Abstract
Cardiopulmonary exercise testing and pulmonary function test are important methods for detecting human cardio-pulmonary function. Whether they could screen vasoresponsiveness in idiopathic pulmonary artery hypertension (IPAH) patients remains undefined. One hundred thirty-two IPAH patients with complete data were retrospectively enrolled. Patients were classified as vasodilator-responsive (VR) group and vasodilator-nonresponsive (VNR) group on the basis of the acute vasodilator test. Pulmonary function test and cardiopulmonary exercise testing were assessed subsequently and all patients were confirmed by right heart catheterization. We analyzed cardiopulmonary exercise testing and pulmonary function test data and derived a prediction rule to screen vasodilator-responsive patients in IPAH. Nineteen of VR-IPAH and 113 of VNR-IPAH patients were retrospectively enrolled. Compared with VNR-IPAH patients, VR-IPAH patients had less severe hemodynamic effects (lower RAP, m PAP, PAWP, and PVR). And VR-IPAH patients had higher anaerobic threshold (AT), peak partial pressure of end-tidal carbon dioxide (PETCO2), oxygen uptake efficiency (OUEP), and FEV1/FVC (P all <0.05), while lower peak partial pressure of end-tidal oxygen (PETO2) and minute ventilation (VE)/carbon dioxide output (VCO2) slope (P all <0.05). FEV1/FVC (Odds Ratio [OR]: 1.14, 95% confidence interval [CI]: 1.02–1.26, P = 0.02) and PeakPETCO2 (OR: 1.13, 95% CI: 1.01–1.26, P = 0.04) were independent predictors of VR adjusted for age, sex, and body mass index. A novel formula (=−16.17 + 0.123 × PeakPETCO2 + 0.127×FEV1/FVC) reached a high area under the curve value of 0.8 (P = 0.003). Combined with these parameters, the optimal cutoff value of this model for detection of VR is −1.06, with a specificity of 91% and sensitivity of 67%. Compared with VNR-IPAH patients, VR-IPAH patients had less severe hemodynamic effects. Higher FEV1/FVC and higher peak PETCO2 were associated with increased odds for vasoresponsiveness. A novel score combining PeakPETCO2 and FEV1/FVC provides high specificity to predict VR patients among IPAH.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is characterized by progressive loss and remodeling of the pulmonary arteries resulting in right heart failure and death. Idiopathic pulmonary artery hypertension (IPAH) is a rare and fatal disease with high mortality rates. IPAH is also a progressive disease that affects the precapillary pulmonary vasculature for which an exact underlying risk factor is unknown.1,2 The acute pulmonary vasodilator test (AVT) during heart catheterization is helpful for the selection of appropriate medical therapy for patients with IPAH. Only the positive responders can probably benefit from long-term use of calcium channel blockers. 3 However, AVT is invasive and expensive, and patients with a lack of monetary resources cannot afford it. To date, noninvasive methods have not been identified to screen out vasodilator-responsive (VR) patients among those with IPAH.
To the best of our knowledge, cardiopulmonary exercise tests (CPETs) and pulmonary function tests (PFTs) are important means to evaluate physiological function, diagnosis, therapeutic effect, and prognosis. 4 Many studies on heart failure and other cardiovascular diseases here and abroad have shown that CPET plays a substantial role in the assessment of disease and prognosis. One study revealed that the CPET characteristics of PAH patients were exercise intolerance and ventilatory inefficiency due to abnormal gas exchange, findings that are characteristic of a ventilation perfusion mismatch. 5 In our center, Chen et al. found that CPET potentially could be used to estimate the severity of Chronic Thromboembolic Pulmonary Hypertension. 6 Zhao et al. showed that CPET could serve as a complementary tool to improve diagnostic accuracy in echocardiography-suspected “PH possible” patients. 7 Yuan et al. found that sex-specific cardiopulmonary exercise testing parameters were predictors of responsiveness among patients with IPAH. 8 However, no analysis of the role of CPET and PFT in predicting vasodilator-responsive patients in IPAH has been published to date.
