Abstract
Pulmonary hypertension is characterized histologically by intimal and medial thickening in the small pulmonary arteries, eventually resulting in vascular “pruning.” Computed tomography (CT)-based quantification of pruning is associated with clinical measures of pulmonary hypertension, but it is not established whether CT-based pruning correlates with histologic arterial remodeling. Our sample consisted of 138 patients who underwent resection for early-stage lung adenocarcinoma. From histologic sections, we identified small pulmonary arteries and measured the relative area comprising the intima and media (VWA%), with higher VWA% representing greater histologic remodeling. From pre-operative CTs, we used image analysis algorithms to calculate the small vessel volume fraction (BV5/TBV) as a CT-based indicator of pruning (lower BV5/TBV represents greater pruning). We investigated relationships of CT pruning and histologic remodeling using Pearson correlation, simple linear regression, and multivariable regression with adjustment for age, sex, height, weight, smoking status, and total pack-years. We also tested for effect modification by sex and smoking status. In primary models, more severe CT pruning was associated with greater histologic remodeling. The Pearson correlation coefficient between BV5/TBV and VWA% was –0.41, and in linear regression models, VWA% was 3.13% higher (95% CI: 1.95–4.31%, p < 0.0001) per standard deviation lower BV5/TBV. This association persisted after multivariable adjustment. We found no evidence that these relationships differed by sex or smoking status. Among individuals who underwent resection for lung adenocarcinoma, more severe CT-based vascular pruning was associated with greater histologic arterial remodeling. These findings suggest CT imaging may be a non-invasive indicator of pulmonary vascular pathology.
Introduction
Pulmonary hypertension (PH) is a heterogeneous disease of the pulmonary vessels, which may be idiopathic, or associated with a broad range of clinical conditions including autoimmune, cardiovascular, and pulmonary disease. 1 Regardless of etiology, PH is characterized histologically by hypertrophy and thickening of the intima and media of the muscularized pulmonary arterioles. 2 These pathologic changes result in progressive luminal narrowing and loss of these smaller distal vessels and leads to the vascular “pruning” that is an angiographic hallmark of PH.2–4
While small vessel remodeling directly contributes to higher vascular resistance and the hemodynamic consequences of PH, these changes are challenging to identify non-invasively, and definitive evaluation for PH currently requires invasive procedures such as right heart catheterization (or rarely, histopathologic examination).2,5 Using image analysis of computed tomographic (CT) imaging, the volume of blood distribution in pulmonary vessels of varying size can be quantified. 6 A relative reduction in the volume of the smaller vessels above the imaging resolution, described as “pruning” on CT, has been used as a radiographic surrogate of the angiographic and histologic changes seen in PH. More severe pruning on CT has been linked to poorer PH-related clinical outcomes in individuals with COPD, including higher pulmonary arterial pressure on catheterization, reduced right ventricular function on cardiac MRI, and greater risk of death.7–9 We recently found that CT pruning also predicted higher all-cause mortality in a general, community-based cohort not selected based on cardiac or pulmonary disease. 10
Despite these clinical associations, it is not clear that CT-based quantification of pruning reflects histologic changes in the pulmonary vessels, particularly in terms of structural changes in the vascular wall. In one prior study of 18 patients, mostly smokers with chronic obstructive pulmonary disease (COPD) who underwent resection for lung cancer, lower small vessel volumes on CT were associated with a reduction in the histological cross-sectional areal density of the pulmonary arteries. 11 However, we previously found that tobacco exposure was unexpectedly associated with higher small vessel pulmonary vascular volumes on CT, 12 despite known histologic evidence of remodeling in smokers even without COPD. 13 As a result, it remains poorly understood whether radiographic differences in the pulmonary vessels are associated with underlying histologic vascular remodeling. To address this knowledge gap, we investigated associations of CT pruning with morphometric changes in the vascular wall on histology in a cohort of patients who underwent surgical biopsy of early-stage lung adenocarcinoma.
Methods
Study population
The study population consists of 138 patients at the Beth Israel Deaconess Medical Center in Boston, MA, who underwent wedge resection or lobectomy for early-stage adenocarcinoma of the lung (i.e. without regional lymph node involvement or distant metastases) between 2005 and 2018. We excluded patients for whom pre-operative CT imaging, spirometry, or original histology slides were not available. This study was conducted in accordance with the amended Declaration of Helsinki. The Institutional Review Board of the Dana-Farber/Harvard Cancer Center (protocol 17-110) approved this study. Informed consent was waived for this study.
