Abstract
For as long as nucleic acids have been utilized to vertically and horizontally transfer genetic material, living organisms have had to develop methods of recognizing cytosolic DNA as either pathogenic (microbial invasion) or physiologic (mitosis and cellular proliferation). Derangement in key signaling molecules involved in these pathways of DNA sensing result in a family of diseases labeled interferonopathies. An interferonopathy, characterized by constitutive expression of type I interferons, ultimately manifests as severe autoimmune disease at a young age. Afflicted patients present with a constellation of immune-mediated conditions, including primary lung manifestations such as pulmonary fibrosis and pulmonary hypertension. The latter condition is especially interesting in light of the known role that DNA damage plays in a variety of types of inherited and induced pulmonary hypertension, with free DNA detection elevated in the circulation of affected individuals. While little is known regarding the role of cytosolic DNA sensing in development of pulmonary vascular disease, exciting new research in the related fields of immunology and oncology potentially sheds light on future areas of fruitful exploration. As such, the goal of this review is to summarize the state of the field of nucleic acid sensing, extrapolating common shared pathways that parallel our knowledge of pulmonary hypertension, in a molecular and cell-specific manner. Principles of DNA sensing related to known pulmonary injury inducing stimuli are also evaluated, in addition to potential therapeutic targets. Finally, future directions in pulmonary hypertension research and treatments will be briefly discussed.
Keywords
Don’t be so gloomy. After all it’s not that awful. You know what the fellow said – in Italy, for thirty years under the Borgias, they had warfare, terror, murder and bloodshed, but they produced Michelangelo, Leonardo da Vinci and the Renaissance. In Switzerland, they had brotherly love, they had five hundred years of democracy and peace – and what did that produce? The cuckoo clock.
∼ Harry Lime (Orson Welles)
Introduction
The apocryphal monologue above, from director Carol Reed’s archetypal 1949 spy-thriller The Third Man, neatly summarizes the key biologic concept that evolutionary pressure can yield beauty in complexity, despite often brutal means. Analogous to this film noir observation is a key concept in molecular biology; organismal detection of proliferative versus danger signaling within an environment as a complicated and evolved form of sophisticated espionage. In order to facilitate health, with successful passage of genetic material, there is arguably no more important cellular function—or more complex—than accurate and rapid processing of information, such as free circulating polynucleotide chains. Functionally cunning, this “spying” on the DNA/RNA of neighboring tissues—as well as the self—is what allows all living creatures to differentiate between: (1) a hostile-takeover leading to programmed or spontaneous cell death (due to injection of foreign RNA or DNA from viral or bacterial infection) versus (2) mitosis or synergistic sharing of genetic material to promote a healthy response to deleterious changes in the environment (such as seen with rapid temperature shifts, circadian light/dark cycles, and seasonal nutrient supply).
Cellular reconnaissance in this manner is particularly important to innate immune cells in appropriately reacting to the presence of free circulating DNA. The concept is consistent with the primordial role for DNA as a danger signal, known long before we discovered its function as the key blueprint of genetic material. 1 The importance of this detection and response is illustrated most prominently by the highly conserved nature of molecular DNA sensors in a large number of organisms from humans to sea anemones and bacteria.2–5 Related, self-DNA sensing is known to be an integral component of many lung inflammatory diseases, 6 including asthma7,8 and pulmonary fibrosis. 9 However, the role of this signaling pathway in pulmonary vascular disease is relatively unknown, despite the well-described consequence of DNA-damage, particularly mitochondrial DNA (mtDNA), in pulmonary hypertension (PH). 10 This is unfortunate, given the multiple drug targets available to regulate cytosolic DNA sensing pathways, a large amount of which are already being explored within the field of cancer and autoimmune disease, 11 that could potentially be applied to re-establishing lung vessel health.
Therefore, the purpose here is to first review in brief the role of DNA damage in PH (Fig. 1), before examining in more depth the pathways recently elaborated upon relating to maladaptive polynucleotide sensing. Finally, we will review novel therapies related to DNA detection, in particular seeking to establish a biologically plausible rationale for application of drugs to patients with a pulmonary vasculopathy (Fig. 2 and Table 1). General nucleic acid—including purinergic and RNA—sensing will not be discussed; however, these topics have recently been broadly summarized elsewhere.12,13 Of note, however, is the fact that free double-stranded RNA detection through toll-like receptor 3 (TLR3) has previously been described as necessary and sufficient for protection against development of PH secondary to chronic hypoxia exposure. 14 The mechanism for TLR3 involvement in pulmonary arterial hypertension (PAH), 15 remains unknown, despite the unexpected benefit of activation having been noted previously in a large vessel injury model. 16 It appears to be a cell-specific effect, however, with expression by pulmonary artery smooth muscle cells resulting in elevated interleukin (IL)-8 levels—promoting endothelin-1 (ET-1) expression 17 and endothelial cell expression blunting alternative double-stranded RNA (dsRNA) signaling leading to canonical inflammatory cascade signaling through nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB). 14 Thus, an apt role for accurate and sometimes lethal cellular foreknowledge in the amplification of abnormal vascular responses within the lung is established.
