Abstract
Background:
A case of mother to child transmission (MTCT) of HIV at a medical center in Washington, DC, resulted in the implementation of universal opt-out rapid testing of patients admitted for delivery. This article evaluates the policy’s efficacy and implementation.
Methods:
We evaluated the implementation using the Reach, Efficacy, Adoption, Implementation, and Maintenance (RE-AIM) framework.
Results:
We could not evaluate decrease in MTCT rate secondary to low sample size (n = 3324) and no true-positive results. Patients not tested (n = 458) were predominately secondary to physician omission (93.7%) and were more likely to be White (p < 0.01) and older (p < 0.01). There was a negative relationship with physician omission over time.
Conclusion:
The policy was successfully implemented with decreasing proportions of patients not tested. Earlier inclusion of testing into standard admission orders and nurse-based approach may have expedited adoption. Given the low incidence of new HIV diagnosis in labor, we were unable to assess decrease in MTCT.
Introduction
Mother to child transmission (MTCT) of human immunodeficiency virus (HIV) is largely preventable through early diagnosis and treatment with antiretroviral therapy (ART), including maternal and neonatal prophylaxis and Cesarean delivery (CD) for high or unknown viral. In the United States, 40–85% of infants who are born with HIV are born to mothers with unknown HIV status. 1 Undiagnosed maternal HIV precludes or significantly delays important measures to prevent MTCT. Efforts to minimize MTCT are of utmost importance in areas like the District of Columbia (DC), where there is an alarmingly high prevalence and incidence of HIV among reproductive aged women (1.9% and 0.087% per year). 2
The National Institutes of Health (NIH), the American College of Obstetrics and Gynecology (ACOG), and the Center for Disease Control and Prevention (CDC) recommend HIV screening as part of the initial prenatal laboratory testing and repeat screening in the third trimester in high-risk individuals and in high-prevalence regions, where prevalence of HIV is greater than 0.1%, such as DC. In high-risk scenarios, if third trimester HIV testing has not been performed, a rapid screen is recommended in labor,2–4 and positive tests should be treated presumptively as a new diagnosis of HIV with a high viral load, until the results of the confirmatory testing are available.1,4–6
Despite of falling national numbers of MTCT, our medical center recently had several neonates who have tested positive for HIV. Although root cause analysis identified maternal adherence as the key risk factor, one of these three cases was found to be secondary to delayed identification of HIV seroconversion late in pregnancy. In light of this, the Women’s and Infants’ Services (WIS) department instituted universal opt-out rapid HIV screening for all patients who are admitted for delivery. 7
Universal, rapid, opt-out HIV testing upon admission for vaginal or CD allows for diagnosis and treatment of new cases of maternal HIV that were not previously diagnosed secondary to lack of prenatal screening or new seroconversion and provides an additional opportunity to prevent MTCT in high-prevalence populations. Diagnosis and treatment of HIV intrapartum does not prevent in utero MTCT; however, maternal intravenous zidovudine (AZT) prophylaxis, CD before rupture of membranes (ROM), and enhanced neonatal antiretroviral prophylaxis with two or three ARVs all independently decrease the risk of intrapartum MTCT.8,9 Due to the time limitation of active labor, intrapartum AZT prophylaxis may not be feasible for mothers who presenting in labor; there is also debate of the benefit (if any) of CD during active labor or after ROM. However, at minimum, the diagnosis of HIV intrapartum allows for immediate postpartum neonatal prophylaxis, which has been shown to decrease perinatal transmission from 25.5% without intervention 10 to 7.1%. 9 Intrapartum diagnosis of HIV also allows for avoidance of breastfeeding to prevent postpartum MTCT and early diagnosis and treatment of cases of neonatal HIV.
This article evaluates the implementation and effectiveness of this intrapartum universal rapid HIV testing program. We also performed a cost-effectiveness analysis; the results are described separately.
Methods
Study setting and design
Our medical center is a 926-bed urban teaching hospital in Washington, DC, and the largest healthcare provider in the Maryland and Washington, DC, region. During the study period, our L&D provided care for a diverse payer mix of patients of private physician and midwifery groups, DC Medicaid and Medicare recipients, and uninsured patients. Our L&D also served as a referral hospital for pregnancies with known fetal anomalies, because of the proximity and relationship with Children’s National Medical Center.
