Abstract
Prognostic stratification of cardiomyopathies represents a cornerstone for the appropriate management of patients and is focused mainly on arrhythmic events and heart failure. Cardiopulmonary exercise testing provides additional prognostic information, particularly in the setting of heart failure. Cardiopulmonary exercise testing data, integrated in scores such as the Metabolism Exercise Cardiac Kidney Index score have been shown to improve the risk stratification of these patients. Cardiopulmonary exercise testing has been analysed as a potential supplier of prognostic parameters in the context of hypertrophic cardiomyopathy, for which it has been shown that a reduced oxygen consumption peak, an increased ventilation/carbon dioxide production slope and chronotropic incompetence correlate with a worse prognosis. To a lesser extent, in dilated cardiomyopathy, it has been shown that the percentage of oxygen consumption peak, not the pure value, and the ventilation/carbon dioxide production slope are associated with a greater cardiovascular risk. Few data are available about other cardiomyopathies (arrhythmogenic and restrictive). Cardiomyopathy patients should be early and routinely referred to heart failure advanced centres in order to perform a comprehensive risk stratification which should include a cardiopulmonary exercise test, with variables and cut-offs shown to improve their risk stratification.
Introduction
Cardiomyopathies represent a heterogeneous group of heart muscle disorders in the absence of conditions such as coronary heart disease, hypertension or valvular disease. 1 Cardiomyopathies may be primary or secondary, including dilated cardiomyopathy (DCM), hypertrophic cardiomyopathy (HCM), restrictive cardiomyopathy (RCM) and arrhythmogenic right ventricular cardiomyopathy/dysplasia (ARVC/D). Prognostic stratification is a cornerstone and a component of paramount importance for appropriate management in these patients, and it is focused mainly on arrhythmic events and heart failure (HF). 2 Recent progress in early diagnosis, biomarker interpretation, comprehensive characterisation and tailored follow-up has allowed a better prognostic stratification of cardiomyopathies.2,3 Despite significant improvements in this field during the past two decades, research is still focused on improving. In this context, cardiopulmonary exercise testing (CPET) represents a valuable tool providing additional prognostic information, particularly in the setting of HF. In all stages and types of HF, from reduced to preserved ejection fraction, CPET data have been demonstrated as useful tools, when added in scores that include clinical, laboratory and other instrumental variables, such as the metabolism exercise cardiac kidney index (MECKI) score (Figure 1).4,5 At the same time, CPET variables are influenced by several factors: among others, recent data suggest for example that obesity may negatively affect peak oxygen consumption (VO2). 6 Therefore, the assumption that their prognostic ability is reproducible to all patients with HF of its aetiology can be misleading in clinical practice. Consequently, CPET data need to be contextualised into specific groups of cardiomyopathies. The main studies analysed are summarised in Table 1.
