Abstract
Background:
The incidence of multidrug-resistant (MDR) organisms is increasing along with mortality. Identifying risk factors for the development of MDR Gram-negative bacilli (GNB) bacteremia could greatly impact patient care and management.
Methods:
Data from the electronic health record of patients with GNB over 13-month period were collected at a single university medical center. Baseline demographic data, risk factor, microbiological data, recurrence of bacteremia, and mortality were recorded.
Results:
A total of 177 patients were included in the analysis. MDR GNB occurred in 46 patients (26%). The mortality rate in the MDR group was 34.8% compared to 13.7% in non-MDR group (
Conclusion:
Our study provides important information about the risk factors for the development of MDR GNB bacteremia and helps prognosticate patient with MDR GNB.
Introduction
Antibiotics remain one of the most significant health achievements to date. However, antimicrobial resistance is regarded as one of the greatest threats to human health. Studies have found resistance genes in 30,000-year-old frozen bacterial DNA in the Arctic and 4-million-year-old bacterial samples recovered from underground caves which were isolated from human exposure.1,2 Enhancing this evolutionary process is antibiotic exposure.
2
The widespread use of antimicrobials among humans and animals along with a globalized society and travel leads to spread of resistant strains worldwide.
3
More than one-third of community-associated infections caused by extended spectrum beta lactamase (ESBL) producing
Methods
This study was a retrospective observational cohort study conducted at the Saint Louis University Hospital in Missouri, USA. Study duration was from 1 July 2013 to 31 July 2014. Study objectives were to identify the incidence, risk factors, and outcome of multidrug-resistant GNB bacteremia. All patients aged 18 years and older with at least one episode of GNB bacteremia during a hospital admission were included in the study. There were no other exclusion criteria. If a patient had multiple admissions for GNB, they were included in the study as different episodes. However, if a patient developed recurrence of bacteremia during the same admission, it was considered as a single-patient episode. Patients were identified by electronic search for positive blood culture demonstrating GNB in the hospital’s microbiology datasets. At the time of study, our hospital was not using rapid diagnostic test to identify the organism. The study was approved by the Institutional Review Board of Saint Louis University Hospital.
Patients’ electronic health records were reviewed. Data collection included the following clinical variables: (a) age, gender, and race; (b) co-morbid conditions; (c) abdominal/thoracic surgery in last 4 weeks; (d) use of antibiotics in last 30 days; (e) source of infection; (f) length of hospitalization ⩾72 h before the onset of GNB; (g) use of a central venous catheter ⩾48 h before the onset of GNB; (h) use of mechanical ventilation for ⩾72 h before the onset of GNB; (i) use of total parenteral nutrition (TPN) ⩾72 h before GNB; (j) antimicrobial resistance patterns in GNB blood culture isolates; (k) antibiotics used to treat the infection (empiric and targeted); (l) appropriateness of empiric antibiotics as well as source control; (m) time to negative blood culture; and (n) mortality within 30 days.
Study definitions
Criteria from the CDC for MDR organism were applied and MDR Gram-negative bacteria were defined as (a) ESBL-producing
Previous antibiotic therapy was defined as systemic antibiotic use for at least 72 h in the preceding 30 days before onset of bacteremia.
Inappropriate empiric antibiotic therapy was defined as treatment regimen that did not include at least one antibiotic that was active
Recurrence of bacteremia was defined as the emergence of a positive blood culture with same GNB after ⩾1 negative blood culture and after an interval of ⩾7 days.
All-cause mortality was defined as death by any cause within 30 days of the onset of bacteremia.
Statistical analysis
Data were analyzed using SPSS version 22. For univariate analysis, odds ratio (OR) and 95% confidence interval (CI) were calculated. Chi-square test was used for
Results
Between 1 July 2013 and 31 July 2014, 180 episodes of GNB occurred. Drug susceptibility data were not available for three patients, so they were excluded from the data analysis.
Patient’s clinical characteristics are described in Table 1. Incidences of diabetes mellitus (DM), human immunodeficiency virus (HIV) infection, end-stage renal disease, and end-stage liver disease were 52 (29.4%), 5 (2.8%), 12 (6.8%), and 28 (15.8%), respectively. In all, 35 (19.8%) patients had hematologic malignancy, 40 (22.6%) had solid organ malignancy, and 51 (28.8%) received chemotherapy. A total of 90 (50.8%) patients had received antibiotics in 4 weeks before the onset of bacteremia. Additional baseline characteristics are described in Table 1.
Baseline clinical characteristics.