We performed a retrospective chart review of all patients admitted to L&D from July 2013 to July 2014. Institutional Review Board (IRB) approval and a full waiver of the Health Insurance Portability and Accountability Act of 1996 (HIPAA) requirements were obtained prior to the start of data collection and extended 1 year later in order to complete the study. We included pregnant women who delivered a viable infant on L&D from 1 July 2013 through 1 July 2014. We excluded pregnant women who did not deliver or who were admitted for a fetal demise or elective termination. Using a password-protected Excel spreadsheet, we collected descriptive data. All data were de-identified prior to statistical analysis. For the analysis, we conducted chi-square tests and Wilcoxon two-sample tests to compare the demographic and obstetric characteristics between patients opting in and out of the test. We considered a p value below 0.05 to be significant. In addition, we used a linear regression model to detect the trend of the weekly HIV screening rate since the implementation. A positive coefficient of weeks since implementation variable indicates a positive trend. All statistical analyses were done by SAS 9.3 (SAS Institute Inc., Cary, North Carolina).
Because of the low overall incidence of MTCT, this initiative was intended as a pilot study for efficacy. By its nature, universal testing is more efficacious at identifying all affected individuals than a risk stratifying protocol over time. Our primary aim was to evaluate the implementation of a universal HIV testing protocol in the urban teaching hospital setting. We used the Reach, Efficacy, Adoption, Implementation, and Maintenance (RE-AIM) 12 framework to evaluate the implementation and to identify key obstacles.
Program development
In accordance with ACOG guidelines, our L&D HIV testing protocol was previously limited to patients with no documented third-trimester HIV result. Testing was done by either enzyme-linked immunosorbent assay (ELISA) or the third-generation rapid antibody test.
In May 2013, after review of the recent cases of MTCT, the WIS department chair circulated a memorandum to all obstetric staff and providers explaining a change in policy to a universal opt-out HIV testing protocol. All patients admitted to L&D in anticipation of delivery were to be screened by the rapid antibody screening method. 6 As the fourth-generation HIV screening test became available, the laboratory replaced the third-generation test with the combined antibody and antigen screening method.
Residents on their 5-week obstetric rotations were responsible for notifying patients of universal rapid HIV testing and placing the order for the test. In accordance with DC law, patients were given the opportunity to opt-out of the screening. Written documentation of assent was not required, other than the general admission consent, unless the patient declined testing. 11
Program evaluations
We used the RE-AIM framework to guide the evaluation for this implementation. The RE-AIM framework is designed to enhance the quality, speed, and public health impact of efforts to translate research into practice in five steps: Reach, Efficacy, Adoption, Implementation, and Maintenance.12,13 Under this structure, we organized the program evaluation as follows.
Reach-intended target population
Under the “Reach” domain, we identified the patients involved in this pilot program and determined their demographic and clinical characteristics. We extracted demographic and clinical data, such as maternal age, race, the status of prenatal history, gestational age at delivery, the status of HIV screening prior to admission of delivery, and the delivery mode from electronic medical records (EMR).
Efficacy or effectiveness
In this domain, we evaluated the impact of our screening intervention on maternal and neonatal outcomes. The primary outcome of this pilot program was to evaluate the effectiveness of universal screening on identification of new HIV cases and the reduction of MTCT. We extracted data from EMR chart review. If new maternal HIV cases had been identified in our target population, we had planned to evaluate prevented cases of MTCT by reviewing the EMR of the neonates to determine HIV status at the time of discharge.
Adoption by target staff, settings, or institutions
Through informational interviews of attending physicians, residents, and nurses, we investigated facilitators and barriers to the adoption of the universal screening protocol.
Implementation consistency and adaptations
We evaluated implementation of the universal HIV screening protocol using two indicators. First, we collected weekly proportion of patients not screened. Secondly, we investigated why patients were not screened during the implementation period. Additionally, for all patients who did not have rapid screening done on L&D, we reviewed the EMR for documentation of opt-out and to assess prior antepartum HIV screening. Informational interviews with the attending and resident physicians and nurses were used to elucidate why no screening was done in patients with either no documentation of opt-out or with canceled orders.
