Abstract
Objective:
To determine factors associated with increased risk of developing cardiovascular disease in a high-risk patient population.
Design:
Cross-sectional analysis of a retrospective cohort study.
Methods:
One-hundred patients at an inner city HIV clinic in 2008 were reviewed. The atherosclerotic vascular disease risk score was calculated using the Pooled Cohort Equation. Chi-square test was performed to identify associations of potential risk factors with elevated atherosclerotic vascular disease risk.
Results:
Eighty-one participants were included in the final analysis. In total, 95.1% were African American, and 38.3% were women. The median atherosclerotic vascular disease risk score was 8.8% and 8.1% in 2008 and 2012, respectively. The medical co-morbidities associated with increased atherosclerotic vascular disease risk were hepatitis C infection (X2 = 3.93; p value = 0.048), elevated triglycerides levels (X2 = 4.0; p value = 0.046), and low albumin (X2 = 4.65; p value = 0.031). There were a higher number of women with known atherosclerotic vascular disease despite lower median atherosclerotic vascular disease risk score compared to men.
Conclusion:
An elevated risk of developing cardiovascular disease persists in high-risk demographic groups of the HIV epidemic even in the current HIV era. There is an unexplained gender disparity and some non-traditional risk factors not accounted for in the Pooled Cohort Equation may be contributing to the excess cardiovascular disease risk observed among HIV-infected patients.
Keywords
Introduction
The World Health Organization projects that HIV/AIDS and ischemic heart disease will represent two of the top three causes of death worldwide by 2030. 1 Affected individuals with HIV are now living longer with improved quality of life due to better and more expanded access to antiretroviral therapy (ART) that has translated into improved survival rates largely from reduction in traditional AIDS-defining illnesses. 2 However, despite effective ART, HIV patients are at increased risk of developing serious non-AIDS illnesses such as cardiovascular disease (CVD). In fact, CVD is currently the third leading cause of death in HIV-infected patients occurring at an unprecedented rate in younger patients as compared to HIV-uninfected patients.3–10
The excess risk of premature CVD observed in HIV-infected patients has been attributed to a variety of epidemiological and biological factors. Studies have identified factors ranging from traditional CVD risk factors to HIV-induced inflammation and immune activation, and/or the contribution of antiretroviral medications (ARVs).5,10–18 However, these studies are heterogeneous in design, and many did not collect information on cardiovascular (CV) risk factors such as smoking and family history.5,19–21 In addition, ART regimens have changed over time, so older studies many not be applicable to current HIV management. Studies with newer ARVs have shown reduced adverse effects with improved lipid profiles, lower fasting glucose and initial decreases in inflammatory markers.22,23 Furthermore, many of the prior studies included primarily Caucasian men and hence generalizability to women and minorities is limited.
The purpose of our study was twofold. The first was to determine whether risk of developing CVD remained elevated in the ART era of changing HIV practice guidelines. The second was to determine the most prominent factors associated with increased risk of developing CVD of a population of HIV-infected individuals primarily composed of African Americans. The new Pooled Cohort Equation, which revised the current version of the Framingham Risk Equation by calculating the 10-year risk of developing atherosclerotic vascular disease (ASCVD), was used in the 2013 American Heart Association/American College of Cardiology cholesterol guidelines and provided a unique opportunity to evaluate the CVD risk in HIV-infected patients. 24
Methods
Study population
For this pilot study, we reviewed the medical records of 100 HIV-infected patients treated at the Evelyn Jordan Center (EJC), a large inner city HIV clinic associated with the University of Maryland Medical Center (UMMC) from 1 June 2008 to 31 May 2012. The objective of the pilot (feasibility) study was to determine the feasibility of conducting a larger retrospective cohort study by extracting data from our clinic records. The intent was to use the experience of the pilot study to adjust our data collection and entry methods in order to improve and streamline the process for the larger retrospective cohort study, if deemed feasible to proceed. The Institutional Review Board of the University of Maryland Baltimore approved this study (HM-HP-00055406), and informed consent from subjects was waived because there were no interactions with study participants.