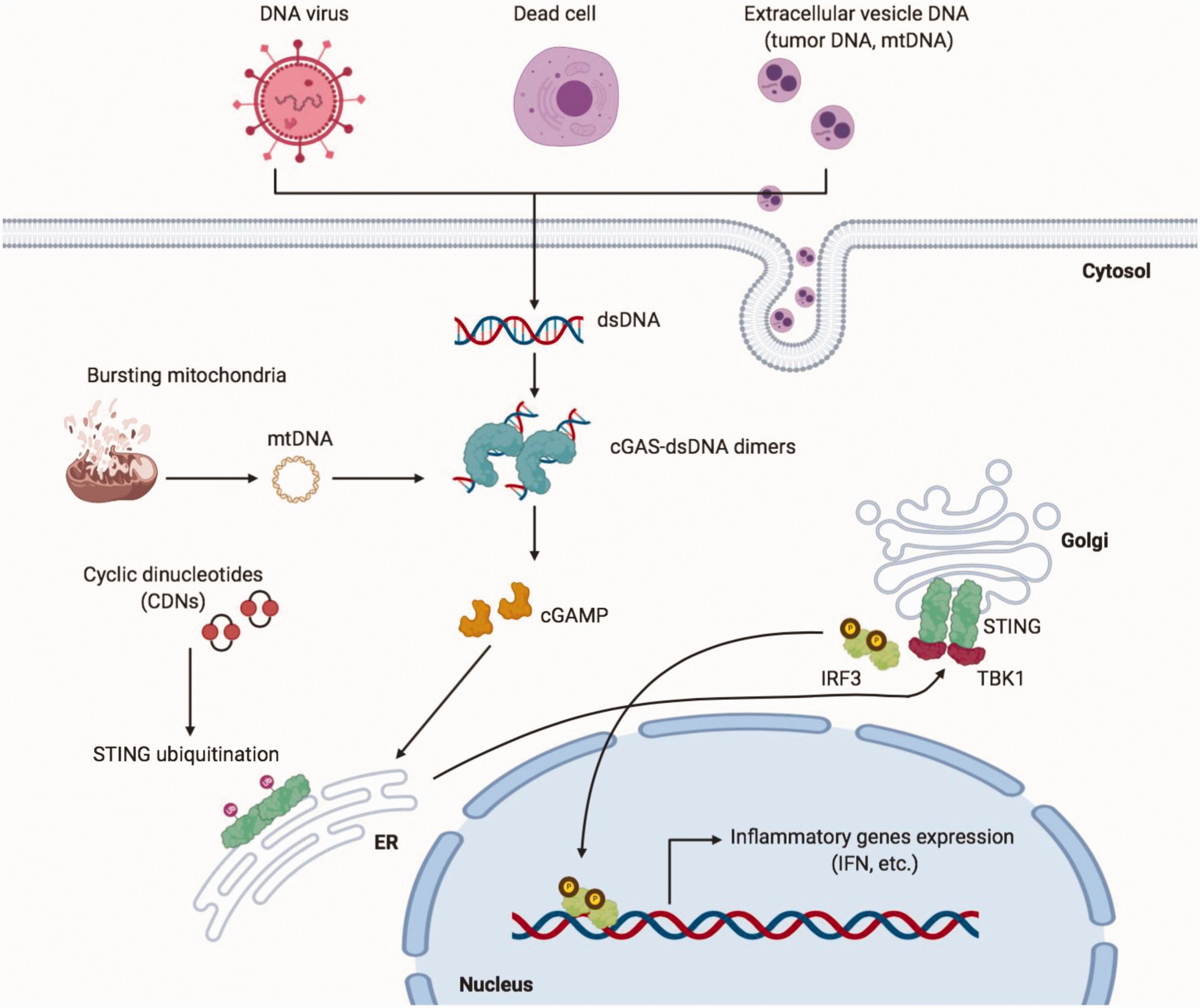
Overview of the cytosolic DNA sensing apparatus.
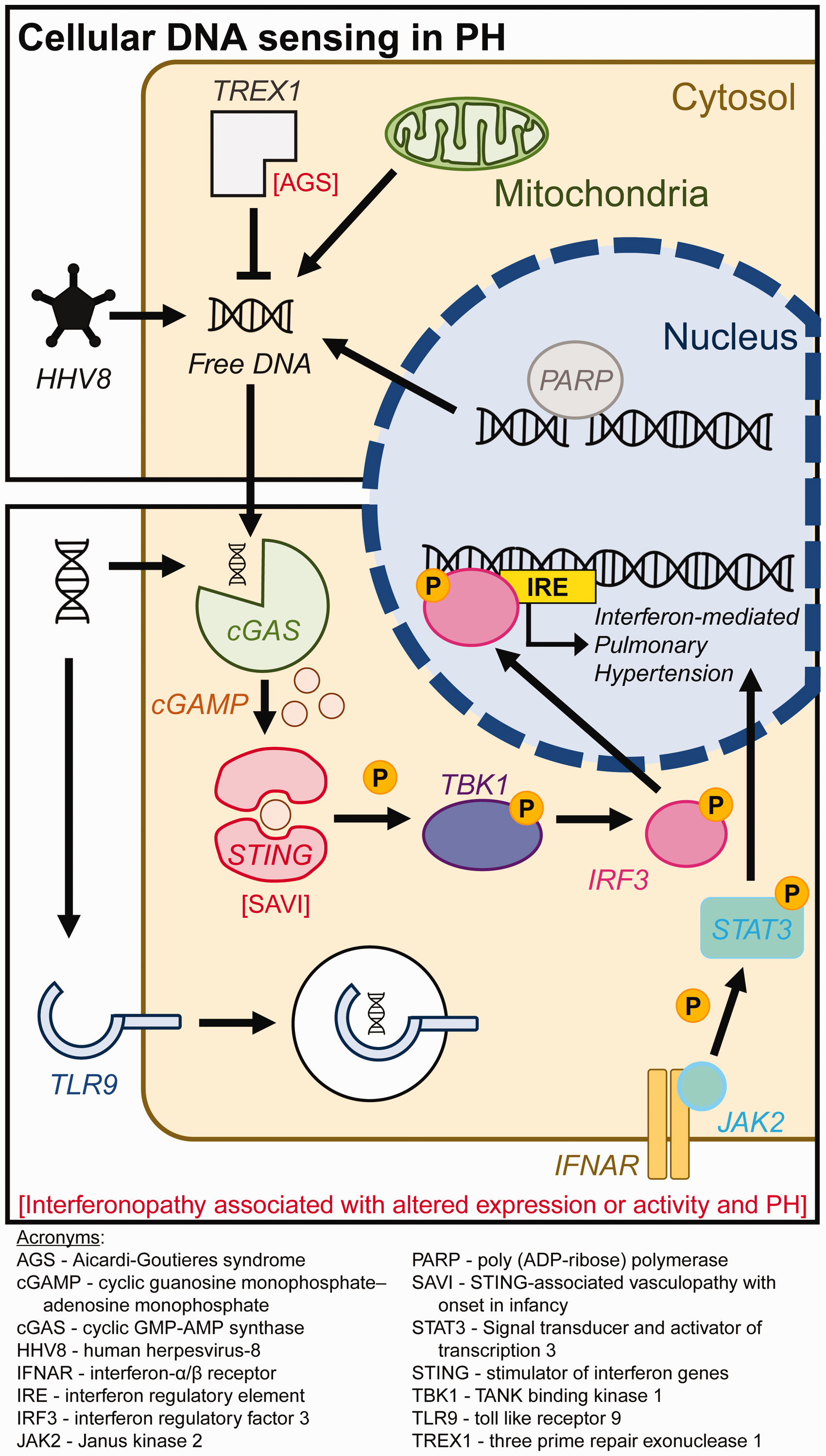
Summary figure detailing relevant pathways to cytosolic DNA sensing in development of pulmonary hypertension (PH).
