Abstract
Introduction:
Intravenous immunoglobulin (IVIG) was reported to be the third most used monotherapy in livedoid vasculopathy (LV). There is currently a lack of randomized controlled clinical trials and no standardized therapeutic regimen for IVIG therapy in LV.
Methods:
We performed a systematic review of the efficacy and safety of IVIG in treating patients with LV using PubMed, Cochrane, and Embase databases.
Results:
Eighty LV patients from 17 articles were included, receiving IVIG therapy at a dose of 1–2.1 g/kg body weight every 4 weeks. The effective rate of IVIG therapy in LV patients was 95% (76/80) in published studies, showing a good clinical response for resolution of pain, skin ulcerations, and neurological symptoms, and reducing the dependence on glucocorticoids and immunosuppressive agents. IVIG therapy was well tolerated, and no severe adverse events were observed.
Conclusion:
Overall, to a certain degree, IVIG is probably a safe and effective treatment alternative for refractory LV patients, which still need to be confirmed by large-scale randomized controlled clinical trials.
Introduction
Characterized by livedo reticularis, ulcerations, and atrophie blanche on the ankles, dorsum of the foot, and lower extremities, livedoid vasculopathy (LV) is considered to be a recurrent thrombo-occlusive vascular disease with an unknown etiology. A major complaint of LV patients is severe pain that seriously affects walking, social activities, daily life, work, and study, and substantially impairs their quality of life. 1 However, the management of LV remains challenging and controversial; there is no standard or first-line therapy for LV and to prevent its frequent relapses. Anticoagulants, antiplatelets, thrombolytics, psoralen and ultraviolet (UV)-A, anabolic steroids, intravenous immunoglobulin (IVIG), and hyperbaric oxygen therapy are the main conventional treatments for LV. Among them, IVIG therapy was evaluated to be the third most used monotherapy, 2 and it provides an alternative for treating patients with refractory LV patients.
Isolated and purified from the plasma of healthy donors, IVIG is a pooled immunoglobulin preparation that is mainly composed of IgG monomers. Small amounts of IgG dimers, IgA, and IgM are also components of IVIG. 3 IVIG neutralizes autoimmune antibodies and interferes with the complement cascade and the cytokine network, and it acts on the immune reaction through T lymphocytes, B lymphocytes, and macrophage levels. 4 Thus, IVIG is a well-tolerated medication that has been primarily used in a variety of autoimmune, inflammatory, and infectious disease, and it has also been administered off-label to patients with immune-related dermatoses that have a severe course. 5
The pathogenesis of LV remains unclear. A proposed therapeutic mechanism for IVIG in LV is as follows:6,7 (1) binding and neutralizing autoimmune antibodies (such as antiphospholipid antibody, lupus anticoagulant, and thrombophilic factors such as anti-β2-GP1 antibody) have been detected in LV patients; (2) inhibiting platelet and lymphocyte activation in LV through modulation of the immune reaction; (3) altered endothelial function in cutaneous blood vessels; and (4) interference with the inflammatory response, complement cascade, and inflammatory cytokine production. In addition, hypercoagulability and thrombosis, inflammation, and its association with coagulation may also play a role in LV etiology.
Levy et al. 8 were the first to describe two intractable LV patients who were successfully treated with IVIG at a dose of 0.4 g/kg body weight for 5 consecutive days every 4 weeks in 1999. Current information on IVIG therapy mainly comprises case reports, case series, and small sample studies. To date, clinical experience with the dosage, frequency, and course of IVIG therapy for LV is insufficient, and there is still a lack of randomized controlled clinical trials and a standardized therapeutic regimen. Here, we performed a systematic review on the safety and efficacy of IVIG to treat patients with LV. The aim of this article is to evaluate the efficacy and safety of IVIG therapy in LV and to provide guidance and evidence on the management of LV.