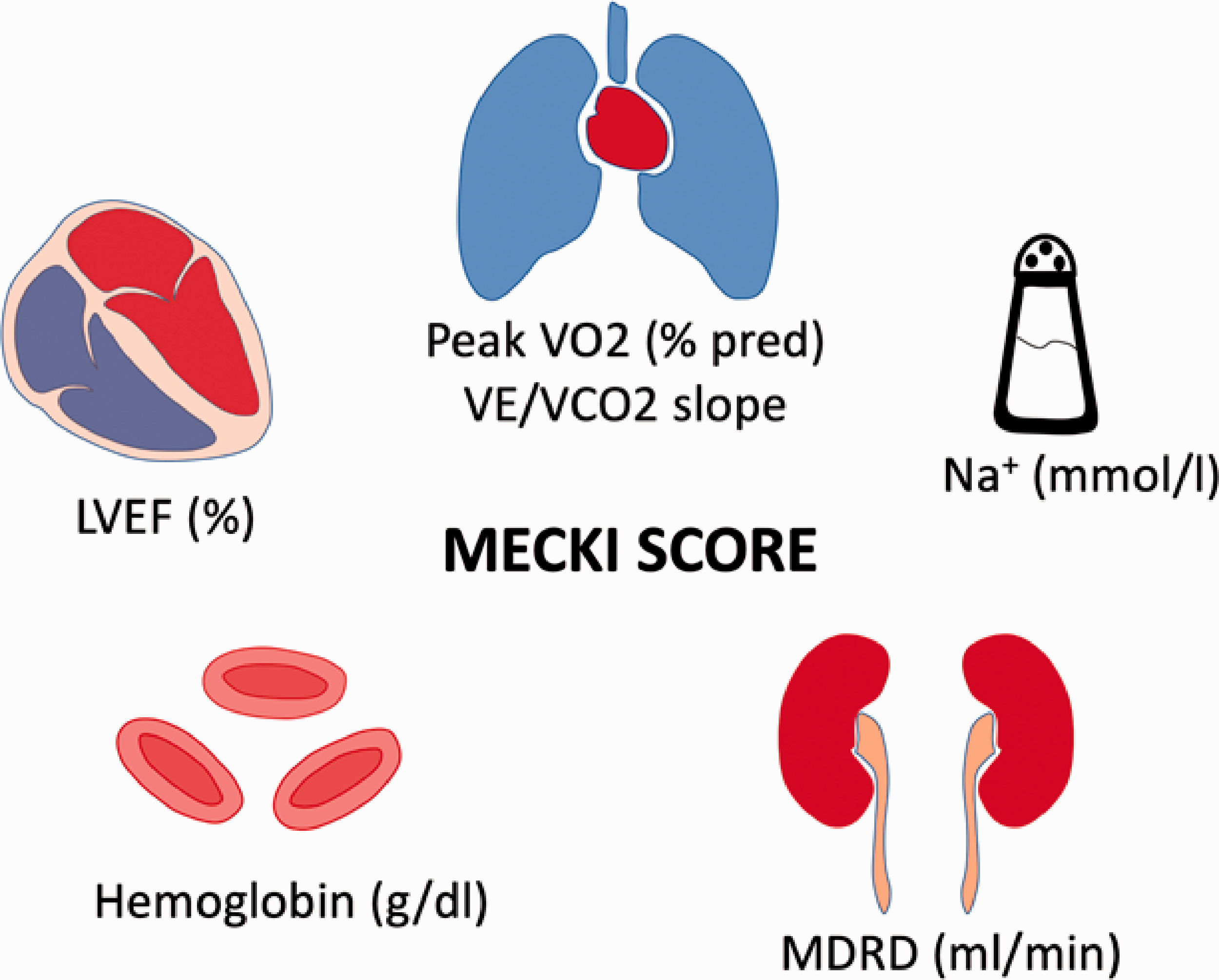
Parameters for the calculation of the metabolism exercise cardiac kidney index (MECKI) score: metabolic exercise (peak VO2% pred, VE/VCO2 slope), cardiac (LVEF), kidney (MDRD, ml/min), index.
Results and main findings of leading studies about CPET in CMPs.