Assessment of histologic pulmonary vascular remodeling
Per institutional histopathologic grossing protocols, at least one tissue section of uninvolved/non-neoplastic lung parenchyma distant from the tumor mass is evaluated for each lung cancer resection specimen; we included only these “uninvolved” sections in this study. Using a digital pathology slide scanner (Aperio, Leica Biosystems, Buffalo Grove, IL), we converted these original hematoxylin and eosin stained histology slides to digital format at 40× magnification. The original slide labels were masked with a study label containing only a unique participant identifier. Once digitized, a single investigator systematically examined the slides (A.J.S.) to manually identify all small pulmonary arteries appropriate for measurement, with guidance from an experienced thoracic anatomic pathologist (P.A.V.). We selected vessels that met the following inclusion criteria: (1) <1.0 mm in diameter 14 with a complete and intact external elastic lamina, (2) patent with no evidence of vascular thrombosis, and (3) properly oriented with a long:short axis diameter <2 to minimize obliquely sectioned or substantially distorted vessels. To minimize misclassification of small pulmonary veins as arteries, we selected muscularized vessels with identifiable elastic lamina in proximity to a respiratory bronchiole. Once the vessels had been manually identified, we used a commercial image analysis software package 15 (MIPAR, Columbus, OH), which employs a semi-automated deep learning-based algorithm, to measure the total vascular area of the small pulmonary artery (i.e. the total cross-sectional area of the vessel including the vascular lumen, endothelial lining, and smooth muscle layers encompassed by the external elastic lamina). We also measured the area of only the smooth muscle layers (i.e. the intima and media), representing the total vascular area excluding the vascular lumen (Fig. 1, shaded area). Given that intimal and medial hypertrophy are one of the characteristic pathologic changes of pulmonary vascular disease, we calculated the relative vessel wall area (VWA%), which was defined as the area of the intima/media divided by the total vascular area (i.e. shaded area divided by the combined shaded plus striped areas from Fig. 1). Higher VWA% values, therefore, indicate greater histologic vascular remodeling. We calculated VWA% for each vessel and averaged them together to generate a mean VWA% for each participant. This approach is similar to those used by prior studies of vascular morphometry.13,16,17
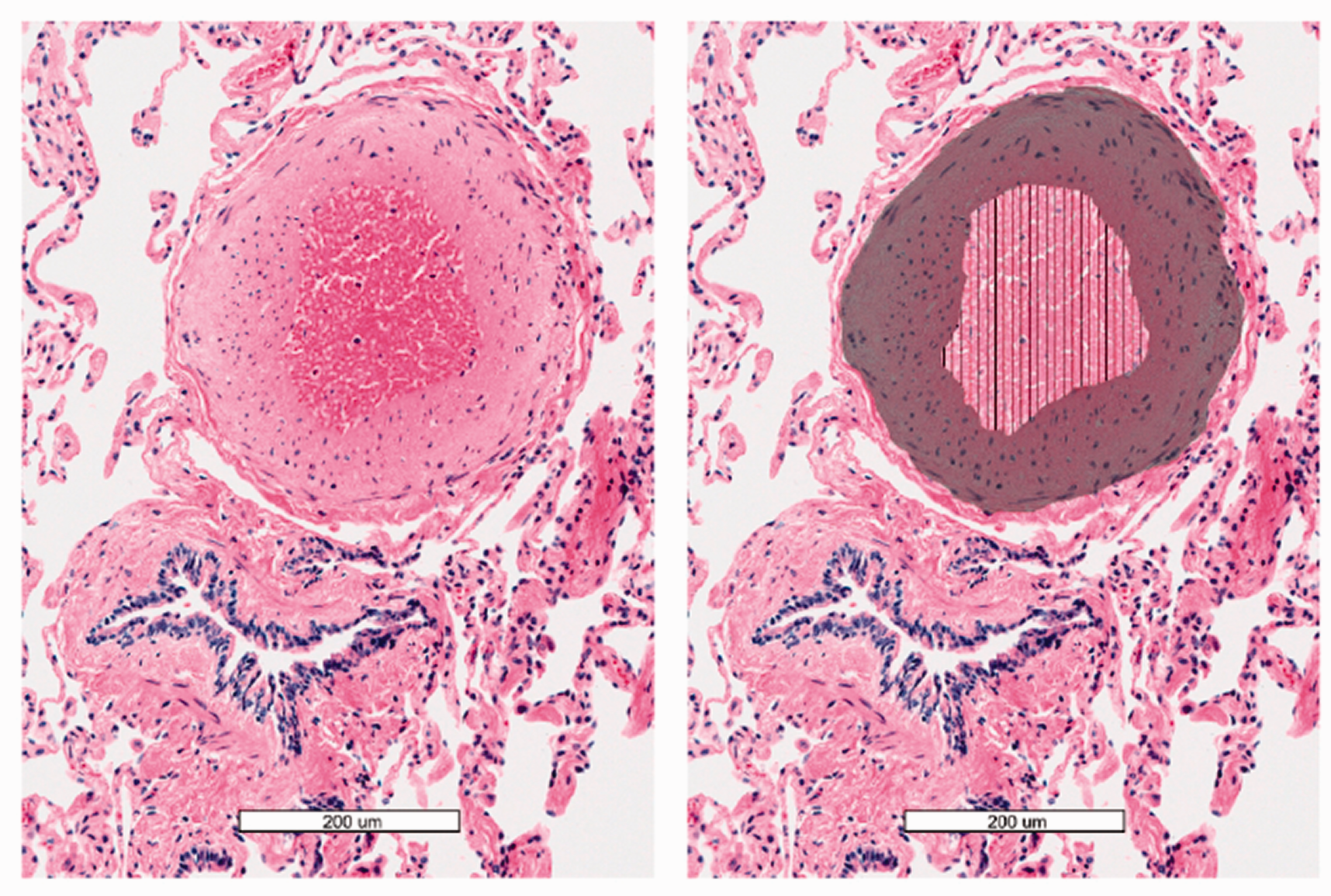
Example of small pulmonary artery, adjacent to respiratory bronchiole (left). Shaded area represents the intima and media layers of the vessel, while striped area represents the vascular lumen (right). VWA% is defined as: shaded area/(shaded + striped area).
