Abstract
Objective:
The purpose of this randomized-controlled pilot study was to explore the effectiveness of a home-based computerized cognitive training intervention in improving cognitive function in a population of older adults with mild cognitive impairment who are living with HIV.
Methods:
In all, 24 participants were enrolled in this study. All study participants were impaired [defined as Montreal Cognitive Assessment (MoCA) score < 26]; 12 were randomly assigned to a computer-training intervention group and 12 to a control group. The intervention group used a home-based computerized cognitive training program for 8 weeks, while the control group received health-related newsletter via email and follow-up phone calls. Cognitive function was measured at study entry, immediately post intervention, and 8 and 16 weeks post intervention
Results:
This study achieved a 92% retention rate, losing two persons from the intervention group. Participants in the intervention group scored significantly higher on cognitive testing immediately post intervention compared to the control group: F(1, 19) = 4.92, p = 0.04. The partial Eta squared of 0.32 indicates a small to moderate effect size.
Discussion:
Cognitive improvement was seen immediately after the intervention, and cognitive improvement was still evident 16 weeks post intervention. Cognitive training could be considered as an option for older adults with HIV experiencing mild cognitive impairment.
Introduction
More than 1.2 million people in the United States, half of whom are 50 years of age and older, are living with HIV. 1 Effective use of combination antiretroviral therapy (cART) has made it possible for persons with HIV to live near-normal lifespan. While this increased lifespan is cause for celebration, it also introduces new challenges in successfully managing HIV disease, including treating problems related to long-term HIV infection and managing age-related comorbidities. Cognitive impairment is a common problem, affecting more than half of adults living with HIV. 2 HIV-associated dementia (HAD) was once a common cognitive problem. While HAD is now rarely diagnosed in the United States, the prevalence of other cognitive problems has increased. These other problems are present even in adults whose HIV viral load is successfully suppressed with effective use of cART.3,4 Risk factors for neurocognitive impairment in adults living with HIV include number of comorbidities, older age, depression, and advanced disease. There may be additional factors for cognitive decline specific to the aging HIV population, including accelerated aging, poor sleep, and fatigue. 5
Cognitive impairment adversely affects quality of life, medication management, and ultimately survival. Previous HIV studies have shown an association between cognitive ability and medication adherence.6–8 Other HIV studies have shown an association between cognitive function and depression. 9 Thus, improving cognitive function can positively impact the lives of older adults living with HIV.
Finding effective methods to improve cognitive function is not a one-size fits-all endeavor. Many HIV studies have reported an improvement in cognitive function with exercise.10,11 In a more recent study of adults living with HIV, a 16-week aerobic exercise program did not produce any significant improvements in cognitive function, and the mean adherence rate to the exercise intervention was 60%. 12 This adherence rate is reflective of numerous other exercise studies in adults with HIV, where high noncompliance and dropout rates reflect an inability or unwillingness to exercise on a routine basis, limiting its use as an effective intervention for a percentage of the population. A meta-analysis of 36 studies of physical activity interventions in adults with HIV found a pooled dropout rate of over 20%. 13
There has been limited research using medications or herbal supplements to improve cognition in adults living with HIV. Both modafinil and methylphenidate have shown some positive effects in treating cognitive problems in this population.14–16 Many older adults, however, worry that medication interactions could interfere with the effectiveness of their HIV medications and are seeking ways to improve their cognitive function without taking any additional medications or supplements. Additionally, many older adults may prefer a more sedentary alternative to exercise for improving cognitive function. A home-based computerized training program, if effective, could be a viable choice for those adults searching for an effective means of improving or maintaining cognitive function with an approach that does not demand physical exercise or require additional medications.
