Abstract
Objective
A systematic review and meta-analysis of randomized controlled trials (RCTs) studying the clinical benefit of chemotherapy with surgical intervention over chemotherapy alone for the treatment of spinal tuberculosis.
Methods
Relevant RCTs were identified by computerized database searches. Trial eligibility and methodological quality were assessed and data were extracted and analysed using odds ratios with 95% confidence intervals. The primary outcome measure was kyphosis angle.
Results
The literature search identified two RCTs conducted in the 1970s and 1980s and a Cochrane Database Systematic Review published in 2006. There were no significant between-group differences in kyphosis angle, bony fusion, bone loss or development of neurological deficit.
Conclusions
There is no obvious statistically significant clinical precedence to suggest that routine surgery will improve the prognosis of patients with spinal tuberculosis.
Keywords
Introduction
Spinal tuberculosis (TB) or Pott disease was first reported in 1779 and represents <1% of all TB cases.1–3 Given the increasing prevalence of TB worldwide, spinal TB is a significant healthcare issue, 4 with the incidence of neurological complications varying between 10% and 43%. 1
The standard management protocol for spinal TB is isoniazid, rifampicin and pyrazinamide chemotherapy for a minimum of 6 months.5–7 In addition, debridement of diseased tissues is routinely performed via an anterolateral extrapleural approach,8–11 transpleural anterior approach8,12,13 or posterior spinal fusion.14,15 Pre- and postsurgical debridement chemotherapy is commonly employed. 16
A review article suggested that surgery in spinal TB is an incidental decision, with no established protocol for the decision-making process and few randomized controlled trials regarding indications for surgery. 17 Surgical intervention decisions are based on: (i) patient age; (ii) presence of comorbidities; (iii) location of bony loss; (iv) relative position of the compressive lesion with regard to the dura, and density of the compressive lesion; (v) number of segments involved; (vi) kyphosis angle; and (vii) region of focal infection,17,18 with the major contraindications being high cost and risk of postoperative complications. 17 It is imperative to perform surgery only if the outcome will be more beneficial than treatment with chemotherapy alone. The objective of the current review of randomized controlled trials was to compare the outcome of chemotherapy alone or a combination of surgery and chemotherapy in patients with spinal TB.
Materials and methods
Data sources and searches
Computerized searches were performed to identify randomized controlled human trials listed in Ovid MEDLINE® (1970 – December 2012), Ovid EMBASE® (1974 – December 2012), SCOPUS (1996 – December 2012), Web of Science® databases including Science Citation Index Expanded™ (1996 – December 2012), Conference Proceedings Citation Index – ScienceSM (1990 – December 2012), Database of Abstracts of Reviews of Effects (DARE) (Issue 3 of 4, July 2012), Cochrane Central Register of Controlled Trials (CENTRAL) (Issue 3 of 4, July 2012) and ClinicalTrials.gov (up to December 2012). Both database-specific controlled vocabulary and general free text terms were used to maximize retrieval. MeSH terms used were: Pott disease; tuberculosis; spinal tuberculosis; tuberculous spondylitis; medical management; chemotherapy; surgery; surgical intervention; randomized controlled trial. Hand searching of key article reference lists was used to locate additional relevant articles.
Assessment of methodological quality
Eligibility assessment and data extraction were both performed independently in an unblended standardized manner by two independent reviewers (X.Z. and B.L.) according to Critical Appraisal Skills Programme (CASP) guidelines. 19 The inclusion of all randomized participants in the final analysis was evaluated and at least 60% completeness of follow-up at each time point was fixed as the threshold. The physicians treating the patients were not blinded either during treatment or follow-up. Scars were visible on X-radiographs of patients included in the studies used in the meta-analysis.
Data extraction, synthesis and analysis
The results of all searches were combined and duplicate articles were removed. Inclusion criteria were: (i) ≥1 year follow-up after initiation of treatment regimen; (ii) confirmed diagnosis of active TB of the thoracic and/or lumbar spine, including the upper sacral vertebra S1; (iii) diagnosis based on X-radiographs showing loss of thin cortical outline and rarefaction of the affected vertebral bodies; (iv) intervention was chemotherapy alone or chemotherapy plus surgery.