CT pulmonary vascular assessment
All participants in our cohort had a pre-operative, thin-slice (≤1.25 mm section thickness) chest CT examination that had been obtained for clinical indications. All CTs were performed without intravenous contrast, at end-inspiration, and in the supine position. From clinical chest CTs, Digital Imaging and Communications in Medicine (DICOM) data were extracted, and these data were anonymized at the point of extraction using the Chest Imaging Platform (www.chestimagingplatform.org) and re-labeled with unique study identifiers. Vascular image analysis was conducted in the Applied Chest Imaging Laboratory at Brigham and Women’s Hospital using Chest Imaging Platform-based software as previously described. 6 After segmenting the lung field and lobes, 18 a scale-space particle approach was used to detect intraparenchymal pulmonary vessels within the limits of CT resolution (Fig. 2). 19 Scale-space particles allow for efficient identification of vascular features, which can be described by measuring second-order spatial derivatives. 6 The method is initialized by placing particle points using a binary mask, which provides initial vessel probability based on the Strain energy method. 20 The method then proceeds towards certain hessian eigenvalues based on reported thresholds for our features of interest (i.e. vessels).6,8 Once optimized, the scale strength of each particle point is a surrogate for the size of the vessel at that location, allowing for volumetric vascular quantification.6,19,21 The volume of the vasculature was computed as a function of the cross-sectional area of the vessels. Measurements of the total volume of all intraparenchymal vessels (TBV) and of the small vessels (defined by cross-sectional area <5 mm2, BV5) were computed from the vascular volume profiles. Vascular measures for the entire pulmonary vascular tree and the specific lobe of the lung from which the surgical biopsy was obtained (i.e. the same lobe used for histologic assessment of pulmonary vascular remodeling). These measures include both arterial and venous volumes. The small vessel fraction (BV5/TBV) represents the relative distribution of blood vessel volume in the smallest, most peripheral blood vessels detectable by CT, with lower BV5/TBV values indicating more severe radiographic vascular pruning.6,12,22,23
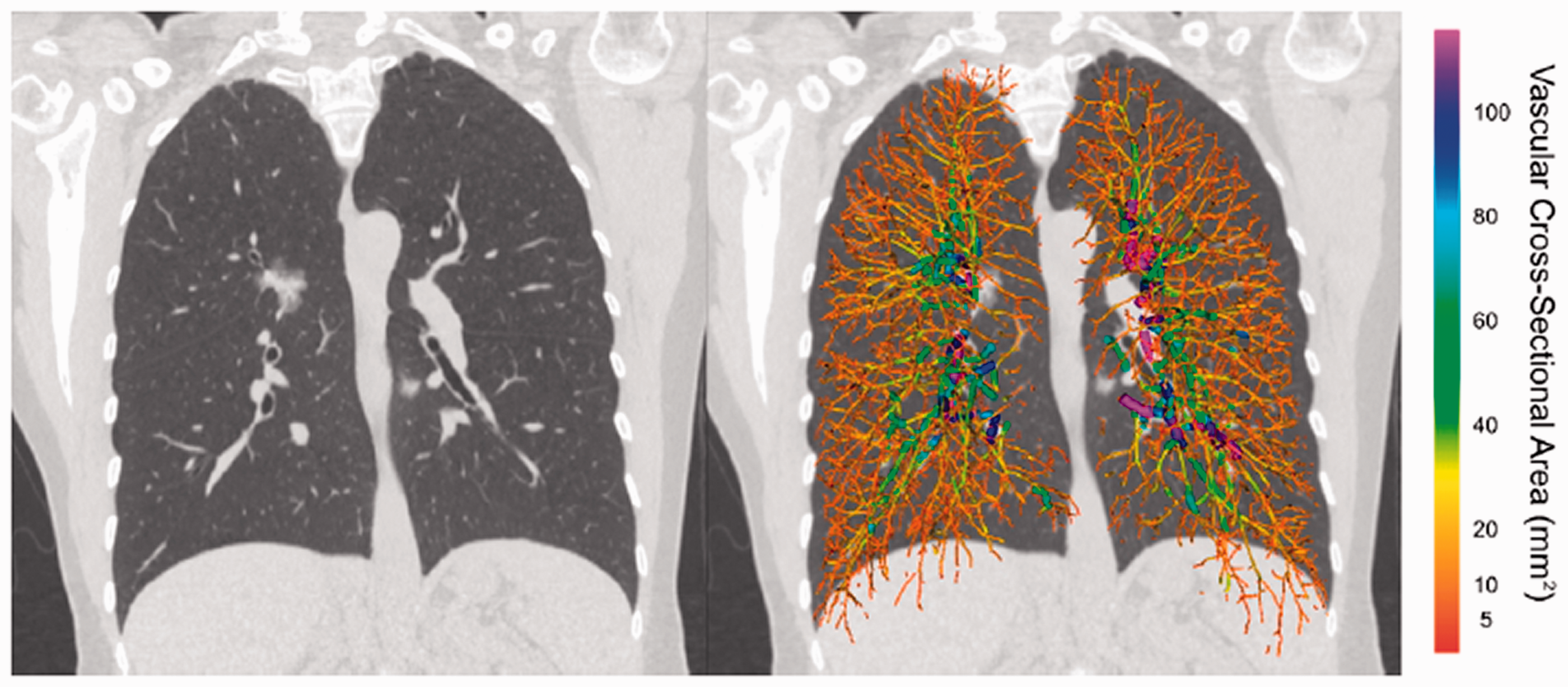
(Left) Coronal CT section demonstrating a right perihilar mixed-density pulmonary nodule, found to be lepidic-predominant adenocarcinoma. (Right) Overlaid volumetric reconstruction of the pulmonary vascular tree. Vessels color-coded by size. In this participant, BV5 = 89.0 mL, TBV = 162.7 mL, and BV5/TBV = 54.7%.
