More than half (52.0%) of the patients had invasive indwelling lines. Regarding kidney function, 851 (68.6%) patients had AKI at various stages, and 95 (7.7%) patients received CRRT. The 14-day and 28-day mortality rates were 15.3% and 23.6%, respectively (Table 2).

Study design.
Clinical characteristics of patients with bloodstream infection.

All data are exhibited as number (%) or median (P25–P75).
AKI, acute kidney injury; BSI, bloodstream infection; CCI, Charlson comorbidity index; CRRT, continuous renal replacement therapy; GCS, Glasgow Coma Scale; KDIGO, Kidney Disease Improving Global Outcomes; MDRO, multidrug-resistant organism; qPitt, quick Pitt bacteremia score; qSOFA, quick Sequential Organ Failure Assessment; SBP, systolic blood pressure; SIRS, Systemic Inflammatory Response Syndrome score.
Association with qPitt individual components and 14-day mortality
Univariate analysis revealed that age, community-onset BSI, indwelling invasive lines, AKI, and four individual variables in the qPitt score (except altered mental status) were significantly associated with 14-day mortality. To further investigate the magnitude of mental status in predicting 14-day mortality, the GCS score was redefined as another potential dichotomous variable and included in the univariate analysis. As a result, we found that GCS ⩽ 8 was a notable variable related to a higher rate of 14-day mortality in univariate analysis (Table 3). Multivariate logistic regression analysis was conducted to determine whether the above-mentioned variables were significant risk factors for predicting 14-day mortality in patients with BSI (Table 4).
Univariate logistic regression analysis for risk factors of 14-day mortality in BSI.

All data are exhibited as number (%) or median (P25–P75).
p Value <0.05.
AKI, acute kidney injury; BSI, bloodstream infection; CCI, Charlson comorbidity index; CI, confidence interval; CRRT, continuous renal replacement therapy; GCS, Glasgow Coma Scale; KDIGO, Kidney Disease Improving Global Outcomes; MDROs, multidrug-resistant organisms; OR, odds ratio; SBP, systolic blood pressure.
Multivariate logistic regression analysis for risk factors of 14-day mortality in BSI.

AKI, acute kidney injury; BSI, bloodstream infection; CI, confidence interval; GCS, Glasgow Coma Scale; KDIGO, Kidney Disease Improving Global Outcomes; OR, odds ratio; SBP, systolic blood pressure.
Development and evaluation of m-qPitt in predicting 14-day mortality
Multivariate analysis revealed that altered mental status had a minor effect on BSI patients’ 14-day mortality as one of the previous qPitt individual components, compared to GCS ⩽ 8. Furthermore, we have already substantiated that the incidence of AKI was significantly associated with 14-day mortality, as described above, and AKI could be detected in a timely manner by monitoring serum creatinine (SCr) levels and urine output at the time of the first episode of BSI. Hence, the modified version of the qPitt score (m-qPitt) was derived with an additional predictive component concentrating on AKI, as well as adjusted for altered mental status (Table 5).
The m-qPitt score.

The worst (highest or lowest) variable is collected in the 24 h leading to and 24 h following collection of index blood culture.
AKI, acute kidney injury; GCS, Glasgow Coma Scale; m-qPitt, modified quick Pitt bacteremia; SBP, systolic blood pressure.
Receiver operating characteristic curve analysis showed that the AUC value was 0.723 (95% CI: 0.683–0.759) for m-qPitt and 0.708 (95% CI: 0.669–0.745, p value 0.093) for qPitt, whereas lower AUC values were observed for qSOFA [0.700 (95% CI: 0.659–0.738, p value 0.128)] and SIRS [0.698 (95% CI 0.658–0.734, p value 0.088)], respectively, which suggested that qPitt had the optimal discrimination power for predicting 14-day mortality in patients with BSI (Figure 2).

Receiver operating characteristic curve analysis of the m-qPitt score for predicting 14-day mortality of critically ill patients with bloodstream infection. The area under the curve was 0.723 (95% CI: 0.669–0.745) for m-qPitt and 0.700 (95% CI: 0.659–0.738) for qPitt.
To select an appropriate cutoff value for m-qPitt, we also analyzed the performance characteristics of m-qPitt for predicting 14-day mortality at different total points. Using the maximal Youden index, the optimal dichotomous m-qPitt cutoff value was determined to be 3 points (Table 6).
Performance characteristics of m-qPitt in predicting 14-day mortality at different cut-off values.