Methods
Study setting and participants
From 1 January 2014 to 1 January 2019 at our Hospital, 245 patients were diagnosed with IPAH, and of these 113 patients were excluded from this study (106 incomplete data and 7 withdrew consent). One hundred thirty-two patients newly diagnosed with IPAH were retrospectively enrolled. IPAH was established according to the 2015 ESC/ERS pulmonary hypertension guidelines and diagnosed by right heart catheterization (RHC) as follows: (I) mean pulmonary arterial pressure (m PAP) ≥25 mm Hg; (II) pulmonary artery wedge pressure (PAWP) ≤15 mm Hg; (III) pulmonary vascular resistance (PVR) >3 Wood Units; and (IV) ruling out other causes of PAH. 9 The exclusion criteria were as follows: (1) connective tissue disease and congenital heart disease and those with portopulmonary hypertension, heritability, drugs and toxins induced, schistosomiasis, and HIV infection; (2) chronic lung disease; (3) chronic pulmonary thromboembolism; (4) pulmonary hypertension due to left heart disease; and (5) IPAH patients who could not tolerate or who refused CPET.
Data collection
All patients underwent blood testing, electrocardiogram testing, PFT, echocardiography, and CPET and diagnoses were confirmed by RHC within two weeks. The study protocol was approved by our Hospital ethics committee, and all participants provided written informed consent. Data were extracted from medical records using a standardized data collection form. All data were checked by two physicians independently and a third researcher adjudicated any difference in interpretation between the two primary reviewers.
Resting pulmonary function test
Each patient underwent resting PFT of forced vital capacity (FVC), forced expiratory volume in 1 s (FEV1), maximal voluntary ventilation (MVV), residual volume (RV), and total lung capacity (TLC) using standard methodology 10 and equipment (Jaeger Corp., Hoechberg, Germany). All resting lung function values were reported in absolute terms and normalized to the percent of predicted value (%pred). Predicted spirometry values were calculated using accepted equations for Chinese individuals. 11
Cardiopulmonary exercise test
CPET was completed within seven days of PFT, within two weeks of RHC, and performed on an electromagnetically braked cycle ergometer (Master Screen CPX, Jaeger Corp., Hoechberg, Germany) using a breath-by-breath system according to the American Thoracic Society/American College of Chest Physicians Statement on CPET. 12 Before each test, the equipment was calibrated in accordance with the manufacturer’s specifications using reference and calibration gases. Standard 12 lead electrocardiograms (ECGs) and pulse oximetry were continuously monitored. Arterial blood pressure (BP) was measured every 2 min with an automatic cuff. The protocol consisted of three minutes of rest, followed by 3 min of unloaded cycling at 55–65 revolutions per minute (rpm), subsequently, a progressively increasing workload of 10 to 25 W/min to the maximum tolerance was conducted, followed by 4 min of recovery. Patients were allowed to stop at any time when they experienced fatigue, dyspnea, chest tightness, or other forms of discomfort during the process. Each anaerobic threshold (AT) was determined by the V-slope method. 13 Measurements included peak workload, peak minute ventilation (VE), peak carbon dioxide output (VCO2), peak oxygen uptake (VO2), peak oxygen pulse (VO2/HR), peak heart rate (HR), peak end-tidal partial pressure of CO2 (PETCO2), and peak breathing frequency (BF). PeakVO2 was defined as the highest 30-s average oxygen uptake in the last minute of exercise, and other peak parameters were calculated simultaneously. The VE/VCO2 slope was obtained by linear regression analysis of the relationship between VE and VCO2. 14 The oxygen uptake efficiency slope (OUES) was computed by a linear square regression from the oxygen uptake on the logarithm of the minute ventilation according to the following equation: VO2 = a × lg VE + b. 15 The oxygen uptake efficiency plateau (OUEP) was determined as 90 s of the highest consecutive values for VO2 (ml/min)/VE (L/min). 16
Acute pulmonary vasodilator test
After diagnostic RHC with baseline hemodynamic indexes, blood gases were measured by a blood gas analyzer (Nova Biomedical) and 20 µg iloprost (Bayer-Schering Pharma) was delivered to patients by a PARI LC STAR nebulizer (PARI GmbH) driven by a PARI Turbo BOY-N compressor (PARI GmbH) within 10–15 min. Then, another set of hemodynamic measurements and blood gases was obtained.17–21 The AVT positive standard met the following standards: m PAP fell more than 10 mmHg to below 40 mmHg without CO decline.22–24 CO was calculated by thermodilution and the cardiac index (CI) was calculated by dividing cardiac output by the body surface area. PVR was calculated as dividing (m PAP-PAWP) by pulmonary blood flow. According to AVT outcomes, patients with IPAH were divided into two groups as follows: the vasodilator responsive (VR) group and the vasodilator-non responsive (VNR) group.