Statistical methods
For our primary analyses, we investigated the relationship between CT vascular pruning (both whole-lung BV5/TBV and lobe-specific BV5/TBV) and histologic remodeling (VWA%) using Pearson correlation and simple linear regression. We then constructed multivariable linear regression models to investigate the association of CT pruning and VWA% with adjustment for age (at the time of surgery), sex, height, weight, smoking status, and total pack-years of cigarette exposure. These covariates were selected a priori, given the known associations with CT vascular metrics and/or histologic remodeling.
In secondary analyses, we performed additional adjustment for FEV1, DLCO, histologic evidence of fibrosis or emphysema (given known relationships between lung disease and pulmonary vascular disease, particularly in populations enriched for smoking), and the number of vessels identified per participant (as an estimate of vascular density). We also added interaction terms to our multivariable regression models to test for evidence of differential associations of histologic remodeling and CT pruning based on sex and smoking status. In exploratory analyses, we also examined if our primary associations differed on the basis of a clinical indicator of pulmonary vascular disease. While invasive hemodynamic assessment was not available for our sample (and therefore a definitive determination of PH cannot be made), 73 participants (53%) had a pre-operative transthoracic echocardiogram (TTE) with Doppler-based estimation of pulmonary artery systolic pressure (PASP) within three years of the index CT examination. Based on guidelines which suggest that PH is less likely in those with PASP < 36 mmHg (assuming right atrial pressure of 5 mmHg), 1 we defined echocardiographic evidence of PH as PASP>36 mmHg and included interaction terms in our primary models to examine if associations of CT pruning and histologic remodeling differed by presence of echocardiographic PH. For linear regression, differences in VWA% are reported with 95% confidence intervals and expressed per standard deviation lower BV5/TBV. For interaction terms, a two-sided p-value of <0.10 for the Wald test was considered statistically significant effect modification. Statistical analyses were performed using SAS 9.4 (SAS Institute, Inc., Cary, NC).
Results
Study participant characteristics
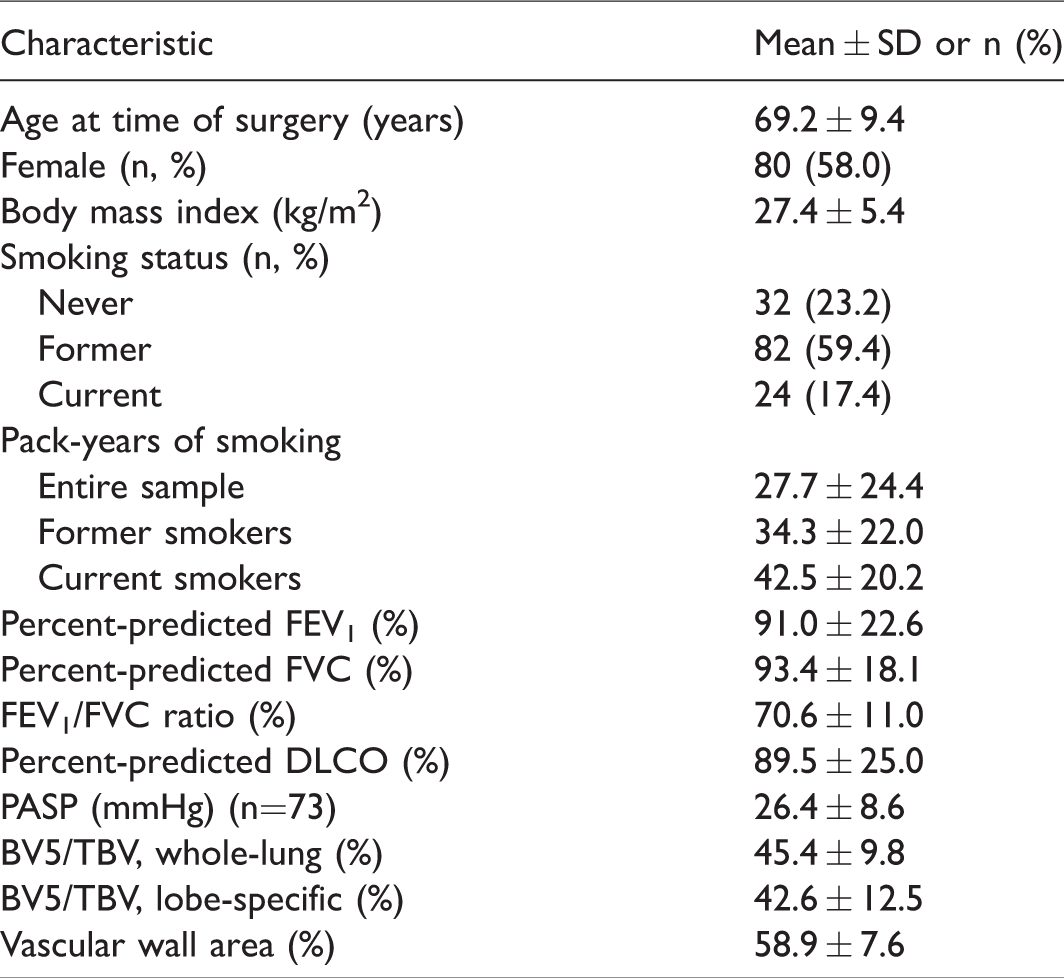
Details of the study cohort (n = 138) are provided in Table 1. The median interval from the CT scan to the date of surgery was 45 days (interquartile range, 37 days). The sample consisted of 80 female participants (58.0%), with a mean age of 69.2 ± 9.4 years. Most of the cohort members were either former or active smokers (59.4% and 17.4%, respectively). Roughly a quarter of participants had been clinically diagnosed with COPD by a pulmonologist (23.2%), while 27 participants (19.6%) met criteria for moderate-to-severe airflow obstruction on spirometry. Average lung function was mildly impaired, with percent-predicted FEV1, FVC, and DLCO of 91.0 ± 22.6%, 93.4 ± 18.1%, 89.5 ± 25.0%, respectively. In the subset of 73 individuals with TTE, the mean PASP was 26.4 ± 8.6 mmHg and seven (9.6%) met echocardiographic criteria for PH. A total of 731 vessels were included for histologic analysis (average of 5.3 ± 1.2 vessels per participant), and VWA% was 58.9 ± 7.6%. Mean BV5/TBV for the whole-lung was 45.4 ± 9.8%, while lobe-specific BV5/TBV was 42.6 ± 12.5%. The distributions of BV5/TBV, lobe-specific BV5/TBV, and VWA% are shown in Supplemental Fig. 1. Overall, values for total BV5/TBV and lobe-specific BV5/TBV were highly correlated (r = 0.82, p < 0.0001).
