Abstract
Invasive fungal infections are increasingly encountered with the expansion of iatrogenic immunosuppression, including not only solid organ and hematopoietic stem cell transplant recipients but also patients with malignancies or autoimmune diseases receiving immunomodulatory therapies, such as Bruton Tyrosine Kinase (BTK) inhibitor. Their attributable mortality remains elevated, part of which is a contribution from globally emerging resistance in both molds and yeasts. Because antifungal susceptibility test results are often unavailable or delayed, empiric and tailored antifungal approaches including choice of agent(s) and use of combination therapy are heterogeneous and often based on clinician experience with knowledge of host’s net state of immunosuppression, prior antifungal exposure, antifungal side effects and interaction profile, clinical severity of disease including site(s) of infection and local resistance data. In this review, we aim to summarize previous recommendations and most recent literature on treatment of invasive mold and yeast infections in adults to guide optimal evidence-based therapeutic approaches. We review the recent data that support use of available antifungal agents, including the different triazoles that have now been studied in comparison to previously preferred agents. We discuss management of complex infections with specific emerging fungi such as Scedosporium spp., Fusarium spp., Trichosporon asahii, and Candida auris. We briefly explore newer antifungal agents or formulations that are now being investigated to overcome therapeutic pitfalls, including but not limited to olorofim, rezafungin, fosmanogepix, and encochleated Amphotericin B. We discuss the role of surgical resection or debridement, duration of treatment, follow-up modalities, and need for secondary prophylaxis, all of which remain challenging, especially in patients chronically immunocompromised or awaiting more immunosuppressive therapies.
Introduction
Invasive fungal infections (IFIs) are increasingly encountered with the expansion of iatrogenic immunosuppression, including not only solid organ transplant (SOT) and hematopoietic stem cell transplant (HSCT) recipients, but also patients with malignancies or autoimmune diseases receiving immunomodulatory therapies. Despite advancement in diagnostic and therapeutic against IFI, their attributable mortality remains high. This high morbidity and mortality can be attributed in part to (1) increasing complex immunosuppressive therapy, (2) global emergence of resistance, (3) limited of access to antifungal susceptibility testing, and (4) antifungal treatment with limited efficacy and significant toxicity profile. This review summarizes previous recommendations and most recent literature on management of invasive mold and yeast infections in adults to guide optimal evidence-based therapeutic approaches.
Part 1: Treatment of invasive yeast infection
Candida spp
Antifungals arsenal
Over the last decade, the epidemiology of invasive candidiasis (ICs) has slowly evolved. Although Candida albicans still predominates, recent epidemiological data of IC have shown a global increase of non-albicans Candida spp.1–3 C. glabrata (Nakaseomyces glabrata) now accounts for a significant proportion (12–30%), part of which is the result of improved identification technologies and selective pressure from fluconazole use.3–6 The international SENTRY Antifungal Surveillance Program reported 8.1% fluconazole resistance among C. glabrata (2006–2016). 3 The rate of echinocandins resistance among C. glabrata was reported at 3.5%, but recent data suggest a higher rate among strains implicated in candidemia.3,7 Resistance strongly impacts mortality as shown in reports of invasive diseases with echinocandins resistant C. tropicalis among patients with hematological malignancies. 8 Thus, given the increasing rate of antifungal resistance, azoles susceptibility testing should be performed for all strains causing invasive disease and echinocandin susceptibility testing should be performed for C. glabrata and C. parapsilosis and for patients previously treated with an echinocandin. 9 Nevertheless, resistance defined by breakpoints may not be the only factor contributing to breakthrough infection as breakthrough fungemia with susceptible strains have been reported in immunocompromised patients with profound and prolonged neutropenia and/or compromised in the skin or mucosal barrier. 10
Management of IC among adults has not significantly changed over the last decade. Echinocandins are the first-line therapy for IC, regardless of species.11–13 Acceptable alternatives include fluconazole or liposomal amphotericin B (LAmB). Isavuconazole (ISA) did not achieve non-inferiority in overall response when compared to caspofungin (CAS) in the ACTIVE trial [60.3% for ISA versus 71.1% for CAS; adjusted difference −10.8 (95% CI −19.9, −1.8)]. 14 Newer triazoles should therefore not be used as first-line therapy. Changing antifungal class may be considered in the setting of breakthrough candidemia. A summary of recommendations is presented in Table 1.
Summary of available and investigational antifungal agents recommended for the treatment of invasive fungal infections.

ClinicalTrials.gov identifier: NCT05178862.
ClinicalTrials.gov identifier: NCT05421858.
ClinicalTrials.gov identifier: NCT03363841.
ClinicalTrials.gov identifier: NCT03059992.
ClinicalTrials.gov identifier: NCT03583164.
ClinicalTrials.gov identifier: NCT04240886.
ClinicalTrials.gov identifier: NCT05037851.
Combination therapy of echinocandin + LAmB if high rates of pan-resistance (e.g. South Asia).
Might be included in study as resistant or refractory IFI cases.
AFST, Antifungal Susceptibility Testing; AmBd, Amphotericin B deoxycholate; CAS, Caspofungin; CM, Cryptococcal meningitis; 5FC, Flucytosine; FLC, Fluconazole; IFI, Invasive fungal infection; ISA, Isavuconazole; LAmB, Liposomal Amphotericin B; PET, Pre-emptive Therapy; POS, Posaconazole; VRC, Voriconazole.