Methods
Search strategy
This systematic review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline. 9 The final literature search was conducted on 7 December 2021 using the PubMed, Cochrane, and Embase online electronic databases for articles concerning IVIG therapy in LV patients. The search terms included the following: ‘livedoid vasculopathy’, ‘livedoid vasculitis’, ‘livedo vasculitis’, ‘atrophie blanche’, ‘white atrophy’, ‘segmental hyalinizing vasculitis’, ‘intravenous immunoglobulin’, ‘IVIG’, and ‘immunoglobulin’. The search strategy used was as follows: (‘livedoid vasculopathy’ OR ‘livedoid vasculitis’ OR ‘livedo vasculitis’ OR ‘atrophie blanche’ OR ‘segmental hyalinizing vasculitis’ OR ‘white atrophy’) AND (‘intravenous immunoglobulin’ OR ‘IVIG’ OR ‘immunoglobulin’). The timeline was from inception of the search engine to December 2021. Articles were restricted to papers that were written in English only. No other filters or automated tools were used in the search process. In addition, all the references of screened articles were searched for further high-quality studies. The ethics approval and informed consent were not applicable in this systematic review.
Study selection
The initial search process yielded 39 articles from the PubMed, Cochrane, and Embase online electronic databases, removing 20 duplicating records. Another two articles were from references of screened articles. Two investigators independently screened the abstracts of all 41 identified records and selected articles for full-text review. The PRISMA flow figure demonstrating the process of literature search and study selection is shown in Figure 1.

The PRISMA flow figure demonstrating the process of literature search and study selection.
The inclusion criteria were as follows: (1) LV patients with a confirmed diagnosis depending on their typical clinical and pathological manifestations in accordance with the main LV diagnostic criteria; 10 (2) LV patients receiving IVIG therapy; (3) studies involving efficacy and safety of IVIG therapy in LV, and recording detailed relevant information like dosage, cycles, frequency, clinical response, and adverse events; (4) article types including research articles, case series, case report, and correspondence; and (5) English articles only.
The exclusion criteria were as follows: (1) uncertain LV diagnosis; (2) details in the general data and about the treatment like dosage, cycles, frequency, clinical response, and adverse events were all vaguely recorded; (3) review articles or editorial articles; or (4) articles written in languages other than English.
According to the inclusion and exclusion criteria, 24 records were excluded due to following reasons: (1) not published in English languages (n = 1); (2) review articles (n = 2); (3) irrelevant subjects (n = 18); and (4) lack details in treatment (n = 3). A total of 17 eligible articles were enrolled in this systematic review, among them 3 articles were studies, and 14 articles were case reports and case series.
We assessed the risk of bias according to the Cochrane handbook of systematic reviews of interventions. Due to enrollment of case reports and case series in this systematic review, overreporting or overestimating of specific results may bias the analysis. However, considering disease rarity and limited evidence on IVIG therapy in LV, we included these case reports and case series.
Data extraction
After carefully screening and reviewing, the following data were extracted from the included studies: first author’s name, year, country, number of patients, mean age, gender, comorbidities, screening for thrombophilic factors, previous treatment, dose, frequency, duration, efficacy of the IVIG therapy, combination treatment, recurrence, and adverse events (Tables 1 and 2). Screening for thrombophilic factors mainly included protein C, protein S, antithrombin III, activated protein C resistance, lipoprotein(a), homocysteine, antinuclear antibody, anticardiolipin antibody, lupus anticoagulant, and several single nucleotide polymorphisms that were related to hypercoagulability, the thrombosis-like coagulation factor V Leiden mutation, and plasminogen activator inhibitor-1 promoter homozygosity.
Demographic and general data of LV patients enrolled.

ASA, acetylsalicylic acid; ASP, aspirin; AZA, azathioprine; COPD, chronic obstructive pulmonary disease; CTX, cyclophosphamide; DM, diabetes mellitus; F, female; GC, glucocorticoid; HBOT, hyperbaric oxygen therapy; HTN, hypertension; LMWH, low-molecular-weight heparin; M, male; MTX, methotrexate; NA, not available; NSAID, nonsteroidal anti-inflammatory drugs; PGE1, prostaglandin E1; PTX, pentoxifylline; PUVA, psoralen and UV-A; RA, rheumatoid arthritis; SLE, systemic lupus erythematosus; tPA, tissue plasminogen activator.
Efficacy and safety of IVIG in the treatment of LV.

ASA, acetylsalicylic acid; ASP, aspirin; CTX, cyclophosphamide; GC, glucocorticoid; LMWH, low-molecular-weight heparin; MTX, methotrexate; NA, not available; NR, no recurrence.
A total of 10 patients responded to IVIG: 9 patients achieved remission after 3 cycles; 1 refractory patient successfully retreated after 6 years with 7 cycles.