Available therapeutics, mode of action and related to DNA sensing pathways.

STING: stimulator of interferon genes; PH: pulmonary hypertension; PD-1: programmed cell death protein 1; PD-L1: programmed death-ligand 1; PMN-MDSC: polymorphonuclear cells; RV: right ventricular; ET-1: endothelin-1; JAK/STAT: Janus kinase/signal transducer and activator of transcription.
DNA sensing and its role in PH
Patients with PAH are known to be predisposed to nuclear and mtDNA damage (recently reviewed elsewhere 18 ). Accordingly, both an increased susceptibility to mutagens—due primarily to malfunction in DNA repair mechanisms—and an increase in baseline systemic DNA damage have been reported in isolated pulmonary endothelial and myeloid cells derived from patients with PAH, 19 including enhanced sensitivity to bleomycin-mediated nuclear injury. In heritable PAH, most commonly due to loss of function mutations in bone morphogenetic protein receptor II (BMPR2), there is known intrinsic genomic instability 20 associated with specific compensatory upregulation in DNA repair enzyme poly (ADP-ribose) polymerase 1 (PARP1) in pulmonary artery smooth muscle cells of patients with disease. 21 Of note, a similar phenomenon of cellular “addiction” to PARP-mediated repair has been described in several malignancies, drawing yet another parallel between PH and cancer biology. 22
Several additional components of the DNA damage response are worth noting, as relevant to development of PH. Lupus Ku autoantigen (Ku70) antibodies have been associated with both PAH 23 and connective tissue diseases that predispose to PH.24,25 As an essential nonhomologous end-joining DNA double-strand break repair enzyme, Ku70-null mice display an increase in DNA damage and is associated with apoptotic resistance as well as PH and emphysema. 26 A delicate balance must be struck, however, as evidenced by the fact that mere inhibition of Ku70, by either decreased phosphorylation 27 directly or epigenetic regulation, 28 results in protection against development of PH. Similarly, expression of the DNA damage response sensors MRE11-RAD50-NBS1 and the E3 ubiquitin ligase UBR5, necessary for promoting endothelial cell health under homeostatic conditions, predisposes to development of PH. 29 Finally, overexpression of Nudix hydrolase 1 (NUDT1), a detoxifying DNA enzyme, results in increased incorporation of oxidized nucleotides into DNA, promoting apoptosis resistance and proliferation secondary to DNA damage 30 ; NUDT1 inhibitors have been shown to block development of PH in the monocrotaline and Sugen/hypoxia rat models.
A hallmark of connective disease, titers of auto-antibodies directed against double-stranded DNA (dsDNA) are known to correlate with development of PH, especially related to scleroderma 31 and systemic lupus erythematosus (SLE). 32 This is consistent with what is known in the DNA sensing literature, where primarily DNA-derived from mitochondria lead to increased interferon production and a worsening of lupus findings using the pristane mouse model of disease. 33 In fact, patients with the monogenic form of SLE are heavily predisposed to an amplified type 1 interferon production cycle, autoinflammatory disease, and PH. 34 The link between autoantibody expression and PAH continues to be explored. 35
mtDNA damage
mtDNA damage has also been associated with BMPR2 mutation, with abnormalities in mitochondrial metabolism and cytosolic mtDNA associated with altered apoptotic response to injury in mutant strains. 36 The group that first described these changes in heritable PAH, later also described a similar phenomenon in another unfortunate experiment of nature; noting a rampant increase in clinically diagnosed methamphetamine-induced PH, they asked whether they could find similar changes in mtDNA in patient samples with disease 37 ? In both circulating bone marrow-derived cell and endothelial cell samples from patients, as well as mice upon induction with methamphetamine, they found an increase in mitochondrial dysfunction with an expected rise in mtDNA damage, subverting the adaptive vascular response to reactive oxygen species (ROS)-mediated injury, and ultimately contributing to PH.38,39 These findings are interesting in light of known sex-specific mtDNA mutations in cardiovascular disease 40 and mitochondrial haplogroup-specific changes 41 associated with development of PH. These data reinforce how influential endothelial cell bioenergetics and mtDNA damage predisposing to vascular injury can be, especially in women, 42 the population primarily diagnosed with and affected by heritable PAH.
Related, if mtDNA damage is involved in the pathogenesis of PH, it stands to reason that protection against injury of nucleotide would prove beneficial to prevention and treatment of disease. As an example, it has been demonstrated in endothelial cells that DNA repair enzyme 8-oxoguanine glycosylase (OGG1) over-activity protects against mtDNA damage and subsequent cell death. 43 OGG1, a glycosylase enzyme that is heavily involved in base excision repair, is induced primarily by xanthine oxidase-induced mitochondrial injury. Therefore, it is thought to be integral in prevention of further mitochondrial dysfunction leading to cellular apoptosis 44 and oxidant-related tissue injury. 45 Regarding translational potential of this protein, OGG1 has thus far been shown to reverse vascular barrier compromise in response to oxidant stress, 46 and hypoxia-inducible factor-mediated endothelial cell injury. 47 OGG1-deficient mice also display an increase in auto-dsDNA antibodies in a lupus model, associated with increased interferon production and worsened objective findings of disease. 33 Ultimately, a balance must be struck between these potentially disadvantageous and beneficial mtDNA injury responses, in order to restore homeostatic cellular function.
