Abstract
Rationale
Mesenchymal stem cell extracellular vesicles (MSC EVs) reverse pulmonary hypertension, but little information is available regarding what dose is effective and how often it needs to be given. This study examined the effects of dose reduction and use of longer dosing intervals and the effect of hypoxic stress of MSC prior to EV collection.
Methods
Adult male rats with pulmonary hypertension induced by Sugen 5416 and three weeks of hypoxia (SuHx-pulmonary hypertension) were injected with MSC EV or phosphate buffered saline the day of removal from hypoxia using one of the following protocols: (1) Once daily for three days at doses of 0.2, 1, 5, 20, and 100 µg/kg, (2) Once weekly (100 µg/kg) for five weeks, (3) Once every other week (100 µg/kg) for 10 weeks, (4) Once daily (20 µg/kg) for three days using EV obtained from MSC exposed to 48 h of hypoxia (HxEV) or MSC kept in normoxic conditions (NxEV).
Main results
MSC EV reversed increases in right ventricular systolic pressure (RVSP), right ventricular to left ventricle + septum weight (RV/LV+S), and muscularization index of pulmonary vessels ≤50 µm when given at doses of 20 or 100 μg/kg. RVSP, RV/LV+S, and muscularization index were significantly higher in SuHx-pulmonary hypertension rats treated once weekly with phosphate buffered saline for five weeks or every other week for 10 weeks than in normoxic controls, but not significantly increased in SuHx-pulmonary hypertension rats given MSC EV. Both NxEV and HxEV significantly reduced RVSP, RV/LV+S, and muscularization index, but no differences were seen between treatment groups.
Conclusions
MSC EV are effective at reversing SuHx-pulmonary hypertension when given at lower doses and longer dosing intervals than previously reported. Hypoxic stress does not enhance the efficacy of MSC EV at reversing pulmonary hypertension. These findings support the feasibility of MSC EV as a long-term treatment for pulmonary hypertension.
Introduction
Pulmonary arterial hypertension (PAH) is a rare disease characterized by marked elevation in pulmonary artery pressure (PAP) and pulmonary vascular resistance. Although the underlying cause of PAH remains unclear, consistent features of the disease include a decrease in the number of peripheral pulmonary vessels and the development of an obliterative vasculopathy in the distal pulmonary arterioles. 1 These changes lead to progressive increases in right ventricular afterload that greatly limits functional capacity and eventually lead to right heart failure and death. Over a dozen medical therapies for PAH have been developed since the beginning of this century, but none attack the pathogenetic mechanisms of the disease and thus, their use has resulted in only modest improvements in pulmonary hemodynamics, functional capacity and survival.2,3 No therapy is curative and there remains a pressing need to develop new therapies that target the underlying pathogenesis of this devastating disease.
Mesenchymal stem cells (MSC), also referred to as mesenchymal stromal cells, are found in the bone marrow and in adipose tissue and demonstrate high capacity for regeneration. They can differentiate into adipocytes, chondrocytes, osteocytes, neurons, and stromal cells, but not hematopoietic cells. MSCs exhibit wide immunomodulatory effects on innate and adaptive immunity including inhibition of T cells, natural killer cells, and B cells.4,5 They also act to maintain hematopoiesis by sustaining a part of the hematopoietic stem cell population in an undifferentiated quiescent state.6,7 They have been shown to impede differentiation of monocytes into antigen-presenting dendritic cells and to modulate polarization of inflammatory macrophages toward an anti-inflammatory phenotype.5–8 Several studies have described their ability to attenuate pulmonary hypertension (PH) in a variety of animal models.9–14 However, in these studies, only small numbers of the administered MSCs can be found in the pulmonary circulation suggesting that their mechanism of action is unlikely to be due to the replacement of injured cells in the pulmonary vascular bed. Instead, MSCs appear to act in a paracrine fashion to modulate the vascular remodeling effects of other vascular cells. Interestingly, many of the immunomodulatory effects of MSC are mediated by the extracellular vesicles (EVs) that they secrete. EVs are microscopic bodies consisting of a cell membrane encapsulating a variety of cytoplasmic proteins and RNA species that vary according to the type of cell that they are derived from. These microspheres are increasingly recognized as an important vehicle for cell-to-cell communications.15–17 Like MSCs, MSC EVs have broad immunomodulatory effects, especially on macrophages where they have been shown to shift macrophage activation from the classical inflammatory pathway (M1) to the alternative anti-inflammatory pathway (M2) and to decrease secretions of inflammatory cytokines.18–20 Recent studies demonstrate strong immunomodulatory effects of MSC EV on alveolar macrophages. For example, MSC EVs increase alveolar macrophage oxygen consumption by enhancing oxidative phosphorylation and suppress secretion of inflammatory cytokines including TGF-β, TNF-α, IL-6, and IL-10. 19 The ability of MSC EV to shift alveolar macrophage activation toward an anti-inflammatory phenotype has been associated with decreased lung injury in models of adult respiratory distress syndrome (ARDS) and bronchopulmonary dysplasia,20,21 but their role in modulating macrophage activity in PH is less well studied. Recently, we found that MSC EV blunt the classical inflammatory (M1) activation of macrophages in vitro and increases the ratio of M2/M1 macrophages in the lungs of rats with PH induced by Sugen 5416 and chronic hypoxia (SuHx-PH). 22
These findings, along with those of others suggest that MSC EV may represent a promising new approach to reversing PH. Indeed, MSC EVs have been shown to prevent and or reverse PH in multiple animal models of PH.21–24 However, the amount of MSC EV used to reverse PH in pre-clinical models would be technically difficult to produce if the same doses were needed in human trials. Presently, it is not known if MSC EVs are effective at reversing PH when they are administered at lower doses or how often they must be given to maintain their beneficial effects. In the present study, we examined the ability of MSC EV to reverse established PH when given at lower doses, longer dosing intervals, and longer periods of time than have been studied previously. In addition, we also examined if the efficacy of MSC EV could be enhanced by exposing MSC to acute hypoxia prior to harvesting their EVs because previous studies suggest that EVs collected from hypoxia-stressed MSC exhibit enhanced immunomodulatory effects.25,26
Methods
Sugen/hypoxia pulmonary hypertension
Male Sprague Dawley rats (Charles River Laboratory, Wilmington, MA) weighing 180–225 g were injected with the VEGF receptor 2 antagonist Sugen 5416 (SU5416, R&D Systems, Minneapolis, MN) at a dose of 25 mg/kg s.c. and placed in hypoxic chambers (10.5% oxygen) for three weeks followed by 1–10 weeks of normoxic (Nx) recovery. Nx control rats were injected with an equal volume of dimethyl sulfoxide (DMSO) vehicle, kept under Nx conditions, and injected with phosphate buffered saline (PBS) at the same time points that SuHx rats were given MSC EV.
Study protocols
For the dose titration studies, rats were given 0.2, 1.0, 5, 20, or 100 µg/kg of MSC EV protein in 500 µl of PBS or 500 µl of PBS vehicle alone by tail vein injection once daily for three days starting the day of removal from three weeks of hypoxia. Rats were sacrificed and PH assessed one week later. For the dosing interval studies, rats were treated with 100 µg/kg of MSC EV or PBS vehicle by tail vein injection once weekly for five weeks or once every other week for 10 weeks starting the day of removal from three weeks of hypoxia. Rats were sacrificed one week after the last injection in the former protocol and two weeks after the last injection in the latter protocol. To assess the relative effectiveness of EV isolated from normoxic (NxEV) or hypoxia-stressed MSC (HxEV), rats were treated with 20 µg/kg of MSC EV or PBS vehicle for three days starting the day of removal from three weeks of hypoxia and sacrificed for measurement of PH two weeks later.