To be eligible for the study, participants had to be at least 18 years old, enrolled in and receiving HIV care at EJC by 1 June 2008 and have at least two routine HIV visits from 1 June 2008 to 31 May 2012. We selected the first 100 charts based on medical record numbers generated in chronological order to determine the feasibility of abstracting and collecting data from these medical charts for a larger retrospective cohort study. One patient had more than one medical record number resulting in 99 eligible patients remaining. Of the 99 eligible participants, 81 had data for every variable required to calculate the ASCVD risk score using the Pooled Cohort Equation. 24 The calculated ASCVD risk score is analogous to the Framingham risk score (FRS) in the prior version of the equation, and it too describes an individual’s 10-year risk of developing CVD. Variables required to calculate the ASCVD risk score are age, gender, race, total cholesterol, high-density lipoprotein cholesterol (HDL-C), systolic blood pressure, diastolic blood pressure, treatment for blood pressure, diabetes, and current smoker.
Data collection
Data were collected for routine HIV visits at 3-month intervals (maximum of four visits per 12-month period) for patients from 1 June 2008 to 31 May 2012. Routine visits were identified by individual abstractors who reviewed clinic notes of all office visits for every subject and decided whether the visit represented a routine HIV clinical visit. A visit was defined as routine if it was a follow-up for HIV, included physical exams and addressed health maintenance issues. Data collected required abstraction of each medical chart in four separate electronic record systems (Medicos, Horizon Patient Folder, Powerchart, and EPIC). Presence of medical diagnoses was based upon documentation in the medical chart after thorough review of all clinical notes in the clinic and hospital medical record systems. Diagnosis of depression was based on mental health provider notes or treatment of depression with appropriate psychiatric medication, and not patient self-report. Coronary heart disease (or CHD event) was defined as one of the following: history of an acute coronary syndrome (e.g. unstable angina), myocardial infarction (MI), clinical or arteriographically proven coronary artery disease (CAD), and ischemic cardiomyopathy. Cerebrovascular disease was defined as stroke or transient ischemic attack. ASCVD was defined as either CHD or cerebrovascular disease. Lab values and ranges were based on assays used by the UMMC reference laboratory, LabCorp.
Statistical methods
For the primary analysis, we performed a cross-sectional analysis of 81 participants in EJC during 2008. A total of 13 participants were excluded from the primary ASCVD risk score calculations because they had prior ASCVD, and the ASCVD risk score calculator is validated for use only in those without known ASCVD. 24 ASCVD risk values were divided into two categories: elevated CV risk, defined as a risk of >7.5% risk of developing ASCVD in 10 years, and low CV risk, defined as <7.5% risk of developing ASCVD in 10 years. Chi-square test of independence was performed to identify associations of potential CVD risk factors with elevated ASCVD risk after removing variables that were part of the ASCVD risk score calculation (age, gender, race, total cholesterol, HDL-C, systolic blood pressure, diastolic blood pressure, treatment for blood pressure, diabetes, and current smoker).
We investigated associations between numerous socio-demographic, behavioral, and clinical factors and ASCVD risk based on an extensive review of the literature in PubMed and Embase over the past two decades. Socio-demographic factors assessed were sex, age, race, educational level, and employment status. Behavioral factors assessed were smoking status alcohol use, illicit drug use, and mode of transmission. Clinical factors evaluated were duration of known HIV diagnosis, ARV use including ARV class, prior opportunistic infections, most recent CD4 cell count, undetectable HIV viral load status, body mass index (BMI), glomerular filtration rate, presence of hepatitis B, hepatitis C, cancer diagnosis, depression, prior CVD that did not qualify as ASCVD, family history of CVD, use of statins, low-density lipoprotein cholesterol, triglycerides, and albumin.
We performed a secondary cross-sectional analysis of the 22 participants who had 2012 study visits and 2012 laboratory data for all variables used in the ASCVD risk score calculator. Given the small sample of participants for this analysis, we did not attempt to identify associations between the potential CV risk factor variables and ASCVD risk scores. Finally, we calculated the ASCVD risk score of those with known ASCVD to assess whether the calculated risk score for these participants correlated with the high-risk score that we would expect in patients with known ASCVD.
The analyses were performed using Stata 12.1 (StataCorp, 2011). To be considered significant, we considered a p value <0.05 using a two-tailed test. No sample size calculations were performed because this was a pilot study undertaken to evaluate the feasibility of a larger retrospective cohort study.
