Abstract
Predisposing factors for the development of a pre-capillary component in pulmonary hypertension associated with left heart disease remain elusive. We report the case of a patient with persistent combined post-capillary and pre-capillary pulmonary hypertension after cardiac transplantation, in whom a rare
Pulmonary hypertension (PH) is common in left heart diseases (LHD-post-capillary PH) and negatively affects outcome.1,2 The trigger of PH-LHD is the passive increase in left-sided filling pressures.
2
However, a small subset of patients may present with significant pulmonary vascular disease defined as combined post-capillary and pre-capillary PH (CpcPH).1,2 The interindividual variability of this superimposed pre-capillary component suggests the presence of risk or protective factors, environmental and/or constitutive.
3
Although genetic factors have been identified in pulmonary arterial hypertension (PAH) as well as in some other forms of PH, genetic data in PH-LHD are scarce.3,4 One study uncovered the presence of 75 variants shared between PAH and CpcPH patients which are present in genes highly expressed in the lungs; however, there is no data on variants in PAH-associated genes in the subgroup of CpcPH patients.
5
We performed targeted gene panel sequencing in a small CpcPH patient cohort and report the case of a patient presenting with persistent CpcPH after cardiac transplantation, in whom a rare
Case description
A 64-year-old patient presented with heart failure with reduced ejection fraction and was diagnosed with nonischemic and nonvalvular dilated cardiomyopathy. He was of Northeast African origin and we had no access to relatives. He underwent his first right heart catheterization at our institution during a scheduled hospitalization for heart transplant evaluation (Table 1). According to guidelines, 1 he was diagnosed with CpcPH based on an elevated pulmonary