For assessment of PH, rats were anesthetized with isoflurane, and right ventricular systolic pressure (RVSP) was measured using a Millar® catheter (AD Instruments Inc., Colorado Springs, CO) inserted into the right ventricle (RV) via the right internal jugular vein. Rats were then sacrificed by exsanguination and the heart and lungs removed. RV hypertrophy was assessed by RV to left ventricle + septum wet weight ratio (RV/LV+S). Pulmonary vascular remodeling was assessed by measuring the muscularization of peripheral pulmonary vessels on histologic sections of the lung as described below. All studies were approved by the Lifespan Animal Welfare Committee at our Hospital (Institutional Animal Care and Use Committee # 5008-19 and #5007-20).
Histologic analysis of lung sections
Lungs were removed en bloc and the trachea and main pulmonary artery cannulated. The airways were infused with 3 ml of 4% paraformaldehyde in PBS and the pulmonary circulation was flushed with PBS. Both injections were performed at a constant pressure of 20 cm H2O. Lungs were kept in 4% paraformaldehyde until embedded in paraffin, sectioned in 5 µm slices, and stained with antibody against rat α-smooth muscle actin (ab5694, Abcam, Cambridge, MA) to assess pulmonary vascular remodeling. Slides were then incubated with the EnVision+ Dual Link System-HRP solution (Agilent Technologies, Santa Clara, CA) containing anti-rabbit immunoglobulins conjugated to peroxidase-labeled polymer. Following chromogenic development, the slides were counterstained with hematoxylin. Images of the staining were then taken by using a Nikon Eclipse E800 microscope (Nikon Instruments Inc., Melville, NY) equipped with a camera and SPOT Advanced 4.7 software (Diagnostic Instruments Inc., Sterling Heights, MI). Muscularization of vessels ≤50 µm was assessed by muscularization index (MI) defined as the total area of the vessel that stained positive for α-smooth muscle actin divided by total cross-sectional area of the vessel. The NIH ImageJ program was used to assess vessel areas.
Mesenchymal stem cell extracellular vesicles
Human MSCs (Lonza group, Basel, Switzerland) were grown in T-175 flasks as per manufacturer’s instructions using EV-free Minimum Essential Media and fetal bovine serum. MSC EVs were isolated at Passage 5 as described previously.27,28 Briefly, the cells were cultured for three weeks in T-175 tissue culture flasks and passaged five times. When the desired number of flasks reach 80% confluency, the flasks were rinsed with PBS and fed 20 ml of serum-free Roswell Park Memorial Institute (RPMI) 160 medium. After 24 h, the medium was harvested and centrifuged for 10 min at 300 × g to remove cellular debris. The supernatant was ultra-centrifuged at 100,000 × g for 1 h. The pellet was resuspended in PBS containing 1% DMSO. A typical MSC EV preparation came from 178 flasks with 1 × 106 cells per flask. The EVs were collected twice over the course of four days. The yield for the two collections was approximately 7 × 1011 EV as quantified by nanoparticle tracking analysis. The total protein content was approximately 900 µg as measured by BCA Protein Assay Kit (Pierce) yielding a concentration of approximately 8 × 108 EV/µg of protein. EVs were then characterized by electron microscopy, particle size distribution using nanoparticle tracking analysis (Nanosight), and expression of cell surface tetraspanins and internal proteins that have been used as EV biomarkers as described previously 28 (Supplemental Figure 1). For experiments that utilized EV isolated from hypoxia-stressed MSC, the same protocol was used except that when flasks reach 80% confluency the flasks were divided into two groups, rinsed with PBS, and fed 20 ml of serum-free RPMI 160 medium. MSCs used for NxEVs were kept under routine Nx conditions, and MSCs used for HxEVs were exposed to 1% oxygen for 48 h before harvesting the EV.
Statistical analysis
Data are shown as mean ± standard error of the mean. Differences between groups were calculated by nonparametric ANOVA (Kruskal–Wallis test) using GraphPad Prism version 6.03 (GraphPad Software, Inc., La Jolla, CA). Data were considered statistically significant at P < 0.05.
Results
Effective of MSC EV dose
Compared with control rats, treatment with Sugen 5416 followed by three weeks of hypoxia and one week of normoxia caused a severe elevation in PAP and marked right ventricular hypertrophy that was evidenced in SuHx-PH rats treated with PBS by higher RVSP (65.7 ± 14.0 vs. 24.5 ± 1.7 mmHg, P < 0.01) and RV/LV+S (0.644 ± 0.038 vs. 0.254 ± 0.01, P < 0.001) (Fig. 1a and b). Administration of MSC EV once daily for three days starting the day after removal from three weeks of hypoxia resulted in a reduction in both RVSP and RV/LV+S one week later as compared to rats treated with PBS vehicle alone. The lowest dose of MSC EV that resulted in a significant reduction in RVSP and RV/LV+S was 20 µg/kg (Fig. 1a and b). There was also a strong trend toward lower RVSP and RV/LV+S in SuHx-PH rats given MSC EV at 5 µg/kg, but the differences were not quite statistically significant. There was a nonsignificant trend toward higher RVSP and RV/LV+S at doses below 5 μg/kg (Fig. 1). Thus, the lowest dose of MSC EV that appears to be effective at reversing PH in the SuHx-PH model appears to be between 5 and 20 µg/kg.
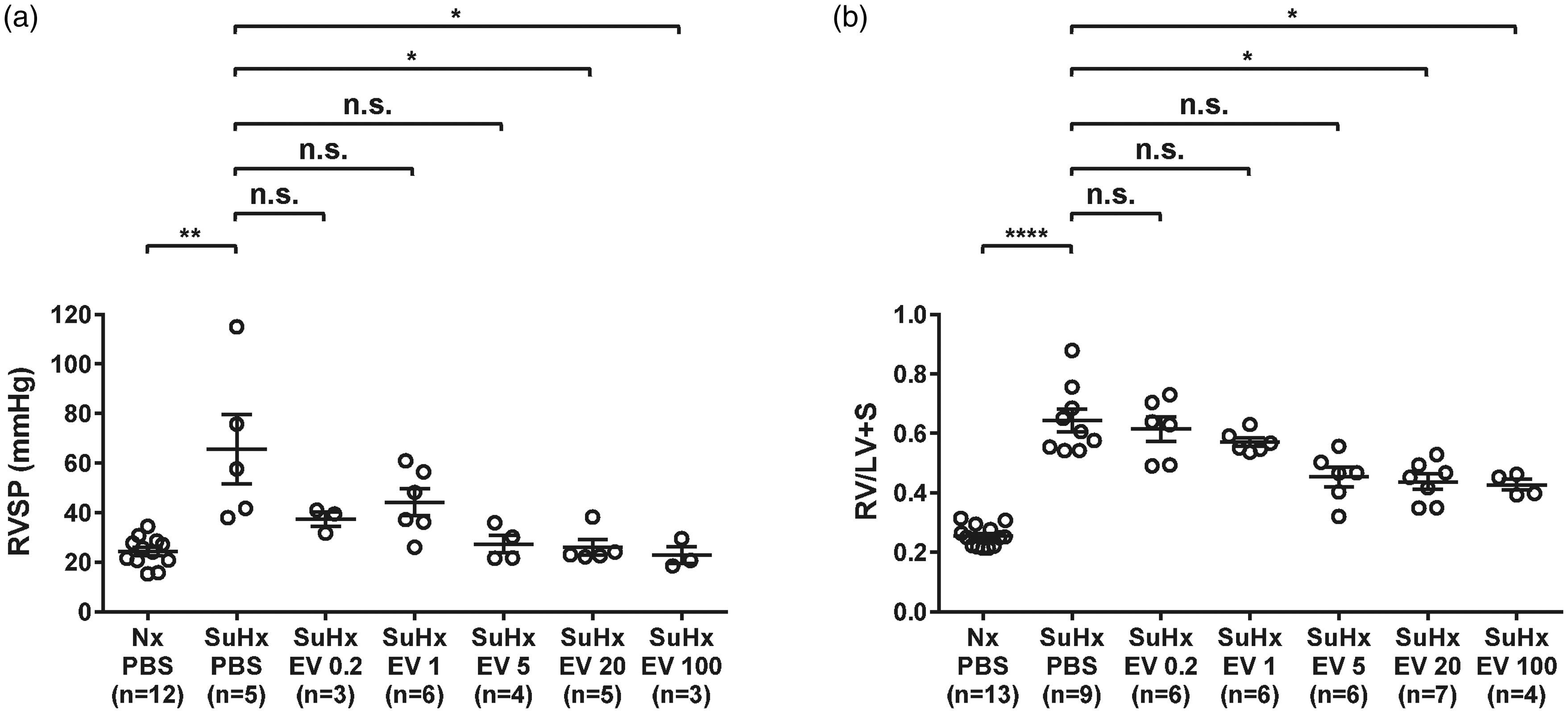
Effect of increasing doses of mesenchymal stem cell extracellular vesicles (EVs) compared to phosphate buffered saline vehicle (PBS) on (a) right ventricular systolic pressure (RVSP) and (b) right ventricle to left ventricle + septum ratio (RV/LV+S) in rats kept under normoxic conditions (Nx) or treated with Sugen 5416 and exposed to three weeks of hypoxia (SuHx). All doses are in micrograms of protein per kg. n = 3–13 per group. *P < 0.05, **P < 0.01, ****P < 0.0001, n.s.: not significant.