Stepdown therapy with fluconazole or voriconazole, based on antifungal susceptibility testing (AFST) results should be considered for patients who have cleared their candidemia. Higher fluconazole dosing should be used for C. glabrata (12 mg/kg daily) based on higher minimum inhibitory concentration (MIC) values (MIC 16–32 μg/mL).11,20,21 Antifungals should be given for a total of 14 days after blood clearance and central lines removed when feasible. 11 Neutropenic patients should have neutrophils recovery prior to discontinuation of antifungal therapy or transitioned to antifungal prophylaxis if indicated.
Adjunctive measures
In case of candidemia, removal of indwelling catheter is important and should be done as soon as possible.11,13 While there are no randomized controlled trial showing the superiority of early catheter removal among candidemic patients, several large observational studies have shown favorable outcome with early catheter removal. 22 When central venous catheter (CVC) removal is not feasible, LAmB and echinocandins should be considered for their effectiveness within the biofilm.13,23
Evaluation for metastatic foci should be performed especially if candidemia is prolonged or refractory to therapy. Although endocarditis is uncommon, its reported incidence among patients with candidemia varies between 2.5% and 11.9% and is sometimes suspected solely based on echocardiography imaging in patient without clinical sign. 24 Routine echocardiography to rule-out endocarditis remains controversial.24,25 The ESCMID guidelines recommend routine transesophageal echocardiography for all patient with candidemia; in contrast, t he Infectious Diseases Society of America (IDSA) guidelines recommends performing an echocardiogram only if endocarditis is suspected in the setting of persistent candidemia, a new heart murmur, heart failure, or embolic phenomena, occurring more frequently among patients with prior endocarditis, valvulopathy, or intravenous drug use.11,13,24,26,27 If endocarditis is confirmed, antifungal treatment should initially consist of LAmB (+/− flucytosine) or high-dose echinocandin, with subsequent stepdown to an azole, if susceptible.11,13 Therapy should be prolonged for at least 6 weeks and surgical valve replacement should be considered.11,13 Lifelong secondary treatment should be considered when surgery is not performed. 11
Ophthalmologic examination is necessary to assess the presence ocular candidiasis. Routine ophthalmologic examination among patients without ocular symptoms is a matter of debate among experts. The American Academy of Ophthalmology recommends against ocular exam for all patients with candidemia given the low reported incidence of true candidal eye diseases (<2%) and suggests referring only those with symptoms or those unable to report symptoms.28–30 On the other hand, both the IDSA and ESCMID guidelines advise for a dilated ocular examination performed by an ophthalmologist within 7 days of candidemia (or after counts recover if neutropenic), based on a higher incidence of ocular diseases (16%), mainly chorioretinitis.11,13 Ocular candidiasis is a challenging complication that warrants prolonged antifungal therapy with an agent with optimal ocular penetration (i.e. fluconazole, voriconazole, or a combination of LAmB and 5FC for 4–6 weeks) and may require intraocular intervention such as vitrectomy and intravitreal antifungal.11,28
