Abstract
Objectives
Persistent oxidative stress is one of the several risk factors that may be associated with the etiology of obesity. The present study is aimed to investigate association between GSTM1 and GSTT1 polymorphisms with obesity and their relationship with plasma lipoproteins, body mass index (BMI) and hypertension.
Design
The GSTM1 and GSTT1 deletion polymorphisms were analyzed by multiplex polymerase chain reaction. The lipoproteins were measured in plasma using commercially available kit and the weight, height, systolic (SBP) and diastolic (DBP) blood pressures by standard procedure of measurements.
Setting
Prince Sultan Military Medical City, Riyadh Saudi Arabia.
Participants
A total of 420 overweight/obese cases and 234 normal weight controls belong to young age Saudis.
Main outcomes measures
GSTM1/GSTT1 polymorphisms may be associated with obesity.
Results
Weight, BMI, low-density lipoprotein (LDL) and SBP were significantly higher while high-density lipoprotein (HDL) was significantly lower in cases in comparison to controls. Frequency of GSTM1+/GSTT1− (OR = 2.70, 95% CI = 1.52–4.81, p = <0.001) and GSTM1−/GSTT1− (OR = 2.43, 95% CI = 1.15–5.15, p = 0.018) was significantly higher in cases as compared to controls. BMI and weight were significantly higher in GSTM1+/GSTT1− and GSTM1−/GSTT1− genotypes, and LDL, DBP and SBP significantly higher in GSTM1−/GSTT1− null genotype while HDL was significantly lower in GSTM1−/GSTT1+ and GSTM1−/GSTT1− genotypes in comparison to GSTM1+/GSTT1+ genotype.
Conclusions
The GSTM1+/GSTT1− and GSTM1−/GSTT1− null genotypes were significantly associated with obesity and have shown relationship with obesity risk factors in cases. Hence, these genes may be associative genetic risk factor for obesity among young age Saudis.
Keywords
Introduction
Overweight and obesity are impaired health conditions characterized by excessive/abnormal fat accumulation. In 2014, World Health Organization (WHO) 1 reports that more than 1.9 billion adults, 18 years and older, were overweight; of these, over 600 million were obese. It is the fifth leading risk for global health and also the major cause for the preventable deaths in Saudi Arabia. 2 The prevalence of overweight/obesity in the age group of 13–18 years in Saudi population was 26.6% and 10.6% respectively. 3
The body mass index (BMI) is extensively used to define obesity in epidemiological studies because of cost effective and easy to calculate. 4 The high BMI is associated with diabetes, hypertension, cardiovascular disease, osteoarthritis, hyperlipidemia, liver steatosis and certain types of cancer.5,6 Despite substantial obesity awareness campaigns, efforts to decrease the energy intake and increase the energy expenditure, basic scientific and clinical investigations, obesity prevalence is still increasing.
Obesity is a complex multifactorial and heterogeneous condition involving complex interaction among genetic, behavioral and environmental factors. However, none of them explains the exact mechanism underlying the cause of obesity. The ethnic prevalence, familial aggregation, twin and adoption studies have strongly recommended the role of genetics in obesity.7,8 Although environmental factors are important, genetics plays considerable role with the heritability estimated as 40% to 70% for BMI variation. 9 The body fat accumulation in monogenic and polygenic form of obesity provides the evidences for strong genetic background in obesity. 10 The monogenic form of obesity is characterized by early onset of the disease and clearly identified phenotype. Currently, more than 200 single gene mutations have been found in 10 well-known genes such as LEP, LEPR, MC4R, BDNF, etc. and are accountable for >10% of monogenic form of obesity. In contrast with monogenic obesity, polygenic obesity implies the additive contribution of many genes which have impact on appetite, metabolism and deposition of fat. The loci architecture underlying the phenotypic changes in polygenic obesity is still being characterized. 8 Till date, there are 97 loci on human genome which have been associated with the risk of obesity. 11 The use of modern molecular biology techniques are helpful in the identification of new genes which could uncover new insights into genetics of polygenic obesity.
The association of oxidative stress, dyslipidemia and hypertension with obesity has been reported in several studies.12–14 Glutathione S-transferases (GSTs), an oxidative stress-related gene, play a critical role in providing protection against electrophiles and products of oxidative stress. The deletion polymorphism of GSTM1/GSTT1 genes may increase oxidative stress and modulate the levels of lipoproteins15,16 and have been associated with risk of obesity and associated clinical outcomes. 17
Therefore, determining the association of deletion polymorphisms of GSTM1 & GSTT1 gene with obesity is on priority research. The present study was planned to investigate the association of GSTM1/GSTT1 gene polymorphisms in the risk assessment of overweight/obesity in young age Saudis.