Effective of extending MSC EV dosing interval
In the dose titration studies described above, MSC EVs were effective at reversing PH after one week when given once daily for the first three days after removal from hypoxia. In order to determine if MSC EVs are effective at reversing PH when given at longer dosing intervals and for longer periods of time, additional studies were conducted in which SuHx-PH rats were given MSC EV at the highest dose studied (100 µg/kg) or PBS vehicle alone once weekly for five weeks or once every other week for 10 weeks, starting the day after removal from hypoxia. In the five-week study, SuHx-PH rats treated with PBS alone developed severe PH as evidenced by an approximate three-fold increase in RVSP and RV/LV+S compared to Nx controls (Fig. 2b and c). However, in rats treated with MSC EV, RVSP and RV/LV+S were not significantly increased from Nx controls (23.9 ± 3.3 vs. 23.7 ± 2.7 mmHg and 0.484 ± 0.056 vs. 0.250 ± 0.011 for RVSP and RV/LV+S, respectively). RVSP and RV/LV+S were lower in rats given MSC EV than those treated with PBS alone, although the difference in RV/LV+S did not reach statistically significance (23.9 ± 3.3 vs. 75.7 ± 7.2 mmHg, P < 0.01 for RVSP, 0.4464 ± 0.0393 vs. 0.6470 ± 0.0628, P = N.S. for RV/LV+S).
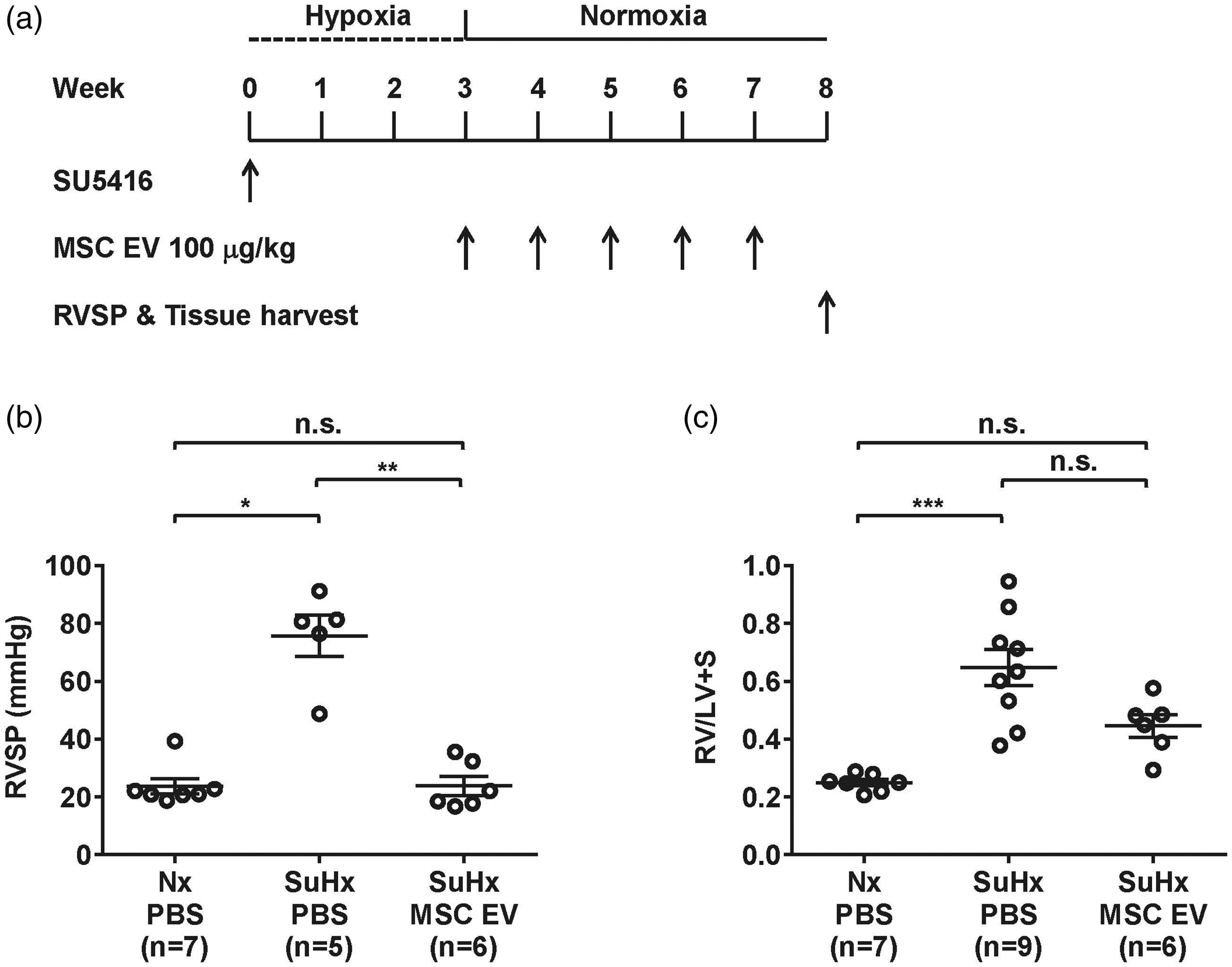
(a) Experimental protocol for reversal of pulmonary hypertension showing timing of administration of mesenchymal stem cell extracellular vesicles (MSC EVs) or phosphate buffered saline vehicle (PBS). (b) Right ventricular systolic pressure (RVSP) and (c) right ventricle to left ventricle + septum ratio (RV/LV+S) were measured five weeks after removal from three weeks of hypoxia (end of week 8 of experimental protocol). Nx PBS: normoxic PBS control; SuHx: rats treated with Sugen 5416 and exposed to three weeks of hypoxia. n = 5–9 per group. *P < 0.05, **P < 0.01, ***P < 0.001, n.s.: not significant.