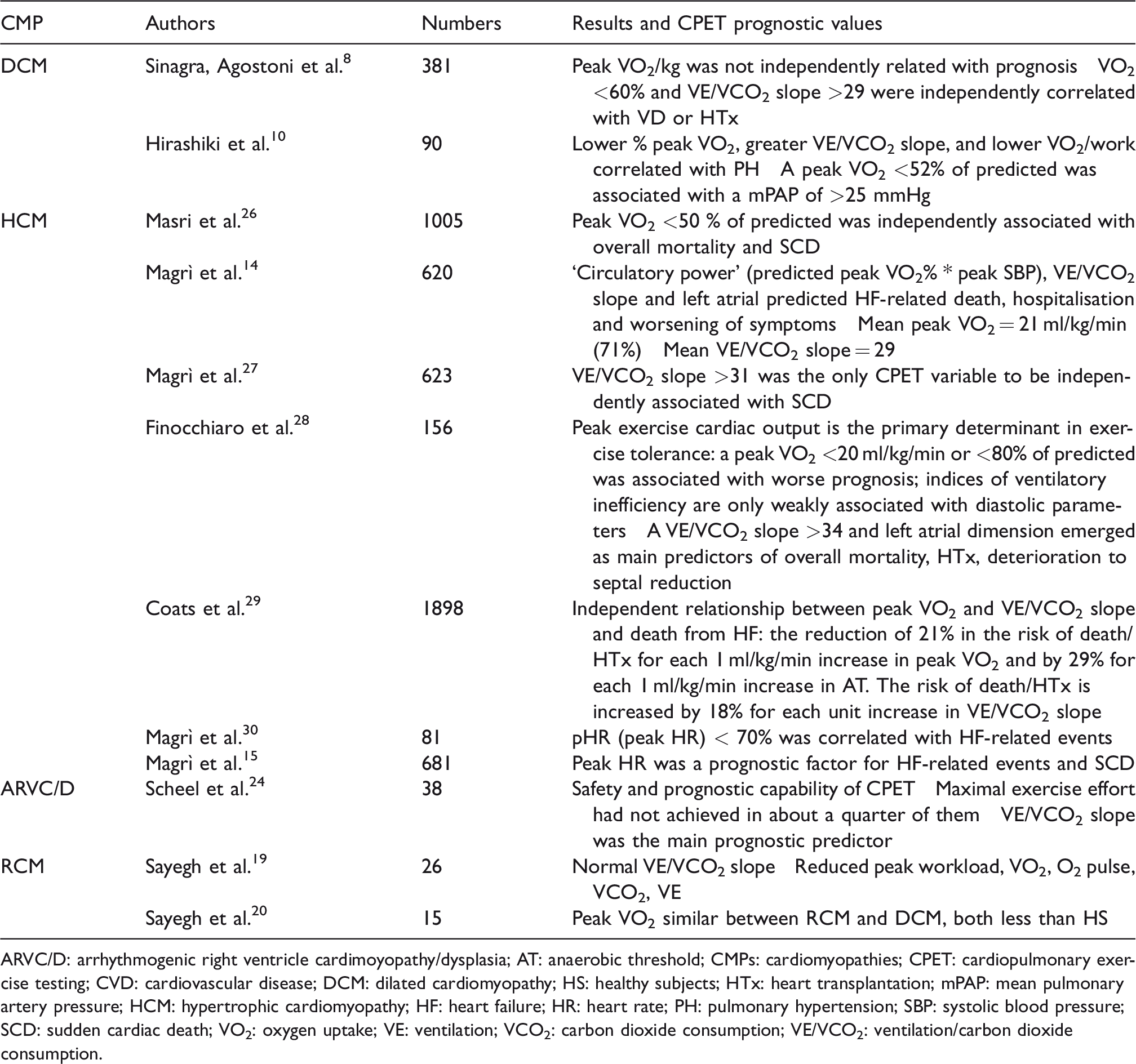
ARVC/D: arrhythmogenic right ventricle cardimoyopathy/dysplasia; AT: anaerobic threshold; CMPs: cardiomyopathies; CPET: cardiopulmonary exercise testing; CVD: cardiovascular disease; DCM: dilated cardiomyopathy; HS: healthy subjects; HTx: heart transplantation; mPAP: mean pulmonary artery pressure; HCM: hypertrophic cardiomyopathy; HF: heart failure; HR: heart rate; PH: pulmonary hypertension; SBP: systolic blood pressure; SCD: sudden cardiac death; VO2: oxygen uptake; VE: ventilation; VCO2: carbon dioxide consumption; VE/VCO2: ventilation/carbon dioxide consumption.