Results
The full text of these 41 articles was screened by two independent reviewers. After the comprehensive literature search and review, a total of 17 articles were included in accordance with the inclusion and exclusion criteria.5–8,11–23 Among them, 2 articles were retrospective studies and 1 article was an open non-controlled prospective trial; other 14 articles were case series and case reports. These eligible articles were from Israel, the United Kingdom, Germany, Spain, the United States, France, Austria, Portugal, Korea, Japan, Turkey, and Romania.
General results
Eighty LV patients from 17 studies, case series, and case reports who received IVIG treatment were included. Among them, 24 patients (30%) were men and 56 patients (70%) were women, with a male-to-female ratio of 1:2.33. The median age of LV patients in this systematic review was 46.64 years, ranging from 17 to 83 years. The comorbidities for LV patients are summarized in Table 3. Hypertension (n = 22) and diabetes mellitus (n = 9) were the first and second most common comorbidities that were recorded in the selected articles. Other documented comorbidities included autoimmune diseases [e.g. systemic lupus erythematosus (n = 3), rheumatoid arthritis (n = 5), and antiphospholipid syndrome (n = 1)], thrombotic diseases [e.g. stroke (n = 1), thrombosis (n = 6), and peripheral arterial disease(n = 4)], neuropathy [e.g. polyneuropathy (n = 3) and mononeuritis multiplex (n = 1)], chronic obstructive pulmonary disease (n = 1), and malignant tumor (n = 6). Screening for thrombophilic factors was conducted in 63 LV patients. Fourteen LV patients (22.22%) had at least one thrombophilic factor that was mentioned above. Most LV patients receiving IVIG therapy were intractable cases, and they had been previously treated with multiple medications but had a poor response or had frequent recurrences. The previous therapeutic medications are mainly divided into the following categories: glucocorticoid, immunosuppressive agents, nonsteroidal anti-inflammatory drugs, anticoagulants, antiplatelet drugs, vasodilator drugs, and other treatments such as hyperbaric oxygen therapy and local plastic surgery (Table 1).
Details about comorbidities in LV patients.

Efficacy of IVIG
Among the 80 LV patients who received IVIG treatment, 75 LV patients responded well to this therapy. In addition, one refractory LV patient was resistant to IVIG therapy in the initial disease episode but was successfully retreated with IVIG for a relapse 6 years later. 6 Overall, the total effective rate of IVIG therapy in LV patients was as high as 95% (76/80) in the published studies. IVIG therapy was generally effective in treating refractory LV patients who were resistant to previous treatment, and it was also effective in patients with new-onset LV when IVIG was used for the initial treatment. Among the five LV patients who had not been prescribed any treatment before they received IVIG therapy for the first time, four of them achieved a favorable clinical response. 6 There was no significant difference in the efficacy of IVIG therapy in LV patients with and without thrombophilic factors.
The curative effect of IVIG therapy in LV is mainly reflected in the following aspects: (1) IVIG acted quickly to resolve pain in LV patients after one to three cycles of treatment, as described in the published studies. In four articles, the earliest pain remission began to appear during the first cycle of IVIG therapy.7,16,20,23 Eighty percent of pain was resolved after two IVIG cycles in a long-term follow-up study of 11 LV patients; 6 (2) ulceration healing usually lagged behind pain resolution. The resolution of skin ulcerations lasted from 1 to 14 cycles in the published studies;6,7,8,12,14,16,19–23 (3) neurological symptoms such as numbness and hypoesthesia caused by ischemic nerve damage are common in LV. 24 However, conventional medications have little influence on LV neuropathy. In addition to the efficacy of IVIG on pain and ulcerations, there was also improvement in dysesthesia along with IVIG treatment maintenance;5,18 and (4) because LV frequently recurs, using IVIG reduced the patient’s dependence on conventional medications such as glucocorticoids and immunosuppressive agents that have multiple severe side effects when used in the long term. Levy et al. 8 described an LV patient who was no longer dependent on cyclophosphamide after six cycles of IVIG therapy.
Frequency and dosage of IVIG
The initial frequency of IVIG therapy was 4-week intervals in most articles,5–8,12,13,16–18,20,22 except for two articles (including two patients) in which the IVIG dose information was vague.11,21 There were also a few LV patients who received IVIG as a single shot or at 2-week, 6-week, and 10-week intervals.7,14,15,19,23 The interval for IVIG therapy was gradually prolonged on the basis of its efficacy and the patients’ response. The details in doses and duration of maintenance phase were limited and mainly depended on clinical experience of dermatologists and disease activity. In maintenance phase, some LV patients paused IVIG therapy directly after receiving 1–3 cycles,17,19,23 or gradually prolonged their intervals for IVIG infusions to 6-week, 8-week, or even 4- to 6-month interval.5,7 Conventional treatments like anticoagulants or antiplatelet medications were gradually applied as combination therapy in maintenance phase.