artery wedge pressure (PAWP >15 mmHg), associated with a pulmonary vascular resistance (PVR) > 3 Wood units (WU) and a diastolic pulmonary gradient (DPG) ≥7 mmHg. He also presented with comorbidities including a metabolic syndrome and continuous positive airway pressure (CPAP)-treated obstructive sleep apnea. He had no history of smoking, no abnormalities on chest imaging, and a normal carbon monoxide diffusion coefficient. A ventilation/perfusion scintigraphy excluded pulmonary embolism. Based on these findings, group 3 or group 4 PH can be excluded.
Hemodynamic evolution and clinical characteristics.
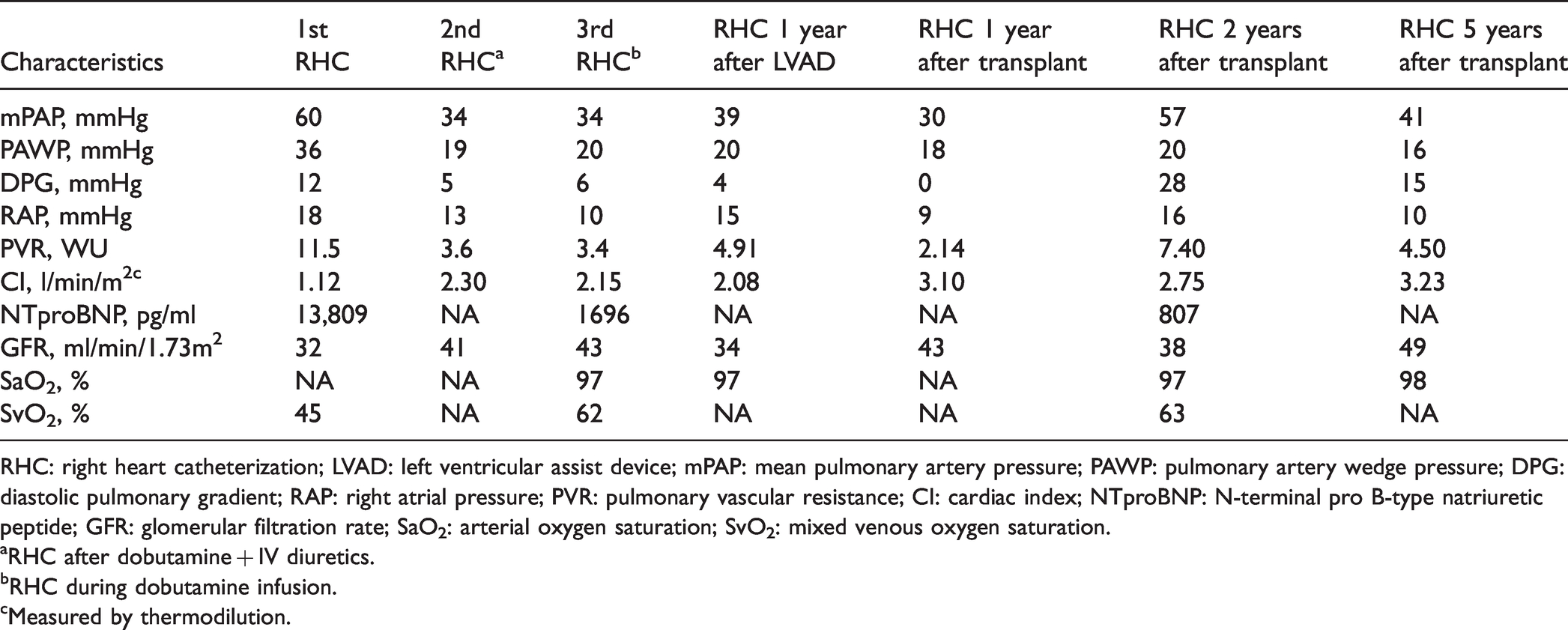
RHC: right heart catheterization; LVAD: left ventricular assist device; mPAP: mean pulmonary artery pressure; PAWP: pulmonary artery wedge pressure; DPG: diastolic pulmonary gradient; RAP: right atrial pressure; PVR: pulmonary vascular resistance; CI: cardiac index; NTproBNP: N-terminal pro B-type natriuretic peptide; GFR: glomerular filtration rate; SaO2: arterial oxygen saturation; SvO2: mixed venous oxygen saturation.
aRHC after dobutamine + IV diuretics.
bRHC during dobutamine infusion.
cMeasured by thermodilution.
His condition worsened until a left ventricular assist device (LVAD) was implanted as a bridge to transplantation. During surgery, he suffered from acute worsening of PH requiring treatment with inhaled nitric oxide. No PAH-approved pulmonary vasodilator was given, as per our standard procedures. One year later, the patient underwent heart transplantation. During the procedure, he suffered from acute right heart failure requiring temporary venoarterial extracorporeal membrane oxygenation. He received a standard immunosuppressive regimen consisting of prednisolone, mycophenolate mofetil, and ciclosporin. After transplantation, his condition greatly improved. However, a CpcPH presentation was observed two years after transplantation, which