Compared to Nx controls, SuHx-PH rats treated with PBS had a significant increase in pulmonary vascular remodeling as demonstrated by a greater than two-fold increase in MI (0.6500 ± 0.0328, vs. 0.3176 ± 0.0238 P < 0.0001) (Fig. 3). However, there was no significant difference in MI between Nx controls and SuHx-PH rats treated with MSC EV (0.318 ± 0.024 vs. 0.484 ± 0.056, P = N.S.). The MI was lower in SuHx rats treated with MSC EV than in those treated with PBS alone, but the difference was not statistically significant (0.484 ± 0.056 vs. 0.650 ± 0.033, P = N.S).
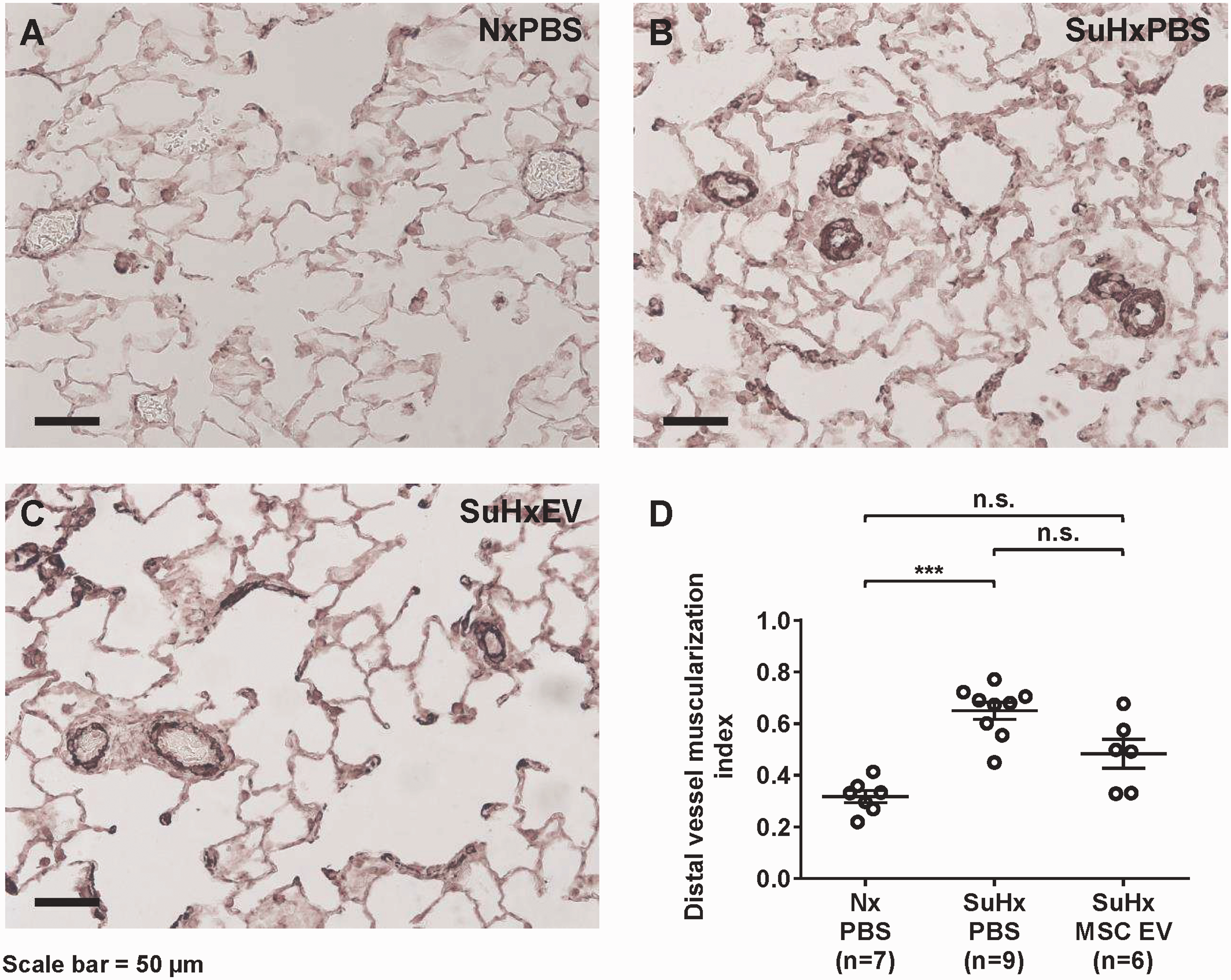
Lung sections stained with an antibody against α-smooth muscle actin showing muscularization (dark brown) of peripheral pulmonary vessels from rats treated with phosphate buffered saline (PBS) or mesenchymal stem cell extracellular vesicles (MSC EVs) once weekly for five weeks following treatment with Sugen 5416 and three weeks of hypoxia (SuHx). (a) Normoxic controls (Nx) treated with PBS. (b) SuHx treated with PBS. (c) SuHx treated with MSC EV. (d) Muscularization of vessels ≤50 µm assessed as total area of vessel staining positive for α-smooth muscle actin divided by total cross-sectional area of the vessel. The NIH ImageJ program was used to assess vessel areas. n = 6–9 per group. ***P < 0.001, n.s. not significant.
In the 10-week study, the severity of PH in the SuHx-PH rats treated with PBS alone was not as great as that seen in the five-week study; however, RVSP and RV/LV+S were significantly increased more than two-fold higher than in control mice (Fig. 4). As in the five-week study, RVSP and RV/LV+S in SuHx-PH rats treated with MSC EV were not significantly higher than in the Nx controls (31.8 ± 3.4 vs. 22.9 ± 2.4 mmHg and 0.367 ± 0.033 vs. 0.263 ± 0.007, P = N.S. for both). RVSP and RV/LV+S were lower in rats given MSC EV than those treated with PBS alone, but the differences did not reach statistically significance (31.8 ± 3.4 vs. 43.1 ± 3.6 mmHg and 0.367 ± 0.033 vs. 0.434 ± 0.043, respectively, P = N.S. for both). Pulmonary vascular remodeling was seen in the SuHx-PH rats treated with PBS as evidenced by higher MI than in Nx controls (0.5503 ± 0.0383 vs. 0.3475 ± 0.0150, P < 0.001) (Fig. 5), but no significant increase in MI was seen between Nx controls and SuHx-rats treated with MSC EV. MI was lower in SuHx-rats treated with MSC EV than SuHx-PH rats given PBS alone, but the difference was not significant (0.436 ± 0.027 vs. 0.550 ± 0.038, P = N.S.) (Fig. 5).