Maintenance of intervention effects over time
On the basis of our early findings of this pilot protocol, we evaluated the logistics of implementing a universal HIV screening policy. We collected information from physicians and nurses to identify the barriers of long-term implementation of the program and their overall satisfaction.
Results
Reach
Basic demographic characteristics are listed in Table 1. During the year of the pilot implementation, there were 3236 live births. The majority were African American (66.0%); 2778 (85.8%) underwent rapid HIV testing on L&D. As seen in Table 1, the patients who were tested were generally younger (28.1 years of age as compared to 29.3, p < 0.001) and of later gestational age [38.9 weeks estimated gestational age (EGA) as compared to 38.3, p < 0.001] than those who were not tested. Of the 308 Caucasian patients, only 78.2% were screened, whereas of the 2140 African American patients, 86.5% were tested at the time of admission; 90.9% of Hispanics and 84.5% of Asian/Pacific Islanders were screened. There was significant racial disparity in taking the rapid HIV test (p < 0.001). There was no statistical difference in the cesarean section rate (p = 0.170), nor gravidity and parity (p = 0.592 and 0.606).
Demographic characteristics.

The p value indicates the result of statistical testing by comparing the group of patients with and without rapid test.
Mean values with standard deviation (in the parenthesis) are estimated for these variables.
Frequency count with column percent (in the parenthesis) is estimated for these variables.
Median with 10th and 90th percentiles (in the parenthesis) is estimated for these variables.
Efficacy
Figure 1 examines the primary outcome of MTCT cases prevented. Of the initial 3324 cases reviewed, 88 were excluded because they either suffered a fetal demise prior to admission or were admissions for elective termination of pregnancy. There were no positive rapid HIV tests done on L&D. One patient was found to be negative by rapid test, but positive by ELISA method discovered after delivery; confirmation testing was negative. Of those who were not screened, 123 (26.9%) had only initial recorded HIV status, 169 (36.9%) had only recent recorded HIV status (within 12 weeks prior to delivery), 57 (12.4%) had no documented status at all, and 22 (4.8%) were known HIV positive patients (Figure 2). All neonatal charts from the 458 patients not tested were reviewed; there were zero cases of MTCT at the time the neonates were discharged from the hospital.
