Abstract
Heart failure with mid-range ejection fraction represents a heterogeneous and relatively young heart failure category accounting for nearly 20–30% of the overall heart failure population. Due to its complex phenotype, a reliable clinical picture of heart failure with mid-range ejection fraction patients as well as a definite risk stratification are still relevant unsolved issues. In such a context, there is growing interest in a comprehensive functional assessment by means of a cardiopulmonary exercise test, yet considered a cornerstone in the clinical management of patients with heart failure and reduced ejection fraction. Indeed, the cardiopulmonary exercise test has also been found to be particularly useful in the heart failure with mid-range ejection fraction category, several cardiopulmonary exercise test-derived parameters being associated with a poor outcome. In particular, a recent contribution by the metabolic exercise combined with cardiac and kidney indexes research group showed an independent association between the peak oxygen uptake and pure cardiovascular mortality in a large cohort of recovered heart failure with mid-range ejection fraction patients. Contextually, the same study supplied an easy approach to identify a high-risk heart failure with mid-range ejection fraction subset by using a combination of peak oxygen uptake and ventilatory efficiency cut-off values, namely 55% of the maximum predicted and 31, respectively. Thus, looking at the above-mentioned promising results and waiting for specific trials, it is reasonable to consider cardiopulmonary exercise test assessment as part of the heart failure with mid-range ejection fraction work-up in order to identify those patients with an unfavourable functional profile who probably deserve a close clinical follow-up and, probably, more aggressive therapeutic strategies.
Keywords
Introduction
The historical dichotomisation of heart failure (HF) patients according to left ventricular ejection fraction (LVEF) in those with reduced LVEF (<40%) (heart failure with reduced ejection fraction; HFrEF) and with preserved LVEF (>50%) (heart failure with preserved ejection fraction; HFpEF) finished definitively with the release of the 2016 European Society of Cardiology heart failure guidelines. 1 Indeed, due to the introduction of the heart failure with mid-range ejection fraction (HFmrEF) category, the ‘grey zone’ of those patients with LVEF ranging between 40% and 49% turned in a well-established subset. In particular, besides the LVEF values, HFmrEF patients were defined according to the presence of at least one of the following criteria: (a) symptoms and signs of HF; (b) elevated levels of natriuretic peptides; (c) relevant structural heart disease (left ventricular hypertrophy or left atrial enlargement) or diastolic dysfunction. 1
Despite the growing interest in the new HF subgroup which accounts for 20–30% of the whole HF population, up to now the HFmrEF category still represents the ‘middle child, neglected and unloved’ within the great HF family.2,3 Indeed, most likely due to its complex phenotype, both the HFmrEF clinical characteristics and risk profile, as well as a definite therapeutic strategy, are far from being elucidated.4–7 In such a context, one of the most relevant and unsolved issue is whether HFmrEF should be considered really as a distinct HF phenotype or just as a transitional step of the progression from HFrEF to HFpEF and vice versa in the continuous HF spectrum. 8 Although some studies, according to a strict pathophysiological viewpoint, suggest considering HF as a unique heterogeneous syndrome with different phenotypes and disease trajectories,9,10 a large body of the literature retains HFmrEF as a real entity with intermediate characteristics between HFrEF and HFpEF both in terms of clinical features and prognosis.5–7,11,12 Accordingly, patients with HFmrEF usually show a higher prevalence of non-ischaemic aetiology as well as comorbidities (i.e. anaemia, hypertension, atrial fibrillation and chronic kidney disease) than those with HFrEF, whereas they are usually younger and more often men compared to those with HFpEF.13,14 Similarly, also with respect to possible differences in outcomes, HFmrEF patients have been shown to have an intermediate risk of all-cause and cardiovascular mortality at one, 5 and 10 years’ follow-up between HFrEF and HFpEF.2,5,15,16 Adding further complexity to the scenario, some previous studies highlighted that those HFmrEF patients who recovered from previous evidence of reduced systolic function, the so-called rec-HFmrEF, show a distinct behaviour. Indeed, a study by Park and colleagues demonstrated that an improvement in LVEF was strongly associated with reduced rates of mortality and hospitalisations, independently from baseline LVEF and current medical therapy. 17 Supporting this datum, another study by Nadruz and colleagues documented a lower risk of cardiovascular death, heart transplantation and implantation of left ventricular assistance devices (LVADs) in rec-HFmrEF patients as compared not only to HFrEF patients but also with respect to those with HFmrEF who never experienced a LVEF lower than 40%, the so called ‘de-novo’ HFmrEF patients. 8 Eventually, most likely due to the above-mentioned open issues and to the lack of specific randomised trials, HFmrEF suffers from a not univocal pharmacological strategy compared to the HFrEF category, usually being undertreated with HF ‘disease-modifier’ drugs, namely angiotensin-converting enzyme inhibitors (ACEi)/angiotensin receptor antagonists (ARBs), β-blockers and mineralocorticoid receptor antagonists (MRAs). 6
Cardiopulmonary exercise test in HFmrEF patients
Cardiopulmonary exercise testing (CPET) represents a cornerstone in the assessment of functional capacity and prognosis of HFrEF patients either as a single CPET parameter (i.e. peak oxygen uptake (pVO2), relationship of ventilation with carbon dioxide production (VE/VCO2 slope)),18,19 as a combination of CPET parameters, 20 or as a part of more comprehensive scores.21–24 Conversely, up to now, few studies challenged the CPET-derived variables with respect to their possible role in HFmrEF clinical management as well as its risk stratification.