Ligand sequestration and stability
Cells can only respond to that which they are capable of detecting, however. Therefore, compartmentalization of substrate—double-stranded mtDNA or nuclear DNA (nucDNA)—through repair or removal is an effective method of preventing potential downstream pathology related to DNA sensing. Effectively, there are two major methods available to prevent accumulation of poly-nucleic acids, through: (1) aforementioned DNA repair enzymes or (2) ligand digestion. The former includes elimination of damaged or senescent mitochondria through mitophagy via direct alteration in mitochondrial membrane permeability—normally tightly regulated in response to stress in order to induce immune tolerance or activation and proliferation—in response to noxious stimuli. 48 A similar process is also involved in the tightly regulated role of nucleic acid chain sensing in response to mitosis, whereby cell cycle does not activate DNA sensing pathways. However, during mitotic arrest, low-level accumulation of nuclear micronuclei—containing nucDNA—can lead to induction of apoptosis through secondary protein alterations in mitochondrial permeability. 49
A second method of nucleic acid sequestration, preventing cytosolic sensing and activation, involves a large family of DNA-targeting enzymes (DNAse) that function in large part to digest dsDNA prior to propagation of the danger signal. Vital to vascular health, deficiency in one of these proteins, DNAse II—functioning within the phagolysosomal compartment—has recently been linked to development of PH in a patient with SLE-like symptoms. 50 Similarly, mutations in TREX1 (or DNAse III) have been strongly linked with autoimmunity and SLE. 51 Functioning primarily within the cell cytosol, TREX1 loss-of-function mutations result in a rare disease entity, Aicardi-Goutieres syndrome (AGS). An illness of constitutive interferon production and signaling associated with development of PH at a young age, AGS is one of several type I interferonopathies linked to pulmonary vasculopathy including: gain-of-function mutations in melanoma differentiation-associated protein 5 (part of the RIG-I-like receptor family associated primarily with dsRNA-sensing), 52 and the related Singleton-Merten syndrome.53,54
Viral nucleic acid sensing
As described in the introduction, DNA sensing pathways are ostensibly established in order to distinguish self versus hostile cytosolic nucleic acid. Relevant to the field of PH, DNA viruses have long been postulated to serve as a potential “second-hit” stimulus in genetically predisposed patients, tipping the scales toward maladaptive vascular repair and development of disease. This hypothesis was initially supported by work demonstrating a higher than expected detection of human herpes virus-8 in the lungs of patients with PAH, 55 but subsequent studies have failed to replicate these findings,56,57 although all have been relatively underpowered to do so.
A portion of the intrigue related to viral detection in patients with PAH, however, may be built on a somewhat faulty premise; that detection of contemporaneous infection—and thus recently acquired viral DNA—in patients is the pathologically relevant sentinel event. A recent publication has colored this assumption in a provocative light, focusing on the role of ancient viruses—conserved DNA sequences of retroviral origin that are vertically transmitted—in aberrant vascular remodeling. 58 In the referenced manuscript, the group propose that PAH is a consequence of sustained immune cell activation due to upregulation of the endogenous retrovirus Human Endogenous Retrovirus-K (HERV-K). Presumably, accumulation of the HERV motifs sustains this chronic inflammatory response, which—related to the above discussion on TREX1—is exactly what has been described in AGS patients with PH; decreased HERV cDNA destruction associated with constitutive innate immune cell activation. 59 Although speculative, one could hypothesize based on these data that the presence of cytosolic DNA—originating from multiple sources—could potentially become a bottleneck in disease progression, and therefore a potent area of study for therapeutic targeting of disease. First, however, we need to have a better understanding of how cell-free DNA is sensed by all cells, and specific cell types in particular.
Sensing of foreign and self-DNA damage in PH
Molecular mechanisms
The evolving story of cytosolic DNA sensing, 60 and self-DNA sensing in primary lung disorders, 6 is complex. To this end, a network of molecular and cellular mechanisms has been described, as relevant to nucleic acid sensing in disorders and disease models with overlapping pulmonary vascular disease pathology. The following is thus a brief overview of mechanistic signaling involved in DNA detection at a tissue and cellular level, with highlighted relevance to PH.
Toll-like receptor 9
The first described cellular dsDNA sensor, 61 TLR9 functions within the intracellular endosomal compartment, and is crucial to prevention of self-DNA recognition, particularly by inflammatory cells. 62 As such, TLR9 is normally described as acting through the canonical inflammatory pathway involving NF-κB, via myeloid differentiation primary response 88 (MYD88). Moreover, TLR9 activation in endothelial cells specifically is associated with an increase in IL-6 paracrine-mediated increase in smooth muscle cell proliferation and pulmonary vascular remodeling. 62 In this particular study, however, effects were predominantly felt to be secondary to hemoglobin–lipid peroxidation and generation of ROS in mice and rats. This must be taken into consideration, however, with evidence that TLR9 stimulation through ligand CpG decreased secondary mediators associated with both protection and susceptibility to PH. 63 An example, TLR9/MYD88 signaling through interferon regulatory factor 1 (IRF1), in synergistic activation with signal transducer and activator of transcription 1 (STAT1), stimulates release of ET-1 in human vascular smooth muscle cells, a known causative factor associated with development of PH. 64
Cyclic GMP-AMP synthase and Stimulator of Interferon Genes
A broader view of cyclic nucleotide-sensing related to nitric oxide signaling (using either cyclic guanosine monophosphate or cyclic adenosine monophosphate intermediates) and associated redox biology is beyond the scope of this discussion, with several reviews on the subject having recently been published, for reference.65–67 However, related to the current discourse is the role of purinergic sensing in PH, which has seen a relative explosion in interest within the past several years. 68 Genomewide RNA studies have previously demonstrated alteration in the purinergic G-protein coupled receptor P2Y-family in patients with PAH and secondary PH, compared to healthy controls. 69 Mechanistically, blocking P2Y1 and P2Y12 receptors blocks adenosine diphosphate (ADP)-induced pulmonary vasoconstriction in pigs. 70 P2X7R—a purinergic receptor for adenosine triphosphate (ATP) and a pattern recognition receptor—has likewise been shown to contribute to PH, through NLR family pyrin domain containing 3 inflammasome activation in monocrotaline-treated rats. 71 Protection against hypoxia-induced PH in mice is likewise afforded through blocking or deletion of CD39, an ectonucleotidase responsible for conversion of nucleotides ATP and ADP to AMP.72,73 Finally, antagonism of the adenosine 2B receptor has demonstrated therapeutic promise, halting progression of bleomycin-induced PH, in particular. 74