Among neutropenic patients with candidemia, abdominal imaging [ultrasound, CT scan, or magnetic resonance imaging (MRI)] should be performed routinely to exclude hepatosplenic candidiasis. Repeat imaging should be done at the time of neutropenia recovery. Follow-up CT imaging should be obtained every 3 months for hepatosplenic candidiasis, and antifungal therapy should be continued until recovery or calcification of the lesions on imaging, which usually takes approximately 6 months. 11
Candida auris
C. auris has emerged as a serious global threat in over 30 countries and its multidrug resistance is associated with mortality as high a 60%.31–33 Several factors contribute to its recent rise, including increase in international travel, drug pressure, and challenges associated with its laboratory identification. 32 C. auris is typically resistant to azoles (90%) and can be variably resistant to echinocandins (2–10%) and amphotericin B (AmB) (8–35%).32,34,35 Extended AFST including azoles, echinocandins, and polyenes should be performed. Although there are no interpretative criteria, results are often inferred from closely related Candida spp. MICs or using the Centers for Disease Control and Prevention (CDC) tentative breakpoints.4,31 In North America where rate of echinocandin resistance to echinocandins is below 5%, echinocandins are the preferred class for initial therapy. Patients with breakthrough infection or with prolonged exposure to echinocandins should be treated with a combination therapy including LAmB with an echinocandin.31,32 In South Asia where the rate of multidrug resistance is higher (up to 30% resistant to three antifungal classes), initial combination therapy (echinocandin + LAmB) is recommended.33,36–38 Removal of CVC is especially important as C. auris is known to colonize skin and the environment which contributes to its nosocomial spread. 31
Future drug options for Candida spp. including C. auris
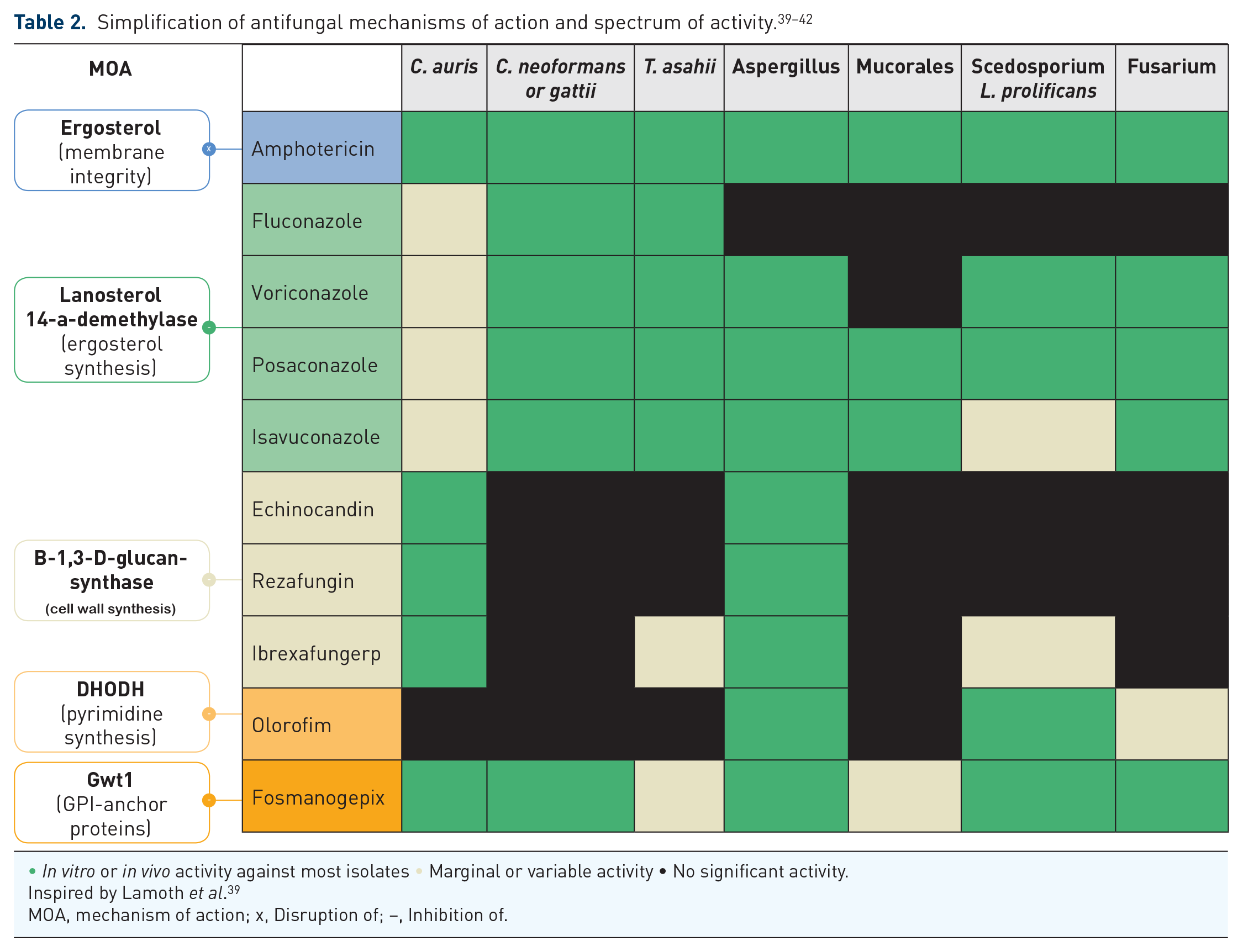
Newer antifungal agents or formulations are being developed and investigated in hope of facilitating management of ICs, including cases of C. auris. The most promising drugs will be briefly discussed here and are included in Table 1 (ongoing studies) and Table 2 (mechanism of action and spectrum of activity).
• In vitro or in vivo activity against most isolates • Marginal or variable activity • No significant activity.
Inspired by Lamoth et al. 39
MOA, mechanism of action; x, Disruption of; –, Inhibition of.
Rezafungin (RZF) is a new echinocandin with prolonged half-life formulated for once weekly intravenous administration. Its pharmacokinetics could eventually permit subcutaneous administration, making it an asset for outpatient management without the need for a CVC. 43 The STRIVE study, a phase II double-blind randomized trial evaluating RZF in comparison to CAS for the treatment of IC documented an overall cure rate (clinical and mycological cure) at 5 days of 62.3% for RZF versus 55.7% for CAS. 15 In the randomized phase III ReSTORE trial that followed, RZF was non-inferior to CAS (+/− stepdown to fluconazole) in global cure at 14 days in the modified intention-to-treat (ITT) analysis [59% versus 61%; −1.1% (95% CI −14.9, 12.7)]. 16
Ibrexafungerp (SCY-078) is a new oral antifungal of the glucan synthase inhibitor class. A phase II study (MSG-10) reported favorable and similar outcomes to fluconazole among six patients who received ibrexafungerp as stepdown therapy for IC. 17 A randomized, double-blind phase III study for treatment of IC is ongoing (MARIO study; ClinicalTrials.gov identifier: NCT05178862). It is also being studied for the treatment of C. auris infection in a single-arm, non-comparative phase III international study; interim results showed partial or complete response in 8/10 patients (CARES trial; ClinicalTrials.gov identifier: NCT03363841).