The total monthly dose of IVIG ranged from 1 to 2.1 g/kg body weight, which could be divided into a high-dose IVIG group (2–2.1 g/kg body weight) and a low-dose IVIG group (1–1.5 g/kg body weight). Here, we demonstrated the detailed information about dosage, duration, treatment outcomes, and adverse events of low-dose and high-dose IVIG groups in Table 4. Sixty-five LV patients [84.42% (65/77)]5,6,7,8,12,14,16–20,22 received high-dose IVIG therapy, whereas 12 LV patients [15.58% (12/77)]5,7,13,15 received low-dose IVIG therapy. The IVIG dose in one LV patient decreased to 1 g/kg body weight after a dramatic clinical response at the initial dose of 2 g/kg body weight, but this led to a less dramatic improvement. 7 Compared with the low-dose IVIG group, 13 the high-dose IVIG group achieved remission in fewer cycles, indicating that it was a more effective LV treatment. 6
Detailed information about dosage, duration, treatment outcomes, and adverse events of low-dose and high-dose IVIG groups.
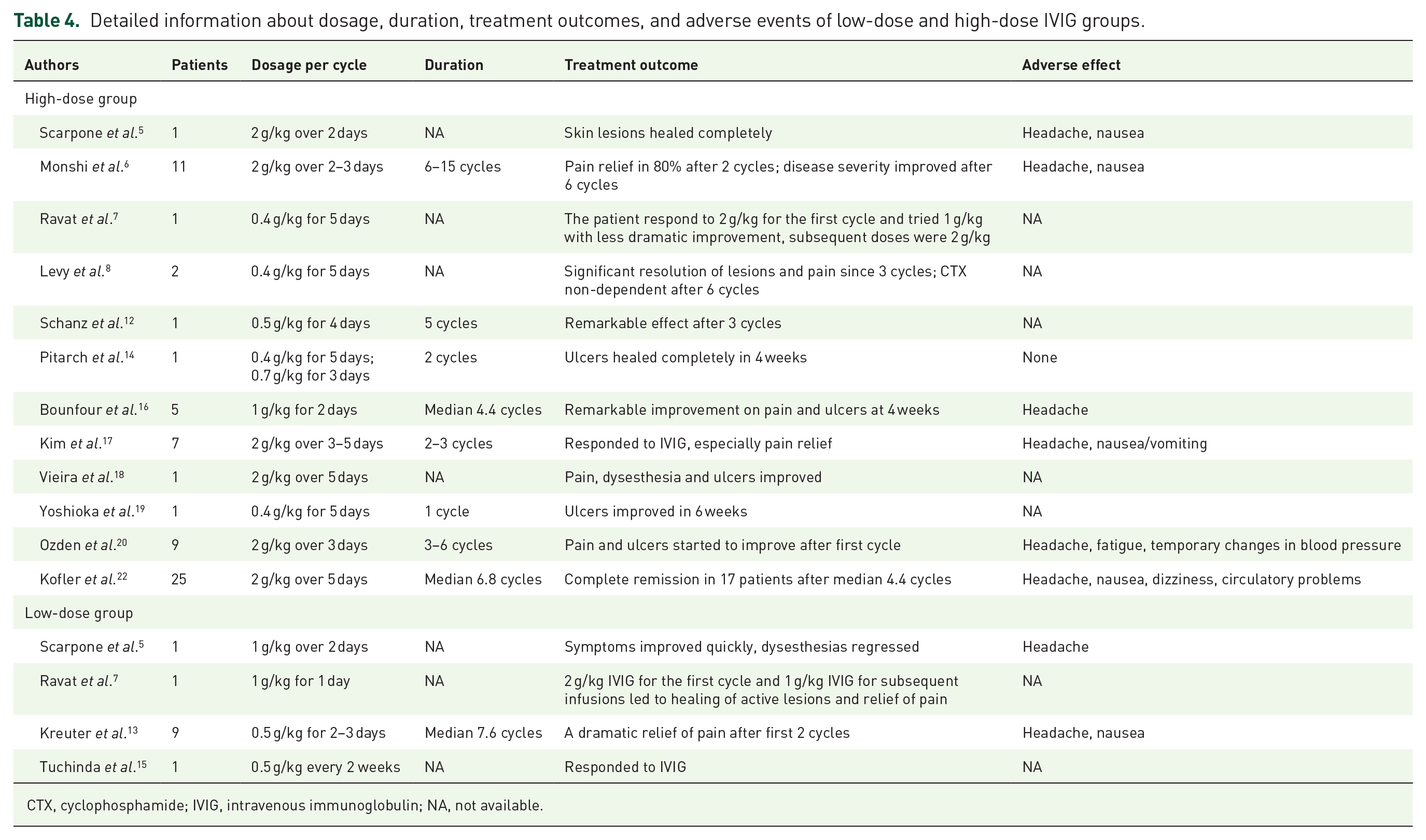
CTX, cyclophosphamide; IVIG, intravenous immunoglobulin; NA, not available.