The home-based cognitive training program implemented in this study offers the opportunity to improve cognitive function in the home, at a time that is convenient, and it can be done in as little as 30 min three times per week. This program and similar programs have been found to have varying degrees of success in older adults with a variety of chronic conditions as a way of improving cognitive function.17,18 In studies specific to adults with HIV, speed of processing training (how quickly the brain processes verbal and visual information) has shown some benefit in improving driving skills and improving activities of daily living. In a small study (n = 20) of adults living with HIV 40 years of age and older, useful field of view, a measure of information processing speed, and visual attention improved after just 10 h of training in a home-based training program. A limitation of this study was the 25% attrition rate after only 12 weeks. 19 In a study of 32 adults living with HIV, 16 with HIV-Associated Neurocognitive Disorder (HAND) and 16 without HAND, computer training was part of a 4-month cognitive rehabilitation protocol that showed improvement in five of eight cognitive domains measured, including learning and memory, abstraction/executive function, verbal fluency, attention, and function. 20
Given these promising results from earlier studies, the goal of this study was to assess overall cognitive improvement in older adults living with HIV who have experienced mild cognitive impairment after an 8-week computerized cognitive training intervention.
Purpose
The purpose of this randomized-controlled pilot study was to explore the effectiveness of a home-based computerized cognitive training intervention in improving cognitive function in a population of older adults with mild cognitive impairment who are living with HIV.
Methods
Research design
A total of 24 participants with mild cognitive impairment were recruited into this randomized-controlled pilot study. Participants were randomly assigned to either the intervention group or the control group. Utilizing a pre-test, post-test design, cognitive function was measured at four distinct time points: at study entry, at the completion of the 8-week cognitive training program, and 8 and 16 weeks after the cognitive intervention was completed. Pre- and post-test measures included the Montreal Cognitive Assessment (MoCA), used to measure cognitive function; the Pittsburgh Sleep Quality Index (PSQI), used to measure sleep quality; and the Center for Epidemiological Studies Depression Scale (CES-D), used to measure depression. All instruments and methods had been used previously in HIV research and had a high degree of validity and reliability.
Participants
Participants were recruited from the Infectious Disease Outpatient Clinic and the Clinical Trials unit of a Midwestern University Medical Center. Inclusion criteria were as follows: (1) adults 50 years and older living with HIV, the age suggested by the National Institutes of health as the cutoff to define older adults in this population; 21 (2) taking antiretroviral therapy (ART) for at least 16 weeks prior to study entry, enough time to reach an undetectable viral load for most people infected; (3) mild cognitive impairment as determined by a score of <26 on the Montreal Cognitive Assessment; (4) the ability to read and speak English, as all written material was in English; and (5) access to a home computer with Internet access, in order to complete the intervention if randomized to the intervention group. Persons were excluded if, in the opinion of the investigators, they could not provide informed consent due to mental impairment.
Instruments
The major study outcome variables were cognitive function, depression, and sleep quality. Demographic data on participants were also collected.
MoCA
The MoCA was used to determine mild cognitive impairment at screening and used at weeks 8, 16, and 24 to measure change in cognitive function post intervention. Three different versions of the MoCA were used to decrease the chance of learning effect. The MoCA is a 30-item assessment tool commonly used to assess cognitive function in adults with HIV.22–24 Originally developed as a means to detect mild cognitive impairment in a clinical setting, this 10-min researcher-administered test assesses several cognitive domains including attention, concentration, executive function, memory, language, and conceptual thinking. The total possible score is 30, persons scoring below 26 are considered to exhibit mild cognitive impairment. 25 The MoCA has been used in some studies to diagnose HAND, although it has been shown to be more suited for detecting mild cognitive impairment.26,27 The MoCA has also been used successfully in longitudinal studies, though none were focused on adults living with HIV.28–30 The MoCA was chosen for its brevity as an assessment tool, making it easily adaptable to the clinical setting.
CES-D
The CES-D 31 is a 20-item self-administered depression questionnaire. Total scores range from 0 to 60 with scores greater than 16 indicating clinically significant depressive symptoms. Scoring for each item is on a 4-point scale ranging from 0 (rarely or none of the time) to 3 (most or all of the time). Scoring on 4 of the items is reversed. The CES-D has reported a Chronbach’s alpha of 0.94. It has been used in many large-scale HIV research studies.32–34 The CES-D assesses depressive symptoms for the past 7 days and takes about 10 min to complete.
PSQI
The PSQI is a 19-item self-report instrument that measures sleep quality over a 1-month period. The PSQI reports a global sleep score and seven component scores. Possible total scores range from 0 to 21, with global scores >5 indicating poor sleep. The overall instrument has a Chronbach’s alpha of 0.83. 35 The PSQI is one of the most commonly used sleep measures and takes about 10 min to complete.