Fosmanogepix (APX001, E1211) is a novel antifungal with a new mechanism of action; it inhibits the Gwt1 enzyme involved in maturation of glycosylphosphatidylinositol or GPI-anchored proteins. It is available in both intravenous and by mouth formulations. Its spectrum of activity is broad and include many resistant yeasts and molds including Candida spp. and Aspergillus spp., with variable activity against Mucorales. 44 Fosmanogepix has however shown higher MIC for C. krusei, potentially secondary to efflux or cell permeability differences. 44 A phase II study (APX001-201) of fosmanogepix as first-line treatment was previously completed with a microbiological cure of 80% in non-neutropenic patients with candidemia.19,44 A phase III clinical trial is currently ongoing for first-line treatment of IC in comparison to standard of care (C4791012; ClinicalTrials.gov identifier: NCT05421858). Fosmanogepix could hopefully be used in the setting of non-krusei IC diseases that have failed or are resistant to fluconazole and echinocandins. It has also shown potent in vitro activity against C. auris, and it was effective in treating invasive C. auris infection in 89% (8/9) of invasive cases in South Africa (Clade III) in a phase II single-arm study. 19
Cryptococcus spp
Cryptococcus spp. (neoformans or gattii) is an opportunistic yeast that can cause severe pneumonia and disseminated disease, including debilitating central nervous system (CNS) infection among immunocompromised hosts. Management of cryptococcal meningitis (CM) has been an evolving topic. Most of the literature on treatment is based on randomized controlled trial (RCT) involving patients living with HIV who still represent most cases worldwide. However, cases are increasingly encountered among non-HIV immunocompromised hosts including patients with hematological malignancies and SOT recipients.45,46
Historically, the treatment of CM in patients with HIV included 2 weeks of AmB and flucytosine, based on data showing improved survival and better fungal clearance with combination therapy compared to AmB monotherapy.47–49 In 2013, a large RCT compared three treatment regimens against CM and observed a survival advantage among patients treated with AmB and flucytosine combination compared to AmB combined with fluconazole or AmB monotherapy, thus confirming that combination therapy is preferred over monotherapy. Subsequently, the ACTA trial confirmed that flucytosine as partner drug with AmB was associated with lower mortality than fluconazole. In addition, the ACTA trial demonstrated that 1 week induction therapy (with combination therapy) was non-inferior to the previously 2-weeks standard. 50 This was particularly helpful for low-income countries and was reflected in previous World Health Organization (WHO) guidelines, which recommended transition to high-dose fluconazole (1200 mg) after completion of 1 week of AmB and flucytosine. Most recently, the AMBITION trial changed the treatment paradigm in low-income settings. This trial showed that a single, high dose (10 mg/kg) of LAmB, given with an oral backbone of fluconazole and flucytosine, was noninferior to the WHO – recommended regimen of 7 days of amphotericin B deoxycholate (AmBd) plus flucytosine in patients living with HIV. 51 Fewer grade 3 or 4 adverse events were seen in the single high-dose LAmB group than in the control group (50.0% versus 62.3%). 51 The WHO has since updated its guidelines to reflect the results of the AMBITION protocol and now recommends a single high dose (10 mg/kg) of LAmB with 14 days of flucytosine (100 mg/kg per day divided into four doses per day) and fluconazole (12 mg/kg), as first line among patients living with HIV. 52 In case of refractory HIV-related CM, salvage therapy with the combination of LAmB, voriconazole, and recombinant interferon-γ (rINF-γ) may be considered. 53 Adjuvant rINF-γ 1b in treatment of CM in AIDS patients may be beneficial in some cases. 54
During induction therapy, repeated lumbar punctures should be done to ensure decrease in the opening pressure and again at day 14 to document clearance of yeast growth to transition to consolidation therapy.48,52 Routine use of adjunctive corticosteroid therapy during the induction phase is not recommended, except for major complication of intracranial hypertension.48,52 Recommended first-line consolidation treatment remains fluconazole (400–800 mg) for 8 weeks, followed by a lower dose (200–400 mg) in maintenance therapy for more than 6–12 months and until immune reconstitution in HIV.48,52
Future drug options for Cryptococcus
New drugs olorofim and rezafungin lack activity against Cryptococcus spp., but manogepix has shown activity in vitro.39,55 In vivo mouse study of encochleated AmB for CM showed promising results. 56
Trichosporon asahii
Trichosporon spp. are opportunistic yeasts that are being increasingly encountered among immunocompromised hosts, especially neutropenic patients. 57 They account for 4.5–20% of non-candida yeasts causing fungemia. 58 T. asahii (formerly T. beigelii) is the predominant species isolated in culture (73–74%).59,60 Trichosporon spp. are usually resistant to echinocandins. 59 High MICs to fluconazole and AmB are also frequently documented; voriconazole appears to have the lowest MICs.57,59,61 Antifungals activity against T. asahii are presented in Table 2.
Despite in vitro data, clinical response to triazoles, mainly voriconazole, seem to be favorable. In a large systematic review of published cases of T. asahii infections (n = 140), clinical improvement with triazoles was superior to AmB (74.1% versus 70.6%; p = 0.015). 57 Combination of a triazole and AmB was not beneficial compared to monotherapy (clinical efficacy 57.9% versus 74.1%; p = 0.25). 57 A large cohort of patients with trichosporonosis (n = 115; 73% T. asahii) reported better survival rate among patients treated with voriconazole compared with other antifungals (p = 0.042). 60 In that study, high MICs to fluconazole correlated with mortality in those receiving fluconazole. 60 Thus, based on in vitro and clinical data, the ESCMID/ECMM guidelines recommend voriconazole as first-line therapy for Trichosporon infection. 58 AmB is an alternative or adjunct in therapy.58,62 Data on the use of posaconazole or ISA are lacking. Duration of fungemia after blood clearance remains uncertain however treatment options, ranging from 6 weeks to 3 months, have been reported.63–65 Antifungal therapy should be continued until resolution of organ disease, if present.