Methods
A total of 420 overweight/obese male (cases) and 234 healthy male (controls) were included in the present case–control study. This study was carried out during the period of 2013 to 2015 at Research Centre, Prince Sultan Military Medical City (PSMMC), Riyadh. Study subjects confirmed their participation by signing an informed consent, and the study was approved by ethics research committee, PSMMC, Riyadh vide project reference number 523 dated 17th June 2013. The blood samples of overweight/obese subjects were collected from Riyadh and its peripheral region. The patients who visited to outpatient clinic for minor illness were recruited as controls. Information on demographic and personal lifestyles was collected with a self-designed questionnaire during face-to-face interviews by the trained researchers. The subjects with the history of smoking, evidence of cancers and metabolic diseases were excluded from both the study groups. The height, weight, systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured according to standardized methods. BMI was calculated as body weight (kg) divided by the square of the measured height (m2). The BMI range of 23–27.5 kg/m2 and BMI > 27.5 kg/m2 were considered as overweight and obese, respectively, as per WHO recommendations for Asian populations. 18
We considered less than 120/80 mmHg as normal, 120/139 mmHg as pre-hypertensive and 140/90 mmHg as hypertensive blood pressures. The subjects included in the study belonged to a relatively homogeneous age group (age range for cases: 18–20 years and controls: 18–32 years), similar in terms of demographical characteristics such as food habits, education, socioeconomic, marital status, etc. After overnight fasting, venous blood samples (3 mL) were drawn in EDTA-coated vials from the forearm vein of all participants. The plasma was separated and stored at −80℃ for biochemical analysis. The aliquots of blood samples were prepared and stored at −80℃ for genomic studies.
Measurement of lipoproteins
Quantification of low-density lipoprotein (LDL) was performed using lactate dehydrogenase reagent kinetic method (United Diagnostic Industry, Riyadh, KSA) according to manufacturer’s instructions. The absorbance was taken at 340 nm against blank for every minute for 3 min, and delta mean/minute for absorbance was calculated. The value of LDL was expressed in mmol/L. The high-density lipoprotein (HDL) cholesterol was quantified by using HDL precipitating reagent as per manufacturer instructions (United Diagnostic Industry). The absorbance was taken at 510 nm against blank using spectrophotometer (Perkin Elmer, USA).
Genomic DNA isolation and genotyping
Genomic DNA for genotyping was isolated from blood by using commercially available QIAamp DNA mini kit (Qiagen, Venlo, Limburg, the Netherlands). The quality of the DNA was checked on agarose gel, and quantitation was done using Nano Drop-2000 (Thermo Fisher Scientific Inc, Waltham, MA). Multiplex polymerase chain reaction (PCR) was performed for GSTM1 and GSTT1 deletion polymorphisms. Briefly, 50 ng of genomic DNA was amplified in a 25-µL multiplex reaction mixture containing 30 pmol of the GSTM1 and GSTT1 primers in a medium consisting of 1.5 mM MgCl2, 200 µmol dNTPs, 2.5 µL 10 × PCR buffer (10 × 500 mM KCl, 100 mM Tris-HCl, pH 9.0) and 1U Taq DNA polymerase. The primers for GSTM1 were 5′ F-GAA CTC CCT GAA AAG CTA AAG C 3′, 5′ R-GTT GGG CTC AAA TAT ACG GTG G 3′ and GSTT1 were 5′ F-TTC CTT ACT GGT CCT CAC ATC TC 3′, 5′R-TCA CCG GAT CAT GGC CAG CA 3′. The human growth hormone (hGH) primers were used as internal control to avoid any false-negative results.
The PCR protocol included an initial melting temperature of 94℃ (5 min) followed by 35 cycles of amplification (2 min at 94℃, 1 min at 59℃ and extension for 1 min at 72℃). A final 10-min extension step (72℃) terminated the process. The final PCR products from co-amplification of GSTM1 (215 bp), human growth factors (330 bp) and GSTT1 (480 bp) were visualized after electrophoresis in ethidium bromide stained 2.5% agarose gel (Sigma-Aldrich, St. Louis, MO). The absence of amplifiable GSTM1 and GSTT1 (in the presence of hGH PCR product) indicates a GSTM1−/GSTT1− (null) genotypes as shown in Figure 1.