Characteristics of study participants (n=138).
Associations of CT pruning and histologic remodeling
In primary analyses, we found that more severe global CT pruning was associated with greater histologic vascular remodeling as defined by relative wall area. The Pearson correlation coefficient between BV5/TBV and VWA% was –0.41 (Fig. 3), and in linear regression models, VWA% was 3.13% higher (95% CI: 1.95–4.31%, p < 0.0001) per standard deviation lower BV5/TBV (Table 2). This association persisted after additional adjustment for age, sex, height, weight, smoking status, and total pack-years. We found similar associations of lower lobe-specific BV5/TBV with higher VWA% (Fig. 3).
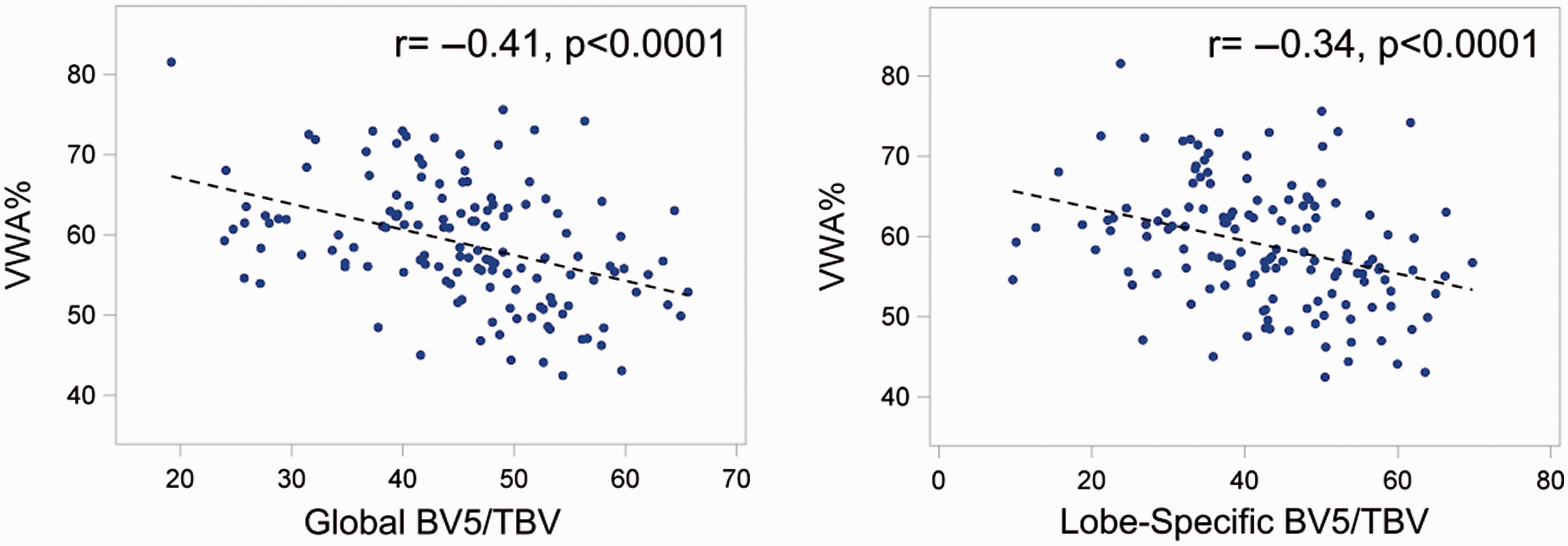
Correlation of vascular pruning on CT (BV5/TBV) with histologic vascular remodeling (VWA%).
Associations of CT pruning and histologic remodeling.