LV patients receiving a dose of 2–2.1 g/kg body weight IVIG therapy could also be classified as a conventional daily dose group (0.4–0.5 g/kg body weight per day) and a high daily dose group (over 0.5 g/kg body weight per day) on the basis of their daily dose of IVIG per cycle. Except for the vague description of the daily dose per cycle in one study, 17 among the LV patients who received high-dose IVIG therapy (2–2.1 g/kg body weight per cycle), 27 of these LV patients received a high daily dose of IVIG that was more than 0.5 g/kg body weight per day, while 31 LV patients received a conventional daily dose of IVIG, which was 0.4–0.5 g/kg body weight per day. Although the sample size is relatively small, LV patients who received a high daily dose of IVIG seemed to achieve faster resolution of pain and ulcerations compared with LV patients who received the conventional daily dose.6,14,16,20
Recurrence
Twenty-three patients experienced recurrence in this systematic review.5,6,16–18,20,22 Among them, the shortest recurrence period after suspending IVIG treatment was less than 3 months, and the longest interval with no recurrence was 8 years. After achieving complete remission, two LV patients had a relapse less than 3 months after discontinuing IVIG therapy. 22 With maintenance IVIG and warfarin treatment, one LV patient relapsed after 8 years, after which she was successfully treated with rituximab. 18
Adverse events
IVIG therapy was well tolerated in most of LV patients. Adverse events that were recorded were moderate. The most common adverse event in IVIG therapy was headache, which occurred in 11 LV patients during IVIG therapy in this systematic review, and these adverse events were usually mild and resolved spontaneously.5,6,13,16,17,20,22 Overall, the incidence of headache for LV patients during IVIG therapy was 13.75% (11/80). Other adverse events included nausea, vomiting, fatigue, dizziness, temporary changes in blood pressure, and circulatory problems.5,6,13,17,20,22
Discussion
In the systematic review, IVIG therapy for LV showed a good clinical response for resolution of pain, skin ulcerations, and neurological symptoms, and it reduced the dependence on glucocorticoids and immunosuppressive agents in both initially treated patients and in resistant patients who were refractory to conventional medications. The total effective rate for IVIG therapy in LV patients was as high as 95% (76/80) in published studies. There was no significant difference in the efficacy of IVIG therapy whether or not LV patients had complications due to thrombophilic factors. To date, a standardized therapeutic regimen for IVIG therapy in LV has not been established. In this systematic review, we compared the efficacy of IVIG between low-dose group (1–1.5 g/kg body weight per cycle) and high-dose group (2–2.1 g/kg body weight per cycle), and between conventional daily dose group (0.4–0.5 g/kg body weight per day) and a high daily dose group (over 0.5 g/kg body weight per day). Although the sample size is relatively small, LV patients with a high daily dose of 2 g/kg body weight per cycle over 2–3 consecutive days every 4 weeks seemed to achieve faster resolution of pain and ulcerations in fewer cycles. From our perspective, IVIG therapy at a dose of 2 g/kg body weight per cycle over 2–3 consecutive days every 4 weeks may be a more effective and more rapid treatment for LV on the basis of published studies. However, the standard administration of IVIG therapy need to be further estimated in large sample studies during long-term follow-up. During the follow-up, the longest interval with no recurrence was 8 years with maintenance IVIG therapy. Generally, IVIG therapy in LV was safe and well-tolerated. A previous study showed that the incidence of adverse events reported in IVIG therapy for LV was about 16% (5/31 patients). 2 Headache was the most common adverse event, accounting for 13.75%. Overall, these adverse events were moderate, and most of the patients achieved spontaneous remission.