Future drug options for T. asahii
Most antifungals in development are unlikely to provide good coverage for T. asahii. Fosmanogepix has shown poor activity in preliminary in vitro studies, although ibrexafungerp showed variable results.66,67 Some cases might be included in the ongoing FURI trial (ibrexafungerp for treatment of refractory IFI; ClinicalTrials.gov identifier: NCT03059992).
Part 2: Treatment of invasive mold infections
Aspergillus spp
Antifungals arsenal
The incidence rate of invasive aspergillosis (IA) has significantly increased in the last two decades with the expansion of transplantation and immunomodulatory therapies. 68 It is the most prevalent invasive mold infection (IMI) (19–43% of all IFI), and Aspergillus fumigatus remains the most frequent isolated species (58%).46,69,70 International surveillance data estimate that the resistance of A. fumigatus to azoles is approximately 1.4–5.8%, with higher rates reported in some European countries. 71 Cryptic Aspergillus spp. within the section Fumigati such as A. lentulus and A. udagawae are more frequently associated with higher azoles MIC values, and efforts should be made to differentiate those from A. fumigatus sensu stricto. 72 A. calidoustus (section Usti) has been associated with intrinsic pan-azole resistance and is therefore encountered more frequently among patients receiving triazoles prophylaxis.72,73 Reduced susceptibility to amphotericin is also described among several species notably A. lentulus and A. terreus. 74 Knowledge of the species may thus be helpful in guiding therapy, and advance in molecular diagnostics could help in this way. Previous guidelines did not suggest routine AFST for Aspergillus spp., however epidemiologic changes in immunocompromised patients, the increased use of antifungal prophylaxis, and emergence of resistance question whether AFST should be routinely performed. 75
Historically, AmB was the agent of choice for treatment of IA. In 2002, a RCT by Herbrecht et al. demonstrated a survival advantage with voriconazole over AmBd among patients with IA [70.8% versus 57.9%; Hazard Ratio (HR) 0.59 (95% CI: 0.40, 0.88)]. 76 Following this seminal trial, voriconazole became the first-line therapy for IA. More recently, the phase III SECURE study showed that ISA was non-inferior to voriconazole for all-cause mortality at 12 weeks [19 versus 20%; −1.0% (95% CI: −7.8, 5.7)] for the treatment of IMIs; encountered cases were mainly IA and half where proven/probable diagnoses. Significantly less adverse events were documented among patients treated with ISA compared to patients treated with voriconazole (42% versus 60% p < 0.001). 77 Similarly, posaconazole was non-inferior to voriconazole in all-cause mortality at 12 weeks [15% versus 21%; −5.3% (95% CI −11.6, 1.0)] while being associated with fewer drug discontinuation secondary to related adverse events. 78 As such, new triazoles are acceptable treatment options against IA and may be associated with fewer adverse events. Liposomal AmB remains an alternative for treatment of IA; however, its safety profile is less favorable due to risk of dose-dependent nephrotoxicity. 79 These recommendations are reflected in Table 1.
Combination therapy with voriconazole and anidulafungin has been compared to voriconazole monotherapy in a study by Marr et al. 80 and did not lead to difference in survival [mortality at 6 weeks 19.3% versus 27.5%; −8.2% (95% CI −19.0, 1.5); p = 0.087]. Of note, in the post-hoc sub-group analysis of patients with positive galactomannan, combination therapy was associated with a survival benefit [mortality at 6 weeks 15.7% versus 27.3%; −11.5% (95% CI −22.7, −0.4); p = 0.037]. As such, combination therapy is not routinely recommended; however, it may be considered for salvage therapy, refractory, or breakthrough IA or infection with known antifungal resistance. 75
Surveillance of short- and long-term azoles associated toxicities is important during treatment as prolonged duration is expected. Azoles use can lead to hepatotoxicity, drug–drug interactions, and QTc prolongation; exceptionally, ISA appears to shorten the QTc and may be an alternative agent for patients with prolonged QT.81,82 Voriconazole is also associated with a wide range of neurological, ocular, and cutaneous toxicities.83,84 Long-term use is associated with development of skin cancers and periostitis.84,85 Posaconazole can cause dose-dependent pseudohyperaldosteronism and hypokalemia. 86 Therapeutic drug monitoring (TDM) has been shown to decrease risk of voriconazole discontinuation secondary to adverse events and should be measured 4–7 days after start of therapy.87,88 However, it is unclear whether TDM correlated with clinical outcome as a recent prospective randomized study reported no difference in outcome when compared to standard dosing.87,89 Although optimal concentration are not perfectly defined, a through between 1–2 and 6 µg/mL is aimed for voriconazole and above 0.5–1.5 µg/mL for posaconazole. 88
Surgery, duration of therapy, and secondary prophylaxis
Surgical resection should be considered for locally invasive disease, infection in proximity to vital structures (e.g. heart, large vessels) or if further iatrogenic immunosuppression is expected. 90 Given the high propensity of Aspergillus spp. to disseminate, assessment of disseminated disease to the CNS and distant occult foci of infection should be considered. 91
Antifungal therapy should be continued until resolution of radiological and clinical disease, which is usually expected to take a minimum of 6 to 12 weeks. Follow-up imaging has limited value in the first 2 weeks of therapy, but is generally recommended at 6–12 weeks to assess disease response and duration of therapy. 75 Decreased in bronchoalveolar lavage galactomannan (GM) value is generally expected on therapy, but heterogeneity of specimens and the invasive nature of procedure limits its value for treatment follow-up. Serum GM trend has been proposed as a prognostic marker for neutropenic patients, and when positive should be repeated to assess treatment response.92,93