Dilated cardiomyopathy
DCM is a primary heart muscle disease characterised by progressive left ventricular or biventricular dilation and dysfunction in the absence of conditions such as hypertension, ischaemic and valvular disease. DCM represents one of the main causes of heart transplantation (HTx) and is associated with an increased risk of arrhythmia-related death. 2 Risk stratification in these patients continues to be challenging and the natural history still remains characterised by a progressive incidence of HF, with significant morbidity and mortality. An accurate clinical assessment of patients with DCM is crucial to identify those who are more likely to die from sudden death or for progressive HF.
Until now, peak VO2 has been used as a pivotal criterion for selecting DCM patients for HTx. Nevertheless, DCM patients represent a peculiar model compared to heart failure with reduced ejection fraction (HFrEF) patients with other aetiologies. They are mostly young, with a lower comorbidity profile and are usually less symptomatic at disease onset. This explains better cardiopulmonary performances at early stages of the disease and a lower risk of events compared to other HFrEF aetiologies. Also, in patients with a long duration of asymptomatic left ventricular dysfunction, adaptive pathophysiological cardiopulmonary mechanisms can lead to better performances on CPET, with a better prognostic impact compared to patients with similar left ventricular dysfunction of different aetiologies (i.e. ischaemic). 7 Although CPET is recognised as an important tool in risk stratification of HF patients, prognostic data in the specific setting of DCM have not been available until a few years ago. The first pioneering study dates back to 2016, led by the cardiology department of Centro Cardiologico Monzino and Trieste Hospital in a large cohort of ‘pure’ DCM patients who consecutively performed CPET. The study findings showed that peak VO2% (the percentage of VO2 obtained compared to the theoretical VO2max of a normal subject of the same age, weight, sex and ethnicity) and the ventilation/carbon dioxide production (VE/VCO2) slope were the strongest predictors of cardiovascular death/urgent HTx. Interestingly, peak VO2/kg was not independently related to prognosis (Figure 2). This finding highlights how in DCM the absolute value of VO2/kg peak does not precisely represent the true impairment of cardiovascular