Statistics analysis
Continuous normally distributed variables are reported as means; interquartile ranges (IQR) or upper and lower quartiles for non-normally distributed variables; categorical variables are reported as percentages. Comparisons between the VR-IPAH group and the VNR-IPAH group were conducted by using chi-square tests for categorical variables, independent-samples Student's t tests for normally distributed continuous variables, and Mann-Whitney U test for non-normally continuous distributed variables. Logistic regression was used to model the probability of diagnosis of vasodilator-responsive patients. Univariate logistic regression was performed in PFT and CPET predictors as the dependent variable was vasodilator responsiveness. Then, a stepwise forward variable selection procedure was used to find independent predictors of vasodilator responsiveness. To establish these scores combining multiple CPET and PFT parameters, we performed a multivariate discriminant analysis adjusted for age, sex, and BMI and determined coefficients for multiplying these values with the measured values of the CPET and PFT parameters. The optimal cutoff value for the combined diagnostic variables were estimated from receiver operating characteristic curve analysis. In all cases, a P value <0.05 was considered statistically significant. The main analysis was performed by SPSS (Statistic Package for Social Science, IBM, Armonk, NY, USA) version 22.0.
Results
Demographic and clinical characteristics of VR-IPAH and VNR-IPAH patients
One hundred thirty-two IPAH patients were enrolled with a mean age of 39 ± 15 years and a mean BMI of 21.93 ± 3.04 kg/m2, 95 (72%) of the patients were female. Seventy (53%) patients were classified as WHO function class (FC) II, and the other patients were classified as WHO FC III. There were 19 VR patients (16 women; mean age 39 ± 16years old) and 113 VNR patients (79 women; mean