Youden index = Sensitivity + Specificity − 1.
m-qPitt, modified quick Pitt bacteremia; NPV, negative predictive value; PPV, positive predictive value; Yi, Youden index.
Lastly, the Kaplan–Meier survival curves showed a notably lower 14-day survival rate in patients with a higher m-qPitt score stratified by our selected cut-off value, namely m-qPitt ⩾ 3 (Figure 3).

The Kaplan–Meier 14-day survival curve of critically ill patients with bloodstream infection stratified by the m-qPitt score at the cut-off value (m-qPitt ⩾ 3). Log-rank test: p value < 0.001.
Application of m-qPitt in predicting 28-day mortality
In comparison with the AUC values of qPitt [0.662 (95% CI: 0.626–0.695, p-value < 0.001)], qSOFA [0.641 (95% CI: 0.605–0.675, p-value < 0.001)], and SIRS [0.636 (95% CI: 0.600–0.670, p-value < 0.001)], the m-qPitt [0.700 (95% CI: 0.666–0.732)] also had an acceptable performance and a favorable discrimination power in predicting 28-day mortality for BSI patients (Figure 4). In addition, the results of the survival analysis for 28-day mortality showed that an individual BSI patient with a total score of m-qPitt ⩾ 3 had a higher risk of death, which was similar to the results for 14-day mortality (Figure 5).
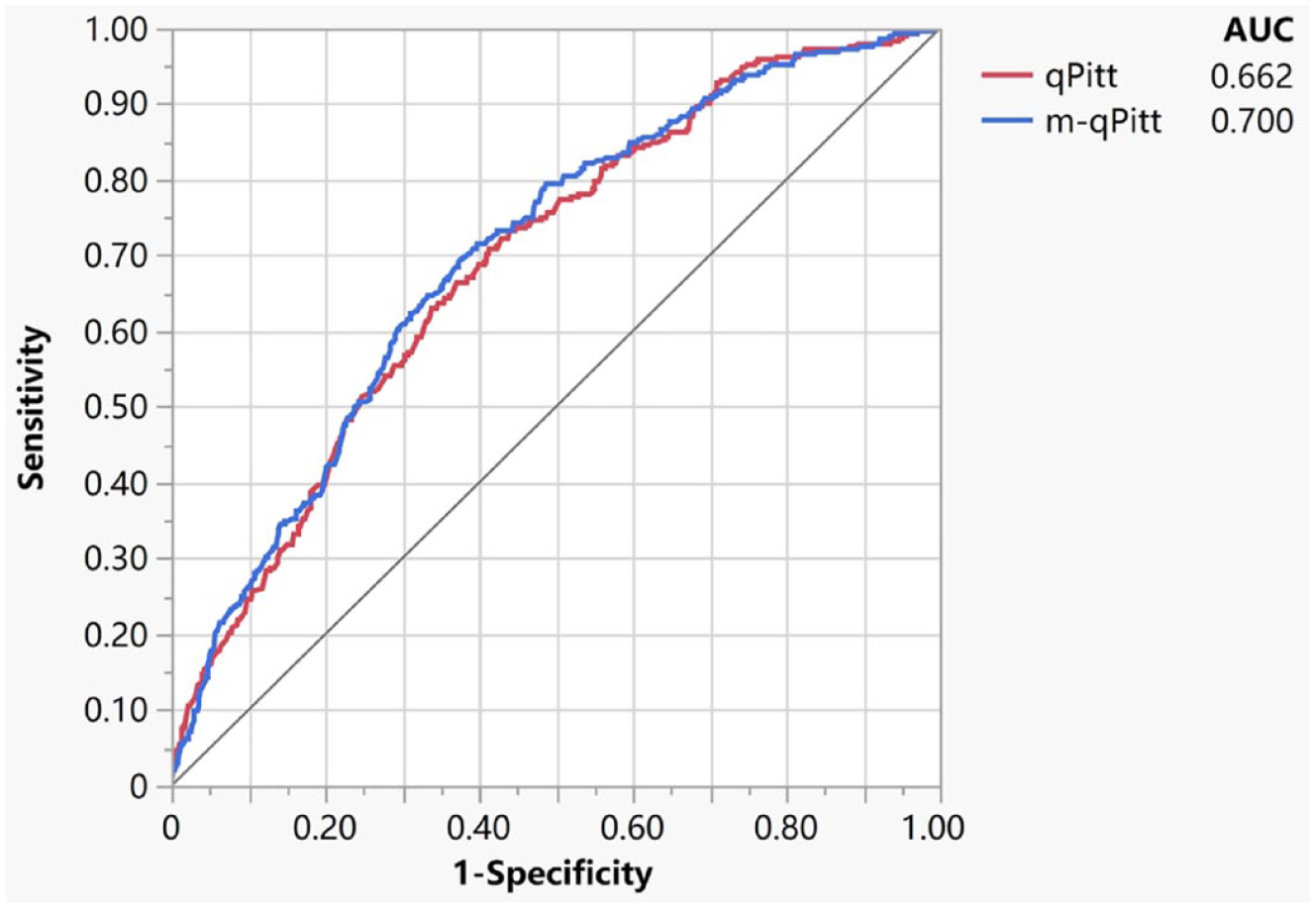
Receiver operating characteristic curve analysis of the m-qPitt score for predicting 28-day mortality of critically ill patients with bloodstream infection. The area under the curve was 0.700 (95% CI: 0.666–0.732) for m-qPitt and 0.662 (95% CI: 0.626–0.695) for qPitt.