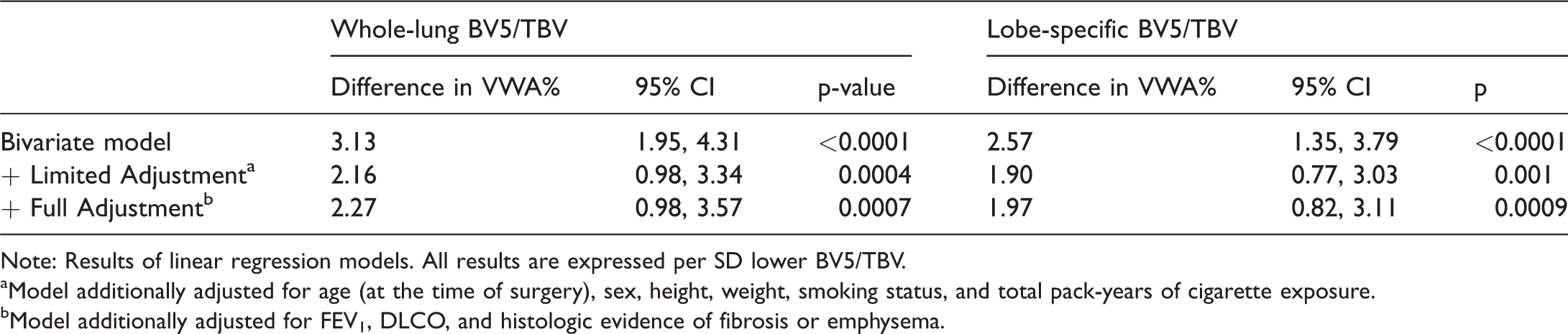
Note: Results of linear regression models. All results are expressed per SD lower BV5/TBV.
aModel additionally adjusted for age (at the time of surgery), sex, height, weight, smoking status, and total pack-years of cigarette exposure.
bModel additionally adjusted for FEV1, DLCO, and histologic evidence of fibrosis or emphysema.
In secondary analyses, we found that additional adjustment for FEV1, DLCO, and histologic fibrosis or emphysema did not substantially alter our primary findings: in fully adjusted models, VWA% was 2.27% higher (95% CI: 0.98–3.57%, p = 0.0007) per standard deviation lower BV5/TBV (Table 2). We found no evidence that the association of CT pruning and histologic remodeling was modified by sex (pinteraction=0.28) or smoking status (pinteraction = 0.72). For example, in stratified analyses, the correlation coefficient between BV5/TBV and VWA% was –0.37 in never-smokers and –0.46 in ever-smokers. In exploratory analyses, in the subgroup of 73 participants with pre-operative TTE data, we found no statistical evidence that the association of CT pruning and histologic remodeling was modified by echocardiographic PH (pinteraction = 0.23). In subsequent stratified analyses, among individuals with (n = 7) and without PH (n = 66), the correlation coefficients between BV5/TBV and VWA% were –0.93 and –0.37, respectively.
Discussion
In this cohort of patients who underwent surgical resection of early-stage lung adenocarcinoma, we found that more severe vascular pruning on CT was associated with morphometric evidence of arterial remodeling in the adjacent uninvolved lung parenchyma on histology. These associations were robust to adjustment for demographic factors, smoking history, and measures of lung disease, and we found no evidence that these relationships differed based on smoking status. Our findings suggest that non-invasive quantification of pulmonary vascular pruning by CT imaging may reflect remodeling of the vascular wall at the histologic level.
PH is a devastating disease that is associated with substantial morbidity and mortality regardless of etiology. Early detection of pulmonary vascular disease may improve prognostication and alter the treatment approach, but early recognition of this disease remains challenging. 5 While histopathologic analysis of biopsy specimens can provide a direct assessment of pulmonary vascular remodeling and right heart catheterization is used to define PH hemodynamically, these are invasive tests that are not practical for detecting PH in its early, clinically silent phase.
Quantitative vascular “pruning” derived from CT imaging may be a non-invasive method of identifying changes in the pulmonary vessels linked to clinically significant PH. For example, more severe pruning on CT has been associated with hemodynamic consequences of PH, including higher pulmonary artery pressures on right heart catheterization and right ventricular dysfunction/remodeling on cardiac MRI in individuals with substantial COPD.7,8 Recently, in both a COPD-specific and a more general population-based cohort, more severe CT pruning at baseline was associated with all-cause mortality during follow-up.10,24 These findings suggest that CT pruning has the potential to be a non-invasive indicator of the presence and severity of pulmonary vascular disease.
While histologic remodeling of the small pulmonary vessels is best characterized in primary pulmonary arterial hypertension, 25 similar vascular changes are consistently observed regardless of PH etiology. Morphometric changes of the vascular walls including medial hypertrophy and intimal fibrosis/thickening have been described in PH due to heart failure, 26 lung disease (including COPD13,27 and idiopathic pulmonary fibrosis 28 ), and chronic thromboembolic pulmonary hypertension, 29 among others. These changes eventually cause narrowing, occlusion, and functional loss of the small pulmonary vessels (i.e. pruning) and culminate in the progressive hemodynamic and clinical consequences of PH.2,5 In addition, thickening of the intima and media is a known feature of systemic arteriopathy in general (particularly for carotid disease). 30
However, although the conceptual basis for CT pruning is clear, it has not been well determined that radiographic metrics directly reflect histologic changes in the pulmonary vessels. This method of CT image analysis can resolve vessels of cross-sectional area up to ∼1 mm2. Therefore, changes in the small pulmonary arteries that are most affected histologically in PH are unlikely to be directly visualized by this technique. However, a prior study by Rahaghi et al. found that differences in small vessel size on CT were associated with histologic alterations. 11 Their study population, which consisted of patients who also underwent surgical resection for lung cancer, was comprised of two separate cohorts: 18 individuals who underwent three-dimensional (i.e. volumetric) CT vascular assessment and 19 who underwent two-dimensional (i.e., areal) CT assessment. Their primary finding was that small vessel volume (normalized for lung volume) was associated with the total cross-sectional area of small pulmonary vessels (as a percent of the area of the field of view) on biopsy specimens, indicating that a reduction in small vessel volume on CT reflects vascular density loss at the histologic level. The results of our study, which found that CT-based pruning was associated with thickening of the walls of the small pulmonary vessels and a resulting loss in luminal cross-sectional area, are likely to represent an analogous and complementary pathologic process to the overall loss of vascular cross-sectional area found by Rahaghi et al.