age 39 ± 15 years old). Patients with VR-IPAH had lower RAP, m PAP, PAWP, PVR, and N-terminal prohormone of brain natriuretic peptide (NT-pro BNP) than patients with the VNR-IPAH group (P < 0.001, P < 0.001, P = 0.04, P = 0.005, and P = 0.02, separately), while there was no difference in age, sex, BMI, WHO FC, or 6-MWD between the two groups (P all ≥0.05; Table 1). There was no significant difference in CO and CI between the two groups (Table 1).
Baseline characteristics of the IPAH patients.
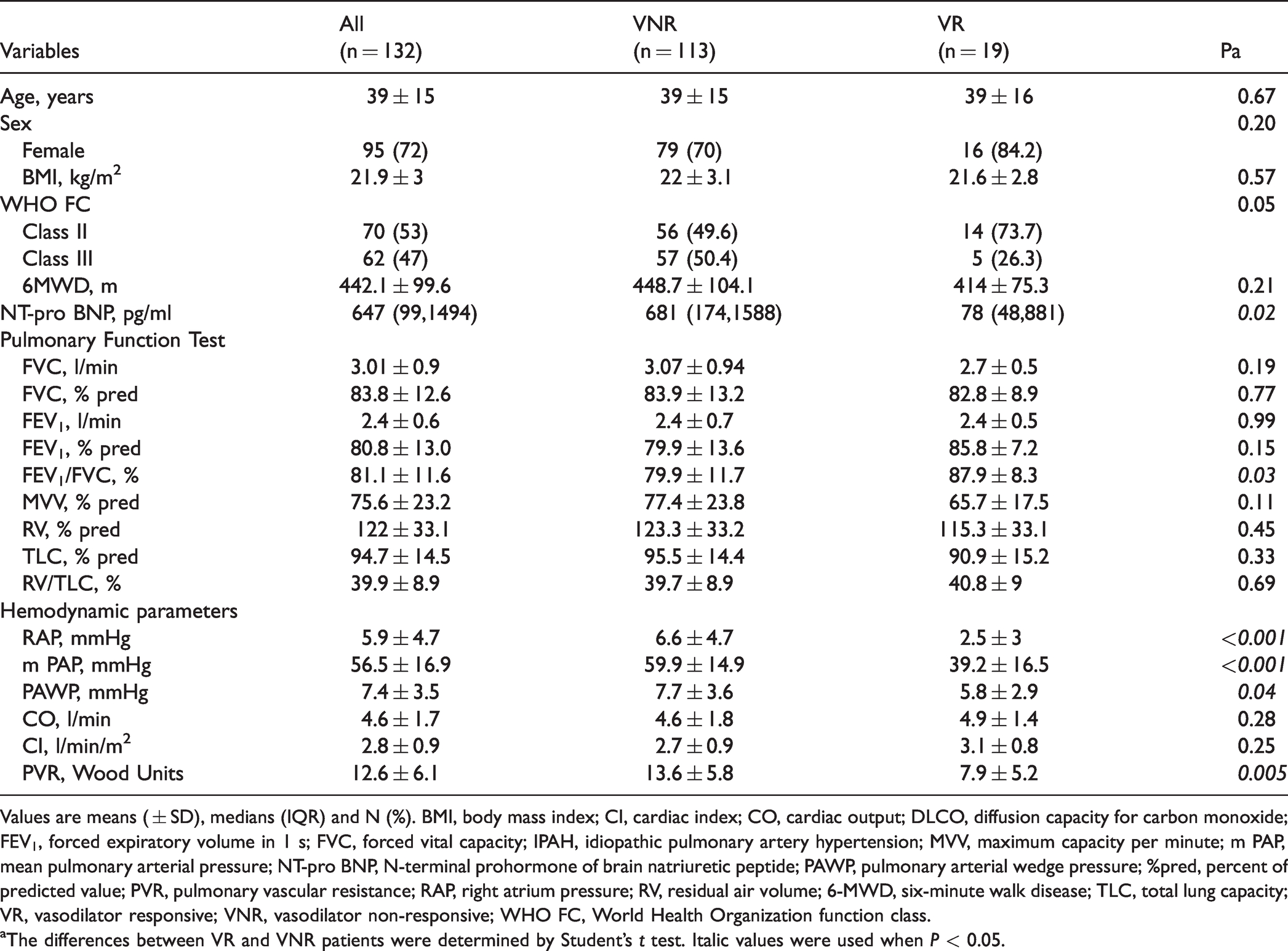
Values are means ( ± SD), medians (IQR) and N (%). BMI, body mass index; CI, cardiac index; CO, cardiac output; DLCO, diffusion capacity for carbon monoxide; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; IPAH, idiopathic pulmonary artery hypertension; MVV, maximum capacity per minute; m PAP, mean pulmonary arterial pressure; NT-pro BNP, N-terminal prohormone of brain natriuretic peptide; PAWP, pulmonary arterial wedge pressure; %pred, percent of predicted value; PVR, pulmonary vascular resistance; RAP, right atrium pressure; RV, residual air volume; 6-MWD, six-minute walk disease; TLC, total lung capacity; VR, vasodilator responsive; VNR, vasodilator non-responsive; WHO FC, World Health Organization function class.
aThe differences between VR and VNR patients were determined by Student’s t test. Italic values were used when P < 0.05.
Evaluation of the diagnostic value of PFT and CPET in vasodilator response capacity of IPAH patients
Patients with VR-IPAH had higher FEV1/FVC values than patients with VNR-IPAH (P = 0.03). However, there was no difference in FVC, FEV1, MVV%, RV, TLC, or RV/TLC values between VNR and VR patients (Table1). The measurements of CPET variables in each group are presented in Table 2. Compared with patients with VNR-IPAH, patients with VR-IPAH had higher AT, PeakPETCO2 and OUEP values (P = 0.04, 0.002 and 0.009, respectively) and lower PeakPETO2 and VE/VCO2 slope values (P = 0.004 and P = 0.03).
