Abstract
Objective
Statins, a class of 3-hydroxy-3 methyl-glutaryl-coenzyme A reductase inhibitors, are widely used for the treatment of atherosclerosis. Less is known about the role of statins in the treatment of vascular complication in systemic sclerosis (SSc). We therefore performed a short-term interventional study with simvastatin in patients with the diffuse variant of SSc and normal lipid profiles.
Methods
Twenty-five patients with diffuse SSc were enrolled and received simvastatin at a daily dose of 20 mg for 28 days. Soluble vascular cell adhesion molecule-1 (sVCAM-1), soluble intercellular adhesion molecule-1 (sICAM-1) and soluble P-, E- and L-selectins were assessed by ELISA prior to treatment and at day 28.
Results
No statistically significant changes in the levels of adhesion molecules were observed: sICAM-1 1011 vs. 1032 ng/mL, sVCAM-1 1225 vs. 1570 ng/mL, sP-selectin 66.7 vs. 66.0 ng/mL, sE-selectin 276 vs. 253 ng/mL and sL-selectin 887 vs. 927 ng/mL prior to treatment and at day 28, respectively.
Conclusions
Markers characterizing vascular activation were not affected by short treatment with low-dose simvastatin in SSc patients, indicating that the endothelial-protective effect of statins may be related to treatment duration and dose.
Introduction
Systemic sclerosis (SSc) is a connective tissue disorder characterized by the presence of immunological abnormalities, widespread fibrosis of the skin and internal organs, and the development of vascular complications. Although skin fibrosis is a hallmark of the condition, it is internal organ damage that contributes to the generally poor prognosis of patients. 1 The mechanisms leading to this extensive internal organ fibrosis are not yet understood, but it is commonly considered that, in parallel with the accumulation of fibrotic tissues, vascular damage and atherosclerosis are at least partially responsible for internal organ ischemia and subsequent end-stage organ failure. 2 Although the prognosis of patients with SSc has improved significantly over the past two decades, atherosclerosis and its fatal complications continue to contribute to premature mortality in this patient population. Therefore, a need remains for novel therapies that can reduce the risk of atherosclerosis-related complications in SSc. Endothelial dysfunction is a common finding in patients with SSc and the process may extend beyond small vessels, thus contributing to typical atherosclerosis and atherosclerosis-related complications, recognized as the leading causes of premature mortality among patients with SSc. It is widely accepted that endothelial cell injury is an early and crucial event in the pathogenesis of SSc. Endothelial damage could originate from viral infections (especially human cytomegalovirus), cytotoxic autoreactive T cells, auto-antibodies against the endothelium and the toxic influence of free radical species on endothelial cells. In SSc patients, however, atherosclerosis may overlap with disease-specific vasculopathy.3,4
Statins, a class of 3-hydroxy-3 methyl glutaryl coenzyme A (HMG-CoA) reductase inhibitors, have proven safe and efficacious for the treatment of atherosclerosis. However, the properties of statins other than their lipid-lowering activity have attracted much attention. These pleiotropic effects cover a wide range of physiopathological activities including cell maturation, immune response regulation, tissue fibrosis and endothelial activity. 5 However, almost all data on these effects are derived from laboratory studies or animal models, with less known about clinical statin activity in SSc populations. Moreover, it is not clear whether promising data from laboratory studies can be translated directly to humans.