Table 1 supplies a brief and schematic overview of all the main published papers dealing with HFmrEF which included a CPET assessment in their own research protocol. Besides some relevant differences in the study construction (i.e. sample size, HFmrEF patients’ characteristics, pre-specified endpoints), what clearly emerges from each of them is that a CPET assessment might represent a useful tool in HFmrEF management.8,25–29 In particular pVO2, either expressed in ml/kg/min or as percentage of maximum predicted, has been found always to be related to the HFmrEF outcome. The underlying reason for pVO2 accuracy in identifying patients at highest risk might derive from its formula, namely the Fick law. 30 Indeed, pVO2 depends not only on the cardiac output but also on the artero-venous oxygen difference, the latter being impaired to a different extent in such a complex and heterogeneous HF setting. However, besides pVO2, also the VE/VCO2 slope, another mainstay in HFrEF risk stratification, has been proved to be significantly associated with a worse outcome. 19 The possible advantage of this CPET-derived parameter in HFmrEF patients, besides its close relationship with left ventricular diastolic dysfunction (i.e. wedge pressure), might be derived from its feasibility in those cases (i.e. elderly and highly comorbid HF patients) in which it is difficult to achieve the metabolic criteria to consider CPET as maximal. 31 Noteworthy, together with pVO2 and the VE/VCO2 slope, many other CPET-derived variables have been investigated in the HFmrEF category. Indeed, Popovic and colleagues demonstrated that also a flattening of the VO2 trajectory during maximal exercise, a marker related to cardiac output impairment and known to be accurate in detecting stress-induced ischaemia, 32 might discriminate those HFmrEF patients with a worse outcome in terms of all-cause death and cardiovascular events. 