Characteristics of CPET parameters of the patient groups.
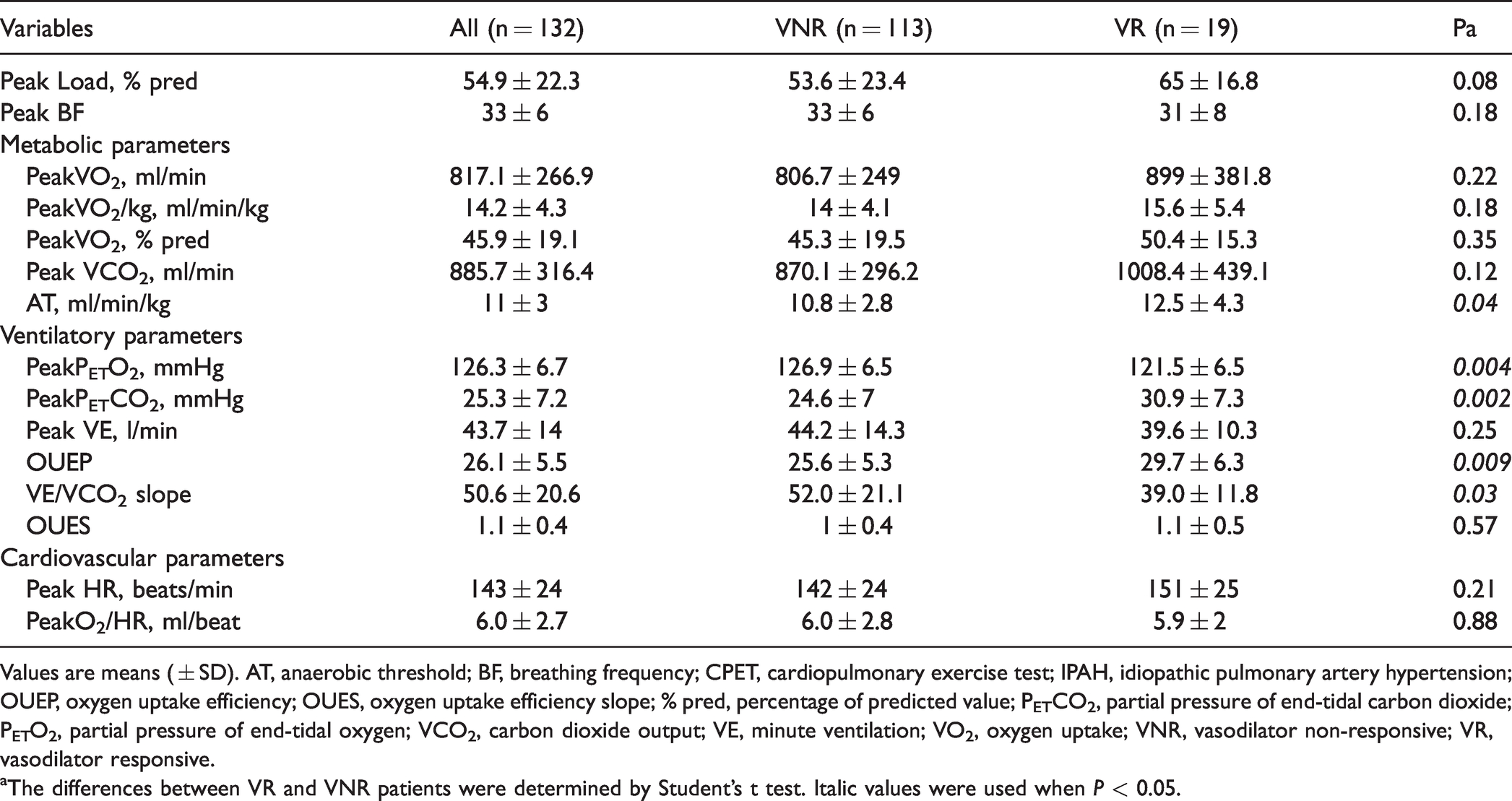
Values are means ( ± SD). AT, anaerobic threshold; BF, breathing frequency; CPET, cardiopulmonary exercise test; IPAH, idiopathic pulmonary artery hypertension; OUEP, oxygen uptake efficiency; OUES, oxygen uptake efficiency slope; % pred, percentage of predicted value; PETCO2, partial pressure of end-tidal carbon dioxide; PETO2, partial pressure of end-tidal oxygen; VCO2, carbon dioxide output; VE, minute ventilation; VO2, oxygen uptake; VNR, vasodilator non-responsive; VR, vasodilator responsive.
aThe differences between VR and VNR patients were determined by Student’s t test. Italic values were used when P < 0.05.
To assess whether PFT and CPET variables allowed discrimination between the VR-IPAH and VNR-IPAH group, univariate logistic regression models were used. The results indicated that the FEV1/FVC, AT/kg, PeakPETO2, PeakPETCO2, OUEP, and VE/VCO2 slope values can predict vasodilator response. Next, multivariate binary logistic regression models were performed, and FEV1/FVC (odds ratio [OR]: 1.14, 95% confidence interval [CI]: 1.02–1.26, P = 0.02) and PeakPETCO2 (OR: 1.13, 95% CI: 1.01–1.26, P = 0.04) values were identified as independent predictors for vasodilator response capacity in IPAH patients after adjusting for sex, age, and BMI (Table 3), which indicated that the higher FEV1/FVC and higher peak PETCO2 were associated with increased odds for vasoresponsiveness. Then, the following new formula combined with PeakPETCO2 and FEV1/FVC was constructed: ln[(p/1–p)] X = –16.17 + 0.123 × PeakPETCO2 + 0.127 × FEV1/FVC, where PeakPETCO2 is given in mmHg, FEV1, and FVC are both given in l/min.
ORs for VR diagnosis are derived from univariate and multivariate binary logistic regression models, identifying independent predictors of VR.