The mechanism of action of statins in the treatment of dyslipidaemias and vascular diseases is at least partially independent of their lipid-lowering properties. The inhibition of MHG-CoA reductase, the key enzyme in the mevalonate synthesis pathway, results in the decreased production of farnesylpyrophosphate and geranylgeranylpyrophosphate, intermediate products in mevalonate synthesis that reduce the prenylation of small GTPases belonging to the Rho and Rac families. Rho and Rac molecules are responsible for key processes such as cytoskeleton formation, cellular trafficking and apoptosis.6,7 Given that vasculopathy is the leading manifestation of SSc, it is likely that statins may exert a beneficial effect similar to that observed in atherosclerosis as endothelium involvement is observed in both conditions. 8 Among the potential biomarkers of SSc, cellular adhesion molecules (CAMs) are of interest as the levels of their soluble forms correlate with endothelial activity and damage and may reflect the activation state of the endothelium. 9 Measurement of the soluble forms of these molecules following statin therapy may therefore provide insight into the function of the endothelium in SSc. Moreover, adhesion molecules such as vascular cell adhesion molecule (VCAM) and intercellular adhesion molecule (ICAM) regulate the activation, adhesion and migration of mononuclear cells to sites of inflammation, so their levels directly correlate with inflammatory activity in the vessel wall. The selectin family of adhesion molecules, consisting of P-, L- and E-selectin, also play important roles in this process. P-selectin is expressed on endothelial cells and is mobilised after activation to the cell surface where it mediates early leukocyte–endothelial interactions. L-selectin is expressed by most circulating neutrophils, monocytes and eosinophils, and is found at high levels in human plasma where it may function as a buffer system, preventing leukocyte rolling at sites of inflammation. Studies on L-selectin levels patients with SSc yielded contradictory results, however, with reduced levels found in some studies10,11 but elevated levels in others.12,13 Soluble E (sE)-selectin is a marker of damaged and/or activated endothelium and has previously been shown to be elevated in patients with SSc compared with healthy controls. 9
The purpose of this study was to determine whether the levels of sICAM-1, sVCAM-1 and sP-, sL- and sE-selectins were altered following short-term simvastatin treatment in normolipidemic patients with SSc but without atherosclerosis.
Patients and Methods
Patients