persists three years later. Of note, chronic treatment regimen remained unchanged. Repeated echocardiograms revealed a normal ejection fraction, no valvular disease, a dilatation of both atriums, and septal dyskinesia. There were no signs of allograft rejection on cardiac biopsies and no significant lesion on his coronary angiogram. There was also no new evidence of group 3 or 4 PH. The patient’s hemodynamic evolution is shown in Table 1.
With the approval of our institutional ethics committee and the patient’s written informed consent, we performed an in-house designed PAH gene panel consisting of eight genes (
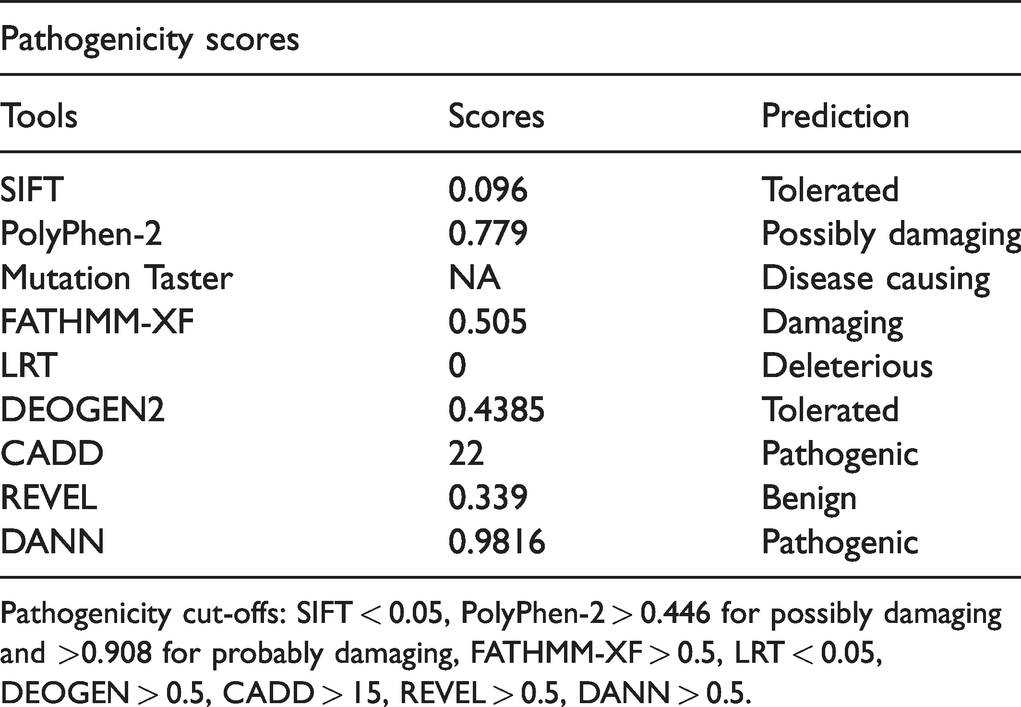
Pathogenicity cut-offs: SIFT < 0.05, PolyPhen-2 > 0.446 for possibly damaging and >0.908 for probably damaging, FATHMM-XF > 0.5, LRT < 0.05, DEOGEN > 0.5, CADD > 15, REVEL > 0.5, DANN > 0.5.
Using the above criteria, we found two nonsynonymous variants in
The
The
In the same study, we also recruited all the other CpcPH patients presenting with a DPG ≥7 mmHg and a PVR > 3 WU, because this group of patients was shown to present with more severe right heart dysfunction based on previous work by our team. 13 The patients’ clinical and hemodynamic characteristics are listed in Table 3. None of them had a positive family history. No variants were found among the five available patients after filtering using the criteria described above.
Characteristics of the other CpcPH patients who underwent genetic testing.
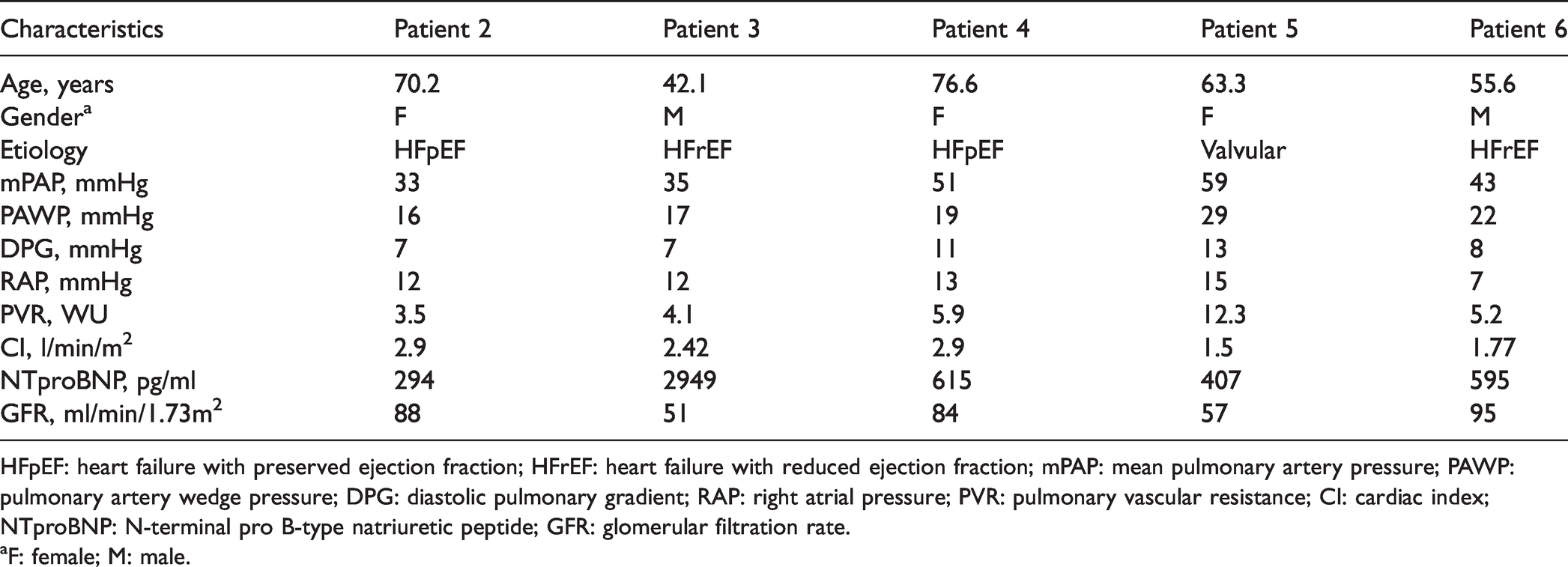
HFpEF: heart failure with preserved ejection fraction; HFrEF: heart failure with reduced ejection fraction; mPAP: mean pulmonary artery pressure; PAWP: pulmonary artery wedge pressure; DPG: diastolic pulmonary gradient; RAP: right atrial pressure; PVR: pulmonary vascular resistance; CI: cardiac index; NTproBNP: N-terminal pro B-type natriuretic peptide; GFR: glomerular filtration rate.