Demographic form
A demographic form was developed to gather data on participant characteristics, including gender, age, ethnicity, education, and health information. Health information included number of medications, comorbidities, years since HIV diagnosis, years taking HIV medications, current HIV viral load, and current and nadir CD4+ T cell count. History of alcohol, tobacco, and drug use was also obtained. This form was completed face-to-face with the participant and medical information verified with information obtained from the participant’s medical record. The most recent HIV viral load and CD4 counts were used for study entry.
Intervention
The computer training was designed as an in-home intervention, using a desktop or laptop computer with Internet connection. BrainHQ, developed by Posit Science (San Francisco, CA, USA) is a commercially available product that provides brain-training exercises designed to improve cognitive performance. Participants were asked to train three times per week for 30 min at a time for 8 weeks. At the 4- and 8-week study visits, participants were asked to self-report the number of times they accessed the program each week and the time spent on the program. The program uses an algorithm, so exercises increase in difficulty as participants’ progress, so exercises constantly change and remain challenging. Exercises are based on the concept of brain plasticity, the idea that cognitive training can stimulate positive changes in cognitive function. BrainHQ has been used in numerous research studies, including studies of adults living with HIV.19,36,37
Control
Participants randomized to the control group followed the same visit schedule as those in the intervention group. Each week participants received a health-related newsletter via email that focused on healthy aging, including tips on diet, sleep, and exercise. Control participants also received a phone call 2 days after the email was sent to confirm receipt of the newsletter and answer any questions participants may have had about the content.
Procedure
Written informed consent was obtained from each study participant prior to performing any study-related procedures. During screening, participants completed Version 7.1 of the MoCA, a short, 30-item researcher-administered test to assess cognitive function. Participants scoring <26 were deemed to have mild cognitive impairment and were eligible to participate in the study. A cutoff score of 26 is the suggested cutoff, demonstrating a sensitivity of 90% and specificity of 87% in detecting mild cognitive impairment. 25 The MoCA has been used in previous HIV studies examining cognition using the cutoff of 26.22,38 All study visits took place in a private consultation room at the AIDS Clinical Trials Unit, housed in the same building as the Infectious Disease Outpatient Clinic.
At study entry, participants were randomly assigned to either the computer-training intervention or the control group. Randomization was accomplished by presenting study participants with sealed envelopes. Each envelope contained a card with ‘group 1’ or ‘group 2’ printed on it. The participant was assigned according to the envelope they chose. At study entry, all participants completed a data form and health-related questionnaires, as well as sleep and depression surveys. Participants in the intervention group were then enrolled in the home-based computerized cognitive training program for a period of 8 weeks. Participants in this group were instructed on how to use the program, were given their password, and then practiced with the program until they felt capable of accessing and using the program at home on their own. Passwords were recorded on study visit sheets and participants were instructed to call the study team for any problems they encountered logging in or using the program. A research assistant blinded to group assignment performed cognitive testing at study visits.
Study team members explained to participants in the control group that they would receive weekly email newsletters related to healthy aging for the next 8 weeks, the period of the intervention. Participants also received follow-up phone calls after the emails were sent to confirm receipt of the newsletter and to answer any questions participants had about the healthy aging-related content.
Each participant returned to the clinic 4 weeks after study entry, at which time they completed a medication adherence survey and updated any health-related information that may have changed since study entry. Participants in the intervention group completed a 30-min training session with the cognitive computerized training intervention at this visit.
All participants returned to the clinic again at 8 weeks to complete an alternative version of the MoCA (version 7.2), a depression survey, and a sleep survey. Participants in the intervention group also completed a final 30-min session with the computerized training intervention at this visit. Follow-up visits were performed at weeks 16 and 24, at which time participants completed versions 7.3 and 7.1 of the MoCA, respectively, along with the depression and sleep surveys.
Statistical analysis
Descriptive statistics were used to summarize demographic and clinical characteristics of study participants. Descriptive, paired t-tests and analysis of covariance (ANCOVA) were used to examine group differences from baseline to post-tests, using baseline scores as the covariate. Assumptions of normality, homogeneity of variance, and linearity were satisfied using ANCOVAs to test for treatment effects. Alpha was set at 0.05.