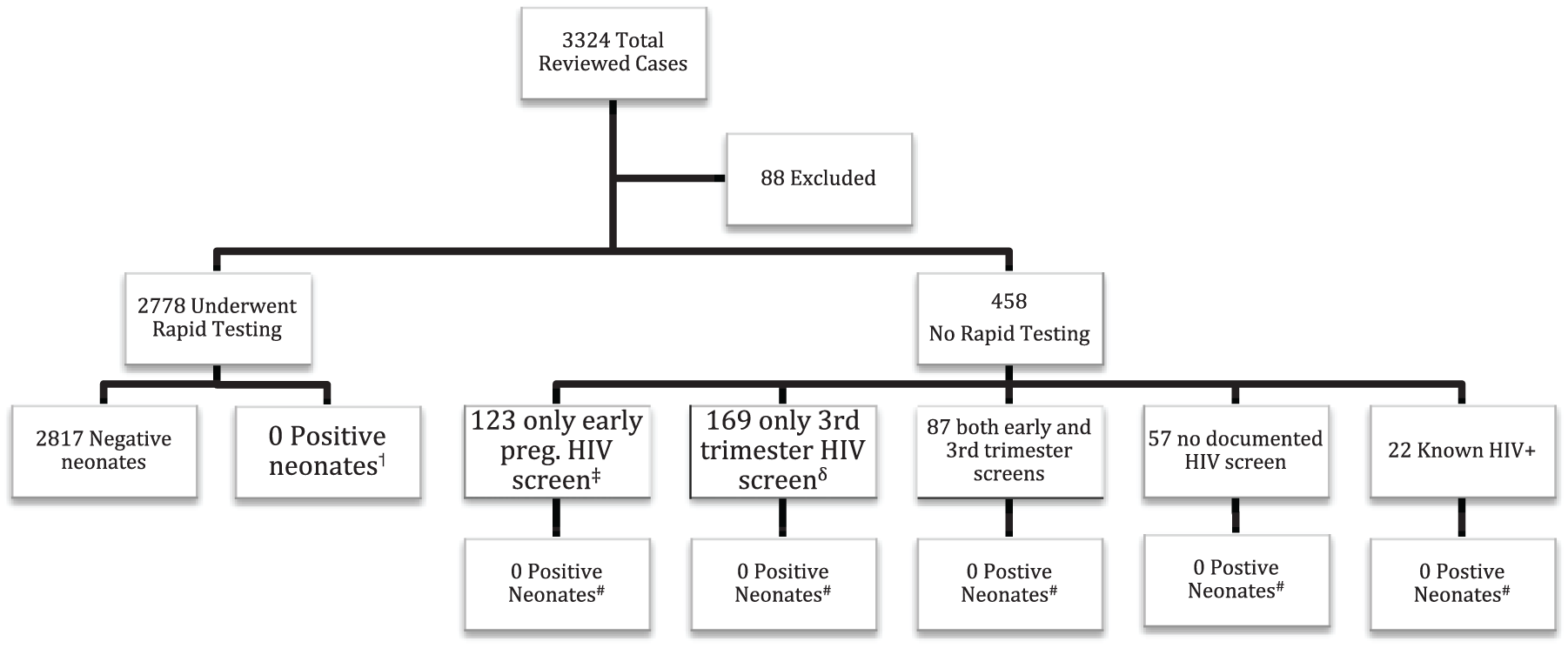
Rapid testing: results and neonatal outcome.

Reasons patients not screened for HIV.
Informational interviews revealed that residents were responsible for inputting all prenatal labs into the L&D EMR system. Patients with prenatal care at outside facilities often presented without their records, thus preventing their input, and contributing to cases without any recorded HIV status.
Adoption
All L&D faculty, staff, and residents were informed of the new protocol by memorandum in May 2013, as well as through announcements at Grand Rounds and L&D morning sign-outs. All providers based at the medical center were adequately informed. 7 Only 7 of 40 residents were actively rotating on L&D at the time of implementation.
In informational interviews, resident physicians stated that while they received the emailed memorandum in May 2013, the new screening protocol was not adequately reiterated at the beginning of their five-week blocks during the study period. At the beginning of their new rotations on L&D they were not initially aware that they needed to order the screening test. The residents universally agreed that unless otherwise documented, a lack of screening was secondary to omission and attributed to a combination of inadequate time and lack of awareness.
Figure 2 depicts why 458 patients were not screened on L&D. The overwhelming majority (93.7%) were not screened secondary to physician omission; 5 (1.1%) were not screened because the order was accidently canceled upon converting the labor orders to the postpartum orders. Only 2 patients (0.4%) had documentation that they declined testing. Only one patient declined testing throughout her pregnancy; 180 of those who were not screened met criteria for screening on L&D even by the previous protocol.
Implementation
To evaluate trends in implementation of the universal rapid HIV protocol, we graphed the proportion of patients screened by week during the study period (Figure 3). The official announcement of policy change came just over 7 weeks prior to our study period. The date the rapid HIV lab test was added to the admission order set correlates to steep upward trend between weeks 23 and 24 (2 December 2013). The linear regression line demonstrates a rise from a mean of 81.7–90.3% by the end of the study period. The downward trend of proportion tested during weeks 16, 21, 23, 28, 37, and 47 correlates to dates of major resident rotation changes.
