Abstract
Introduction:
Ethambutol (EMB) is an anti-mycobacterial agent that is most commonly used in combination with other anti-tuberculosis (TB) drugs in the treatment of TB. Studies have shown that primary resistance rates of
Methods:
Several databases, including Medline, Embase, and Iranian databases, were searched from March 2000 to January 2016 to identify studies addressing EMB-resistant TB in Iran. Comprehensive meta-analysis (V2.2, Biostat) software was used to analyze the data.
Results:
Of the 112 records identified from the databases, 10 studies fulfilled the eligibility criteria. The pooled prevalence of primary EMB-resistant TB was estimated at 4.2% [95% confidence interval (CI) 1.8–9.0]. No evidence of publication bias was observed among the included studies (
Conclusion:
Results of systematic review and meta-analyses indicated that effective strategies to minimize the acquired drug resistance, to improve the drug susceptibility testing (DST) capability, and to control the transmission of resistance should be attached importance for control of TB in Iran.
Introduction
Tuberculosis (TB) caused by
Method
Literature search and inclusion criteria
To identify relevant studies, we conducted a literature search in the bibliographic databases Medline (via PubMed), Web of Science, Embase, and Iranian databases from March 2000 to January 2016. Key words used in the search included ‘tuberculosis’, ‘primary drug-resistance’, ‘ethambutol’, and related terms. Only studies that used standard methods for drug susceptibility testing (DST) of
Data extraction
Data were extracted using an extraction form by the first author and were verified by the second author. The following data were extracted: first author, publication year, study setting, enrollment time, number of patients investigated, drug resistance profile, human immunodeficiency virus (HIV) status, and distribution of age and sex in the study population.
Quality assessment of studies
Quality assessment of included studies was performed using checklist provided by the Joanna Briggs Institute [2014].
Statistical analysis