Cyclic dinucleotide detection, we now know, originated in bacteria, with bacterial Stimulator of Interferon Genes (STING) being particularly adept at response to invader generation cyclic di-GMP. This is juxtaposed to the refined mammalian response primarily to the secondary intermediate cyclic GMP-AMP (cGAMP), and is believed to represent a prokaryotic defense against bacteriophages and viral invaders, cellular “spies”. 75 Humans do have some retention of the cellular machinery to respond to bacterial cyclic di-GMP, however, such as the oxidoreductase RECON-mediated activation of inflammatory lynchpin, NF-κB. 76
First described in a small cohort of six patients in 2014, the STING-Associated Vasculopathy with onset in Infancy (SAVI) syndrome illustrates the negative sequelae related to unmitigated cytosolic DNA sensing, with systemic inflammation, cutaneous vasculopathy, and—most relevant to this discussion—development of early pulmonary fibrosis and PH. 77 In this disorder, a germline mutation in STING (also referred to as TMEM173) causes constitutive homodimerization and activation of the protein, independent of the specific secondary messenger cyclic dinucleotide produced by cGAMP synthase (cGAS) upstream, which normally initiates recognition of free DNA. Thus, SAVI—another member of the interferonopathy family of diseases which includes the aforementioned AGS—results in elevated basal levels of type I interferons, including interferon (IFN)β. Propagation of intracellular inflammatory signaling pathways is then increased, such as phosphorylation of STAT1 and STAT3. This occurs primarily in peripheral blood mononuclear cells (CD14+ monocytes), and not T lymphocytes. However, lung disease has been demonstrated to develop independently of type I interferon signaling—and cGAS detection of dsDNA—relying in large part on the immunophenotype of T cells, in at least one study. 78 Likewise, STING activation in vascular endothelial cells has been shown to be associated with tissue-specific effects such as elevated apoptosis, a phenomenon dramatically reduced by treatment with Janus kinase (JAK) inhibitors. 77 However, the myeloid and lung changes do appear to be independent of classical interferon-stimulated gene activation through phosphorylation of transcription factor interferon regulatory factor 3 (IRF3) by TANK-binding kinase 1 (TBK1), in some of the hyper-activating mouse mutant models. 79
Of note, STING gain-of-function mutation does not necessarily have to occur in the dimerization domain of the gene in order to manifest severe PH, 80 though these patients will often still respond well to JAK inhibition. 81 A similar mutation has also been demonstrated to predispose mice to gamma herpes virus T cell-mediated pulmonary fibrosis, similar to patients diagnosed with SAVI, and effect which is myeloid-cell dependent. 82 PH, however, was not assessed in this report, though mild elevation in pulmonary pressures would be expected following degree of lung fibrosis.
STAT protein family
As described previously, STAT signaling is necessary for appropriate and maladaptive inflammatory responses in PH. 64 Inhibition of STAT1-induced cytokine production—in particular—has been associated with pharmaceutical treatment of PH, reducing phosphorylation and nuclear recruitment of the transcription factor, leading to improved pulmonary pressures. 83 Related, STAT1/3 inhibition with broad tyrosine kinase-inhibitor imatinib has been shown protective against the development of monocrotaline-induced PH in rats. 84 STAT3 signaling especially has received a large amount of interest in the field of pulmonary vascular disease. 85 However, these data are primarily viewed through a smooth muscle cell-specific lens, due both to oncogenic mediator expression by this cell type 86 and reversal of monocrotaline associated PH with use of relatively specific STAT3 inhibition.
The relationship between cytosolic DNA sensing and STAT-activation is in a nascent stage. From investigation of SAVI patients, and murine modeling, we know that STING can suppress STAT3 activation tonically along with IFNβ/IL-6 production via alternative phosphorylation of a TBK1 on serine-residue 754 (Ser754). 87 Interestingly, IL-6-mediated STAT3 intracellular signaling is associated with familial PAH due to mutations in BMPR2. 88 Although not felt to manifest in a cell-specific manner, exosome delivery of micro RNA (miRNA) influencing STAT3 activation as a causative agent in PH suggests a paracrine/endocrine mechanism of disease development. 89 Potentially related immune cell contribution has been demonstrated to be affected by upstream IL-6 blockade, leading to protection against heritable PH modeling in mice. 90 These changes in turn correlate with decreased T-helper cell 17 accumulation in the lung, as well as a decreased in the M2 polarization of pulmonary macrophages.
STAT5a/b differential activation has been hypothesized to contribute to sex-specific manifestation of animal and human PH.91,92 contributing to BMPR2 mis-localization and subsequent dysfunction. 93 STAT6, on the other hand, has been shown to be associated with T cell-mediated effects on vascular smooth muscle cell proliferation in murine PH pathology. 94 This mechanism is felt to be through indirect signaling via soluble mediators—similar to schistosomiasis-induced PH. 95 In particular, the Th2 cytokines IL-4 and IL-13, acting via STAT6, in both the bleomycin-induced pulmonary fibrosis 96 and chronic hypoxia-induced PH 97 models, have been demonstrated to act in hypoxia-mediated pulmonary vascular remodeling through the well-characterized mediator hypoxia-inducible mitogenic factor (FIZZ1/RELMα). 98 Specifically, IL-13 has a direct effect on STAT6 associated smooth muscle cell proliferation via the IL-4 receptor (IL-4R), 99 a process that closely mirrors that of cGAS/STING activation via cyclic dinucleotide signaling. 100 Unsurprisingly, this pathway is especially relevant to DNA sensing, as activation of STAT6 by STING is critical for antiviral innate immunity, 101 requiring JAK-independent TBK1 phosphorylation. Of course, by definition activation or inhibition of any STAT family member is likely to have potentially profound side effects, because these are central regulators in the immune system, and in many tissue homeostatic activities. Additionally, the different STAT proteins—of which there are only seven mammalian STAT proteins (1, 2, 3, 4, 5a, 5b, 6)—have quite distinct areas of biologic activity so in the case that one of them is blocked, others do not necessarily compensate for the missing function.