The Kaplan–Meier 28-day survival curve of critically ill patients with bloodstream infection stratified by the m-qPitt score at the cut-off value (m-qPitt ⩾ 3). Log-rank test: p-value < 0.001.
Discussion
To the best of our knowledge, this is the first comprehensive investigation to evaluate the clinical efficacy of qPitt in predicting mortality in patients with Gram-negative and Gram-positive BSI. The 14-day and 28-day mortality rates in our study were 15.3% and 23.6%, respectively, while similar results were observed in previous studies.2,21,22 We identified significant risk factors related to 14-day all-cause mortality in patients with BSI and developed the m-qPitt, a practical and reliable scoring system with four components (hypothermia, hypotension, respiratory failure, and cardiac arrest) from the original qPitt, which had notably better discrimination ability than the original qPitt and other scoring systems in predicting 14-day and 28-day mortality. An m-qPitt ⩾ 3 may be a suitable cut-off value in the clinical decision-making process to distinguish BSI patients with a high risk of death, which could provide reliable evidence for early antimicrobial treatment to improve life-saving potential as much as possible. Interestingly, m-qPitt had a significantly better performance in predicting 28-day mortality than in predicting 14-day mortality in critically ill patients in terms of the results of the Delong test.
One of the main highlights of the present study was the discovery that AKI played an important role in predicting mortality in patients with BSI. The incidence of AKI was shown to increase the risk of all-cause mortality 2.65 times in patients with BSI. This was similar to the conclusions of several previous studies, in which deteriorated renal function was a crucial prognostic factor affecting mortality in patients with severe infections, including BSI.23–25 Westgeest et al. 9 conducted a multicenter retrospective cohort study and found that AKI development was highly associated with 30-day mortality in patients with S. aureus bacteremia. Moreover, patients with CRAB complex bacteremia had poor clinical outcomes when they had AKI. 26 Therefore, it is recommended to evaluate the occurrence of AKI in m-qPitt since dynamic surveillance of SCr level and urine output volume is available and rapid by point-of-care testing in daily clinical practice. Nevertheless, evaluating SCr levels and urine output volume remains more time-consuming than evaluating other physiological parameters. To extend the scope of m-qPitt application as much as possible, it is vital to shorten the delay time to identify the occurrence of AKI by utilizing novel rapid techniques or new exogenous biomarkers.
The GCS is widely considered an effective standard for evaluating the severity of unconsciousness because of its objectivity and convenience. 27 We also sought to emphasize the importance of the GCS score in predicting BSI mortality. According to our findings, we have modified the original qPitt component from altered mental status to GCS ⩽ 8 to improve the predictive performance of m-qPitt, whereas GCS ⩽ 8 is usually used to define coma. 28 This means that a much more seriously altered mentation might be preferable for predicting BSI patient mortality. However, most patients with GCS ⩽ 8 will probably be considered critically ill and transferred to the ICU. Therefore, quite a few non-ICU patients may not meet the criteria of GCS ⩽ 8, whereas we have only enrolled ICU patients in our study. Another arrestive point is the potential overlapping phenomenon of patients with GCS ⩽ 8 and patients with respiratory failure requiring mechanical ventilation. Mechanical ventilation is usually accompanied by the use of sedatives and neuromuscular blocking agents, which can iatrogenically induce the emergence of GCS ⩽ 8. Given that altered mental status was still adopted in several scoring systems for critically ill patients (such as qSOFA), the aforesaid perspectives should be demonstrated in our next-step studies to determine whether GCS ⩽ 8 would be the suitable cut-off value for both ICU and non-ICU patients with BSI, as well as distinguishing GCS ⩽ 8 from respiratory failure requiring intubation in other ways, including but not limited to performing GCS score evaluation prior to mechanical ventilation.