27 Eventually, a more recent study by Rovai and colleagues, on behalf of the metabolic exercise combined with cardiac and kidney indexes (MECKI) research group, confirmed a similar rate of exertional oscillatory ventilation (EOV) to those observed in HFrEF and its worse impact on prognosis in a large cohort of rec-HFmrEF patients. 28
List of all published studies (retrospective) dealing with HFmrEF which included a CPET assessment.
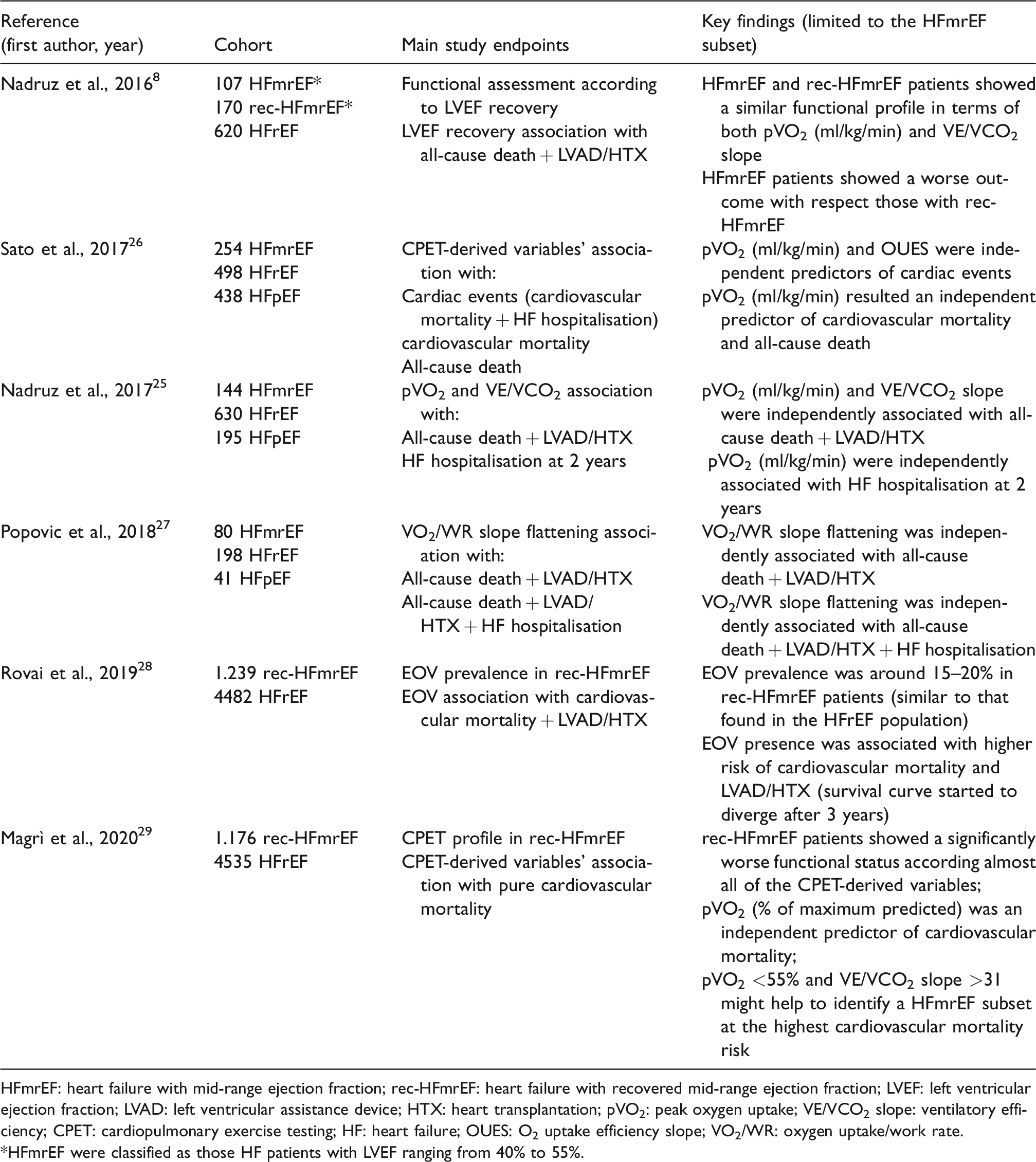
HFmrEF: heart failure with mid-range ejection fraction; rec-HFmrEF: heart failure with recovered mid-range ejection fraction; LVEF: left ventricular ejection fraction; LVAD: left ventricular assistance device; HTX: heart transplantation; pVO2: peak oxygen uptake; VE/VCO2 slope: ventilatory efficiency; CPET: cardiopulmonary exercise testing; HF: heart failure; OUES: O2 uptake efficiency slope; VO2/WR: oxygen uptake/work rate.
*HFmrEF were classified as those HF patients with LVEF ranging from 40% to 55%.
Insights from the MECKI score research group in rec-HFmrEF
A recent Italian multicentre study, conducted by the MECKI score research group, sought to characterise a large cohort of stable rec-HFmrEF (
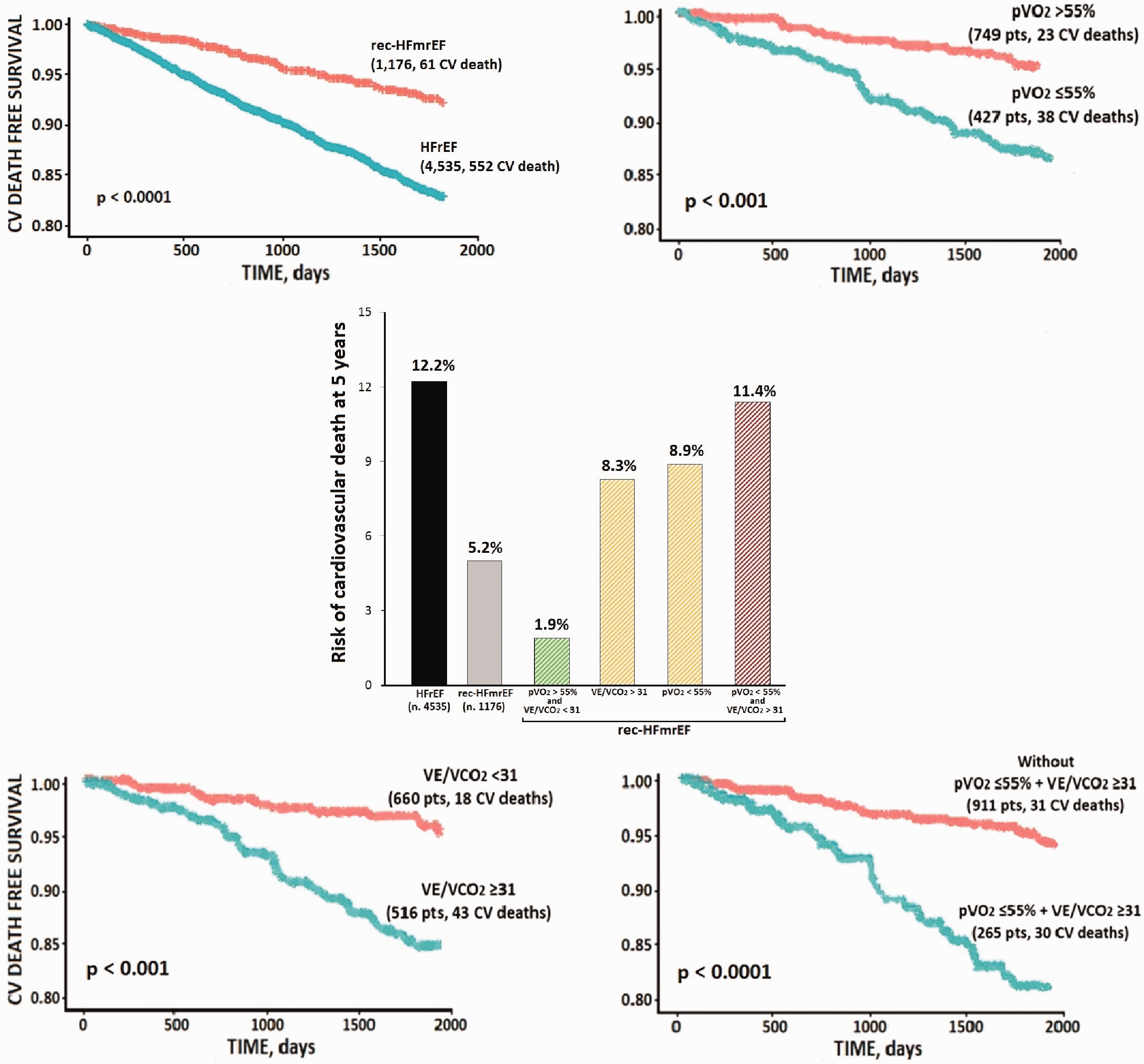