Consecutive patients were recruited from the scleroderma clinic at the Department of Internal Medicine and Rheumatology Medical Faculty in Katowice, Poland, and screened for eligibility for inclusion in the study (Figure 1). Diagnosis of SSc was established according to the 2013 American College of Rheumatology revised criteria. 14 All patients suffered from diffuse SSc as established according to le Roy classification criteria. 15 Clinical manifestations of SSc included Raynaud’s phenomenon, skin damage including pitting scars and active ulcers, and interstitial lung disease (defined as ground-glass opacities, sub-pleural reticulation with or without pleural irregularities, traction bronchiectasis and/or honeycombing on routine high-resolution CT scans). All patients were characterized by antinuclear antibody positivity.
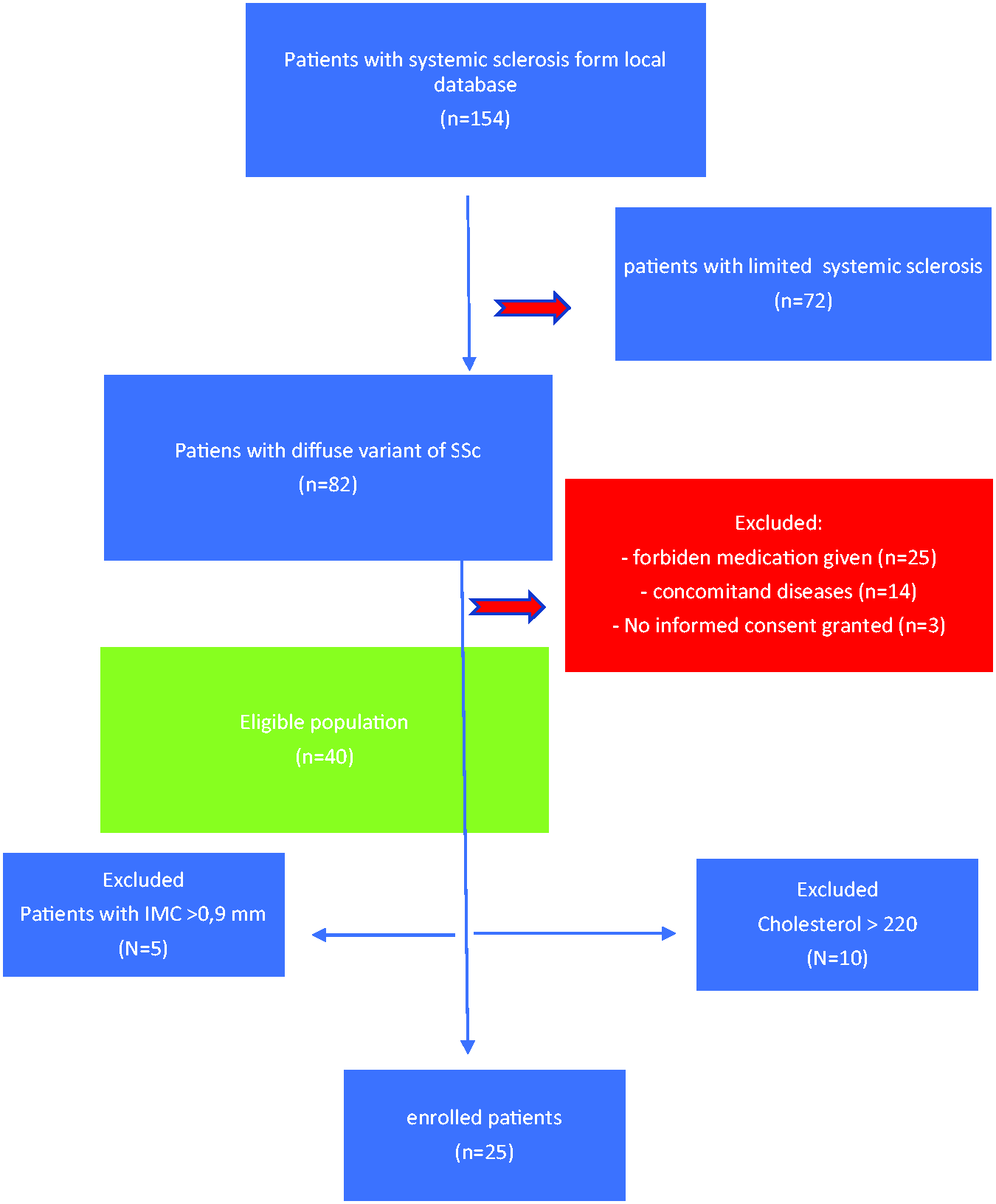
Patient recruitment flow chart
Ultrasound and echocardiography measurement
To assess the pure pleiotropic effect of statin (independent of antiatherosclerotic activity), patients without atherosclerosis with an intima–media complex (IMC) measurement < 0.9 mm were recruited to the study. Depending on dose, disease-modifying rheumatic drugs (DMARDs) and steroids may exert additional beneficial or toxic effects on endothelial function. Therefore, to reduce the direct influence of cytostatic and anti-inflammatory drugs on the endothelial bed, patients were permitted to take DMARDs and immunosuppressants at standard doses (steroids, a dose not exceeding 10 mg/day; for methotrexate, < 25 mg/week; mycophenolate mofetil, < 2.0 g/day; azathioprine, < 200 mg/day).16,17 Patients were excluded from the study if they were receiving prostanoids, vasoactive drugs or endothelin-1 antagonists, or had undergone B-cell depletion therapy in the 12 months preceding enrolment. We also excluded patients with cardiac insufficiency (ejection fraction < 50%), pulmonary hypertension or renal failure, and patients with contraindications for statin therapy. Patients with clinically overt atherosclerosis were also excluded from the study. Digital ulcers (DU) are recognized as a disease-related feature of SSc, therefore patients with DU were eligible for inclusion if they did not require treatment with vasoactive drugs, which interfere with endothelial activity and prevent the assessment of changes in endothelial structure after statin treatment. Patients who had received statins or other antilipemic drugs for at least 6 months prior to enrolment were also excluded. Pulmonary artery pressure was assessed by ultrasound as described previously. 18 It has recently been proposed that patients with echo-measured pulmonary artery pressure > 41 mmHg can be diagnosed as having pulmonary arterial hypertension. 19 As the current study aimed to assess the pleiotropic effects of statins, patients with cholesterol profile abnormalities (total cholesterol level > 220 mg/dL and/or LDL > 130) were excluded. This study was approved by the local bioethical committee at the Medical University of Silesia, Katowice, Poland (approval number KWN-6501-96/I/08), and written informed consent was obtained from all patients prior to participation.