Cellular mechanisms
Endothelial cells
Human pulmonary artery endothelial cell migration has previously been shown to be regulated directly through interferon-signaling, via IRF3. 102 Despite a single study’s finding that interferon alpha (IFNα) is capable of reversing PH associated with the combination of vascular endothelial growth factor receptor antagonist (Sugen 5416) and chronic hypoxia exposure, 103 there is strong evidence for type I interferon signaling as being detrimental in the pathogenesis of pulmonary vascular disease. 104 It is worth noting that in the former manuscript, there was no significant difference in levels of interferon in the circulation of patients with PAH, compared to matched healthy controls. In the latter report, however, endothelial cells grown from patients with PAH were more sensitive to effect of interferon-stimulation than donor controls. Moreover, interferon alpha receptor type 1 (IFNAR1) null mice were protected from the deleterious effects of chronic hypoxia exposure, specifically noted to display a decrease in whole lung ET-1, which is known to be associated with worsened disease outcomes. These findings are consistent with human “experiments” using IFNβ as treatment for patients with multiple sclerosis, where development of PH is a well-described, although rare, complication. 105
Of note, endothelial cell proliferation is promoted specifically by mtDNA detection through cGAS/STING, via effects on canonical HIPPO-signaling pathway regulation of transcription factors yes-associated protein 1 (YAP)/TAZ. 106 The latter is especially relevant in light of recent findings demonstrating that patients with PAH have aberrantly activated HIPPO-mediated transcription factors YAP/TAZ signaling within pulmonary vessels, specifically vascular smooth muscle cells. 107 Such paracrine effects may ultimately explain how type 1 interferon induction is capable of specialized recruitment of certain myeloid-derived cells, through elaboration of chemokines such as CX3CL1 and CCL5, within the pulmonary circulation. 108
Myeloid cells
Bone marrow-derived cells have previously been described to play unique roles in development of pulmonary vascular disease. 109 Related, cytosolic DNA sensing through cGAS/STING signaling is known to tightly regulate the innate immune response preventing development of chronic inflammatory disorders. 110 For example, plasmacytoid dendritic cells—a unique DC sub-population that specializes in interferon production—have been described to increase interferon signaling primarily through cGAS/STING activation. 111 This cell-type has likewise been shown to be correlated with progression of elevated pulmonary pressures in patients with PAH.112,113
More recently, a sub-population of myeloid-derived cells has been described that is characterized as immunosuppressive, facilitating protection against T-cell activation, primarily. These myeloid-derived suppressor cells (MDSCs), phenotypically similar to either monocytes (M-MDSC) or polymorphonuclear cells (PMN-MDSC), are evolutionarily conserved immature bodies that are liberated from the bone marrow during periods of stress and induction of emergency myelopoiesis, and are linked to development of a wide swathe of illnesses, from cancer to autoimmune diseases. 114 Our own group has recently described these cells as playing a necessary and sufficient role in the development of PH secondary to chronic hypoxia or interstitial lung disease, 115 with evidence of increased circulating levels of myeloid-derived cells in the blood of patients with PAH. 116 Importantly, in response to cellular DNA breakage via application of external radiation, STING is necessary to facilitate accumulation of MDSC at tumor bed sites, through CCR2-mediated chemoattraction of primarily M-MDSC. 117 Although there is evidence that this results in a pro-immunosuppressive effect—and thus progressive growth of malignancy—it is a complicated response with at least one group having shown that high amounts of MDSC STING stimulation associated with ligand infusion results in tumor regression. 118 In part, this may be explained by an indirect and differential response to downstream inflammatory signaling, such as STAT activation. For example, MDSC STING activates suppressor of cytokine signaling 1, itself a potent inhibitor of STAT3 signaling, necessary for facilitating progression of the DNA Epstein-Barr Virus-associated malignancy nasopharyngeal carcinoma. 119 This is an exceedingly complex regulatory network, however, with STAT3 in turn serving to downregulate STING signaling through ligand inhibition, 120 and playing a role in cytosolic DNA sensing related to immune checkpoint expression. 121 Ultimately, more work remains to determine the specific role STING plays in the immunosuppressive fate of MDSC in a variety of illnesses. 122
Exposures and therapy
Exposures
A host of clinical and bench-side stimuli of systemic inflammation due to DNA sensing pathway activation are linked to the development of chronic lung diseases, such as idiopathic pulmonary fibrosis (IPF) or chronic obstructive pulmonary disease. Below summarizes some of these exposures, and how they may relate primarily to progression of associated PH (World Health Organization Group 3 PH).
Pulmonary fibrosis-inducing agents
Although TLR9 has previously been linked to development of rapidly progressive IPF,123,124 the majority of research into the role of DNA sensing has focused on cGAS/STING. For example, silica inhalation is known to induce pulmonary fibrosis in patients as well as mice. Pulmonary cell death induced by silica leads to release of dsDNA resulting in STING activation, which in turn mediates pulmonary fibrosis, a process inhibited by treatment with DNAse I therapy. 9 The same study went on to confirm that in patients with silicosis and silica-associated interstitial lung changes, there was an in increase in circulating levels of dsDNA, which correlated with myeloid-derived cell activation in sputum and bronchoalveolar lavage fluid. These findings are prescient in relation to PH induced by crystalline silica, specifically involving the dangerous dyad of endothelial dysfunction and inflammation, contributing to pulmonary vascular remodeling. 125 The mechanism for this phenomenon has only recently been elucidated, with extracellular oxidative stress—associated with depression in the activity of superoxide dismutase 3—leading to increased severity of PH upon either silica 126 or bleomycin 127 exposure.
These conclusions are interesting in comparison to what is known regarding the previously referred to SAVI and interferonopathy patient findings, where often the first and most deadly complication brought to clinical attention are actually the interstitial lung changes. 128 Broadly, mutations resulting in elevated STING signaling are associated with worse pulmonary fibrosis in both human and murine models of disease. 129 As a further point of interest, it has recently been demonstrated that a decrease in STING levels of circulating myeloid cells is associated with acute exacerbations of IPF. 130 It is unclear if this is a compensatory response to acute injury, as another study demonstrated that herpes viral infection alone in mice with a SAVI-mutation (N153S) was enough to develop severe type 1 interferon-mediated fibrosis. 82 Pulmonary vasculature changes were not addressed in any of these studies, however.