Cardiovascular mortality in HFrEF and rec-HFmrEF patients: insights from the metabolic exercise combined with cardiac and kidney indexes (MECKI) score research group. Kaplan–Meier estimator of cardiovascular mortality at 5 years for left ventricular ejection fraction in the overall population (rec-HFmrEF vs. HFrEF) (upper left panel) and for peak oxygen uptake (peak VO2 ≤55%) (upper right panel), ventilatory efficiency (VE/VCO2 slope ≥31) (bottom left panel) and both cut-off values (bottom right panel) in the rec-HFmrEF sample. The incidence rate of cardiovascular mortality at 5 years in the overall HFrEF and rec-HFmrEF groups and in the rec-HFmrEF subgroups categorised according to cut-off values of peak VO2 and VE/VCO2 slope (central panel). Modified from Magrì et al. 29 rec-HFmrEF: heart failure with recovered mid-range left ventricular ejection fraction; HFrEF, heart failure with reduced left ventricular ejection fraction; peak VO2: peak oxygen uptake; VE/VCO2 slope: ventilatory efficiency.
Conclusions and perspectives
The clinical management of the early defined HFmrEF category most likely represents one of the main challenges in the near future for HF specialists. Indeed, a number of issues remain still open and, in particular, it is difficult to stratify the HFmrEF risk. In such a context, growing evidence suggests that reduced pVO2 and increased VE/VCO2 slope values, as well as other CPET-derived features, just known to characterise those HFrEF patients at the highest risk (i.e. VO2/work rate flattening, anaerobic threshold not identified, EOV presence) (Figure 2), are also able to predict a poor outcome in the HFmrEF class. Thus, waiting for possible dedicated clinical trials, it is very reasonable to include the above-mentioned CPET-derived variables in the routine work-up of these patients and to focus on and to manage strictly at least those HFmrEF patients with an unfavourable CPET profile.