IMC measurement
IMC measurements were performed during the screening period according to the guidelines of the American Society of Echocardiography using a Toshiba ultrasound machine (Toshiba CoreVision Pro, Toshiba America Medical Systems, USA) equipped with an 8–13-MHz linear transducer. 20 The site of IMC measurement and IMC identification were as proposed in the Mannheim Consensus. 21 Specifically, longitudinal and transverse images were obtained and, in the absence of plaques, IMC measurement in the common carotid artery was performed in a 10-mm region located 10 mm below the carotic bulb. Using ultrasound, IMC was identified as a double-line pattern on the far walls of the common carotid arteries in a longitudinal image. IMC images consisted of two parallel lines representing the leading edges of two anatomical boundaries: the lumen–intima and the media–adventitia interfaces. Arterial scans were manually obtained by the same blinded, experienced operator, with 10 measurements recorded for each side. Patients were excluded if plaques or IMC measurements > 0.9 mm were observed. Laboratory testing included the measurement of sICAM-1, sVCAM-1, sE-selectin, sP-selectin and sL-selectin levels, and was performed twice, prior to simvastatin treatment and after 28 days of treatment. Simvastatin was administered daily at a fixed dose of 20 mg.
Blood samples were obtained in the morning after at least 8 hours of fasting, clotted for 30 minutes and then centrifuged for 15 minutes at 1500 ×g. Samples were frozen immediately at −80°C until laboratory assessment. The serum concentrations of sVCAM-1, sICAM-1 and selectins were measured using commercial ELISA microwells kits (IBL International, Hamburg, Germany; catalogue numbers BE 59011, BE59061, BE59051, and BE59081 for sICAM-1, sE-selectin, sVCAM-1, and sP-selectin, respectively; and Biovendor, Modrice, Czech Republic; catalogue number RMBS 206R for sL-selectin). Antinuclear antibodies were assessed using ELISA kits produced by Generic Assay (Dahlewitz, Germany). Assays and tests were carried out according to the manufacturer’s instructions.
Statistical analysis
Statistical analyses were performed using STATISTICA 9.0PL (StatSoft Poland). The results were presented as means ± standard deviation. Distribution of variables was evaluated by the Shapiro–Wilk test. To compare differences between two dependent groups (before and after treatment), the non-parametric Mann–Whitney U test was used. Results were considered statistically significant if they had a p-value < 0.05. All tests used were two-tailed.
Results
Twenty-five patients (22 women, 3 men) with diffuse SSc were enrolled in the study, and their characteristics are presented in Table 1. The mean age of patients was 55.2 years and disease duration from first non-Raynaud’s symptom was 57 months. Four patients were smokers, while three were ex-smokers. All patients were positive for antinuclear antibody, and the antibodies were further identified as anti-topo I (Scl-70) in 20 patients (75%), anti-centromere in 4 patients (16%) and anti-Ro-52 in 3 patients (12%). Patients presented with typical scleroderma-related symptoms, which are summarized in Table 1. Nineteen patients were treated with steroids (76%), 10 with methotrexate (40%), 4 with azathioprine (16%) and 3 with mycophenolate mofetil (12%).
Clinical characteristics of patients

Data are presented as mean ± standard deviation or number (percent), except where otherwise indicated. BMI, body mass index; IMC, intima media complex; mRSS, modified Rodnan skin score; CK, phosphocreatine kinase; LDL, low density lipoproteins; NS, non-significant.
All patients completed the study and no treatment-related side effects were reported during the study period. No significant changes in serum concentrations of sICAM-1, sVCAM-1, and sE- and sP-selectins were observed after 28 days of simvastatin treatment. Although the levels of sE- and sP-selectin were numerically lower after 28 days, the values did not achieve statistical significance. Additionally, there was a trend towards elevated levels of sL-selectin at the end of the study, but again the changes were not significant. Statin therapy resulted in a reduction in total and LDL cholesterol levels by the end of the study period, indicating good adherence by patients to the treatment protocol. All results are summarised in Table 2.