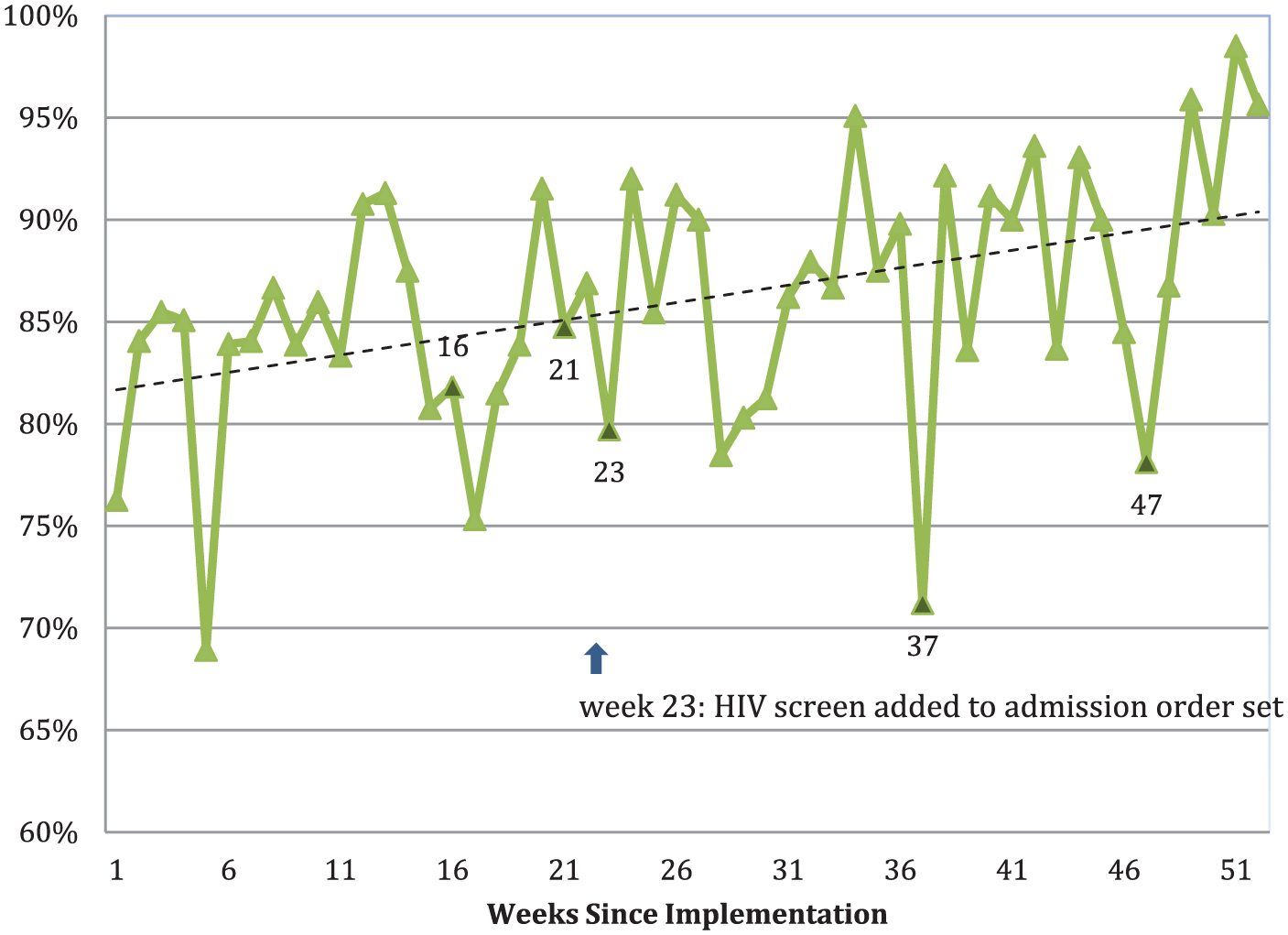
Weekly proportion of patients screened for HIV.
Informational interviews with the attending and resident physicians and nurses to elucidate why screening was not done in patients without documentation of opt-out or canceled orders confirmed our suspicions that the majority of these cases were likely secondary to physician omission.
Maintenance
Through informational interviews, resident physicians stated that the nursing staff began collecting the blood sample for the HIV screening test whether or not the order had been placed. If the order was not placed, nurses would remind residents to place the order or document that the patient declined screening. Nurses voiced that they accepted the universal screening policy as a new standard of care and advocated for process by which the residents would automatically order the screening test.