Gel picture showing GSTM1 and GSTT1 deletions. M-100 bp marker, Lane 1, 3, 6, 7 and 8: GSTM1 and GSTT1 present, Lane 2: GSTT1 deletion, Lane 4 and 5: GSTM1 deletion and Lane 9: GSTM1 and GSTT1 both deletion.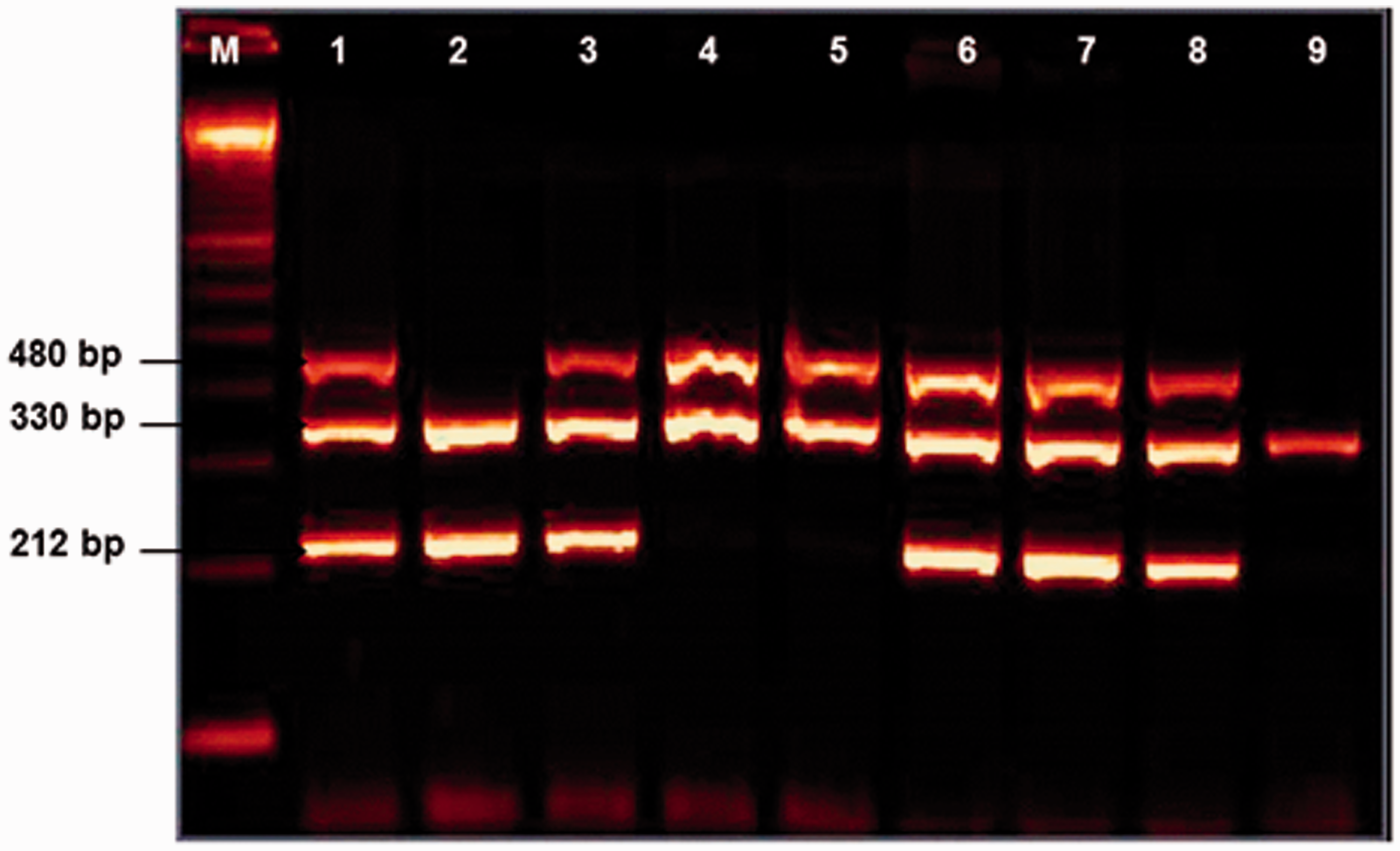
Statistical analysis
Statistical software SSPS for windows version 21.0 (Chicago, IL) was used for statistical analysis. The χ 2 test was used to compare genotypic and allelic frequencies between cases and controls, and values were presented as crude odds ratio (OR) and 95% confidence intervals (CI). Independent t-test was used to analyze significant difference of weight, BMI, height, HDL, LDL, DBP and SBP between cases and controls and in different genotypic groups. The value of p < 0.05 was considered to denote significance.