DM, diabetes mellitus; ESLD, end-stage liver disease; ESRD, end-stage renal disease; GI, gastrointestinal; GNR, Gram-negative rod; GU, genitourinary; HIV, human immunodeficiency virus; TPN, total parenteral nutrition.
Out of 177 patients with GNB, 46 (26%) patients had MDR GNB (Table 2), 90 (50.8%) patients had received antibiotics in 4 weeks before the onset of bacteremia, and 130 (73.4%) patients had received appropriate antibiotics. Median duration of antibiotics was 14 days. Median duration of bacteremia was 1 day with the range of 1–7 days. In all, 14 (7.9%) patients developed recurrence of bacteremia during same hospitalization. Overall mortality in our cohort was 19.2%.
Study outcome.

MDR, multidrug resistant.
The most frequently isolated organism was
Organisms with MDR
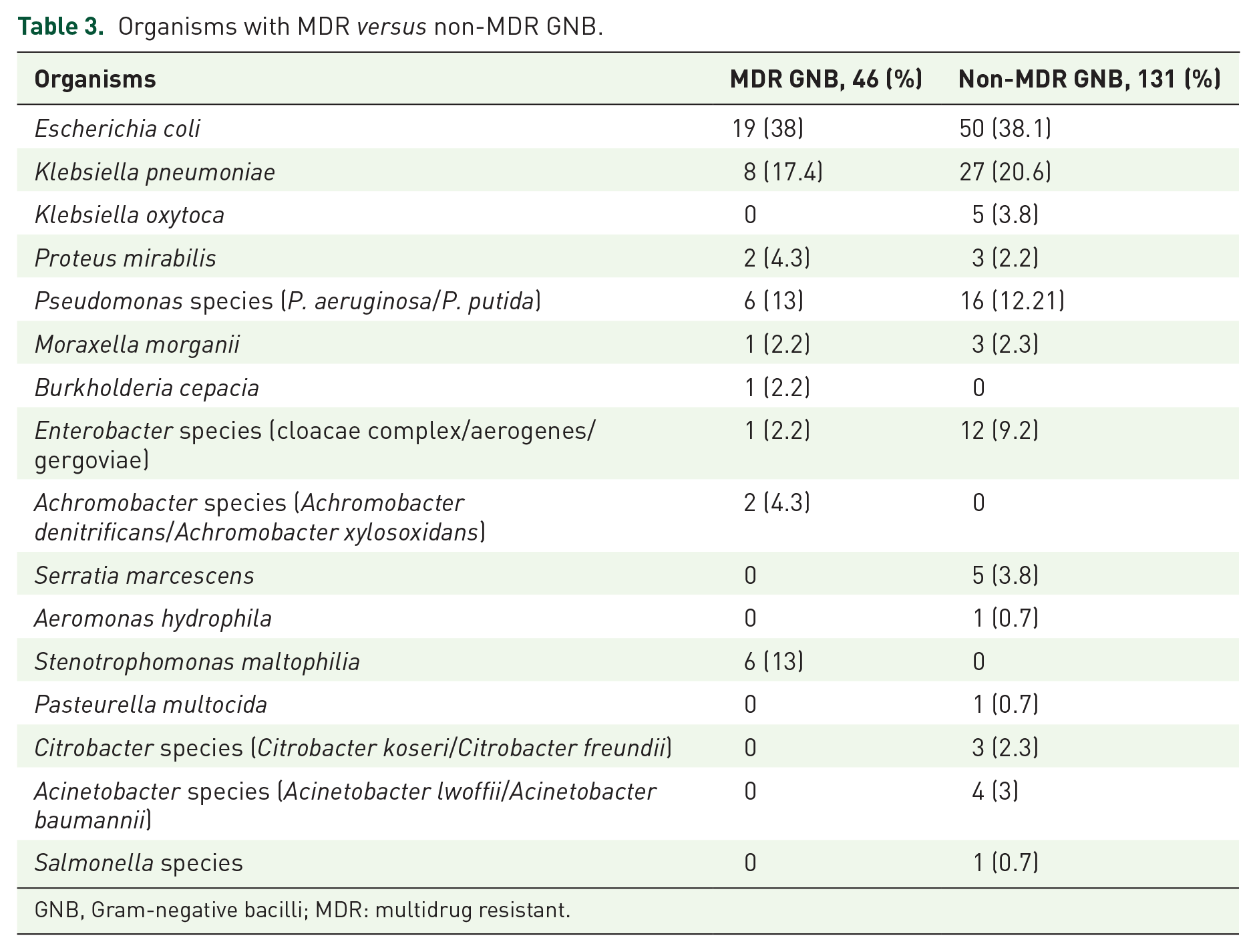
GNB, Gram-negative bacilli; MDR: multidrug resistant.