Meta-analyses were performed using Compre-hensive Meta-Analysis version 2.2 (Biostat). Heterogeneity in the reported effects was examined using a fixed and a random effects model. Heterogeneity between studies was measured using
Results
Of the 112 records identified from the Medline (Via PubMed), Web of Science, Embase, and the Iranian database searches, 21 were considered for evaluation of the full manuscript. Of the full-text articles reviewed, 10 studies fulfilled the eligibility criteria (Table 1). Figure 1 shows why records were excluded based on the assessment of title/abstract and full-text articles. The final dataset included data from different TB centers of Iran.
Characteristics of studies included in the meta-analysis.

EMB: ethambutol.
Any EMB resistance.
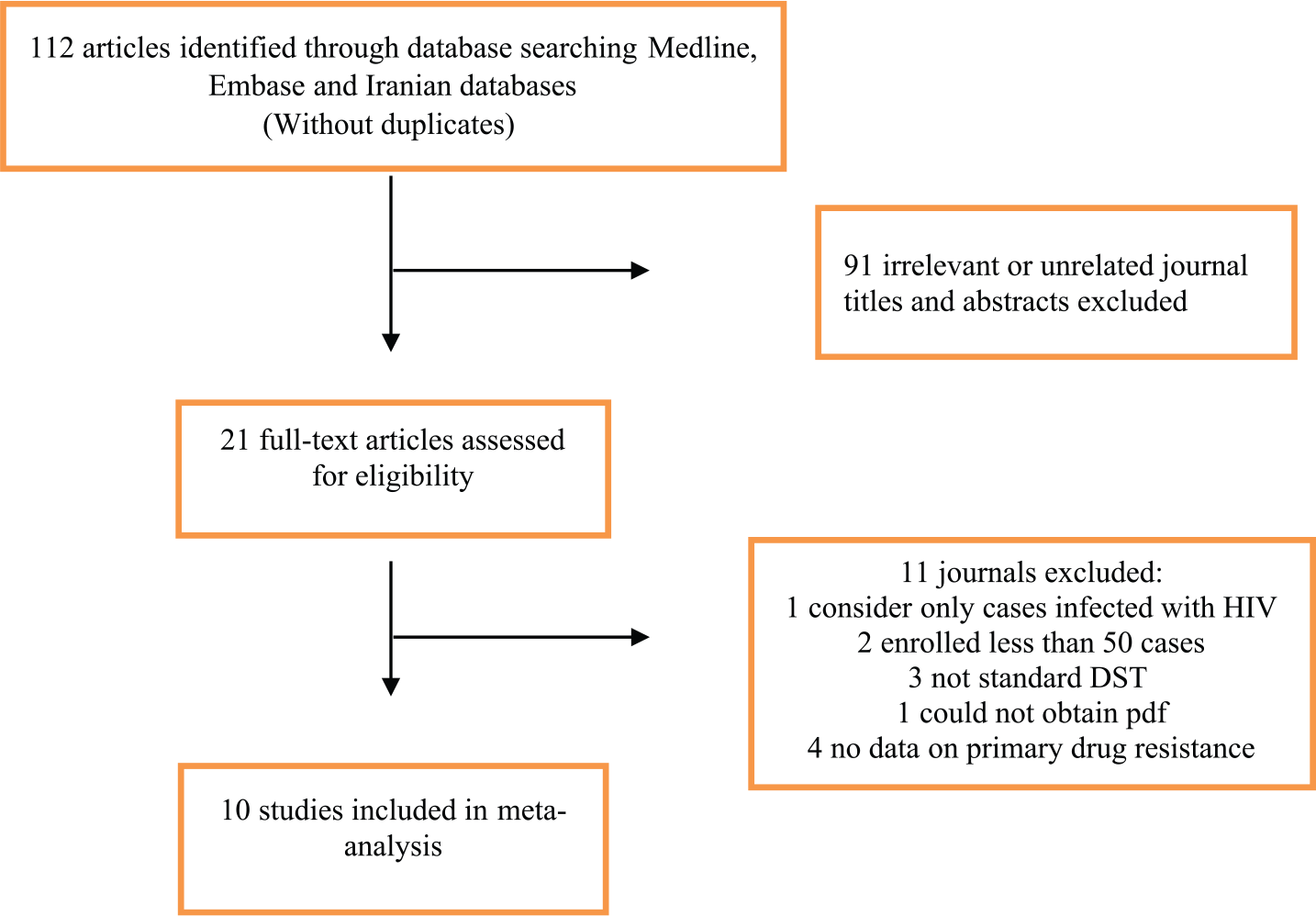
Flow diagram of search strategy.
Of the 10 studies, the pooled prevalence of primary EMB-resistant TB was estimated at 4.2% [95% confidence interval (CI) 1.8–9.0]. The results were found to be frequently heterogeneous (
Meta-analysis of prevalence of primary EMB resistance in Iran.
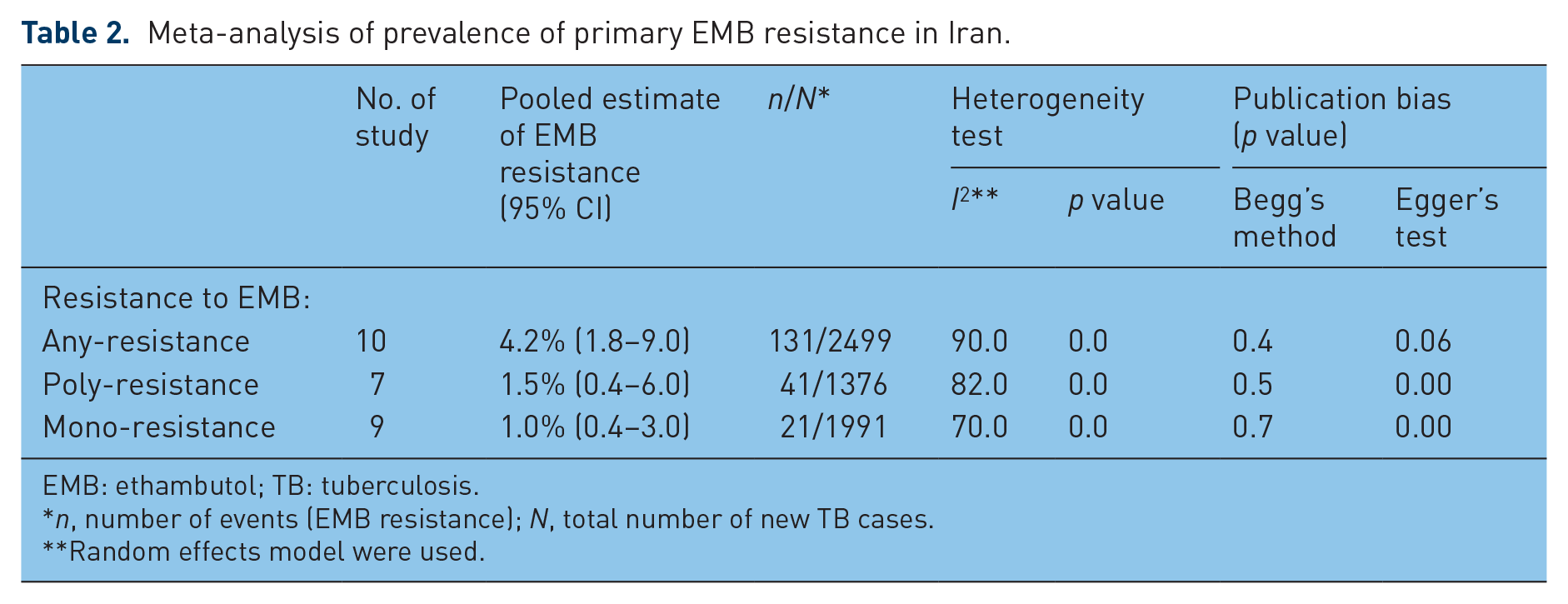
EMB: ethambutol; TB: tuberculosis.
Random effects model were used.

Forest plot of the meta-analysis on any EMB-resistant cases.
As shown in Table 2 and Figure 3, no evidence of publication bias was observed (

Funnel plot of the meta-analysis on any EMB-resistant cases.
Discussion
EMB is one of the key components of the first-line anti-TB drugs and is added to the current treatment regimen for TB as a protection against unrecognized resistance to other core drugs (INH, RIF, and PZA) [Horsburgh et al. 2015]. Increasing resistance to EMB is associated with increased risk of unsuccessful TB treatment outcome and acquiring resistance to other anti-TB drugs [WHO, 2011]. Therefore, understanding the current situation of primary EMB-resistant TB is necessary for the programmatic management of TB cases in Iran. Studies have shown that primary resistance rates of