fitness of DCM patients. A predicted VO2 less than 60% and a VE/VCO2 slope greater than 29 were associated with poor prognosis in the study population. In this cohort of ‘pure’ DCM patients, these cut-points were slightly different and more accurate than those previously described in CPET among HFrEF patients in prognostic stratification. In view of the above, the importance of an aetiological classification upstream of HF was emphasised to differentiate specific cut-points for the identification of high-risk patients. 8 Among the cardiomyopathies, DCM is probably the one that best fits in the characterisation given by the MECKI score as it has been validated on HFrEF patients.
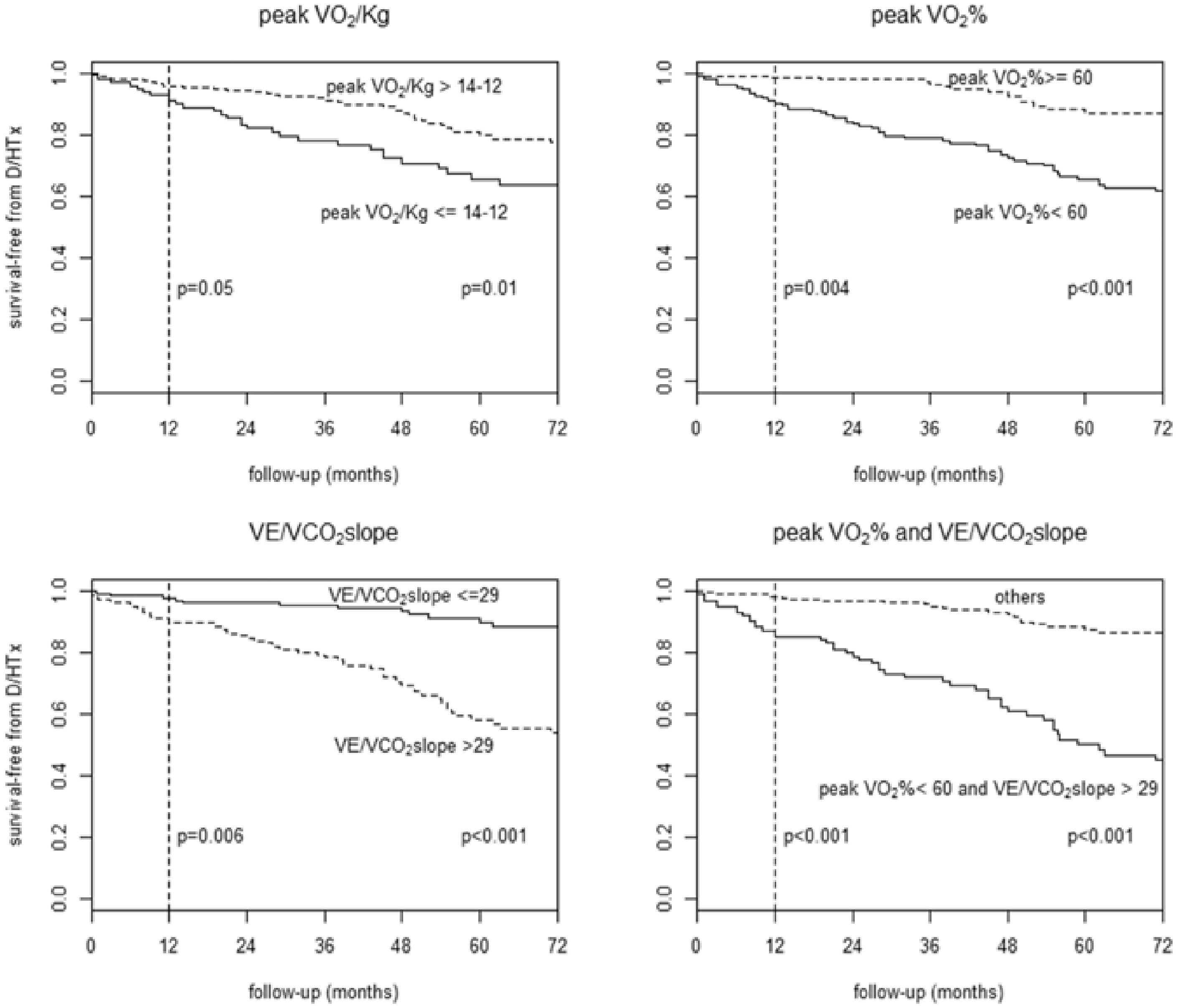
Survival free from cardiovascular death/heart transplantation in dilated cardiomyopathy (DCM) patients. Kaplan–Meier survival curves stratified by peak VO2/kg (14–12 mL/kg/min refers to the classically used cut-points for peak VO2, respectively, in the presence or absence of beta-blocker therapy, upper left panel), peak VO2% cut-point 60%, upper right panel), VE/VCO2 slope (cut-point 29, bottom left panel), combination of peak VO2% (cut-point 60%) and VE/VCO2 slope (cut-point 29, bottom right panel). D/HTx: cardiovascular death/heart transplantation; peak VO2/kg: peak of oxygen consumption per kg; peak VO2%: percentage of predicted oxygen consumption; VCO2: carbon dioxide production; VE: ventilation. Reproduced from Sinagra et al., 8 with permission.
In recent years there has also been growing emphasis on the role of pulmonary hypertension (PH) in the prognosis of HF patients with DCM. 9 In this setting, PH secondary to left ventricular dysfunction is frequent and is a consolidated predictor of mortality and morbidity. In a work by Hirashiki et al. %peak VO2 was the best predictor of PH in DCM patients and a reduction below 52% was strongly related to the presence of mean pulmonary arterial pressure (mPAP) greater than 25 mmHg. Likewise, VO2 peak, VE/VCO2 slope and VO2/work were other variables related to the presence of PH in DCM patients. 10 Within the DCM spectrum there are also different pathophysiological mechanisms; in a few cases several factors such as excessive alcohol consumption, 11 arrhythmias 12 or drugs (e.g. anthracycline) can unmask the DCM phenotype. Further studies will be needed to understand if these cardiomyopathies can express themselves differently in CPET.