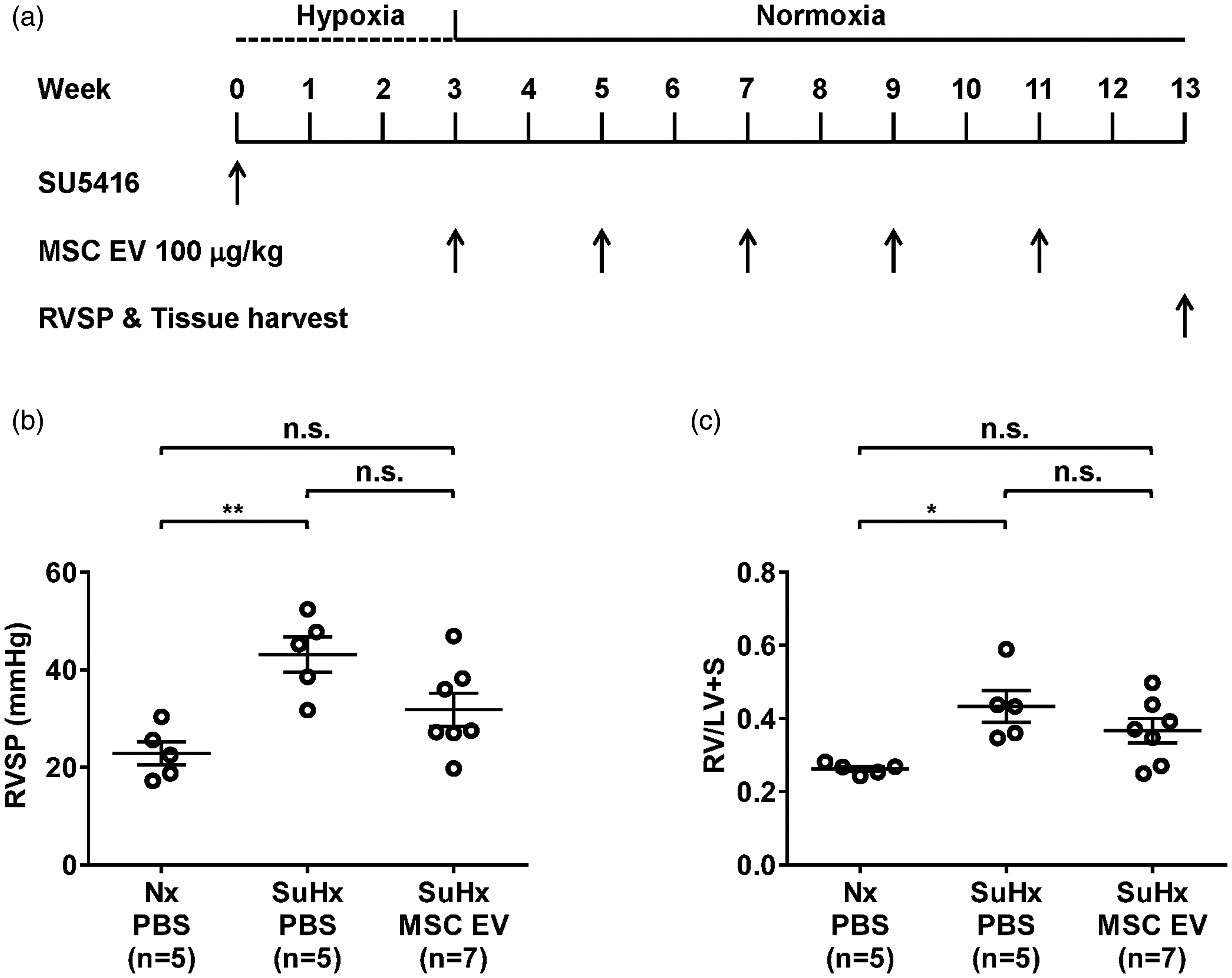
(a) Experimental protocol for reversal of pulmonary hypertension showing timing of administration of mesenchymal stem cell extracellular vesicles (MSC EVs) or phosphate buffered saline vehicle (PBS). (b) Right ventricular systolic pressure (RVSP) and (c) right ventricle to left ventricle + septum ratio (RV/LV+S) were measured 10 weeks after removal from three weeks of hypoxia (end of week 13 of experimental protocol). Nx PBS: normoxic PBS control; SuHx: rats treated with Sugen 5416 and exposed to three weeks of hypoxia. n = 5–7 per group. *P < 0.05, **P < 0.01, n.s.: not significant.
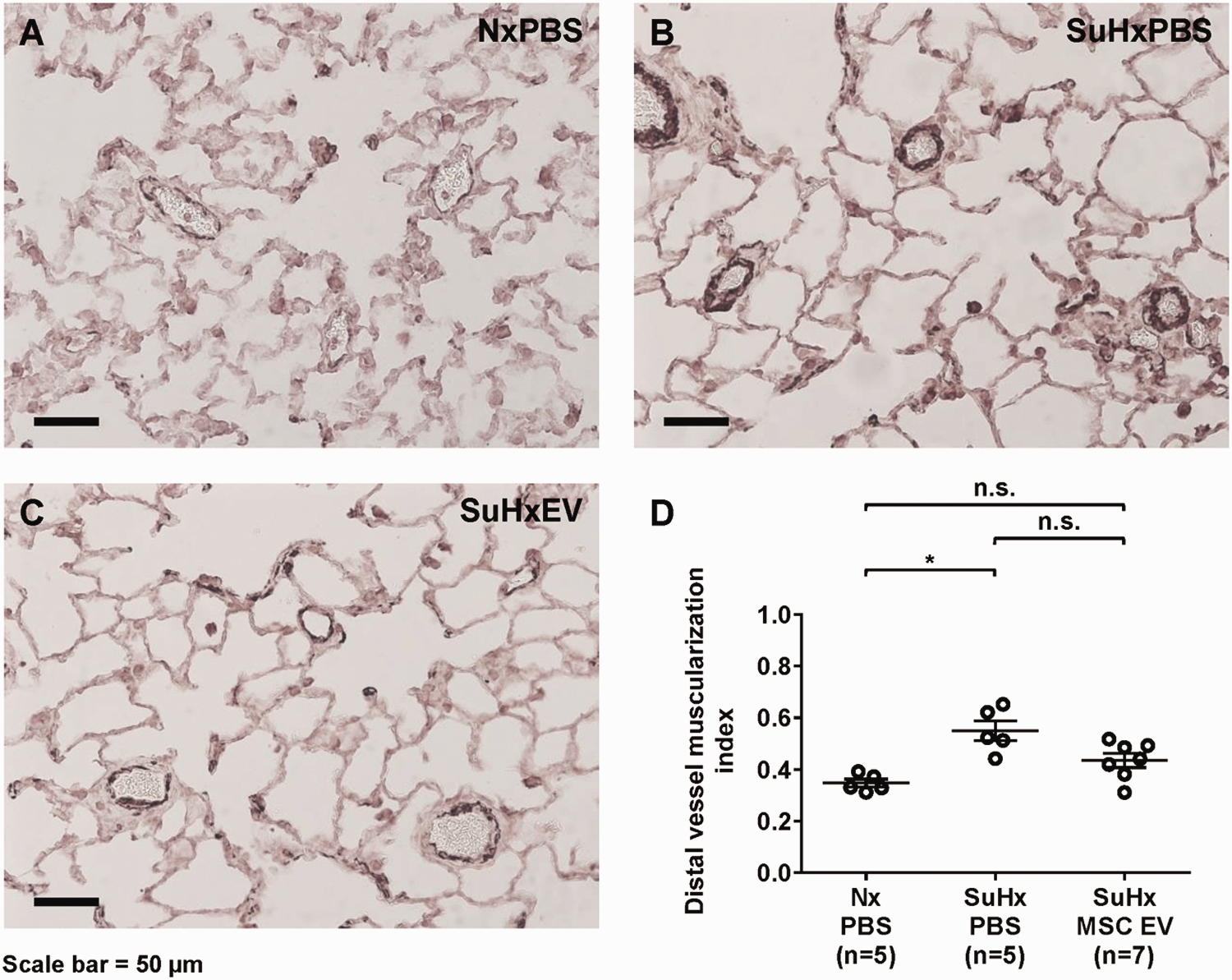
Lung sections stained with an antibody against α-smooth muscle actin showing muscularization (dark brown) of peripheral pulmonary vessels from rats treated with phosphate buffered saline (PBS) or mesenchymal stem cell extracellular vesicles (MSC EVs) once every two weeks for 10 weeks following treatment with Sugen 5416 and three weeks of hypoxia (SuHx). (a) Normoxic controls (Nx) treated with PBS. (b) SuHx treated with PBS. (c) SuHx treated with MSC EV. (d) Muscularization of vessels ≤50 µm assessed as total area of vessel staining positive for α-smooth muscle actin divided by total cross-sectional area of the vessel. The NIH ImageJ program was used to assess vessel areas. n = 5–7 per group. *P < 0.05, n.s.: not significant.