Results
Sample characteristics
Two participants, both randomized to the intervention group, did not complete the study, resulting in a lost-to-follow-up rate of 8%. One of these participants could not be located to ascertain reasons for not completing the study and the second informed the researchers he did not feel he had the time required to complete the intervention and withdrew from the study. Demographic data on these two participants were similar to that of participants who completed the study. As data were not collected for either participant after the week 4 study visit, their data were not used in the analysis.
A total of 22 participants (92%) completed the study (Table 1). Average age of study participants was 56 years [standard deviation (SD) = 5.15 years]. The age for participants in the intervention group was slightly older than that of the control group, 58 (SD = 6.5) years versus 54 (SD = 3.0) years, but the difference was not significant (p = 0.10). The majority of participants in both groups were male (100% in the intervention group and 75% in the control group), again, this difference was not significant (p = 0.082). The majority were African American, 50% in the intervention group and 67% in the control group. Years of education were similar between the two groups (p = 0.54), with the intervention group reporting 14.6 (SD = 1.7) years of education and the control group reporting 14.1 (SD = 2.1) years. There were no significant differences in drug (p = 0.33) or tobacco (p = 0.11) use between the groups. The only variable that differed significantly between the two groups was current use of alcohol (p = 0.04). Of the intervention group, 40% reported current alcohol use, while 83% of the control group reported current alcohol use. Data were not collected about the amount of alcohol, drugs, or tobacco used by study participants. The only data collected were if the use was current, in the past, or never.
Demographics of study participants (n = 22).

CES-D: Center for Epidemiological Studies Depression Scale; PSQI: Pittsburgh Sleep Quality Index; SD: standard deviation.
Overall, participants in the study had well-controlled HIV infection. Only 2 (0.09%) had detectable viral loads. Current CD4 counts, which were similar between groups, had a mean of 602 (SD = 437) in the intervention group and a mean of 539 (SD = 284) in the control group. Nadir CD4 counts were also similar, with a mean of 183 (SD = 153) in the intervention group and a mean of 237 (SD = 228) for the control group. Years since HIV diagnosis was also similar, with a mean of 19 years (SD = 6.5) for the intervention group and a mean of 18 years (SD = 9.2) for the control group. Number of comborbidities was also similar between groups, with 4.5 (SD = 2.27) in the intervention group and 4.7 (SD = 3.6) in the control group.
Sleep and depression scores were collected at baseline and were not significantly different between groups. Mean scores on the PSQI for both groups were above the 5.0 cutoff indicating poor sleep. The intervention group had a mean of 8.2 (SD = 3.85), and the control group reported a mean of 8.9 (SD = 4.93). On the CES-D, the intervention group had a mean score of 18 (SD = 12.06) and the control group reported 16.1 (SD = 10.80). Scores for both groups were indicative of clinically significant depressive symptomology.
Effect of intervention
On the basis of the scores on the MoCA, we found statistically significant improvement in cognitive performance after just 8 weeks of the computer-training intervention. Furthermore, we found that these gains were seen 16 weeks after completing training. Participants randomized to the intervention group used the cognitive training program between 4 and 16 h over the course of 8 weeks (M = 10.48, SD = 3.71). There was statistically significant improvement in the intervention group on total MoCA scores immediately after the cognitive intervention (week 8) compared to the control group: F(1, 19) = 4.92, p = 0.04. The partial Eta squared of 0.32 indicates a small-to-moderate effect size.
Analyzing component scores, statistically significant improvement was seen in Executive Function in the intervention group compared to the control group at week 8: F(1, 19) = 9.09, p = 0.007, with a small-to-medium effect size of 0.32. Changes in the memory and attention components improved, but were not significantly different in the intervention group compared to the control group: F(1, 19) = 3.07, p = 0.096 and F(1, 19) = 2.23, p = 0.152, respectively, and a small effect size of 0.14 was seen for each.