Finally, DCM behaves as a dynamic disease with a need for continuous re-stratification of risk during follow-up: to date, there is no clear consensus on how often is reasonable to repeat a CPET in order to monitor its changes during DCM patients’ follow-up. It seems reasonable to propose to repeat it every 1–2 years.
Hypertrophic cardiomyopathy
HCM is an inherited genetic disease characterised by left ventricular hypertrophy unexplained by abnormal loading conditions 13 and represents the most common cardiomyopathy occurring in nearly 1:500 of the general population. Sudden cardiac death (SCD), albeit rare (0.5–1% per year), is a catastrophic event, caused by fatal arrhythmias in generally young men (between the third and fourth decade of life). Currently, in a subset of HCM patients, SCD is still an unpredictable complication and may represent the first manifestation of the disease. In light of this, the assessment of arrhythmic risk is a cornerstone of management in HCM patients. However, recent studies point to the growing role of HF and stroke-related death as important causes of morbidity and mortality in the long term. Recently, the European Society of Cardiology (ESC) guidelines included in the indications for the execution of CPET also the evaluation of the severity and causes for exercise intolerance, the assessment of systolic blood pressure changes during exercise and the early identification of candidates for septal reduction procedures. 14
In these patients, exercise limitation is primarily due to the inability to increase stroke volume secondary to left ventricular diastolic dysfunction, chronotropic incompetence and left ventricular outflow obstruction.15–17 For these pathophysiological features of HCM, the oxygen pulse curve (a derived CPET parameter reflecting stroke volume) may display an early flattening due to an inability of the left ventricle to increase the stroke volume, as shown in Figure 3. 14 This finding, in those subjects without chronotropic incompetence, is associated with a compensatory heart rate increase, and its earliness correlates with the prognosis. 