Effect of hypoxic stress on MSC EV efficacy
To determine if the efficacy of MSC EV on reversing PH could be enhanced by stressing MSC, additional studies were conducted comparing the effects of standard MSC EV that are collected from MSC grown under Nx conditions (NxEV) to those of EVs collected from MSCs that had been stressed by exposure to 48 h of hypoxia prior to collection (HxEV). The concentration of EVs in pooled culture media was greater in MSC exposed to hypoxia than in MSC kept in Nx conditions (protein concentration = 0.273 vs. 0.497 mg/ml, total EV number measured by Nanosight reading: 4.68 × 1011 vs. 6.88 × 1011, for Nx and hypoxia-stressed MSCs, respectively). SuHx-PH rats were given once daily injections of NxEV or HxEV at a dose of 20 µg/kg or an equal volume of PBS alone for three days starting the day after removal from three weeks of hypoxia. Severity of PH was assessed two weeks after removal from hypoxia (Fig. 6a).
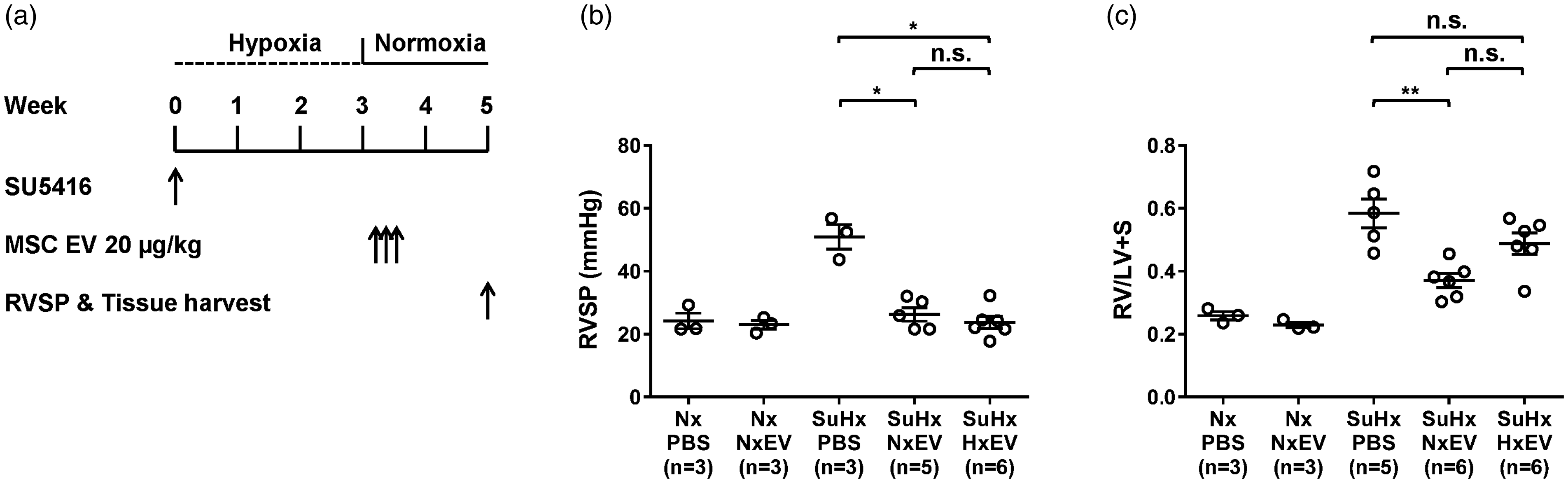
(a) Experimental protocol for reversal of pulmonary hypertension showing timing of administration of mesenchymal stem cell extracellular vesicles (MSC EVs) or phosphate buffered saline vehicle (PBS). Rats were left in normoxic conditions (Nx) or treated with Sugen 5416 followed by three weeks of hypoxia (SuHx) before receiving PBS or EV isolated from normoxic or hypoxia-stressed MSCs (NxEV, HxEV). (b) Right ventricular systolic pressure (RVSP) and (c) right ventricle to left ventricle + septum ratio (RV/LV+S) measured two weeks after removal from three weeks of hypoxia (end of week 5 of experimental protocol). n = 3–6 per group. *P < 0.05, **P < 0.01, n.s.: not significant.
As in the other experiments, SuHx-PH rats treated with PBS had a greater than two-fold increase in RVSP (51.0 ± 3.8 vs. 24.2 ± 2.5 mmHg, P < 0.001) and RV/LV+S (0.5843 ± 0.0463 vs. 0.2587 ± 0.0129, P < 0.001) compared to Nx controls (Fig. 6b and c). Both NxEV and HxEV reduced RVSP to baseline levels making it difficult to exclude the possibility that HxEVs were more effective than NxEVs at lower pulmonary artery (PA) pressure. However, RV/LV+S was significantly lower in rats given NxEV compared to SuHx-PH rats treated with PBS, but RV/LV+S was not significantly reduced in rats treated with HxEV (0.4879 ± 0.0341 vs. 0.5843 ± 0.0463, P = N.S.). In the SuHx-PH rats, both NxEV and HxEV reduced pulmonary vascular remodeling as evidenced by a significant reduction in MI compared to rats treated with PBS alone (0.4651 ± 0.0210 and 0.4706 ± 0.0248 vs. 0.7184 ± 0.0276, respectively, P < 0.05 for both) (Fig. 7), but there was no difference between rats treated with NxEV vs. HxEV.