LV is a thrombo-occlusive vascular disease of the lower extremities, where hypercoagulability and thrombosis play major roles in its pathogenesis. At least one thrombophilic factor was detected in more than one-fifth of LV patients in this article. The therapeutic mechanism of IVIG for treating patients with LV remains unclear. It is speculated that neutralizing autoimmune antibodies, modulating the immune and inflammatory responses, and altering endothelial function are the likely therapeutic mechanisms of IVIG in LV.25,26 However, the anticoagulation effect of IVIG is undefined and not fully recognized. Even thrombotic events that occur in arteries, veins, and intracranial vein sinuses are considered to be severe delayed adverse effects of IVIG treatment with an approximate incidence of 1–16.9%. 27 Although IVIG therapy was shown to be the third most commonly used monotherapy for treating patients with LV, 2 IVIG combined with anticoagulants and antiplatelet drugs such as warfarin, low-molecular-weight heparin, and clopidogrel was used in seven studies, case series, and case reports in this systematic review.5,6,16,18–20,22 Currently, there has been no report of thrombotic events that were caused by IVIG therapy for LV.
Considering the high cost and relatively complex administration mode of IVIG, we do not recommend IVIG as the first-line therapy or initial treatment for LV. However, IVIG therapy provides an alternative treatment for refractory LV patients or LV patients with contraindications to conventional treatment. IVIG should be administered in the acute ulcerative stage of refractory LV and to prolong intervals or switch to conventional medications when achieving remission. Because hereditary and acquired thrombophilic factors were detected in LV, we suggest using preventive procedures to prevent thrombotic events in IVIG therapy for LV, including combination therapy with anticoagulants and antiplatelet drugs, normal saline hydration before and after infusion, and a slow infusion rate. 28
There are some limitations in this systematic review. The sample size is relatively small, and case series and case reports were the main sources for this article. Case series and case reports tended to reported LV cases where patients were successfully treated by IVIG, and thus, the effectiveness of IVIG therapy was overestimated. And there was no control group for the safety and efficacy evaluation of IVIG therapy. Hopefully, standardized score and index could be applied in future clinical studies and randomized controlled clinical trials to evaluate the clinical response of IVIG therapy in LV more objectively.
Conclusion
In conclusion, we found that IVIG therapy was effective in 95% of LV patients in the published studies. IVIG showed a good clinical response for resolution of pain, skin ulcerations, and neurological symptoms, and it reduced the dependence on glucocorticoids and immunosuppressive agents. Overall, to a certain degree, IVIG therapy for LV was safe and well tolerated. Our article provides clinical evidence and guidance for IVIG therapy in the management of LV, which requires further confirmation by large-scale randomized controlled clinical trials.
Supplemental Material
sj-docx-1-taj-10.1177_20406223221097331 – Supplemental material for Efficacy and safety of intravenous immunoglobulin for treating refractory livedoid vasculopathy: a systematic review
Supplemental material, sj-docx-1-taj-10.1177_20406223221097331 for Efficacy and safety of intravenous immunoglobulin for treating refractory livedoid vasculopathy: a systematic review by Yimeng Gao and Hongzhong Jin in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-2-taj-10.1177_20406223221097331 – Supplemental material for Efficacy and safety of intravenous immunoglobulin for treating refractory livedoid vasculopathy: a systematic review
Supplemental material, sj-docx-2-taj-10.1177_20406223221097331 for Efficacy and safety of intravenous immunoglobulin for treating refractory livedoid vasculopathy: a systematic review by Yimeng Gao and Hongzhong Jin in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-3-taj-10.1177_20406223221097331 – Supplemental material for Efficacy and safety of intravenous immunoglobulin for treating refractory livedoid vasculopathy: a systematic review
Supplemental material, sj-docx-3-taj-10.1177_20406223221097331 for Efficacy and safety of intravenous immunoglobulin for treating refractory livedoid vasculopathy: a systematic review by Yimeng Gao and Hongzhong Jin in Therapeutic Advances in Chronic Disease
Footnotes
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China (82073450); CAMS Innovation Fund for Medical Sciences (CIFMS) No. 2021-I2M-1-059; National Key Research and Development Program of China (2016YFC0901500).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