Statistical analyses
The outcomes (kyphosis angle, bone loss, bony formation and neurological deficit) of the collected manuscripts were synthesized in piloted forms independently and in duplicate (X.Z. and B.L.) and formed the basis for meta-analysis, which was performed according to Cochrane Collaboration and the Quality of Reporting of Meta-analyses (QUORUM) guidelines. 20 Odds ratios (OR) were used to analyse all dichotomous outcome measures, and data were presented with 95% confidence intervals (CI). The risk of bias of each relevant article was assessed using the 12 criteria recommended by the Cochrane Back Review Group. 19 Criteria were scored ‘yes’ (criterion met), ‘no’ (criterion not met) or ‘unsure’ (not enough information to make the decision). Articles that met six of the 12 criteria were considered to have a low risk of bias. Potential publication bias was tested using a funnel plot. Data were compared using χ2-test. Statistical analyses were performed using SPSS® version 18.0 (SPSS Inc., Chicago, IL, USA) for Windows®. P-values <0.05 were considered to be statistically significant.
Results
The literature search identified 35 potentially relevant articles, of which 2816,21–47 were excluded (Table 1, Figure 1). In addition, a systematic review published in 2006 was identified,
17
which analysed seven papers reporting on two randomized controlled trials (total n = 331) (Table 2).48–54 Both trials were initiated by the British Medical Research Council (MRC) Working Party on Tuberculosis of the Spine, with one performed in cooperation with the Indian Council of Medical Research (ICMR). These trials formed the basis of the current analysis.
Schematic diagram of literature search performed in the current systematic review and meta-analysis of the management of spinal tuberculosis. Characteristics of studies excluded from a review of the medical management of spinal tuberculosis. ICMR, Indian Council of Medical Research; MRC, Medical Research Council. Characteristics of studies included in a review of the medical management of spinal tuberculosis. ICMR, Indian Council of Medical Research; MRC, Medical Research Council.

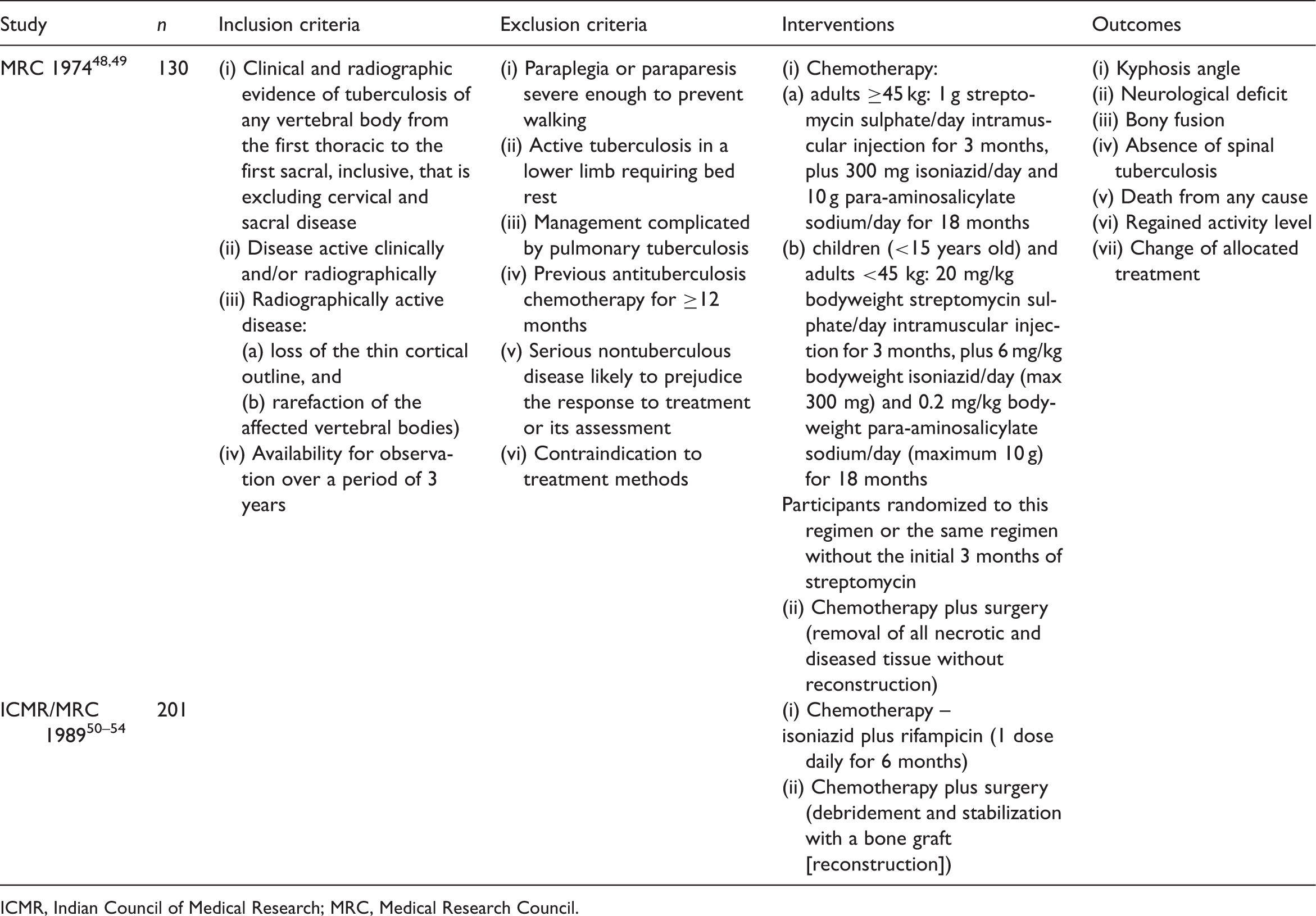
Characteristics of patients included in two randomized controlled trials comparing chemotherapy and chemotherapy plus surgery for treatment of spinal tuberculosis.