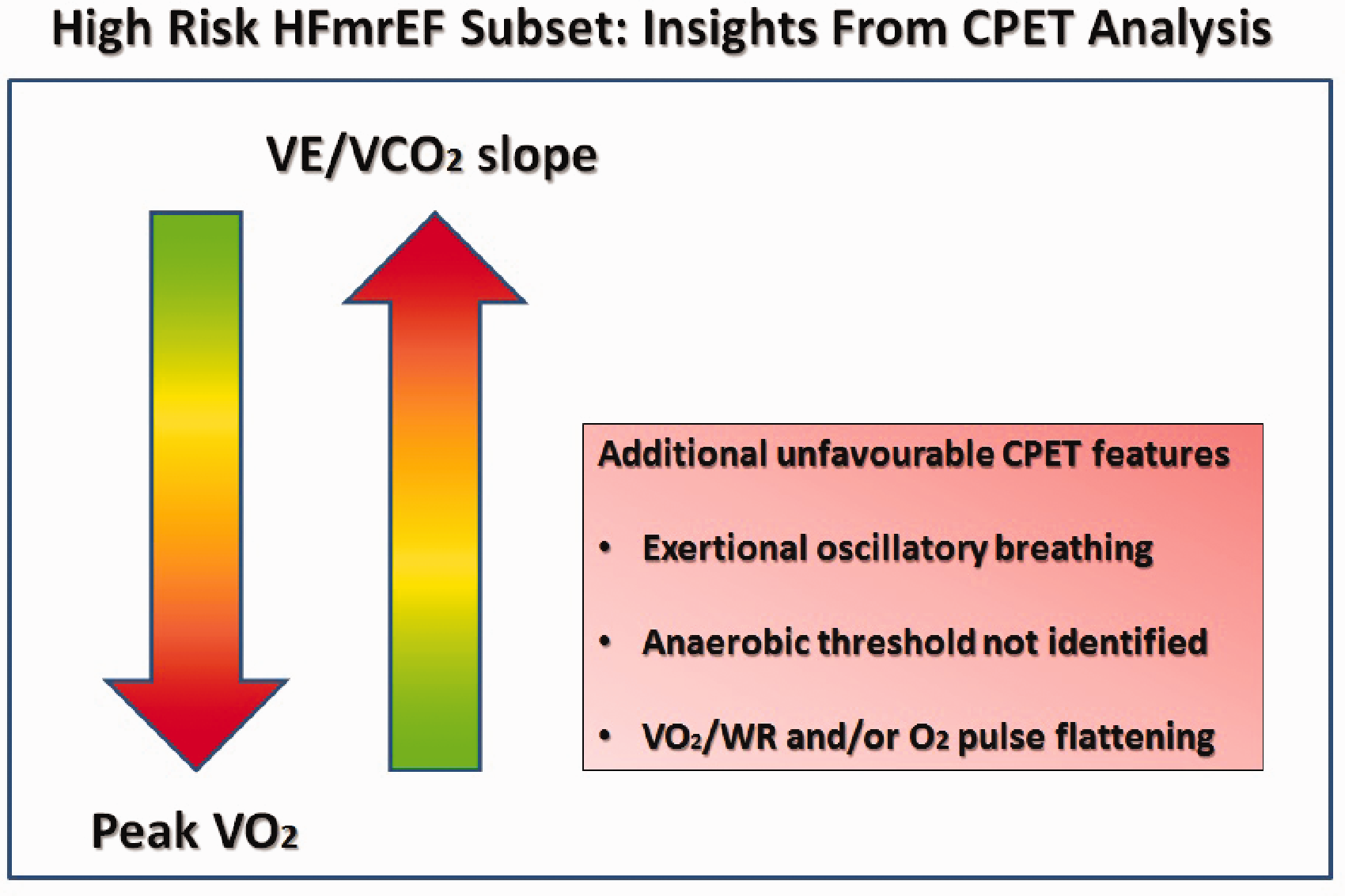
Cardiovascular risk assessment in HFmrEF according to CPET-derived parameters. HFmrEF: heart failure with mid-range left ventricular ejection fraction; CPET: cardiopulmonary exercise test; peak VO2: peak oxygen uptake; VE/VCO2 slope: ventilatory efficiency; VO2/WR: oxygen uptake/work rate.
Footnotes
Author contribution
DM and GG contributed to the conception or design of the work. DM and GG contributed to the acquisition, analysis, or interpretation of data for the work. AB and CD drafted the manuscript. GP, MC and MS critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work ensuring integrity and accuracy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