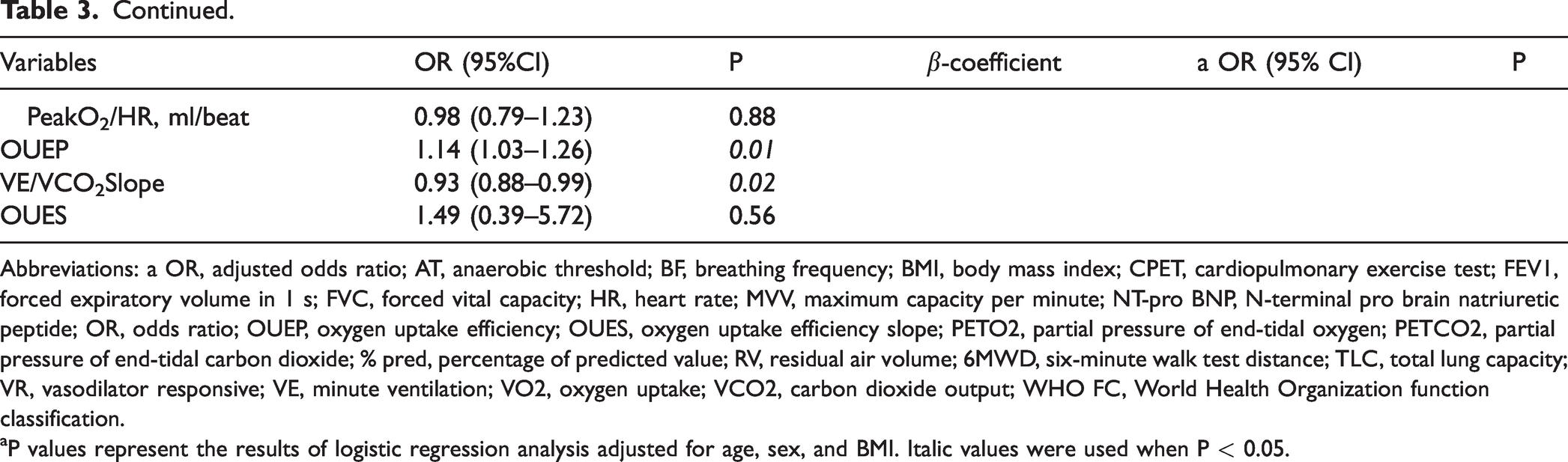
Abbreviations: a OR, adjusted odds ratio; AT, anaerobic threshold; BF, breathing frequency; BMI, body mass index; CPET, cardiopulmonary exercise test; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; HR, heart rate; MVV, maximum capacity per minute; NT-pro BNP, N-terminal pro brain natriuretic peptide; OR, odds ratio; OUEP, oxygen uptake efficiency; OUES, oxygen uptake efficiency slope; PETO2, partial pressure of end-tidal oxygen; PETCO2, partial pressure of end-tidal carbon dioxide; % pred, percentage of predicted value; RV, residual air volume; 6MWD, six-minute walk test distance; TLC, total lung capacity; VR, vasodilator responsive; VE, minute ventilation; VO2, oxygen uptake; VCO2, carbon dioxide output; WHO FC, World Health Organization function classification.
aP values represent the results of logistic regression analysis adjusted for age, sex, and BMI. Italic values were used when P < 0.05.
The optimal cutoff value of this model for the detection of VR was −1.06 (area under the curve: 0.8, P = 0.003, probability value ≥ cutoff = VR, Fig. 1), with a specificity of 91% and sensitivity of 67%.