In many low- and middle-income settings, due to inadequate laboratory capabilities for DST, most of the MDR-TB cases are treated based on the standardized regimens that are recommended by WHO and the Union [Caminero and Van Deun, 2013; WHO, 2014]. These regimens use drug resistance survey data from a representative patient population but do not use DST results from the individual patient’s isolate [Caminero and Van Deun, 2013; Daley and and Caminero, 2013]. However, there are conflicting opinions about drug-resistant cases other than MDR-TB, largely because of the paucity of strong evidence. Definitive randomized or controlled studies have not been performed to determine the best treatment regimens for various patterns of drug resistance [WHO, 2011]. Treatment of these cases with first-line drugs alone will likely result in failure. Thus, when a decision has been made to modify standard chemotherapy, the most effective regimen should be chosen to maximize the likelihood of cure. In this regard, different regimens have been suggested by WHO for common patterns of mono- and poly-resistant TB [WHO, 2014]. For example, for EMB- and INH-resistant TB, the WHO Guide recommends a regimen with 9–12 months of RIF, PZA, and levofloxacin [WHO, 2014]. Some experts also recommend using a second-line injectable agent for treatment [Rich, 2013; Varaine and Rich, 2013]. It should be taken into consideration that, if there is a high probability that the effectiveness of any of the first-line drugs has been compromised, the patient should be treated with an MDR-TB regimen [WHO, 2014]. Therefore, using rapid DST methods to evaluate the status of resistance to anti-TB drugs during therapy is crucial for treatment of patients with mono- and poly-resistant TB.
The heterogeneity of estimates between studies and limitations associated with potential publication bias is considered a limitation of this study.
In conclusion, this review provides an overview of prevalence of primary EMB-resistant TB in Iran. Our results suggest that effective strategies to improve the DST capabilities, to minimize the acquired drug resistance, and to control the transmission of resistance should be attached importance for control of TB in Iran.
Footnotes
Acknowledgements
We also would like to thank from the “Clinical Research Development Center of Baqiyatallah hospital” for their kindly cooperation.
Funding
This study was supported by Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