Previous antibiotic use (OR: 3.3, 95% CI: 1.6–6.8), urinary catheter as a source of infection (OR: 3.7, 95% CI: 1.3–10.4), and hospitalization ⩾72 h before the onset of GNB bacteremia (OR: 2, 95% CI: 1–4.2) were the significant factors for the MDR bacteremia (Table 4). In a multivariate analysis model, DM (OR: 2.8, 95% CI: 1–4.88), previous antibiotic use (OR: 2.93, 95% CI: 1.25–6.87), and urinary catheter as a source of infection (OR: 5.96, 95% CI: 1.78–19.94) were the significant risk factors for the MDR GNB bacteremia (Table 5).
Univariate analysis for MDR GNB bacteremia.

CI, confidence interval; GNB, Gram-negative bacilli; GNR, Gram-negative rod; MDR, multidrug resistant; OR, odds ratio.
Multivariate analysis of risk factors for MDR GNB bacteremia.

DM, diabetes mellitus; ESRD, end-stage renal disease; GNB, Gram-negative bacilli; MDR: multidrug resistant; TPN, total parenteral nutrition.
End-stage liver disease (OR: 2.89, 95% CI: 1.19–7), solid organ malignancy (OR: 3.7, 95% CI: 1.67–8.31), central venous catheter as a source of infection (OR: 0.18, 95% CI: 0.04–0.81), intra-abdominal infections (OR: 3.47, 95% CI: 1.6–7.52), hospital stay of ⩾72 h before the onset of GNB (OR: 2.4, 95% CI: 1.1–5.3), use of mechanical ventilation for ⩾72 h before the onset of GNB bacteremia (OR: 4.37, 95% CI: 1.46–13), inappropriate empiric antibiotics (OR: 4.42, 95% CI: 2–9.7), and MDR GNB (OR: 3.34, 95% CI: 1.52–7.33) were all significant factors for mortality. In a multivariate analysis model, end-stage liver disease, (OR: 3.64, 95% CI: 1.07–12.3), solid organ malignancy (OR: 3.64, 95% CI: 1.25–10.56), intra-abdominal source of infection (OR: 3.66, 95% CI: 1.14–11.73), inappropriate empiric antibiotics (OR: 7.59, 95% CI: 1.68–34.34), and urinary catheter as a source of infection (OR: 5.68, 95% CI: 1.37–23.5) were the significant risk factors for mortality from GNB (Table 6).
Multivariate analysis for mortality in patients with GNB bacteremia.
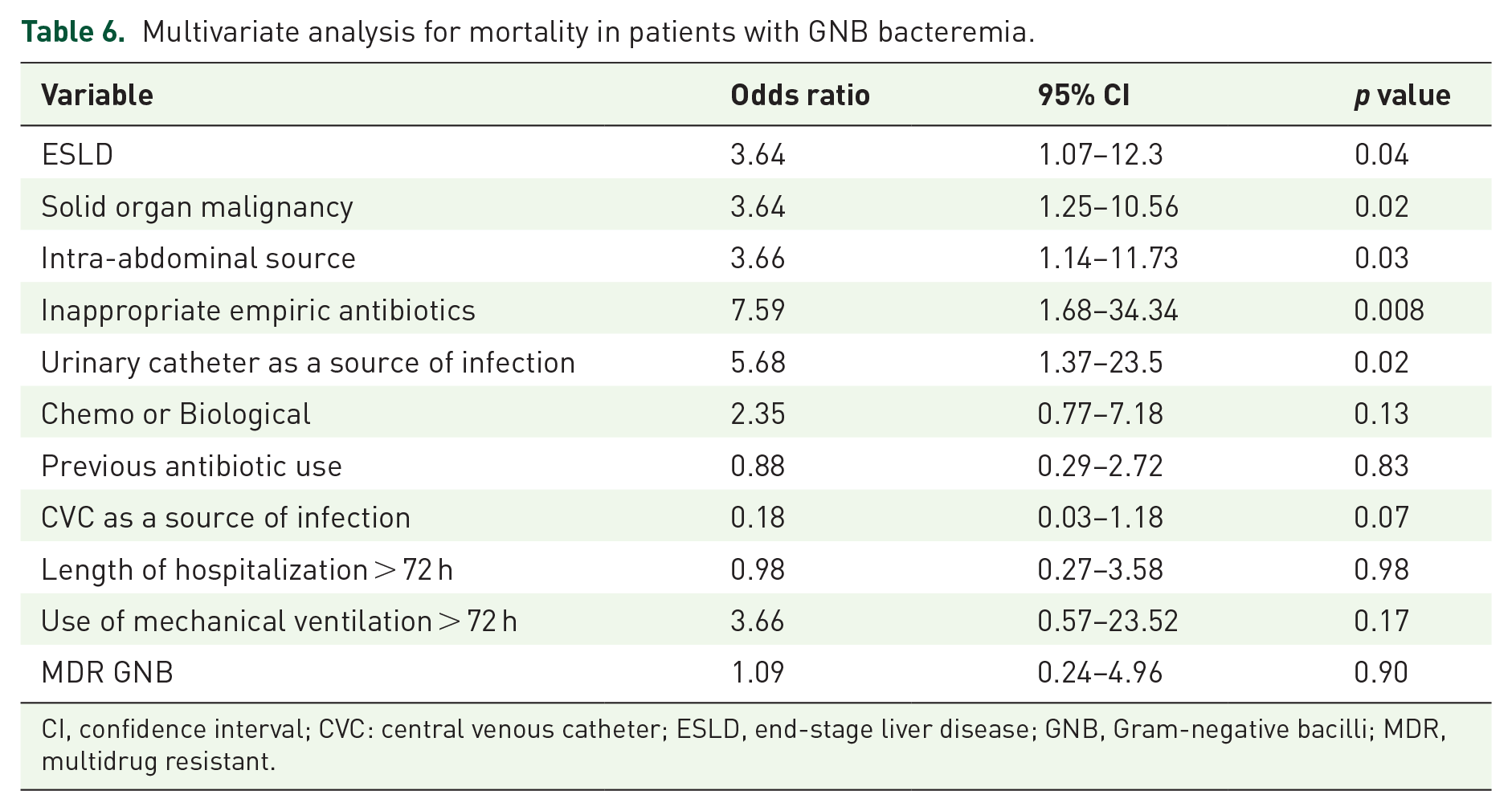
CI, confidence interval; CVC: central venous catheter; ESLD, end-stage liver disease; GNB, Gram-negative bacilli; MDR, multidrug resistant.
Discussion
The prevalence of MDR among GNB is rapidly increasing. Quantification of the prevalence and the common antimicrobial resistance patterns of MDR GNB isolates recovered from patients at hospital admission, as well as identification of patients with a high risk of harboring MDR GNB, would have important implications for patient care.
13