Results
The 81 participants were primarily African Americans (95.1%) and included 31 women (38.3%), and the median age was 52 (6) years (see Tables 1 and 2). The most common risk factor for HIV acquisition was injection drug use (IDU) (60.5%) followed by heterosexual contact (32.1%). The majority of patients had been diagnosed with HIV for over 10 years at study entry (86.4%), and the median CD4 cell count was 361 (317) cells/mm3. The majority of patients were receiving ART (87%), and more than half of those receiving ART were virologically suppressed with viral loads below limits of assay detection (55.5%). Twenty-one patients were receiving atazanavir, and two patients were receiving darunavir and raltegravir. None were receiving maraviroc. The remainder of patients was receiving older ARVs. The majority of patients were co-infected with hepatitis B and/or C (>70%). Almost half (45.7%) were overweight with BMI > 25, and diabetes mellitus was present in 20 patients (24.7%). Baseline characteristics were not significantly different in the 18 patients who were excluded when compared to the 81 who were included (data not shown).
Socio-demographic characteristics of HIV-infected participants.

IQR: interquartile range; IDU: injection drug use; MSM: men who have sex with men.
Clinical characteristics of HIV-infected participants.

ARVs: antiretroviral medications; PIs: protease inhibitors; NNRTI: non-nucleoside reverse transcriptase inhibitor; NRTI: nucleoside reverse transcriptase inhibitor; IQR: interquartile range; BMI: body mass index; GFR: glomerular filtration rate; CHD: coronary heart disease; ASCVD: atherosclerotic vascular disease; CVD: cardiovascular disease; HDL: high-density lipoprotein; LDL: low-density lipoprotein.
The median ASCVD risk score in the primary analysis of patients seen in 2008 (N = 68) was 8.8% consistent with an elevated risk of developing ASCVD in 10 years (see Table 3). However, ASCVD risk score was significantly higher in men than women (10.8% vs 4.0%; p value < 0.001) This gender differential was observed with or without ASCVD. The ASCVD risk score of the five men with ASCVD (27.3%) was significantly higher than the ASCVD risk score of the eight women with ASCVD (12.7%). Therefore, despite the greater absolute number of HIV-infected women having developed ASCVD by 1 June 2001, their calculated ASCVD risk score was less than half that of their male counterparts who had developed ASCVD. Notably, 5 of 55 men (9.1%) and 8 of 39 women (20.5%) had known ASCVD at study entry. Finally, in the secondary analysis, the median ASCVD risk score in 2012 was 8.1%, and the gender differences in ASCVD risk scores persisted (see Table 3).
Median Framingham risk score by gender in HIV-infected participants.
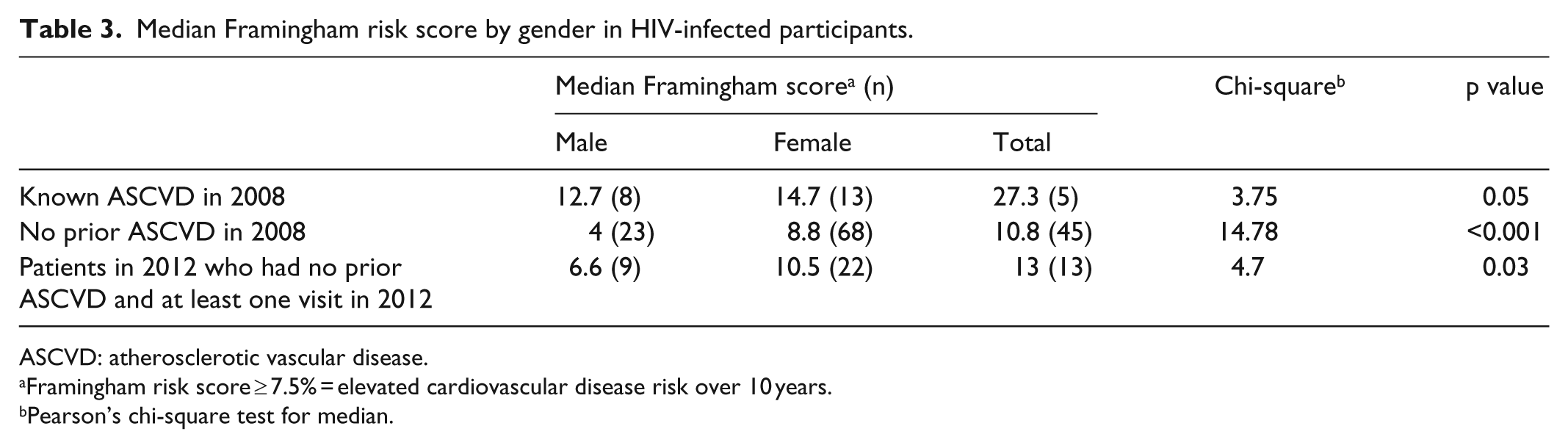
ASCVD: atherosclerotic vascular disease.