Because of concern for relapse, secondary prophylaxis may be considered in specific patient population.94,95 Most data supporting this strategy have been reported from retrospective studies where prophylaxis may decrease relapse among patients on chemotherapy or HSCT.96,97 Despite lack of robust data, both American (IDSA) and European (ESCMID-ECMM-ERS) guidelines give consideration for secondary prophylaxis in cases of persistent or subsequent immunosuppression.75,98 The American Society of Clinical Oncology and IDSA Clinical Practice Guideline update recommends prophylaxis with mold-active oral triazole (posaconazole, voriconazole, and ISA) or a parenteral echinocandin in patients experiencing extended periods of neutropenia and at >6% risk for IA. 99 When azoles are contraindicated due to toxicity or drug interactions, inhaled AmB or liposomal AmB may be considered. 100
Future drug options for Aspergillus spp
New antifungals in the pipeline with activity against Aspergillus spp. include: olorofim, fosmanogepix, ibrexafungerp, and opelconazole (see Table 2). Olorofim is the first agent of the new antifungal class named Orotomides; it inhibits the fungal dihydroorotate dehydrogenase enzyme involved in pyrimidine biosynthesis. 40 It was developed for both intravenous and oral administration. 40 It has good activity against Aspergillus spp. including azole-resistant isolates, but lacks activity against Mucorales and yeasts. 40 The FORMULA-OLS phase IIb study (ClinicalTrials.gov identifier: NCT03583164) assessing olorofim for the treatment of difficult-to-treat IMIs reported encouraging interim results with 44% treatment success (complete or partial response) and 14% all-cause mortality at 6 weeks. Tolerance was good with abnormal liver enzyme being the most commonly reported adverse event (8%) (ClinicalTrials.gov identifier: NCT03583164). Successful overall response was seen in 47% of IA cases. 101 The OASIS phase III study is currently evaluating olorofim in comparison to LAmB among patients with refractory IA or intolerant to azole therapy (ClinicalTrials.gov identifier: NCT05101187).
Fosmanogepix is also studied for the treatment of difficult-to-treat IMI and IA (ClinicalTrials.gov identifier: NCT04240886; AEGIS study). The FURI trial is evaluating the efficacy and safety of ibrexafungerp in a non-comparator single arm study for the treatment of refractory IMI, IC, and IA (ClinicalTrials.gov identifier: NCT03059992). Interim results reported improved or stable clinical status among 50% (5/10) of IA cases. 102 Opelconazole (PC945) is a new triazole agent optimized for inhaled formulation, with very limited systemic absorption and few CYP3A4 interactions, that will target non-Niger Aspergillus spp. 103 A phase II study is ongoing to look at its safety in the preemptive therapy setting in lung transplant recipients (ClinicalTrials.gov identifier: NCT05037851). Like with the inhaled formulation of amphotericin, the use of inhaled opelconazole could limit systemic toxicities of alternative therapies among transplant recipients with an indication for anti-mold prophylaxis and other non-neutropenic hosts with IA. Encochleated Amphotericin B (CamB; MAT2203) is a novel nanoparticle-based encochleated formulation of amphotericin that is protected from gastrointestinal degradation and therefore has higher oral availability and fewer systemic toxicities. 104 As such, CamB may become an interesting oral treatment option against IA; however, there are no clinical studies to supports its use for this indication to date.
Mucorales
Mucormycosis can cause life-threatening invasive rhinocerebral or pulmonary disease in both immunocompromised and immunocompetent hosts. Risk factors include diabetes, malignancy, bone marrow and organ transplantation, IV drug use, and deferoxamine therapy. 105 AFST are limited by the absence of recognized breakpoints or epidemiological cutoff values for Mucorales and when done, MIC/MEC correlation with clinical outcome is unclear. AmBd has the most favorable in vitro activity against Mucorales. 106 Available antifungal arsenal consists of AmB and newer triazoles. Early surgical debridement and prompt antifungal therapy are key elements in the management of this infection.
Antifungal arsenal
First-line therapy for mucormycosis is LAmB. Liposomal formulation is favored over the deoxycholate formulation due to its improved safety profile and similar efficacy.105,107,108 Optimal dosing remains uncertain, but higher dose have been associated with better outcome. 109 The non-comparative pilot study AmBiZygo has assessed the efficacy and tolerability of higher dose of LAmB. Doses of 10 mg/kg were associated with improved outcome compared to the results of the DEFEAT Mucor study where lower doses of LAmB were used (7.5 mg/kg/day).109,110 However, high-dose LAmB was associated with high rates of nephrotoxicity (40%). 109 Current guidelines recommend LAmB at a dose of at least 5 mg/kg per day for mucormycosis without CNS involvement, and 10 mg/kg/day in cases of CNS involvement.111,112
ISA and posaconazole are active against Mucorales. ISA was assessed in a phase III open-label non-comparative RCT for the treatment of mucormycosis (VITAL study). Outcomes were comparable to those of matched historical controls from the FungiScope registry treated with AmBd (mortality rate of 33.3% versus 41.3%, respectively). 113 Moreover, ISA has several advantage over LAmB including a good safety profile (most common side effects were gastrointestinal complaints), availability in both oral and intravenous formulations, fewer drug–drug interactions, and lack of need for TDM. Currently, there are no RCT comparing posaconazole to AmB for first-line therapy of mucormycosis. Data derived from cases prospectively included in international registries have documented a 40–67% survival rate among patients treated with posaconazole.114,115 Posaconazole as salvage therapy has been associated with favorable response ranging from 63% to 80%.115–117 As such, ISA and posaconazole are acceptable alternatives for treatment of mucormycosis, when LAmB is not tolerated, or in the setting of refractory disease. 111 Additionally, the oral formulations for both these triazoles make them suitable stepdown options. Caution should be exerted in the setting of ocular or CNS involvement due to concern of low penetration of these compartments of both posaconazole and ISA.118–121 Voriconazole lacks activity for Mucorales and should not be used for treatment of mucormycosis.