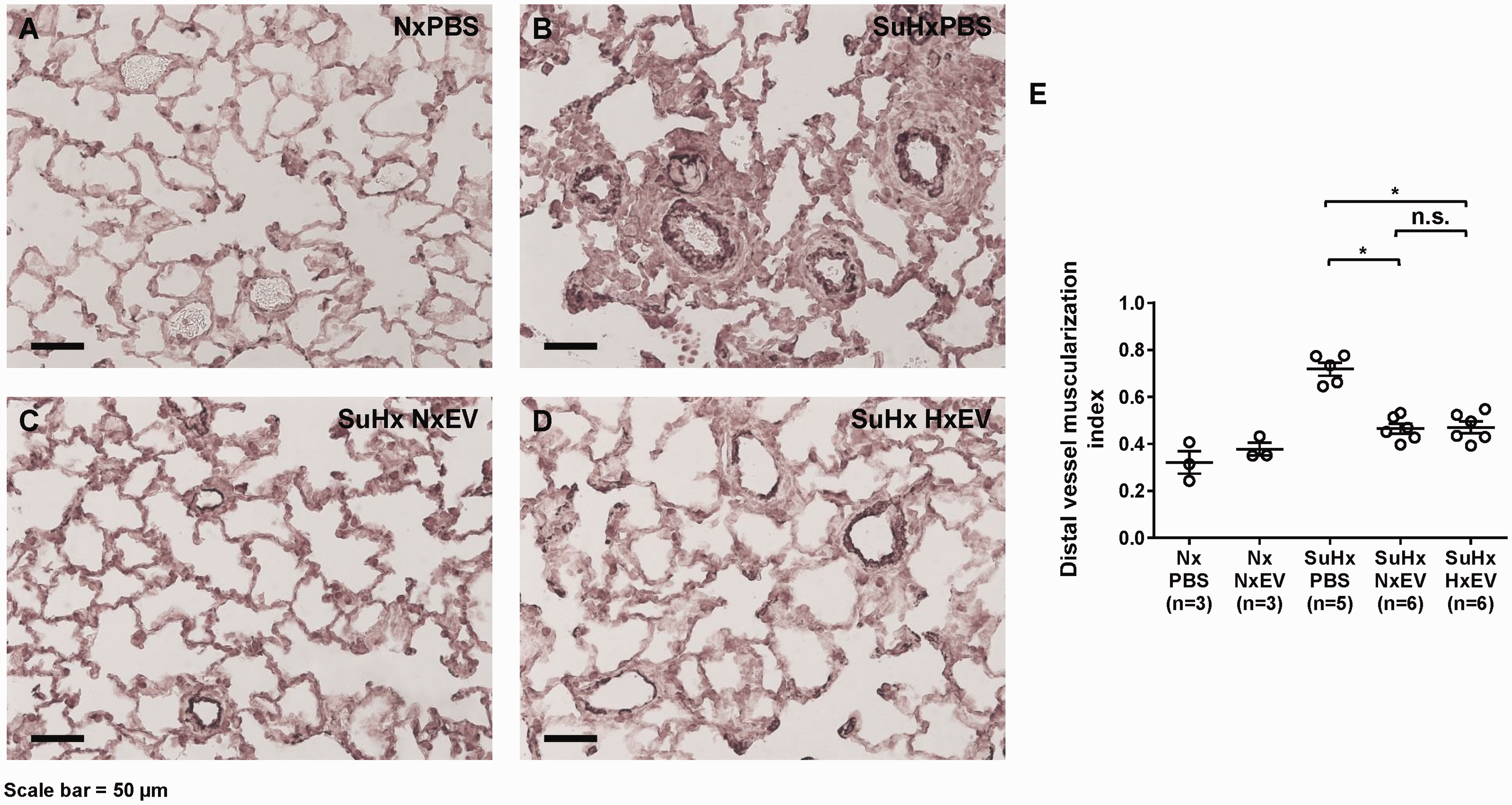
Lung sections stained with an antibody against α-smooth muscle actin showing muscularization (dark brown) of peripheral pulmonary vessels from rats treated with phosphate buffered saline (PBS) or EV isolated from normoxic or hypoxia-stressed MSCs (NxEV, HxEV) for three days following treatment with Sugen 5416 and three weeks of hypoxia (SuHx). Control rats were kept in normoxic conditions and treated with PBS or NxEV. (a) Normoxic controls (Nx) treated with PBS. (b) SuHx rats treated with PBS. (c) SuHx rats treated with NxEV. (d) SuHx rats treated with HxEV. (e) Muscularization of vessels ≤50 µm assessed as total area of vessel staining positive for α-smooth muscle actin divided by total cross-sectional area of the vessel. The NIH ImageJ program was used to assess vessel areas. n = 3–6 per group. *P < 0.05, n.s.: not significant.
Discussion
Although several studies have now demonstrated the efficacy of MSC EV in blunting and reversing PH in several pre-clinical models, few data exist regarding what dose of MSC EVs must be given or how often they must be given to achieve and maintain this effect. Furthermore, to our knowledge, no studies have examined the effect of MSC EVs on PH for more than four weeks after administration (Table 1). This information is important for the development of MSC EV as a therapeutic agent for the treatment of PAH in human subjects. In previous studies, we found that MSC EVs were effective at reversing SuHx-PH in rats when given at a doses of 5, 20, or 100 µg/kg once daily for three days starting the day after removal from hypoxia. 22 Interestingly, these doses were 1/200, 1/50, and 1/10 of the dose we had used to reverse PH in mice treated with monocrotaline. 27 In the present study, we did not see a significant reduction in RVSP or RV/LV+S in rats given MSC EV at the 5 µg/kg dose, although there was a strong trend toward a decrease and the mean values of RVSP and RV/L+S were nearly identical to those seen in the statistically significant 20 and 100 µg/kg treatment groups. At doses lower than 5 µg/kg, however, there was a strong trend toward reduced efficacy. Thus, the minimum effective dose of MSC EV on reversing PH in SuHx-PH rats appears to be in the 5–20 µg/kg range which is considerably lower than doses that we and others have used in previous studies22–24,27,29–31 (Table 1).
Previous studies evaluating the effect of mesenchymal stem cell-derived extracellular vesicles on pulmonary hypertension.
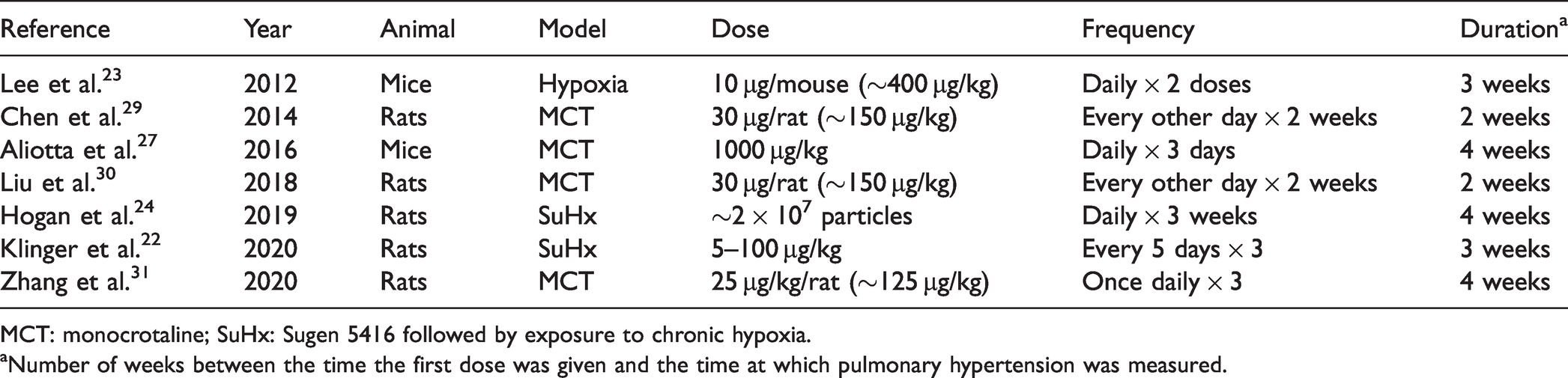
MCT: monocrotaline; SuHx: Sugen 5416 followed by exposure to chronic hypoxia.
aNumber of weeks between the time the first dose was given and the time at which pulmonary hypertension was measured.
In addition to determining if MSC EV could be effective at reversing SuHx-PH when given at lower doses, we also sought to determine if they could reverse PH if given less frequently. In our previous studies, 22 we found that once daily injection of MSC EV at 100 µg/kg for three days significantly decreased RVSP and RV/LV+S for up to two weeks after the first dose was given. We also found that administration of the same dose once daily every fifth day was equally effective at reversing PH for up to two weeks. However, when we examined pulmonary hypertensive changes three weeks after giving MSC EV once daily for three days, RV/LV+S was no longer significantly reduced compared to SuHx-PH rats given PBS alone, although RVSP remained significantly reduced and unchanged from Nx controls. 22 Other investigators have given MSC EV daily or every other day (Table 1).