Results
Characteristics of controls and cases.

Unpaired “t” test was applied for analysis of quantitative variables like age, weight, height, BMI, etc., *p < 0.05 is significant.
BMI: body mass index; LDL: low-density lipoprotein; HDL: high-density lipoprotein; SBP: systolic blood pressure; DBP: diastolic blood pressure.
Genotypic distribution of GSTM1 and GSTT1 genes in controls and cases.
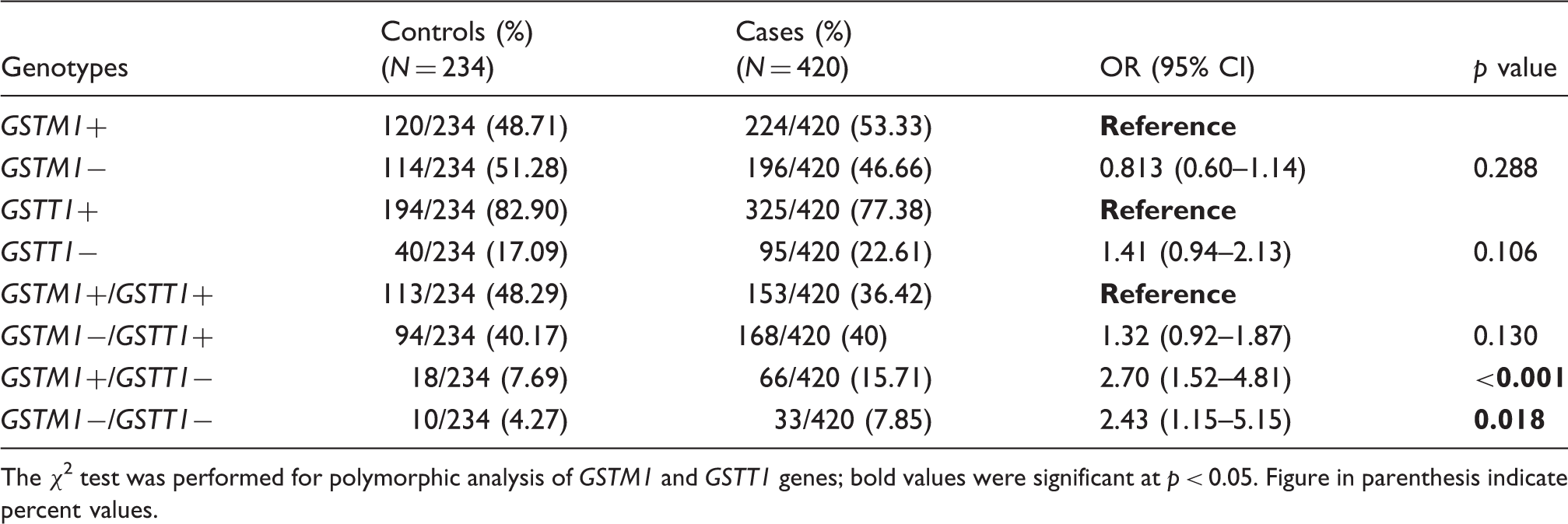
The χ 2 test was performed for polymorphic analysis of GSTM1 and GSTT1 genes; bold values were significant at p < 0.05. Figure in parenthesis indicate percent values.

Histogram showing the comparison of LDL and HDL levels in GSTM1+/GSTT1+ with GSTM1−/GSTT1+, GSTM1+/GSTT1− and GSTM1−/GSTT1− (null). Independent t-test was applied to analyze the data. p < 0.05 was considered as significant.

Histogram showing the comparison of DBP and SBP in GSTM1+/GSTT1+ with GSTM1−/GSTT1+, GSTM1+/GSTT1− and GSTM1−/GSTT1− (null). Independent t-test was applied to analyzethe data. p < 0.05 was considered as significant.

Histogram showing the comparison of BMI, weight and height in GSTM1+/GSTT1+ with GSTM1−/GSTT1+, GSTM1+/GSTT1− and GSTM1−/GSTT1− (null). Independent t-test was applied to analyze the data. p < 0.05 was considered as significant.