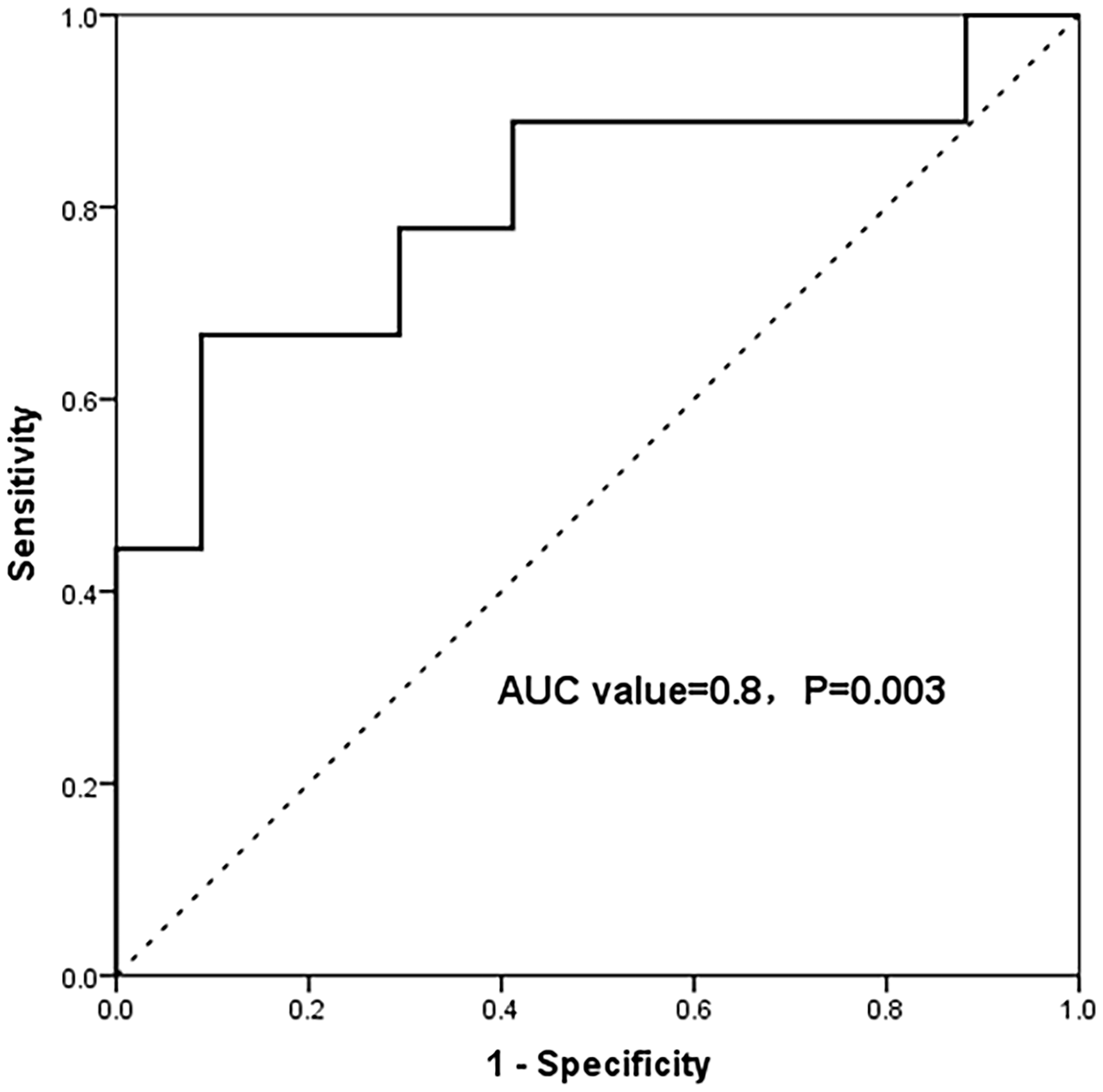
ROC curve for the determination of VR in IPAH patients.
Discussion
The purpose of our study was to assess the predictive value of CPET and PFT to screen for vasodilator-responsive patients among IPAH patients. Our results demonstrated that (I) compared with VNR-IPAH patients, VR-IPAH patients had less severe hemodynamic effects (lower RAP, m PAP, PAWP, and PVR values) and lower NT-pro BNP values; however, we found no difference in CO and CI, demographic characteristics (age, sex, and BMI), WHO FC, and 6-MWD between the two groups and (II) higher FEV1/FVC and higher peak PETCO2 values were associated with increased odds for vasoresponsiveness. A new formula (= –16.17 + 0.123 × PeakPETCO2 + 0.127 × FEV1/FVC) was constructed according to the results of the multivariate binary logical regression model to predict VR patients among those with IPAH.
In our study, VR-IPAH patients had lower RAP, m PAP, PAWP, and PVR scores than patients with VNR-IPAH. The mechanism that caused lower RAP, m PAP, PAWP, and PVR scores in VR-IPAH patients is unclear. Arena et al. reported that increased PVR could increase physiologic dead space, which was reflected by decreased ventilatory efficiency. 4 Yi Tang et al. had described that ventilatory efficiency in VNR-IPAH group was lower than that in the VR-IPAH group even though PVR, peak VO2, and peak O2 pulse levels were similar between the two groups. In our analysis, the outcomes were similar to those observed in Yi Tang’s study, which may be due to that increased PVR was based on vascular tone rather than intimal proliferation leading to greater recruitment under perfused microvasculature and downstream capillary surface area under the condition of exercise in patients with VR-IPAH.25,26
In addition, we found that patients with VR-IPAH had higher FEV1/FVC and peak PETCO2 levels than VNR-IPAH patients, and these values were associated with increased odds for vasoresponsiveness. In a wide range of conditions, FEV1/FVC is an indicator for ventilation, and PETCO2 is considered to be a good indicator for evaluating the ventilation/perfusion relationship. 27 The mechanism by which FEV1/FVC and PeakPETCO2 are related to vasoresponsiveness is unknown. Variations in PETCO2 have been shown to reflect changes in both cardiac output and pulmonary blood flow. 27 Patients with cardiac disease have been shown to have an abnormally low PETCO2 during exercise, especially those with an impaired response of cardiac output during exercise. 28 In addition, PETCO2 reduction is inversely correlated with the elevation of m PAP in PAH, 29 and reduced values of PETCO2 have high diagnostic accuracy to identify the likelihood of pulmonary vasculopathy in patients with PAH.5,30 Therefore, we assume that the higher the PETCO2 value is, the better the pulmonary blood flow will be and the less severe the patients’ pulmonary vasculopathy is, resulting in better vascular elasticity and vasoresponsiveness in VR-IPAH patients. However, further research is needed to confirm our hypothesis.