Tobacco smoke inhalation
Cigarette smoke is well described as injurious to pulmonary barrier function primarily due to epithelial cell injury and concurrent release of self-DNA and activation of the cGAS/STING pathway. 131 Downstream interferon signaling and neutrophilic infiltrates then contribute to tobacco smoke-related fibrosis and emphysematous changes, with IFNAR1-antibody acting to inhibit phenotypes in exposed mice. Complicating the story, another group has uncovered that STING is actually decreased in settings of cigarette-exposure-mediated DNA damage and release, resulting in worsened emphysema, though—again—a compensatory effect cannot be ruled out. 132
Of course, cigarette smoke is also known to induce DNA damage, through promotion of oxidative stress, in vascular endothelial cells directly. 133 Likewise, nicotine alone is capable of independently inducing DNA damage via similar mechanism, in isolated in vitro studies. 134 These factors may ultimately contribute to chronic nicotine-induced pulmonary vascular remodeling, 135 with prevention of tobacco-smoke associated PH secondary to emphysema having previously been demonstrated in response to antioxidant/vasodilator administration. 136
Ionizing radiation
Finally, application of external radiation (either ambient or therapeutic) acts to increase interferon activity through STING activation, 137 an exposure known to induce acute pneumonitis and chronic lung fibrosis. 138 Interestingly, mtDNA released by tumor cells elicits a STING priming effect in circulating innate immune cells, responsible for the abscopal response of distant tumor burden after treatment. 139 Although the pathology of disease is complex, the latter phenomenon may in part explain downstream protection against direct and indirect radiation-induced pulmonary fibrosis through blocking TBK1 phosphorylation and IRF3 nuclear translocation. 140
While broad thoracic radiation has been demonstrated to result in development of PH in sheep, 141 reprogramming of circulating myeloid cells in response to soluble factors of vascular injury may explain why even partial lung irradiation in rats results in out-of-field changes to pulmonary vessels and PH. 142 Particular attention must be paid to both indication for radiation and etiology of PH, however, given that low dose irradiation in patients with PH associated with myelofibrosis may actually remit both diseases. 143 This, again, suggests a focus on myeloid-derived cells in patients with PH of any origin.
Therapy
Given the dearth of curative PH therapies, the benefit of exploring cytosolic DNA sensing as a viable pathway in disease augmentation lies in the variety of translational pharmaceutical therapies under investigation for targeting specifically DNA damage and release, proximal and distal to the cGAS/STING pathway. A full review of drugs that directly target cGAS/STING has been summarized recently, 144 therefore we include only those related pathways with previously established connection to DNA sensing and PH.
PARP inhibitors
As mentioned, PARP is an enzyme necessary for accurate DNA base excision repair. Inhibitors of PARP have shown promise clinically in treatment of BRCA positive breast and ovarian cancers, acting to induce a STING-dependent antitumor response and proliferative cell death; this occurs in a synergistic manner with combination immune checkpoint blockade. 145 The interaction is complex, however, with cGAS acting to suppress homologous recombination through the PARP-complex, promoting tumor growth. Thus, effects on cGAS signaling enable unfettered PARP activity allowing the protein to be an attractive target for oncologic treatment itself. This is an important concept related to potential PH treatment applications, as overexpression of PARP allows PAH smooth muscle cell proliferation and resistance to apoptosis with an induced return to homeostasis resulting in protection against PH. 146 Furthermore, PARP inhibitors can induce a STING-dependent immune response, due to accumulation of toxic DNA double-stranded break, inducing antitumor immunity complemented by concurrent upregulation of the immune checkpoint proteins programmed cell death protein 1 (PD-1) and programmed death-ligand 1 (PD-L1). 147 Thus, the STING pathway can potentially be utilized to exploit cellular addiction to both repair mechanisms (PARP) and quiescent re-programming. “Olaparib for PAH: a Multicenter Clinical Trial (OPTION)”, an open label Phase 1b clinical trial examining the use of the only orally available PARP1 inhibitor in patients with PAH will hopefully shed light on future advanced phase clinical trials in this area of study (ClinicalTrials.gov Identifier: NCT03782818).
PD-1/PD-L1 inhibitors
The latter is a reoccurring theme in the literature, whereby STING activation enhances antitumor immunity through induction of immune checkpoint protein expression, increasing susceptibility broadly to inhibitor blockade. 148 In particular, cGAS is known as the essential component for the antitumor effect of immune checkpoint blockade, wherein cGAS deficiency renders immune checkpoint inhibitors non-efficacious in a disease model of melanoma. 149 However, priming the STING pathway with a combination of cGAMP and anti-PD-L1 antibodies enhanced antitumor response greatly. This is especially provocative in light of our own group’s recent description of the necessary role for PD-L1 in development of Group 3 PH models of PH, 150 though caution must be exercised in patients with suspected autoimmune disease.
miRNA targets
In particular, hypoxia is known to influence miRNA transcription, increasing miR27 specifically in human pulmonary artery endothelial cells, inducing PH through downregulation of PPARγ.151 Relevant to cytosolic DNA sensing is that miR27 is suppressed in certain types of cancer, inhibiting STING signaling and blunting T lymphocyte response malignancy. 152 Although a potentially minor component, the findings do give further credence to study of miRNA signaling related to cGAS/STING activation, given the multitude of conceptual overlap between PH and cancer immunotherapy. 153
CD47 blockade
STING is ultimately necessary for facilitating CD8+ T cell mediated anti-proliferative effects, in particular in conjunction with CD47 blockade. 154 CD47 is a phagocytosis inhibiting receptor—the “don’t eat me” protein—that when absent increases phagocytic consumption; increased expression of CD47 is in turn linked to poor prognosis in cancer patients. Again, partly mediated though a coordinated cellular response with immune checkpoint expression, the response to enhanced CD47 expression occurs unsurprisingly in a dendritic cell-dependent manner, privileging mutational burdens in the tumor microenvironment through promotion of adaptive cell immunosuppression. 155 This is interesting in light of the fact that CD47 is known to have higher expression in the lungs of patients with PAH, with blockade of the protein preventing development of PH in hypoxia-exposed animals, 156 although no DNA-damage response pathway analysis was performed. Supporting empiric data demonstrated normalization of pulmonary pressures in CD47–/– mice, as well as ex vivo demonstration of decreased ET-1 production by human pulmonary artery endothelial cells treated with a neutralizing CD47 antibody. 157 Finally, in a model of sickle cell disease-associated PH severity of pulmonary vascular disease correlated with levels of CD47. 158 This study also demonstrated that mice with myeloid cell deficiency of CD47 were conferred protection against development of PH, associated with decrease ROS generation and a decreased DNA damage response. Thus, CD47 regulation through cytosolic DNA sensing in innate immune cells remains a plausible—and thought-provoking—novel method of treating the underlying pathophysiology predisposing to pulmonary vascular disease.