Another intriguing point in our study was that a higher mortality rate was observed in patients diagnosed with community-onset BSI, which has rarely been mentioned in the literature. One possible reason is that we could not accurately differentiate community- and healthcare-associated BSI from community-onset BSI owing to the lack of corresponding information to match the criteria for classifying healthcare-associated BSI from the MIMIC-IV database. 29 Accumulating evidence suggests that healthcare-associated BSI has distinct characteristics and epidemiology, with underlying diseases, sources of infection, causative pathogens, and clinical outcomes resembling those observed in hospital-onset BSI rather than community-associated BSI.30–33 Hence, we did not add this potential variable to m-qPitt for BSI mortality prediction.
Considering the significance of the immune system in our understanding of the pathophysiological mechanism of infection, it is not surprising that an immunocompromised status would be a biologically feasible and widely acknowledged risk factor for increasing mortality in critically ill patients with BSI. 34 However, we discovered that an immunocompromised status did not significantly influence the mortality of patients with BSI in the current study. Another multicenter cohort study including 907 patients with S. aureus bacteremia also found no significant difference between immunosuppressed and immunocompetent patients in terms of overall in-hospital mortality. 35 Not coincidentally, it has been reported that those who had sepsis and septic shock with an immunocompromised status did not have a higher mortality rate than non-immunocompromised patients. 36 Battle and Al-Hasan 37 thought that immunocompromised patients would more easily capture the clinicians’ attention and receive aggressive empirical antimicrobial treatments, which could be a plausible explanation for this phenomenon. This paradox may also be related to immunosuppression, which could inhibit the development of the sepsis cascade by lowering the pro-inflammatory chemokine and cytokine responses.38–40 Further clinical research is necessary to validate this perceived contradiction between immunosuppressed status and a lower mortality rate in a larger population.
Based on the results of earlier studies, bacteremia caused by MDROs can increase mortality, length of stay, and readmission rates.41–44 Notably, we found that the 14-day mortality in BSI caused by MDROs was slightly but not significantly higher than that ascribed to non-MDROs in our study. Dinh et al. conducted a retrospective study of BSI patients with spinal cord injury to identify the clinical risks of MDROs. They also found no significant differences in BSI mortality between patients infected by MDROs and non-MDROs. 45 This could be interpreted as a decrease in bacterial metabolic fitness stemming from antibiotic resistance, which could possibly attenuate virulence in pathogenic bacteria.46–48
Our study had several limitations. First, owing to the retrospective nature of the MIMIC-IV database, the quality of records could be influenced by missing data and input errors. A well-designed prospective study with a larger sample size should be conducted to validate the efficacy of m-qPitt. Second, potential contaminants of blood cultures were not identified and excluded from our study, such as coagulase-negative Staphylococcus or Corynebacterium species, which could threaten the reliability of our modified scoring system. Third, we did not evaluate the appropriateness and delay of antimicrobial treatment regimens, which might be another vital contributing factor to the final results. Fourth, laboratory parameters of hepatic function were not measured in this study because the current hepatic-associated scoring system could only reflect the liver function of patients with specific hepatic diseases. It is also inaccurate and inconvenient to evaluate hepatic function by monitoring the fluctuations in several hepatic parameters. In addition, there are other AKI classification criteria, including risk, injury, failure, loss of kidney function, and end-stage kidney disease classification and acute kidney injury network classification, besides KDIGO, which should be compared in our further investigation.
Conclusion
We identified multiple risk factors that may affect the survival of BSI patients through multivariate analysis. To enhance the predictive performance of qPitt, a novel scoring system (m-qPitt) has been optimized and developed based on our results and the original qPitt, which has acceptable capability and reliability for predicting mortality in critically ill patients with BSI. This could provide adequate evidence for the preliminary judgment of BSI prognosis and the early identification of high-risk BSI patients by clinicians and pharmacists. More importantly, the development of m-qPitt indicated that the incidence of AKI of any degree of severity was a crucial prognostic factor contributing to BSI mortality. Large-scale prospective studies should be conducted to investigate the clinical efficacy and applicability of m-qPitt for more BSI patients.