To our knowledge, no published study has indicated a noninvasive method to screen for patients with VR-IPAH. For the first time, we have shown that CPET combined with PFT could be used to help identify VR-IPAH patients. The main previously reported CPET characteristics in PAH patients were exercise intolerance and ventilatory inefficiency due to abnormal gas exchange, findings that are characteristic of a ventilation perfusion mismatch. 5 PETCO2 was correlated with exercise capacity and cardiac output during exercise, and the PETCO2 level reflects an impaired cardiac output response to exercise in cardiac patients diagnosed with heart failure. 28 Yi Tang et al. reported that ventilatory efficiency in patients with VR-IPAH is better than that in patients with VNR-IPAH. 31 Similarly, our study revealed that the ratios of FEV1/FVC, peakPETCO2 and OUEP were higher, while the VE/VCO2 slope was lower in the VR-IPAH group than in the VNR-IPAH group. This result indicated that the VR-IPAH group could have better ventilation function and implied a better capacity for oxygen uptake and exercise tolerance. Additionally, we found that the AT value was higher in the VR-IPAH group than in the VNR-IPAH group, indicating an earlier development of lactic acidosis in VNR-IPAH patients. For the first time, we found that higher FEV1/FVC and higher peak PETCO2 values were associated with increased odds for vasoresponsiveness after adjusting for age, sex, and BMI. In addition, for the first time, we constructed a novel index combining PeakPETCO2 and FEV1/FVC values to predict VR patients among IPAH patients.
Limitations
One of the main limitations of this study is that there is no validation cohort for this novel developed formula. Another limitation in our study is the relatively small number of patients. All data were obtained at a single center; thus, there is a possibility for selection bias. Furthermore, the accuracy of the formula needs to be further verified by other centers, including a larger patient sample size. The CPET indicators we studied are not sufficiently comprehensive, and we did not analyze the resting time variables.
Conclusions
Compared with VNR-IPAH patients, VR-IPAH patients had less severe hemodynamic effects (lower RAP, m PAP, PAWP, and PVR levels). Higher FEV1/FVC and higher peak PETCO2 values were associated with increased odds for vasoresponsiveness. A novel formula (= –16.17 + 0.123 × PeakPETCO2 + 0.127 × FEV1/FVC) confers high specificity to predict vasodilator-responsive patients among IPAH patients.
Footnotes
Acknowledgements
We thank all of the investigators, fellows, nurses, and research coordinators who participated in this study.
Author contributions
Ci-Jun Luo, Lan Wang, and Jin-Ming Liu designed the study. Ci-Jun Luo, Hong-Ling Qiu, Chang-Wei Wu, and Jing He contributed to conducting the research, analyzing the data, and preparing the article, and are the co-first authors. Ping Yuan, Qin-Hua Zhao, Rong-Jiang, Su-Gang Gong, Wen-Hui Wu, Jian Guo, and Rui Zhang contributed to data acquisition and statistical analysis. Lan Wang and Jin-Ming Liu contributed to study supervision. Jin-Ming Liu, Lan Wang, Ping Yuan, Rong Jiang, Wen-Hui Wu, and Qin-Hua Zhao contributed to the acquisition of funding. All authors contributed toward data analysis, and critically revising the paper, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by the Program of Natural Science Foundation of Shanghai (grant number 18ZR1431500) and Program of the National Science and Technology Information System of the People’s Republic of China (grant number 2018YFC1313603).
Ethical approval
The protocol was approved by Shanghai Pulmonary Hospital ethics committee, and all patients provided written informed consent.