Adhesion molecules before and after simvastatin treatment in patients with systemic sclerosis

Data are presented as mean ± standard deviation. NS, non-significant.
Discussion
The purpose of this 28-day interventional study was to assess the influence of simvastatin on endothelial activation in normolipidemic patients with SSc. To identify a pleiotropic effect independent of the lipid-lowering properties of statins, only normolipidemic SSc patients were included. The unique population of SSc patients comprised only patients with diffuse SSc with a high prevalence of Scl-70 antibodies. Scl-70 antibodies were observed in 10%–40% of the non-selected SSc patients. The high prevalence of antibodies associated with diffuse skin involvement and a more aggressive disease expression in this study is likely attributable to the selection of patients with this form of the disease. 22 As markers of endothelial function, levels of sVCAM-1, sICAM-1 and selectins were determined as their plasma concentrations strongly correlate with endothelial damage. Moreover, the levels of CAMs are known to be elevated in SSc and indicate endothelial activation in the disease state.9,11 The beneficial effect of statin therapy on endothelial function is well established, although little is known about their effect in patients with SSc. Among the 13 controlled and observational studies of statins in SSc to date, only 256 SSc patients were enrolled, limiting the ability to confirm the role of statins as concomitant therapy for SSc. 23 Statins have recently been shown to exert an effect on endothelial function in a murine model of SSc. 24 However, the high doses of simvastatin administered in this study do not correspond to clinical use. A number of studies have demonstrated the efficacy of statins in the treatment of atherosclerosis, which can, at least in part, be explained by the pleiotropic activities of statins 25 and may contribute to their effects on endothelium. In parallel with endothelial activation, increased levels of soluble CAMs were observed, suggesting that CAMs may function as markers of endothelial activation. 26 Among the few studies that have examined this mechanism to date, contradictory results have been reported. Del Papa at al. observed a significant reduction in ICAM, VCAM and E-selectin after 3 months of simvastatin treatment. 27 In agreement with this finding, Abou-Raya and Kuwana observed decreased levels of ICAM and E-selectin in two studies with atorvastatin treatment for 6 and 4 months, respectively.28,29 In a 24-month extension study, Kuwana showed a significant reduction in markers of endothelial injury (sVCAM-1 and sE-selectin), although the first significant changes in CAM levels were observed not the 12-month timepoint. 30 Similar to the results of the present study, Furukawa et al. 31 failed to show any change in the levels of adhesion molecules in an 8-week study with pravastatin. This suggests that the effects of statins are subject to a time lag, and that changes may not be apparent before 3–4 months of treatment. The results obtained in the study indicate that the effect of statin on endothelium may not be entirely cholesterol independent. By definition, the pleiotropic effects of statins are independent of their lipid-lowering properties and should manifest before lipid changes become apparent. 32 In the present study, however, changes in cholesterol levels were observed without any change in the levels of markers of endothelial activation. It remains unclear where some statins might not have pleiotropic activities, as statins can differ in their effects on absorption, tissue binding and solubility. 33
Some limitations of this study included the small sample size, short treatment duration and low dose of simvastatin used. The purpose of the short duration was to identify the pleiotropic potential of simvastatin shortly after treatment initiation and long before it exerts an effect on the lipid profile. The low dose of simvastatin was used to determine whether it exerts immunomodulatory effects in patients with SSc. Given that few SSc patients to date have been treated with statins, their effects in this patient population cannot be confirmed. Although statin treatment may represent a beneficial concomitant therapy in patients with SSc, the conflicting clinical and pre-clinical obtained to date preclude the formulation of guidelines on when and how to use statins for the treatment of SSc in clinical settings.
Footnotes
Acknowledgement
The author wish to express thanks to Laura Coates PhD, University of Oxford, for English editing assistance.
Declaration of conflicting interest
The author declares that there is no conflict of interest.
Funding
The work was has been supported by the Polish Ministry of Education (grant number 402 267136).