Framingham risk score ≥ 7.5% = elevated cardiovascular disease risk over 10 years.
Pearson’s chi-square test for median.
The medical co-morbidities associated with increased CVD risk were hepatitis C infection (X2 = 3.93; p value = 0.048), elevated triglycerides levels (X2 = 4.0; p value = 0.046), and low albumin (X2 = 4.65; p value = 0.031) (see Table 4). There were no significant associations between ASCVD risk and education, employment status, alcohol use, illicit drug use, or mode of transmission (Table 4). Notably, specific ARVs were not associated with the elevated FRS. We found other associations that were noteworthy though they were not statistically significant: history or active hepatitis B (p value = 0.072), depression (p value = 0.157), protease inhibitor (PI) use (p value = 0.207), and non-nucleoside reverse transcriptase inhibitor (NNRTI) use (p value = 0.247) (see Table 4).
Association of risk factors with Framingham risk score among HIV-infected participants.

IDU: injection drug use; MSM: men who have sex with men; ARVs: antiretroviral medications; PIs: protease inhibitors; NNRTI: non-nucleoside reverse transcriptase inhibitor; NRTI: nucleoside reverse transcriptase inhibitor; BMI: body mass index; GFR: glomerular filtration rate; ASCVD: atherosclerotic vascular disease; CVD: cardiovascular disease; LDL: low-density lipoprotein.
Discussion
In this study, we identified several risk factors for CVD not included in the Pooled Cohort Equation in a population of predominantly African American HIV-infected patients: hepatitis C, hypertriglyceridemia, and hypoalbuminemia. Furthermore, we demonstrated that an urban cohort of HIV-infected patients with a median age of 52 years and consisting predominantly of African Americans, with a large percentage of women, and majority IDUs have a high 10-year risk of developing ASCVD. The ASCVD risk remained high in 2012 despite changing HIV practice guidelines. Finally, HIV-infected patients women had a higher burden of ASCVD than men even though their ASCVD risk scores were only half those of their male counterparts in this study population.
Our study identified several risk factors for elevated CVD risk that are not included in the current Framingham Risk Equation. Identification of unaccounted factors that may be associated with increased CVD risk in the HIV-infected population is important for designing future derivation and validation studies to more accurately predict CVD risk, since there is a robust body of the literature suggesting that current risk scoring tools may be suboptimal among HIV-infected patients. 25 Our finding of hepatitis C as a CV risk factor was consistent with that of the Veterans Aging Cohort Study–Virtual Cohort Study which found HIV/hepatitis C co-infected male patients had a significantly higher risk of CHD compared to HIV-monoinfected patients (adjusted hazard ratio, 1.93; 95% confidence interval (CI), 1.02–3.62). 26 The Data collection on Adverse events of Anti-HIV Drugs (D:A:D) cohort study showed no association between hepatitis B or hepatitis C co-infection and development of MI, but their study population did not include as large a proportion of minorities, women, and IDUs as ours. 27 Although elevated triglyceride levels have been found to be an independent risk factor for MI in the general population in several large studies, to our knowledge, only one study in HIV-infected individuals demonstrated that hypertriglyceridemia was independently associated with an increased risk of MI (relative risk (RR), 1.17; 95% CI, 1.06–1.29) after adjustment for CVD risk factors.28–31
Our finding of hypoalbuminemia as a risk factor for elevated CVD is supported by several epidemiological studies in the general population which have shown that low serum albumin concentration is associated with a higher risk of morbidity and mortality from CVD particularly in elderly and end-stage renal disease patients.32–38 In HIV-infected persons, a retrospective analysis of the Department of Veterans Affairs HIV Clinical Case Registry demonstrated lower serum albumin levels were strongly associated with 1-year mortality and development of ASCVD and heart failure, but this population did not include women. 39 Proposed pathogenic mechanisms have included chronic inflammation, anemia, and accumulation of uremic toxins in end-stage renal disease patients, overnutrition-induced metabolic derangements particularly in younger patients, inflammatory-mediated vascular dysfunction particularly in elderly patients, antioxidant effects, and reduced cholesterol efflux from cells.36,38,40–43 This is interesting given the well-described elevated inflammatory state of HIV-infected persons both on and off ART. 11
The preponderance of the literature suggests that there is an increased burden of CVD in HIV-infected individuals. Our study confirms the elevated risk of developing CVD in a population that included high-risk demographic groups underrepresented in many other studies but disproportionately represented in the HIV epidemic. 44 These results are consistent with the results of the Veterans Aging Cohort Study–Virtual Cohort Study which included 82,459 male veterans and which demonstrated an increased risk of incident MI among HIV-infected compared with uninfected veterans (hazard ratio, 1.48; 95% CI, 1.27–1.72) after adjusting Framingham risk factors. 45 Additionally, numerous other cohorts have found increased risk of CVD as measured both in overt or subclinical CVD in HIV-infected compared to uninfected individuals though most of these studies reflected use of older ARVs.5,8,9,46–48 More recent analyses of the D:A:D cohort and the California Kaiser Permanente database demonstrated declining rates of MI in HIV-infected persons compared to their earlier reports, but these study populations do not include as large a proportion of African Americans, women, or IDUs as ours, and so these trends may not apply to these high-risk demographic groups.2,49 Finally, many of these studies report not only excess risk but also accelerated disease onset which is consistent with our study findings of elevated CV risk in a relatively younger age group than in the general population.