15 Furthermore, an increase of pulmonary pressures, the worsening of mitral regurgitation and the impaired peripheral oxygen uptake due to a low mitochondrial density in skeletal muscle can contribute to a reduction in exercise capability. In haemodynamically stable patients the VO2/work slope, a marker of aerobic work efficiency, is usually preserved. An important pathophysiological feature in these patients is exercise-induced myocardial ischaemia, which can be highlighted during CPET by a sudden flattening of the VO2/work slope, in which the analysis of repolarisation on ECG can often be inconclusive. From a prognostic point of view, several studies have been conducted. They are summarised in detail in Table 1. In HCM patients, a reduced VO2 peak (i.e. <50%) and high VE/VCO2 slope are associated with overall mortality and SCD. Moreover, chronotropic incompetence emerged as an important prognostic factor for HF-related events. 15
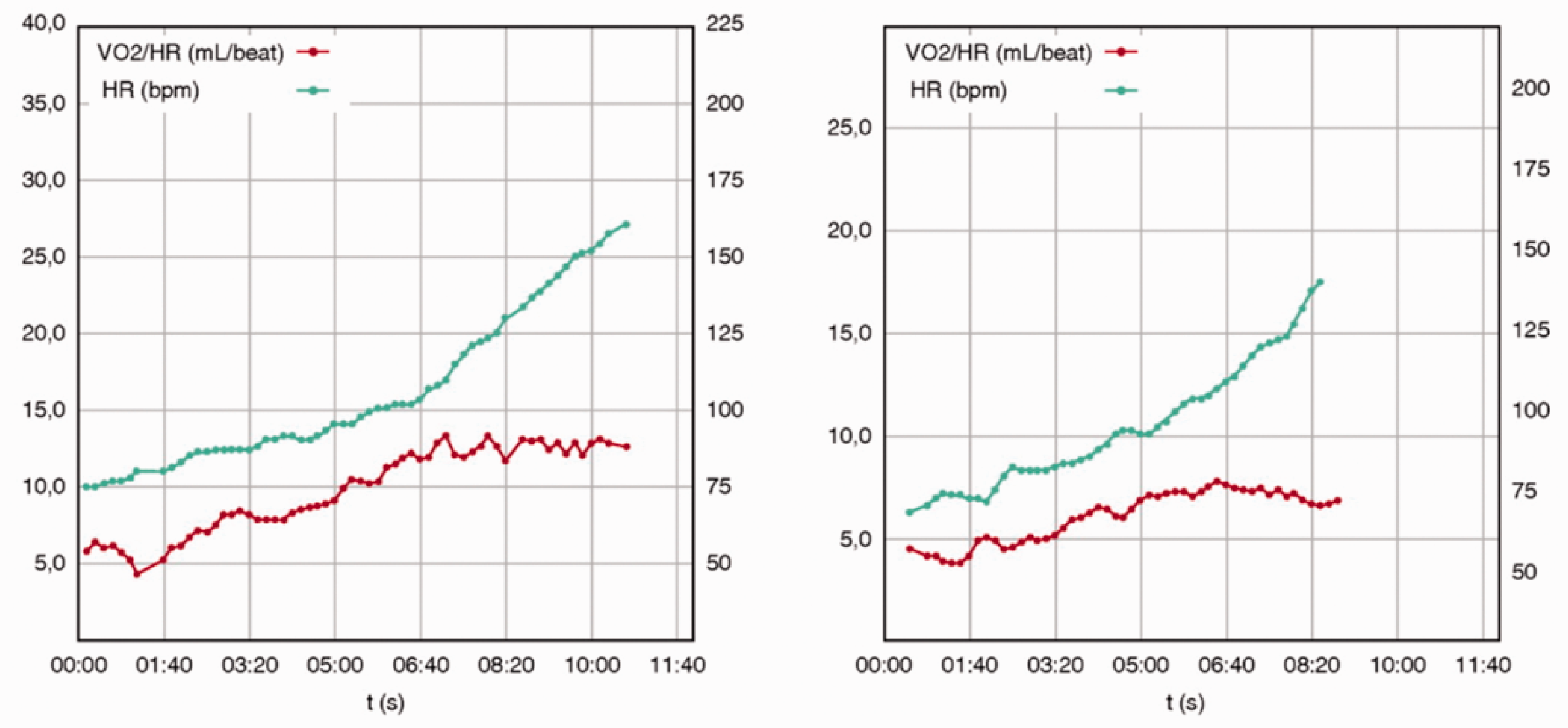
Relationships between oxygen pulse (VO2/HR) versus heart rate (HR) in a patient with asymptomatic hypertrophic cardiomyopathy (HCM) (left column) and in a patient with New York Heart Association (NYHA II) mildly symptomatic HCM with end-stage phase (right column). Although different in magnitude, both patients showed a reduced VO2 peak (70% and 55%), an early oxygen pulse flattening with a compensatory HR increase. Reproduced from Magrì and Santolamazza, 14 with permission.
Restrictive cardiomyopathy
RCM is a myocardial disease characterised by an increase of myocardial stiffness that leads to impaired ventricular filling. 18 Many aetiologies can lead to an RCM phenotype, such as infiltrative disorders (amyloidosis, Gaucher disease, Hurler syndrome, fatty or glycogen infiltration, Fabry disease, haemochromatosis). 18 Hypertrophy is not typical, although in the case of some infiltrative diseases (e.g. cardiac amyloidosis) and storage diseases (e.g. Fabry disease) an increased left ventricular thickness might be present. Systolic function is usually preserved, although it can be reduced in advanced stages of the disease. To our knowledge, investigations focused on the role of CPET in RCM are lacking.