Because of its potentially fulminant progression and high mortality, using combination of antifungals that may have synergistic effects is tempting when treating mucormycosis. Multiple combinations have been studied, generally with LAmB as a backbone with addition of an echinocandin or a triazole. Although echinocandins are considered ineffective against Mucorales, one retrospective study suggested improved survival among patients with cerebral mucormycosis treated with LAmB and CAS compared to patients treated with LAmB alone (100% versus 45%; p = 0.02). 122 In contrast, Abidi et al. 123 did not observe better outcome with this combination therapy. Combination of azoles with LAmB has also been attempted with conflicting results. A large cohort of hematologic patients with mucormycosis treated with different combination therapies reported no difference in outcome between combination therapy and monotherapy. Taken together, data supporting the use of combination therapy are scant and conflicting. Nevertheless, given the high mortality associated with mucormycosis, combination therapy with LAmB and posaconazole or an echinocandin may be considered in cases of progressive disease.62,111,124
Surgery, follow-up modalities, duration of therapy, and secondary prophylaxis
Prompt and aggressive surgical debridement with clear margins is crucial to cure mucormycosis, especially for cutaneous and rhinocerebral diseases. 111 Several surgical interventions are often necessary for optimal control. Extension work-up with a cerebral, sinus, and lung CT are recommended to evaluate disease extent and guide surgical management. Cerebral and orbital MRI should be performed in the presence of neurological symptoms. 111
Correction of underlying risk factors or reduction of immunosuppression when feasible should be done (e.g. hyperglycemia in a diabetic patient). Hyperbaric oxygenation and granulocytes infusion in neutropenic patients have been proposed, but no evidence strongly supports their use.125,126 Deferasirox is not recommended. 110
Duration of therapy should be guided by clinical responses and continued until complete resolution of infection, based on clinical and radiological imaging. 111 Treatment is often prolonged to 6–12 months.111,116 Follow-up imaging is advised during treatment and to guide surgical interventions. Evidence for secondary prophylaxis is limited, but prophylaxis may be considered for neutropenic patients, those treated for graft versus host disease and on a case-by-case basis based on immunosuppressive status. 111
Future drug options for Mucor spp
There are few antifungals in development for treatment of mucormycosis. Oteseconazole and PC-1244 are two azoles that have showed in vitro or in vivo activity in animal models against Mucorales species.127,128 Fosmanogepix is not active against Mucorales with the exception of Mucor spp.44,129,130 Ibrexafungerp has no or weak activity against Mucorales spp. 131
Scedosporium spp
Scedosporium spp. is an ubiquitous hyaline mold that can cause severe pulmonary or disseminated infections in SOT, HSCT recipients as well as in patients with chronic pulmonary diseases receiving immunomodulators.132–134 Trauma in the setting of environmental disaster such as tsunami and near drowning incidents are risk factor among immunocompetent hosts. 134 Management of scedosporiosis is challenging because of antifungal resistance (see Table 2). 112 Scedosporium spp. is intrinsically resistant to AmB and variably resistant to echinocandins. 134 Voriconazole is the most active agent against Scedosporium spp., followed by posaconazole.135–137 In contrast, higher MICs have been reported with ISA, and it should not be used against scedosporiosis.138,139
There is no RCT for the treatment of scedosporiosis, and most data are derived from in vitro and observational cohort studies. Several studies have reported better outcomes with voriconazole compared to AmB.134,140,141 A recent observational cohort study of 209 cases of scedosporiosis showed a survival benefit at 42 days for voriconazole monotherapy compared to AmB (mortality 11.3% versus 58.8%; p < 0.001). 134 Combination therapy has only been reported anectodally.134,142 As such, voriconazole monotherapy is the recommended treatment for scedosporiosis.62,112,143,144
Despite medical therapy, scedosporiosis can be refractory, and therapy is limited by toxicity or intolerance. In such circumstances, antifungals combination for salvage therapy have been used despite the lack of robust data. Various combinations have been used and include voriconazole backbone combined with either terbinafine, echinocandin, miltefosine or AmB with variable outcomes.142,145–157 In case of intolerance or resistance to voriconazole, posaconazole has been used with anecdotal success.158,159
Lomentospora prolificans
Lomentospora prolificans (formerly Scedosporium prolificans) is closely related to Scedosporium spp. but is now recognized as phylogenetically distinct. 160 Clinical presentation and predisposing factors can be similar, but L. prolificans distinguished itself by its multidrug resistance (resistant to AmB, echinocandin, and azoles) and the lack of effective antifungal therapy (see Table 2).136,157,161 Thus, it is associated with high mortality (47–78%), especially in cases of disseminated diseases (88%).133,162 Current guidelines recommend using voriconazole in combination with terbinafine based on the retrospective analysis of a cohort of 41 patients with lomentosporiosis, where this combination therapy was associated with higher treatment success compared to other antifungal regimens (63% versus 29%; p = 0.053).62,112,143,144,163,164 Other combinations with posaconazole, echinocandins or LAmB may be considered in case of refractory diseases.62,143,144
For both scedosporiosis and lomentosporiosis, surgical debridement should be considered when feasible, especially in case of CNS infection.133,165 Optimal duration of therapy is unknown, but should be at least until clinical and radiological resolution of diseases and potentially until recovery of immunocompromised state if reversible.