We are aware of only one other study to examine the relationship between volumetric CT measures of the pulmonary vasculature and histologic changes in the pulmonary arterioles, with the Rahaghi study being the other. 11 While not directly relevant to histologic changes, Zhai et al. also found that CT-based methods of quantifying pulmonary vascular morphology were able to accurately capture pulmonary vessels using a 3D-printed vascular phantom which included vessels as small as 0.2 mm (200 µm) in diameter. 31 Our study’s strengths include a relatively large cohort size, which is one of the largest to investigate the correlations of radiographic and histologic pulmonary vascular morphology. In addition, our secondary models were able to account for demographic factors, smoking history, and lung disease in our analyses. Importantly, our sample also represents a somewhat different population than the Rahaghi study, which consisted mostly of smokers with more severe COPD. For example, in their three-dimensional/volumetric cohort, 13/18 participants (72%) were clinically diagnosed with COPD and had a median percent-predicted FEV1 of 65% (compared to 23% with COPD in our study, with a normal average percent-predicted FEV1 of 91% in our study). 11 We also utilized an innovative method of vascular image analysis, which allowed for a volumetric assessment of the CTs for all participants, and the use of both lobe-specific and whole-lung vascular data. Finally, we used a semi-automated histologic analysis method to measure the entire two-dimensional area of the vascular wall, which may allow for a more detailed and comprehensive assessment of remodeling compared to a one-dimensional measurement of wall thickness alone.
Our study has several limitations. The CT data utilized for our vascular analysis were clinically acquired over a period spanning 14 years (2005–2018) using different model CT scanners, although our image analysis method controls for reconstruction kernels to minimize any potential effects related to these differences. Also, remodeling identified on histology may not represent vasculopathy present in other parts of the lung that were not sampled on biopsy. However, we believe that our ability to also examine CT vascular pruning specific to the resected lobe reassures that this was not a major source of error in our results. The histologic samples used in our study were not processed specifically to avoid tissue contraction, although all samples were fixed and processed in the same standard method per our institutional protocols. In addition, our selection for morphometric assessment included criteria to exclude substantially distorted or misoriented vessels, which we believe mitigates this source of measurement artifact to some degree. Specific vascular staining was not performed on our histologic samples and we are thus unable to evaluate changes in the media or intima separately; however, given that both changes in the intima and media have been reported in prior studies of pulmonary vascular disease, 2 we believe that our analysis, which reflects the combined changes in both layers, remains of interest. Finally, while histologic remodeling is well documented in all major categories of PH, we do not have invasive hemodynamic data for our sample and we are unable to definitively determine the extent to which the participants in our study are affected by PH – limiting our ability to draw firm conclusions about whether the CT-histologic associations observed in this study are relevant in those with clinical PH. However, we performed exploratory examinations in a subgroup of our cohort with echocardiography, which notably revealed that the correlation of BV5/TBV and VWA% was much stronger in those with evidence of PH (–0.93, compared to –0.41 for the sample as a whole). While limited by a very small sample size, these additional analyses provide some reassurance that the link between radiographic vascular pruning and histologic remodeling may be relevant to hemodynamically significant pulmonary vascular disease, although additional research in larger and better defined cohorts is necessary to verify this finding.