Discussion
Reach
While the overall reach was excellent, there was a statistically significant bias toward screening patients of color and younger patients (Table 1). Considering that physician omission was responsible for 93.7% of why these patients were not tested (Figure 2), bias in screening may reflect provider awareness of the higher incidence of HIV in younger women and women of color versus and/or reflect provider prejudices that these patient populations may have received less prenatal care or are more sexually promiscuous. Other characteristics such as gravidity and parity did not play into whether or not a patient was tested.
Efficacy
Despite the high incidence of HIV in Washington, DC, the efficacy of this program could not be evaluated given our lack of true-positive tests or MTCT cases during this period (Figure 1). Universal screening will eventually identify new HIV cases. This is demonstrated in all universal screening studies done in the emergency department (ED) setting, where new cases have been identified in 0.4–1.2% of those screened.6,14,15 This will eventually prevent new cases of MTCT; our pilot study, however, evaluated only 1 year of this program. Furthermore, the ED studies included only patients who had not been screened within the last year, while the overwhelming majority of our patients had first- and/or third-trimester HIV screening (Figure 1).
Adoption
Our pilot study period began 7 weeks after L&D implemented a universal HIV screening protocol for pregnant patients admitted for delivery. At the initiation of the pilot study period, compliance with universal screening was greater than 80% (Figure 3). By the end of the pilot, over 90% of our patient population was being screened. Given this trend, we anticipate that the proportion of patients screened or offered screening will approach 100% within the next calendar year, thereby eliminating the 458 missed opportunities for screening noted in our pilot year (Figure 1). As adoption of this protocol improves, not only will seroconversions late in pregnancy get recognized, those 180 who should have been screened on L&D anyway are more likely to be screened.
As shown in Figure 3, downward trends of the proportion of patients screened were noted to occur during the shifts in rotating residents, as residents previously rotating through other hospitals returned. Adoption would likely have been further improved with better communication with the rotating residents. Though inadequate time was cited by the residents as a major barrier, this is not likely to change and is a universal obstacle. The shifts that were not accompanied by downward trends are likely a result of detailed handoffs and communication between the staff and the residents, as well as potentially less buy weeks.
As suggested in the ED literature, implementation strategies need to acknowledge the barriers of provider awareness and time constraints in order to be able to minimize them. 16
Implementation
Implementation was also aided by addition of the rapid HIV screening test in the labor admission order set. The legal requirement to allow a patient to opt out, however, meant that the order had to be separately and therefore cognizantly selected by the provider. While seemingly minute, this additional mouse-click likely led to the high number of physician omissions. A better method may have been to order automatically, but have an additional check-box after the fact to note that the patient had been counseled and had not opted out.
To eliminate physician omission, and 93.7% of missed screening opportunities (Figure 2), the task of ordering the rapid HIV screening test and counseling the patient could be delegated to the nursing staff. ED literature demonstrates that the nursing staff is overwhelmingly supportive of screening initiatives and tends to be more focused on task-based protocols than the physicians, who are dealing with acute care issues unrelated to HIV screening. In Establishing an ED HIV screening program: lessons from the front lines, the authors propose a system in which opt-out counseling is done as a part of routine triage. 17 Already, the L&D nursing staff is universally collecting demographic data and screening for domestic violence while filling out admission forms. Addition of universal HIV opt-out counseling documentation with an automated order to the admission forms would allow this to be done as part of the admission process, thus eliminating physician omission and ensuring proper counseling.
Maintenance
As time progressed, maintenance of the program shifted away from the rotating residents and toward the more permanent nursing staff who accepted the protocol as a new standard of care. Though not yet suggested by Figure 3, the likely effect of this will be less variability in the week-to-week proportion of patients who are not tested. This further supports the nurse-based admission implementation strategy as described above.
Conclusion
Our pilot study of universal rapid HIV screening on labor and delivery at our medical center, as evaluated in the RE-AIM framework, represents a successfully implemented protocol. With maintenance of this program and continued improvement in implementation strategy, we will notice a trend toward 100% inclusion of our high-risk population. As we work to eliminate missed screening opportunities, we hope to prevent cases of MTCT. Using this pilot program as well as an ongoing cost benefit analysis, we hope to help other institutions in communities with high HIV prevalence implement this program with a goal to identify all late maternal HIV conversions and prevent MTCT.
Footnotes
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