There are many fewer studies assessing CVD risk in HIV affected women than in men. Our finding that there is a higher number of women with known ASCVD despite having half the median ASCVD risk score compared to their male counterparts is consistent with prior studies, but ours included more minorities and IDUs than most others. For example, Triant et al. 6 found that HIV-infected women in Boston had higher rates of MI even in premenopausal age groups than their male counterparts and Lang et al. 8 found that the standardized morbidity ratio of HIV-infected women in France for CHD risk was 2.7 versus 1.4 in HIV-infected men. Given our study findings showing lower ASCVD risk scores in women with known ASCVD than in men, it may be problematic to assume that the current version of the FRS equation (Pooled Cohort Equation) is the best suited to assess CVD risk in HIV-infected women. This highlights the need to identify factors that may be explain the unaccounted differential CVD risk in HIV-infected women compared to men.
There are several important limitations to our study. First, the small sample size limited our ability to detect other significant associations. Nonetheless, we identified several risk factors for developing CVD that are not currently included in the Pooled Cohort Equation that may help explain the observed increased rate and accelerated onset of CVD among HIV patients. We are reassured that these findings were consistent with reported data from some large prospective cohorts. Second, we were unable to meaningfully adjust for multiple covariates in the same model due to the limited number of patients with complete covariates assessment. However, the aim of this pilot study was exploratory, and therefore we conducted descriptive analyses and presented unadjusted analyses. As no multivariable analyses were done, it is possible that due to the number of comparisons conducted some of our significant findings occurred by chance. However, we were reassured similar findings have been reported in some larger studies, albeit among often differently distributed demographic groups. We are currently conducting a larger, retrospective cohort study which is adequately powered to detect associations between these variables and ASCVD risk. Third, the retrospective design did not allow capture of all data relevant to CV risk such as anthropometric measures, accurate assessment of lipodystrophy, and measurement of select markers of inflammation and immune activation. Additionally, the cross-sectional nature of the study did not allow us to identify change of CV risk factors associated with change of ASCVD risk over time at the individual level. Finally, the majority of our study population was receiving older ARVs, so we were not able to identify CVD risk factors associated with newer ARVs.
There are also important strengths to our study. First, our study population consists of large percentages of certain high-risk demographic groups that are often underrepresented in larger HIV cohorts. Second and importantly, we were able to capture relevant confounding variables of CV risk missing in numerous other studies due to our comprehensive and detailed data collection techniques. Finally, our study investigated elevated CVD risk in a HIV clinic population utilizing the most recently updated 2013 Framingham Risk Equation which is timely and relevant with easy applicability to the clinical setting.
In conclusion, our study findings confirm an elevated risk of developing CVD in high-risk demographic groups of the HIV epidemic at a relatively young age in the current HIV era. We also observed an unexplained disparity of ASCVD risk scores based on gender and identified some non-traditional risk factors not accounted for in the Pooled Cohort Equation that warrant validation in larger studies particularly prospective HIV and HIV–hepatitis C co-infected cohorts that have adequate representation of minorities and women. Further studies investigating pathogenic mechanisms underlying these observations are urgently needed.
Footnotes
Acknowledgements
The authors acknowledge the following for the contributions to data collection for the article: Rawan Faramand, Ashley Laleker, Pankti Patel, and Alexandra Ward. All authors critically reviewed the manuscript and approve the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the University of Maryland, Baltimore Institutional Review Board. Protocol Number: HP-00055406.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was not sought for the present study because it was waived by the Institutional Review Board.