aF: female; M: male.
Discussion
The PH-LHD patient population is heterogeneous, and the factors leading to the development of a pre-capillary component are incompletely understood. The hemodynamic definition of the pre-capillary component in PH-LHD has been debated over time.
2
In the current PH guidelines, CpcPH is defined by a DPG ≥7 mmHg and/or a PVR > 3 WU.
1
However, our group recently showed that patients with high DPG
It is now well known that a genetic predisposition underlies some forms of PAH. The transforming growth factor-β superfamily signaling pathway is the main pathway implicated in PAH and
There are rare reports of underlying mutations or polymorphisms in other forms of PH such as CTEPH.
3
In a cohort of 49 CTEPH cases, 7 exonic missense variants were found in
To our knowledge, this is the first reported study of variants in known PAH-associated genes in a CpcPH patient cohort. We highlighted a rare
Our study has limitations inherent to the small sample size and the lack of a control population. This study is monocentric and took place in a tertiary referral center where only extreme phenotypes undergo genotyping. Also, this study has insufficient power to study frequent variants, potential new genes, or variations in noncoding DNA. Moreover, the fact the
In conclusion, we did not find causative variants with strong effects in PAH-associated genes in our small CpcPH patient cohort using a candidate gene-panel approach. However, we cannot exclude the hypothesis that rare PAH-associated variants could modulate the pre-capillary component in certain CpcPH patients. Studies in bigger patient cohorts using genome-wide genetic tests and a control population are needed.
Footnotes
Author contributions
JLV, AB, and MA designed the study. LC and AB recruited patients. LC and SC collected clinical data. All authors contributed substantially to the analysis and interpretation of the data. LC drafted the manuscript, and all authors revised it critically for important intellectual content. All authors approved the final version of the article.
Ethical approval
The Hospital-Faculty Ethics Committee of Hôpital Erasme approved this study (no. P2014/461 and P2018/639).
Guarantor
Antoine Bondue.
Acknowledgments
The authors wish to thank Smits G, Badoer C, Desmyter L, and Dewachter L for their help with data analysis and interpretation throughout the project and N.O. Stitziel for sharing unpublished data.
Declaration of Conflicting Interests
Outside the submitted work:
AB received consultancy fees from Amicus, Baeyer, Boehringer Ingelheim, Sanofi, Pfizer, and Alnylam; speaker fees from Pfizer, Sanofi, and Alnylam; and reports travel grants from Abbott, Pfizer, and Takeda. JLV received consultancy fees from Actelion, Bayer, Bial Portela, PhaseBio, Respira Therapeutics, United Therapeutics, and SoniVie and reports speaker fees and travel grants from Actelion, Acceleron, Bayer, Merck, Novartis, and United Therapeutics. LC reports travel grants from Sanofi and Pfizer. SC received an ERS PAH Short-Term Research Training Fellowship (STRTF 2014-5264) supported by an unrestricted grant by GSK and the international grant “Cesare Bartorelli” in 2014 funded by the Italian Society of Hypertension. CD and MA have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fonds Erasme pour la Recherche Médicale (LC), by the Foundation for Cardiac Surgery (LC and AB), and by the FNRS (grant number J.0011.19F) (AB). JLV and AB are co-holders of an Actelion chair for Research in Pulmonary Hypertension.