JAK/STAT inhibitors
With respect to interferonopathies, such as the discussed AGS and SAVI, JAK/STAT inhibitors have shown tremendous efficacy at symptom improvement in a limited number of patients with this rare form of autoimmune disease. Although one study has shown that treatment with immunobiologic agent baricitinib resulted in improved clinical outcomes in patients with SAVI, AGS, and Chronic Atypical Neutrophilic Dermatosis with Lipodystrophy and Elevated Temperature syndrome were improved, the authors did not report outcomes related to any change in pulmonary pressures as a result of treatment. 80
Regarding the PH literature on the subject, another JAK/STAT inhibitor (ruxolitinib) has been demonstrated to improve pulmonary pressures associated with myelofibrosis, 159 attributed primarily to decrease in circulating interferon mediated cytokines (IL-6, IL-8, IL-4, and TNFα). 160 Interestingly, the same group demonstrated that JAK inhibition acted primarily through downregulation of STAT3 activity, in PAH vascular lesions, imparting decreases in diseased endothelial cell survival, migration, and angiogenesis. 161 There is consequently a biologically plausible rationale for further exploration of this drug class in the treatment of patients with PH of many etiologies.
Caution must be exercised, however, with respect to lessons learned from studies regarding imatinib, a biologic agent with overlapping kinase inhibitor function. Though imatinib showed early promise in Phase II trial analysis, 162 when empirically tested as add-on therapy in patients with PAH (IMPRES; a randomized, double-blind, placebo-controlled clinical trial), serious adverse events were more common in the experimental-arm, with expected higher discontinuation rates, including an increased risk of subdural hematoma in patients receiving anticoagulation. 163 Unfortunately, these effects persisted upon long-term analysis, despite some derived benefit in right ventricular function being noted in those able to tolerate drug. 164 There remains hope, though, as despite the potential for off-target effects leading to development of PH in oncology patients receiving chemotherapy, 165 more recent work has described successful reversal of PH predisposing smooth muscle cell proliferative activity with use ruxolitinib, promoting ameliorative reversal effects on vessel remodeling.166,167
Future directions
Therapeutic development
Multiple promising immunotherapeutic drugs targeting cellular DNA sensing are under development, primarily in the field of cancer research. This includes cGAS/STING-directed treatments 168 that could also prove promising in reversal of pulmonary vascular disease. Specifically, there are numerous pathways shared between the DNA sensing apparatus and known PH-associated signaling pathways that could potentially still be exploited for novel therapies.
Absent in melanoma 2/IRF3
Inflammasomes are emerging as a viable target in PH-directed therapy.169,170 A necessary component for appropriate inflammasome response in cells is through absent in melanoma 2 (AIM2), a vital protein in bacterial and viral DNA liberation that thus directly participates upstream in STING activation and stabilization of IRF for interferon response element transcription. 171 AIM2 has likewise been implicated in smooth muscle cell migration related to development of atherosclerosis in Apoe–/– mice. 172 In a similar manner, IRF3 specifically has been implicated in human pulmonary artery endothelial cell migration in vascular regeneration related to PH pathology. 102 Thus, these proximal and distal elements of cell DNA handling may yet prove to yield targets regarding application to pulmonary vasculopathies.
TBK1
Of special note, IRF3 is phosphorylated by TBK1 in a final step prior to translocation to the nucleus with binding to the IRE. 173 However, the kinase undergoes regulation by several canonical inflammatory networks, including tonic inhibition of TBK1 downstream of STAT3, through differential phosphorylation of serine residue at position 754 in the transactivation domain of the intracellular signaling protein. 87 Thus, TBK1 can also function to inhibit interferon-signaling through suppression of cytokine IL-6 signaling. With the potential to finetune the response of such an important signature of cytokine and chemokine release, either up- or downregulating depending on post-translational modification, TBK1 represents a ripe area to be explored in models of PH.
Conclusions
Despite progress in study of PH related to DNA damage and signaling, many questions remain such as: What is the role of interferon-independent cGAS/STING signaling? How does cytosolic DNA sensing in non-immune cells in the lung contribute to disease pathogenesis? What is the contribution of classical genetic mutations predisposing to PAH in interferon potentiated disease? While rigorous study design and testing of translational pharmaceuticals are still necessary to safely bring any therapy from bench-to-bedside, our hope is that by taking advantage of current knowledge in sister fields of research (oncology and immunology, in particular), we can streamline delivery of possibly curative drugs to a currently undertreated population.
Of course, several of the connections between polynucleotide signaling and PH are ultimately inference at this stage. The relevance for continued exploration of DNA sensing is potentially bolstered, however, by examining further the initial cinematic conceit; “The Third Man” did not originate as a suspense-laden film noir, instead derived from Aristotle’s “Third Man Argument”. The discourse focuses on the infinite regress of forms whereby the description of X (in this case, X as a perfectly functioning pulmonary vascular bed) is self-predicated upon the existence of X, begging yet another description of X and X, ad infinitum. Such a paradox of endlessly defining the perfect form can be paralleled in the possible delay in bringing safe novel therapeutics applied to an incurable disease state, while awaiting the full “perfect” mechanistic understanding of disease. We simply cannot afford to wait.
Footnotes
Contributorship
A.J.B., A.P., H.G., C.R.M., and F.P. wrote the manuscript, and L.J. wrote and edited the manuscript.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the National Institute of Health (NIH) K08 HL144085, R01 HL142776, R01 HL142887 (A.J.B.), R01 AI110606, and R21 AI1322865 (L.J.).