At 8 weeks post intervention (week 16 of study), total MoCA scores for the intervention group were not significantly different than the control group after controlling for entry MoCA scores F(1, 19) = 0.14, p = 0.71. Executive Function was also no longer significantly different, F(1, 19) = 1.83, p = 0.19. There was no longer a statistically significant difference in attention between groups F(1, 19) = 0.031, p = 0.86.
At 16 weeks post intervention (week 24 of study), as in week 8 post intervention, there were no longer any significant differences between groups on the total MoCA score or component scores of Executive Function or Attention.
Total scores on the MoCA showed statistically significant improvement at each measured time point compared to the screening MoCA in the intervention group. Scores dipped from 24.1 to 23.2 between immediate post intervention and 8 weeks post intervention before rebounding to the week 8 level (immediate post intervention) at 16 weeks post intervention. The control group score changes improved, but were not significant at any of the measured time points (Table 2).
MoCA scores at each time point for intervention and control groups.
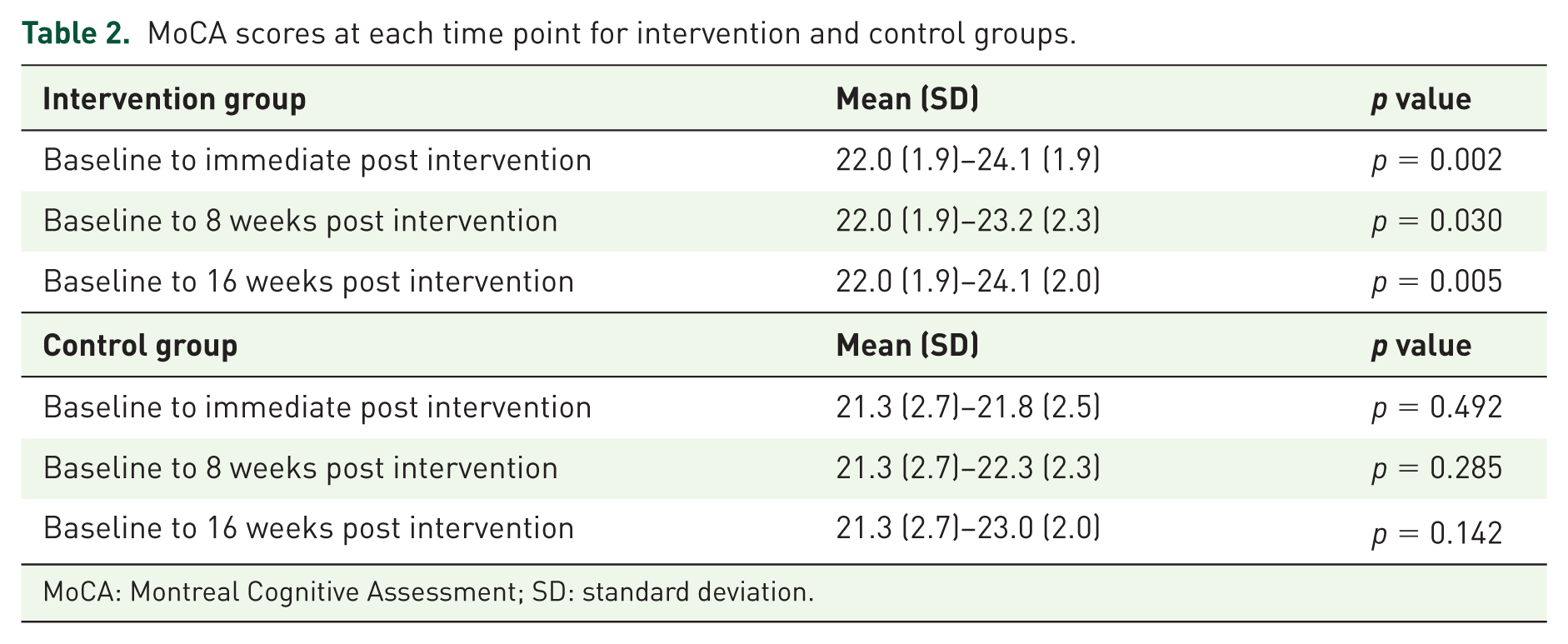
MoCA: Montreal Cognitive Assessment; SD: standard deviation.