Discussion
In the present study, deletion polymorphism of GSTM1 and GSTT1 genes was evaluated for genetic susceptibility to the risk of obesity/overweight and their association with lipoproteins, BMI and hypertension. The present study demonstrates that the frequency of GSTM1− (null) genotype is 51.28% in controls and 46.46% in cases. Our observed frequency of GSTM1− in controls is corroborated with many others Arabic populations such as Bahrainis, Tunisian, Lebanese 19 and Egyptian. 20 However, the frequency of the GSTM1− was higher than Indian, 21 Japanese 22 and African 23 populations. The GSTT1− null genotype frequency was higher in cases (22.61%) than controls (17.09%). The frequency of GSTT1− null genotype in the present study is comparatively lower than the other Arab populations that varied from 20% to 37%.19,24 while in line with Turkish, 25 Indian 26 and French 27 populations. The difference in GSTT1− null genotype frequency in comparison to other Arab populations may be attributed to the different environmental conditions and natural selection processess. Moreover, Saudi population being a closed and isolated society with a high rate of consanguinity (inbreeding) may also contribute for the different GSTT1− null genotype frequency.
Our results demonstrated that the frequency of GSTM1+/GSTT1+ and GSTM1−/GSTT1+ was higher in controls while GSTM1+/GSTT1− and GSTM1−/GSTT1− null genotypes frequencie were significantly higher (OR = 2.70, p < 0.001 and OR = 2.43, p = 0.018) in cases as compared to controls, respectively. The deletion in these genes may relate to significantly high levels of oxidative stress, 15 resulting in oxidative damage to macromolecules and tissues which may contribute to obesity. 16
We observed significantly (p < 0.05) lower values of HDL in GSTM1−/GSTT1+ and GSTM1−/GSTT1− while LDL was significantly higher in GSTM1−/GSTT1− null genotype as compared to GSTM1+/GSTT1+ genotype. This allows us to infer that deletions in GSTM1/GSTT1genes may contribute to obesity related complications such as dyslipidemia. The influence of deletion polymorphisms of GSTM1/GSTT1 on serum HDL and LDL in the present study is in agreement with earlier report. 16 Amer et al. 28 reported significantly high LDL cholesterol in patients with GSTT1− null genotype as compared to those with the GSTT1+ genotype in type-2 diabetes mellitus (TDM). Furthermore, Afrand et al. (2015) have reported significantly low HDL in GSTT1− null genotype as compared to GSTT1+ genotype in metabolic syndrome. 29
Our findings suggested that the cardiovascular risk may increase in individuals with GSTM1−/GSTT1− null genotype due to significantly high DBP and SBP as compared to other genotypic groups. There are numerous studies that have reported a significant association of GSTs gene polymorphism on susceptibility to increased arterial pressure.12,30,31 Tang et al. 32 reported that GSTM1−/GSTT1− subjects have higher C-reactive protein and fibrinogen and lower total antioxidant status in comparison to patients with GSTM1+/GSTT1+ genotype. Further, they suggested that GST polymorphisms may modify the levels of oxidative stress and inflammation in Chinese coronary artery disease patients. The significantly high value of BMI, LDL, DBP and SBP in GSTM1−/GSTT1− null genotype reflects association of BMI with lipoprotein and hypertension, as reported in previous research studies.33,34 BMI has been used as a marker for identifying dyslipidemia in obesity cases. 35 The high BMI in GSTM1−/GSTT1− null genotype has been supported by the previous study where GSTM1 null genotype is significantly associated with high BMI in TDM patients. 36 The possible mechanism could be the deletion in GSTM1/GSTT1 genes resulting in a deficiency in the protein product, which is related to the total loss of enzymatic activity. 37 This could cause improper detoxification of xenobiotic, reduced defense ability against oxidative stress and free radical-mediated cellular damage 38 and subsequently may influence the individual susceptibility to obesity. The deletion of GSTM1/GSTT1 may also enhance the risk of obesity by modulating detoxification of genotoxic secondary metabolites.
Our finding may be useful for better understanding of obesity patho-physiology and its risk prediction. The differences in our finding with published data could indicate role of these genotypes distinct to overweight/obesity in Saudi population. This could help in unraveling the possible molecular mechanism underlying obesity. Further studies on oxidative stress and antioxidant-related genes polymorphism and their expression profile will be required to validate our findings.
Footnotes
Acknowledgement
The authors thank S. Sadaf Rizvi for the help with laboratory work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Institutional ethical approval has been taken to conduct this research study vide project reference number 523 dated 17th June 2013.
Guarantor
AA is the guarantor for all the content presented in this paper.
Contributorship
HAA, MMA and TA performed clinical examinations, collected demographic data and read the manuscript. MM, MMA and TA performed genotyping and biochemical analysis. HAA, MM and MMA analyzed the results and drafted the manuscript. AA designed the study, supervised and gave final approval for the manuscript to be published. All authors checked interpreted results and approved the final version.