Overall, our study’s findings suggest that CT pruning may indicate pathologic changes in the small pulmonary vessels, even in those without a substantial burden of lung disease or exposure to tobacco smoke. While the presence of PH conclusively predicts poor outcomes regardless of etiology, there is no effective, non-invasive, and widely available method of screening for this deadly condition.5,32 Doppler echocardiography has long been considered the initial test of choice for non-invasive investigation of suspected pulmonary hypertension due to its convenience, lack of radiation exposure, and its ability to assess multiple hemodynamic and structural elements related to PH.33,34 However, echocardiographic assessment of PH is known to be technically challenging and operator dependent, with reproducibility and reliability that may vary from one operator to another.5,34,35 A recent meta-analysis of 27 studies evaluating echocardiographic measures of PH against gold-standard right heart catheterization demonstrated a pooled sensitivity of 85% and specificity of 71%, which was inferior to the performance of cardiac MRI (92% and 86%, respectively). 33 Cardiac MRI offers distinct advantages, as it does not require radiation exposure, is not operator dependent, provides excellent spatial resolution of right ventricular morphology, and correlates well with invasive assessment of PH.33,34,36 However, cardiac MRI is not yet widely available outside of specialized centers, is relatively costly, and cannot be utilized in patients with magnetic objects, which limit its use as a screening tool for PH.34,36,37
In comparison, CT imaging is already widely obtained when evaluating individuals with cardiac or respiratory symptoms, and our method of quantifying pruning on CT can be applied retrospectively to these data. Our findings may therefore support the application of image analysis to existing images as a practical method of identifying patients with early pathologic changes in the pulmonary vessels without requiring any additional testing or radiation exposure. However, given the well-recognized risks of radiation exposure due to CT scans, 38 further research is necessary before CT pruning can be recommended for the primary purpose of screening for pulmonary vasculopathy. Among non-invasive tools, a particular strength of CT-based modalities of vascular quantification is that CT allows for global assessment of the pulmonary vascular tree, and thus may potentially be superior in capturing earlier vasculopathy. Tools such as echocardiography, cardiac MRI, and even right heart catheterization do not directly visualize abnormalities within the pulmonary vessels, but rather detect downstream hemodynamic consequences of vascular loss, which occurs only after much of the pulmonary circulation has already been occluded. 5 Our finding that the association of radiographic pruning and histologic remodeling was similar for global and lobe-specific CT measures provides additional reassurance that global vascular assessment reflects vascular changes observed at a single site. Finally, this method of quantifying CT pruning is reproducible 6 and can detect vascular changes that occur after surgical or medical therapy.39,40 Future research is necessary to determine the role that CT pruning may have in screening and prognostication for those with PH, particularly in its early stages. This may include the integration of multiple imaging modalities, 41 as studies have demonstrated that combining CT-based measures of pulmonary hypertension with echocardiography, for example, may be superior to either method in isolation.42,43
In conclusion, in this study of individuals who underwent resection for early-stage lung adenocarcinoma, more severe pulmonary vascular pruning on CT was associated with greater small vessel pulmonary arterial remodeling on histology. These findings provide additional insight into the relationship between the pulmonary vessels’ radiographic appearance and underlying histologic vasculopathy and suggest CT imaging may be a non-invasive indicator of pulmonary vascular disease.
Supplemental Material
sj-pdf-1-pul-10.1177_20458940211061284 - Supplemental material for Vascular remodeling of the small pulmonary arteries and measures of vascular pruning on computed tomography
Supplemental material, sj-pdf-1-pul-10.1177_20458940211061284 for Vascular remodeling of the small pulmonary arteries and measures of vascular pruning on computed tomography by Andrew J. Synn, Constance De Margerie-Mellon, Sun Young Jeong, Farbod N. Rahaghi, Iny Jhun, George R. Washko, Raúl San José Estépar, Alexander A. Bankier, Murray A. Mittleman, Paul A. VanderLaan and Mary B. Rice in Pulmonary Circulation
Supplemental Material
sj-pdf-2-pul-10.1177_20458940211061284 - Supplemental material for Vascular remodeling of the small pulmonary arteries and measures of vascular pruning on computed tomography
Supplemental material, sj-pdf-2-pul-10.1177_20458940211061284 for Vascular remodeling of the small pulmonary arteries and measures of vascular pruning on computed tomography by Andrew J. Synn, Constance De Margerie-Mellon, Sun Young Jeong, Farbod N. Rahaghi, Iny Jhun, George R. Washko, Raúl San José Estépar, Alexander A. Bankier, Murray A. Mittleman, Paul A. VanderLaan and Mary B. Rice in Pulmonary Circulation
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Authors’ contribution
AJS, MAM, and MBR designed the study with contributions from and the approval of all the other co-authors. AJS performed all statistical analyses with assistance and oversight from MBR. CDMM and AAB assisted in the acquisition and interpretation of the radiographic data. SYJ assisted in data collection and statistical analysis. PAV and IJ provided expertise regarding analysis of histologic remodeling and interpretation of these results. GRW, RSJE, and FNR contributed to radiographic image analysis and assisted in the interpretation of these results. All authors contributed to and approved the final version of the article.
Ethical approval
The Institutional Review Board of the Dana-Farber/Harvard Cancer Center (protocol 17-110) approved this study. Informed consent was waived for this study.
Guarantor
AJS.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by grants to Dr. Synn from the American Lung Association (CA-626548) and the National Heart, Lung, And Blood Institute (F32 HL143819). Rahaghi is supported by the NIH (grant 1K23HL136905). Washko is supported by the NIH (grants R01 HL122464 and R01 HL116473), and reports grants and other support from Boehringer Ingelheim, Genentech, Quantitative Imaging Solutions, PulmonX, Regeneron, ModoSpira, BTG Interventional Medicine, Janssen Pharmaceuticals, Toshiba, GlaxoSmithKline, outside the submitted work. Dr. Washko's spouse works for Biogen which is focused on developing therapies for fibrotic lung disease. San José Estépar is supported by the NIH (grants 1R01 HL116931 and R01 HL116473), and reports personal fees from Toshiba, Boehringer Ingelheim, Eolo Medical, and Leuko Labs, outside the submitted work. He is also a founder and co-owner of Quantitative Imaging Solutions which is a company that provides image based consulting and develops software to enable data sharing. Bankier reports personal fees from Spiration, Daiichi, CRICO, and Elsevier, outside the submitted work. VanderLaan reports consulting fees from Gala Therapeutics, Caris Life Sciences, and Intuitive Surgical, outside the submitted work. Mittleman reports grants from the NIH, US EPA, during the conduct of the study; grants from PCORI, Kellogg Foundation, and US National Institute of Justice, outside the submitted work. Rice is supported by the National Institute for Environmental Health Sciences (grant K23 ES026204), the American Thoracic Society Foundation, and the American Lung Association.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