Scores for the memory component did not improve from baseline to week 8 in the intervention group; however, they showed statistically significant improvement 8 and 16 weeks post intervention compared to baseline scores. This result was not significantly different from the control group, however, as their scores also improved from baseline. Attention scores improved at each time point for the intervention group; however, the improvement was not statistically significant. Attention scores for the control group remained similar at each time point, exhibiting no improvement over the course of the study (Table 3).
Total and component scores of MoCA by group at each time point.
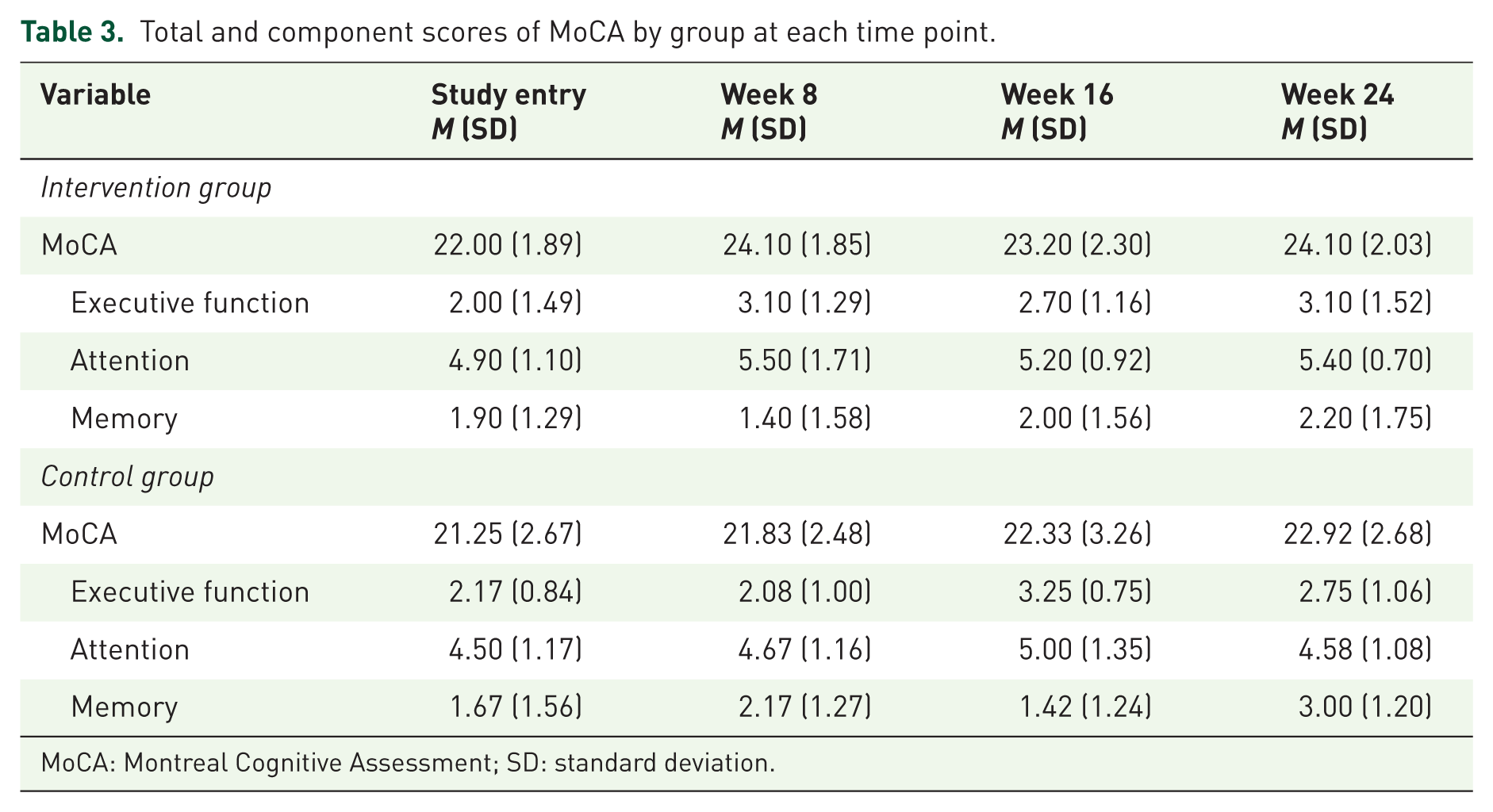
MoCA: Montreal Cognitive Assessment; SD: standard deviation.