Data presented as n of patients or mean (SD not available).
Includes patients available at 3-year follow up.
ICMR, Indian Council of Medical Research; MRC, Medical Research Council.
Comparison of mean kyphosis angle in two randomized controlled trials comparing chemotherapy and chemotherapy plus surgery for treatment of spinal tuberculosis.

Data presented as mean (SD not available).
ICMR, Indian Council of Medical Research; MRC, Medical Research Council.
Both trials reported on the neurological status of the participants. No participants who were free from neurological deficit on entry developed any deficit, and there were no statistically significant changes over time in the numbers of patients with neurological deficit (n = 23 at entry, n = 23 at 18 months, n = 23 at 3 years and 20 at 5 years).48–54
ICMR, Indian Council of Medical Research; MRC, Medical Research Council.
Publication bias within the studies were assessed both visually, using a funnel plot (Figure 2), as well as the 12 criteria recommended by the Cochrane Back Review Group
19
(Figure 3). There was no evidence of publication bias on the funnel plot, and the number of criteria met was 9/12 in both studies. All of the studies included in the current meta-analyses were thus considered to have a negligible risk of bias. None of the included studies had an adequate description of withdrawals or drop outs (Figure 3).
Funnel plot of studies included in the current meta-analysis of randomized controlled trials comparing chemotherapy and chemotherapy plus surgery for treatment of spinal tuberculosis. There was no evidence of publication bias. Overall quality of the evidence for each outcome in studies included in the current meta-analysis of randomized controlled trials comparing chemotherapy and chemotherapy plus surgery for treatment of spinal tuberculosis (MRC 197448,49 and ICMR/MRC 198950–54). Quality was assessed using an adapted GRADE approach, as recommended by the Cochrane Back Review Group.
19
The quality of the evidence for a specific outcome was based on the study design, risk of bias, consistency of results, directness (generalizability), precision (sufficient data) and potential bias for the reporting of results across all studies that measured that particular outcome. ITT, intention to treat.

Discussion
The objective of this systematic review and meta-analysis was to compare chemotherapy plus surgery with chemotherapy alone for treatment of spinal TB. There was no statistically significant benefit of routine surgery, and surgery had no effect on kyphosis angle. The incidence of progressive kyphosis and the kyphosis angle at study entry were high (>30°) for all participants.48–54 Spinal surgeons generally consider kyphosis >30° to be unacceptably high and an indication for surgical correction. 18
The current review revealed no between-group difference in bony fusion, which is often considered the best evidence of healing.46,47 Data on the speed of bony fusion were not provided in either trial, and it was therefore not possible to assess differences during early phases of treatment. The amount of bone is considered important for the stability of the spine; as both studies excluded patients with >3 U total bone loss, the role of surgery in these more severe cases could not be assessed. A small number of participants had neurological deficit at study entry, but there were no statistically significant between-group differences in the improvement of this deficit. Deterioration of neurological deficit or persisting deficit with spinal cord compression can be an indication for surgery, 17 and a small group of patients (n = 5/130) in the chemotherapy group required surgery to decompress the spinal cord.48–52
The current analysis has several limitations, the most important of which is the availability of only two relevant studies, severely restricting the value of the meta-analysis findings. In addition, there was inadequate follow-up data for the MRC trial48,49 at any time point, and at 10 years in the ICMR/MRC trial.50–54 Finally, both trials were performed almost three decades ago and their current clinical relevance is difficult to determine given the advances in medical and surgical management of spinal TB. It is therefore imperative that randomized controlled trials are initiated to assess the benefits of surgery in spinal TB in the current setting. Several small-scale non-randomized studies have been performed more recently,21-47 but the results of the present analysis suggest that routine surgery cannot be recommended unless within the context of a large, well conducted randomized controlled trial.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Acknowledgements
We thank everyone who helped with this study.