In the present study, we demonstrated that MSC EV given once weekly significantly reduced RVSP and prevented the increase in RV/L+S and muscularization of pulmonary arteries as assessed by MI in the SuHx-PH rats for up to five weeks after removal from hypoxia, suggesting that their beneficial effect can be maintained for an extended period of time when given once weekly. In fact, beneficial effects of MSC EV were seen for up to 10 weeks in SuHx-PH rats treated with 100 µg/kg MSC EV every other week. In these studies, RVSP and RV/LV+S and the pulmonary vascular MI were not significantly lower in SuHx-PH rats treated with MSC EV than in rats given PBS vehicle alone, but none of these variables were significantly increased compared to nomoxic controls in the MSC EV group, whereas all of these indices of PH were significantly elevated in the SuHx-PH rats treated with PBS alone. It is possible that the loss of a significant difference between treated and untreated animals was due to an attenuation in the degree of RVSP and RV/LV+S in the untreated rats by week 10. In the SuHx-PH rats given PBS alone, RVSP at week 5 was 75.7 ± 7.2 mmHg compared to 43.1 ± 3.6 at week 10. Similarly, RV/LV+S was 0.647 ± 0.063 in the PBS-treated rats at week 5, but only 0.4335 ± 0.0431 at week 10. The reduced severity of the degree of PH by week 10 may have resulted in our study being under powered to detect significant differences in RVSP, RV/LV+S, and MI between SuHx-PH rats treated with MSC EV vs. those treated with PBS alone. It is also possible that significant differences in RVSP, RV/LV+S, and MI between rats given MSC EV and those given PBS would have been seen if they were treated every week in the 10-week study instead of every other week. Although the minimally effective dosing interval and the maximum duration of effect remain uncertain, the results from the present study suggest that MSC EV, given at a dosing interval of every 1–2 weeks are capable of attenuating pulmonary hypertensive and vascular remodeling in the SuHx model of PH for a considerably longer period of time than the 2–4 weeks that have been previously studied (Table 1). These findings suggest that the beneficial effects of MSC EV treatment can be sustained long term and support the development of MSC EVs as a potential new therapy for the treatment of PAH.
Another approach to improving the efficacy of MSC EVs in the treatment of PH is to enhance the potency of the EVs that are used. Data have been accruing to suggest that the anti-inflammatory effects of MSC EV can be increased by stressing MSC prior to EV harvest. Lo Sicco et al. 26 reported that exposure of adipose-derived MSC to 1% oxygen for 48 h prior to EV harvest increased the quantity of EVs recovered by about 30%. They also reported that EVs isolated from hypoxic MSC had significantly altered expression of 68 microRNAs compared to EVs isolated from Nx MSC EV including many that have been implicated in inflammation, proliferation, and differentiation pathways. Furthermore, they found that injection of HxEV into areas of cardiotoxin-induced muscle injury increased the number of M2 macrophages compared to injection with NxEV. Considering the increase in the number of lung M2 macrophages that we observed in our previous study of SuHx-PH rats treated with MSC EV, 22 we questioned whether administration of HxEV would have an enhanced effect at reversing PH. In the present study, we found that hypoxic stress increased the number of EVs in the culture media of MSC as previously reported. HxEVs were just as effective at reversing the SuHx-induced increase in RVSP as NxEV. Whether or not HxEVs were more effective than NxEVs could not be determined as both types of MSC EVs resulted in complete normalization of RVSP two weeks after administration. However, there was no indication that HxEVs were any more effective than NxEVs at reversing right ventricular hypertrophy as assessed by RV/LV+S or muscularization of peripheral vessels as assessed by MI. In fact, RV/LV+S tended to be higher in SuHx-PH rats treated with HxEV than in those treated with NxEV and was not significantly lower in SuHx-PH rats treated with HxEV than in SuHx-PH rats given PBS alone. Thus, our findings do not suggest that exposing MSC to hypoxic stress prior to harvesting EVs results in an EV preparation that is more efficacious for reversing SuHx-PH.
Our findings are limited to the preparation of MSC EVs that were used in this study. We isolated EVs from MSC culture media by high-speed centrifugation as described in our previous studies.22,27 This technique results in the collection of EVs of all sizes and does not exclude contamination by large proteins in the culture media. However, our results are very similar to those of other investigators who have examined the effect of only the exosome fraction of MSC EVs on reversing PH in rats using the SuHx-P model. 24 Those studies used MSC EVs that were isolated using size exclusion chromatography instead of ultracentrifugation. Thus, we believe that the effects observed in the present study are mediated by MSC EVs and not by other components of the culture media that may be associated with the pelleted fraction of EVs. Finally, the present study was performed in adult, male rats and thus we cannot exclude the possibility of sex-related differences in response to MSC EVs.
Summary
The findings from the present study add to the growing body of evidence that MSC EVs are highly effective at reversing increases in pulmonary arterial pressure, RV hypertrophy, and pulmonary vascular remodeling in rodent models of PH. Our findings demonstrate that the dose of MSC EV needed to achieve these effects is considerably lower than what has been used in previous studies. Furthermore, the ability of MSC EV to reverse PH can be sustained for at least five weeks after starting treatment in the SuHx-PH rat model when given at weekly intervals and some attenuation of pulmonary vascular remodeling can be seen as long as 10 weeks after starting treatment when given every two weeks. We found no evidence that the ability of MSC EV to reverse PH can be enhanced by hypoxic stress of the MSCs prior to EV collection. Together, these findings suggest that MSC EVs are effective at long-term reversal of PH and have the potential to be developed as a unique therapy for treatment of pulmonary vascular disease.
Supplemental Material
sj-pdf-1-pul-10.1177_20458940211046137 - Supplemental material for Effect of dose, dosing intervals, and hypoxic stress on the reversal of pulmonary hypertension by mesenchymal stem cell extracellular vesicles
Supplemental material, sj-pdf-1-pul-10.1177_20458940211046137 for Effect of dose, dosing intervals, and hypoxic stress on the reversal of pulmonary hypertension by mesenchymal stem cell extracellular vesicles by James R Klinger, Mandy Pereira, Michael Del Tatto, Mark S Dooner, Sicheng Wen, Peter J Quesenberry and Olin D Liang in Pulmonary Circulation
Footnotes
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JRK’s institution received financial support for this study from United Therapeutics as an investigator sponsored study. JRK’s institution also receives support from Lung International, a subsidiary of United Therapeutics for his role as the principle site investigator for a clinical trial in PH. The other authors declare that there is no conflict of interest.
Authors’ contributions
JRK: Substantial contributions to study conception and design, analysis and interpretation of data, drafting and revision and final approval of manuscript and agreement to be accountable for all aspects of the work. MP, MDT, MSD, SW: Contributions to study design, acquisition, analysis and interpretation of data, revision and final approval of manuscript. PJQ: Substantial contributions to study conception, interpretation of data, revision and final approval of manuscript and agreement to be accountable for all aspects of the work. ODL: Substantial contributions to study conception and design, data acquisition, analysis and interpretation of data, revision of final manuscript and final approval of manuscript, and agreement to be accountable for all aspects of the work.
Ethical approval
This study was approved by the Lifespan Animal Welfare Committee at Rhode Island Hospital (Institutional Animal Care and Use Committee # 5008-19 and #5007-20).
Guarantor
JRK and ODL.
Acknowledgements
We would like to thank the Flow Cytometry Core and the Vesicle Core of the COBRE Center for Stem Cells and Aging at Rhode Island Hospital and Brown University.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants from the National Institutes of Health P20 GM119943, and the American Heart Association Transformational Project Award 18TPA34110329 and United Therapeutics Corporation.
Supplemental Material
Supplemental material for this article is available online.