Discussion
The purpose of this study was to examine the effectiveness of using a computerized training program to improve cognitive function in older adults living with HIV who exhibited mild cognitive impairment. In this study, overall cognitive function scores improved, as did executive function scores. Score changes for the memory component were not significant at week 8 compared to the entry scores, but at 8 and 16 weeks post intervention, scores were significantly improved. Attention scores improved, though not statistically significant in the intervention group, while the control group scores showed very little change from baseline. Memory results were not significantly different between groups, as both groups improved from baseline.
Cognitive function, including executive function, attention, and memory, has been shown to be an important predictor of medication management in older adults.39,40 These findings were also reported with adults living with HIV even in middle age.6,7 Because adults living with HIV may exhibit many symptoms associated with aging earlier than HIV-negative adults, including cognitive impairment, finding an intervention that could improve cognitive function or slow cognitive decline will make successfully managing HIV throughout the aging process easier.
Retention in the study was 92%. Participants received reminder phone calls the day before each of their scheduled appointments; in addition, they were compensated for each of their study visits. If participants missed an appointment, they received up to three phone calls to reschedule the appointment. These methods proved very effective in retaining study participants. It should be noted, however, that not all participants in the intervention group completed all training sessions over the course of 8 weeks. The amount of time participants used the computer-training program at home ranged from 4 to 16 h, and this suggests that computer training may not be an intervention all people would continue on their own. Computer training could be one option offered to patients to improve cognitive function, but other interventions, such as exercise or pharmacological interventions, may be better suited for some.
The prevalence of depressive symptoms and sleep problems in this population was somewhat of a surprising result. Of the 22 study participants, 19 (86%) had a global sleep score >5 on the PSQI, indicating poor sleep quality, and 12 (55%) had scores on the CES-D of 16 or greater, indicating clinically significant depressive symptoms. Sleep problems and depressive symptoms can adversely affect cognitive function in adults living with HIV and may have been contributing factors to the cognitive impairment experienced by study participants.5,41 Future studies should examine the effects of improving sleep and treating depression as additional methods of improving cognitive function in this population.
Limitations
The small sample size was a limitation of this study. However, significance was seen even with the small sample size, suggesting the need for further research with larger sample sizes. Two participants did not complete the study, and both were from the intervention group. This may suggest that computer training may be a viable option for some, but other options may need to be considered for others. Offering patients several choices of ways to improve cognitive function allows patients to choose options that work best for them. It is unclear why MoCA scores in the intervention group fell slightly 8 weeks post intervention from the scores immediately after the intervention before rising back to levels seen immediately post intervention at the 16-week post-intervention visit. A longer follow-up period in future studies would help determine how long the positive effects of the intervention could be sustained.
A nonactive control group was another limitation of this study. Future studies could compare cognitive training with other interventions designed to improve cognitive function.
Conclusion
Successful aging can be a challenging endeavor for people living with HIV. Cognitive impairment, physical and psychological comorbidities, polypharmaceutical issues, and the stigma of living with HIV present imposing obstacles to effectively managing HIV disease throughout the lifespan. Finding practical interventions, such as computer-based cognitive training, offers older adults an option that may improve cognitive function and does not require additional medications or added healthcare visits. Looking for opportunities to involve patients in successfully managing their disease can improve health outcomes and help persons to age successfully in spite of their HIV infection. While this study showed improvements in several cognitive areas, longer longitudinal studies are needed to assess the clinical significance of these changes.
Footnotes
Author notes
The content is solely the responsibility of the authors and does not necessarily represent the official view of the NIH.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by a grant from the Office of Nursing Research at Goldfarb School of Nursing at Barnes-Jewish College and the Washington University Institute of Clinical and Translational Sciences grant UL1TR000448 from the National Center for Advancing Translational Sciences (NCATS) of the National Institutes of Health (NIH).
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