Future drug options for Scedosporium spp. and L. prolificans
Olorofim has excellent activity against both Scedosporium spp. and L. prolificans. 40 In vitro and animal studies have reported encouraging results.40,166 Successful cases of prolonged use (10–12 months) without adverse events have been reported.167,168 Olorofim for treatment of difficult-to-treat IMI is currently studied in an RCT (FORMULA-OLS study) (ClinicalTrials.gov identifier: NCT03583164). Interim results reported successful overall response at 6 weeks in 55% and 53% of scedosporiosis and lomentosporiosis, respectively. 101 Fosmanogepix also appears promising in treating scedosporiosis and lomentosporiosis. In vitro data suggest potent activity of the drug against Scedosporiosis spp. and L. prolificans.41,44,130,169 Murine models studies also show prolongation of survival in immunocompromised mice with scedosporiosis. 170 In vitro data for ibrexafungerp suggest only modest activity against both species. 131
Fusarium spp
Fusarium spp. is the second non-Aspergillus mold causing human infections. 171 Cases of fusariosis are most commonly seen in the immunocompromised population with prolonged neutropenia or severe T-cell immunodeficiency.172,173 The Fusarium genus encompasses three complexes: Fusarium solani complex, F. oxysporum complex, and F. fujikuroi complex. 172 Multidrug resistance is common and antifungals resistance patterns vary significantly between complexes. 174 F. solani complex, which accounts for more than half of clinical diseases, typically shows high MICs to all azoles but may retain lower MIC to AmB (see Table 2).172,175–177 Thus, effort to make good speciation and perform antifungal susceptibility testing should be made, even in the absence of Clinical and Laboratory Standards Institute (CLSI) or European Committee on Antimicrobial Susceptibility Testing (EUCAST) criteria.
Optimal treatment of fusariosis remains uncertain given lack of clinical trials. Historically, AmB was the treatment of choice. Despite concerns of poor in vitro activity, clinical improvement with LAmB have been reported (partial or complete response in 46–82%).178–180 Voriconazole appears to have similar clinical efficacy (overall response in 45–63%), making it a suitable treatment option.180–182 In one of the largest retrospective cohort of fusariosis (n = 233), survival probability at 90 days was 60% for patients receiving voriconazole and 48% for patients receiving LAmB (2001–2011). 180 Recent guidelines therefore recommend using either agent as first-line therapy for fusariosis.112,143,144 However, given the great variability of susceptibility to antifungals, some experts suggest starting an empirical treatment combining both LAmB and voriconazole.62,112,144 In severe or refractory cases, combination therapy with voriconazole with either terbinafine or AmB may be considered based on in vitro and anecdotal reports.180,183 In vitro data have shown more frequent synergy between voriconazole and terbinafine, but published clinical experience has only shown modest success compared to case reports treated with LAmB and voriconazole.184–188 Posaconazole could be considered for refractory cases if susceptibilities are known.112,176,189 The current experience with ISA is not encouraging as nine fusariosis cases treated with ISA in the SECURE and VITAL trials showed treatment failure in most. 190 Echinocandins should not be used as they lack activity against Fusarium.112,176
Surgical debridement, including that of the potential primary skin source, should be considered when feasible and effective therapy should be continued until resolution of disease.62,112,143,144 Correction of underlying risk factors or reduction of immunosuppression should be attempted, including consideration of granulocytes infusion in neutropenic patients although data are limited.112,143,173,191
Future drug options for Fusarium spp
Among novel agents, olorofim and fosmanogepix are promising for the treatment of fusariosis. Olorofim demonstrates good in vitro activity against Fusarium spp., including more resistant strains of F. solani (MICs 0.25–1 mg/L and 1–4 mg/L at 50% and 100% inhibition). 192 In vitro and in vivo data from murine models with disseminated fusariosis have also shown encouraging results for the use of Fosmanogepix (survival 100% at day 14 with E1210 versus 20% for control; p < 0.05). 193 ibrexafungerp, like other glucan synthase inhibitors, lacks activity against Fusarium spp. 131
Conclusion
We have reviewed the management of most important invasive yeast and mold infections focusing on available antifungals and the literature that supports their use. Significant changes in immunocompromised hosts, increase in international travel, and widespread use of antifungal prophylaxis are all contributing to the changing epidemiology of IFI, such that resistant species are more commonly encountered, and treatment is becoming more challenging. Ongoing studies evaluating novel antifungal agents should provide critical information on the roles of these new molecules in the management of resistant or refractory cases. Development of other new antifungal classes might be critical and should be supported by collective and coherent action plans on a global scale with WHO, IDSA, ESCMID, and Mycoses Study Groups.