Our study found that 26% of GNB at the institution are MDR and case fatality rate was 34%. Gudiol
Our study provided important risk factors for the development of MDR GNB and predictors for mortality. DM, urinary catheter as a source of infection, and previous antibiotic use predicted the development of MDR GNB similar to prior studies.
Michalopoulas
Significant risk factors for morality in our study included the following: (a) end-stage liver disease, (b) solid organ malignancy, (c) intra-abdominal source of infection, (d) urinary catheter as a source of infection, and (e) inappropriate empiric antibiotic therapy. Our study is unique as our hospital is a major liver transplant and cancer center.
Lye studied 675 patients with GNB bacteremia; 301 had MDR GNB bacteremia. Risk factors for higher mortality included the following: male gender, higher APACHE II score, higher Charlson comorbidity index, ICU stay, and the presence of concomitant pneumonia.
19
A meta-analysis of 32 studies performed by Rottier concluded that ESBL was associated with higher mortality, but after adjustment for inadequate empiric antibiotics, it was found that inadequate empiric antibiotic was responsible for most of the mortality attributed to the ESBL-producing organisms.
20
Blot reported that age, ‘high-risk’ bacteremia (intra-abdominal and lower respiratory tract source of infection leading to bacteremia), higher APACHE II score, acute renal failure, and
Limitations for our study include the following: (a) single-center study, (b) retrospective analysis of clinical data, (c) relatively smaller sample size, (d) small number of patients with pneumonia which is considered a risk factor for mortality and MDR bacteremia in other studies, and (e) time to source control was not addressed in this study, which can impact the mortality rate.
In summary, the incidence of MDR GNB is increasing and associated with high mortality. Independent risk factors like DM, presence of urinary catheters, and receipt previous antibiotics were predictors for the development of MDR GNB bacteremia. Risk factors like end-stage liver disease, solid organ malignancy, intra-abdominal infection, urinary catheter as a source of infection, and inappropriate empiric antibiotics therapy were associated with higher mortality. MDR bacteremia was not associated with the recurrence. Further studies are needed to define the optimal strategy for the empirical treatment of patients at the risk for MDR GNB bacteremia.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