Sayegh et al. 19 conducted CPET on 26 patients with endomyocardial fibrosis: they were mostly women, with pure diastolic dysfunction. The authors demonstrated a normal VE/VCO2 slope and respiratory exchange ratio achievement. On the other hand, reduced stroke volume parameters (peak workload, VO2, oxygen (O2) pulse, VCO2 and VE) have been described. Given that the left ventricular ejection fraction (LVEF) in these patients was preserved, the reason for these findings is presumably to be attributed to poor peripheral perfusion.
The same authors also compared CPETs of 15 patients with RCM, 10 patients with DCM and 10 healthy subjects finding that peak VO2 was similar between both cardiomyopathies but lower compared with healthy subjects, showing the same reduction in functional capacity between RCM and DCM. 20 In the broader setting of HF and preserved ejection fraction, both peak VO2 and the VE/VCO2 slope are able to provide incremental prognostic information regarding all-cause mortality and for incident HF hospitalisation, beyond relevant classical clinical covariates. 21 These findings support the notion that CPET is a robust albeit underutilised tool for risk stratification in heart failure with preserved ejection fraction (HFpEF) as well.
Accordingly, the low sample size of these studies does not allow us to generalise these results widely.
Arrhythmogenic right ventricular cardiomyopathy/dysplasia
ARVC/D is a rare genetic cardiomyopathy (incidence from 1:2000 to 1:5000) characterised by fibrofatty replacement of cardiomyocytes, resulting in right ventricular dysfunction and life-threatening ventricular arrhythmias. Despite the poor incidence, the typical ARVC/D onset is between the third and fifth decade of life, not rarely with SCD, which remains a fearsome presentation. 22 According to recent studies, there is a phenotypic overlap between involvement of the right ventricle, the left ventricle and DCM. 23 Further studies are needed to outline its characteristics and their relationship with CPET. Nevertheless, in recent years an early diagnosis and intervention (e.g. with implants of cardiac implantable defibrillators) have improved the life expectancy of ARVC/D patients. Consequently, other progressive manifestations such as right and left-sided HF became important features in the natural history of the disease. Robust data on the role of CPET for prognostic stratification in ARVC/D are still lacking. In the first and recent study, Scheel et al. 24 demonstrated both the safety and prognostic capability of CPET in a large ARVC/D cohort, although the maximal exercise effort was not achieved in about a quarter of the patients who performed a submaximal test (i.e. maximal beta-blocker therapy, fear of developing arrhythmias by effort). In this cohort the VE/VCO2 slope proved to be the main prognostic predictor of transplant-free survival, confirming the close relationship between impaired right ventricular function, VE/VCO2 and symptomatic HF. 25 Despite these promising data encouraging the use of CPET in ARVC/D patients, further studies will be needed to clarify the prognostic role of CPET in these cohorts of patients.
Conclusions
Currently, the risk stratification in cardiomyopathies is a very actual issue. CPET represents an important prognostic tool to stratify both the arrhythmic risk and HF-related events. CPET-derived variables and existing cut-offs showed their best prognostic ability when tailored according to the specific cardiomyopathies, suggesting a more personalised approach. Cardiomyopathy patients should be early and routinely referred to HF advanced centres in order to perform a comprehensive risk stratification which should include a CPET.
Footnotes
Acknowledgements
This paper is dedicated to the memory of Professor Fulvio Camerini, foremost expert in cardiomyopathies, outstanding clinician and scientist. The author(s) would like to thank Fondazione CR Trieste, and FINCANTIERI for their support. They are also grateful to all the healthcare professionals for the continuous support to the research and clinical management of patients and families with cardiomyopathies followed in the HF outpatient clinic and cardiomyopathy centre of Trieste.
Author contribution
All author(s) contributed to the conception or design of the work. All authors contributed to the acquisition, analysis, or interpretation of data for the work. All authors drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work ensuring integrity and accuracy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
