Abstract
Pulmonary hypertension (PH) is an incurable disease that often leads to right ventricular hypertrophy and right heart failure. This study investigated single versus combined therapy with sildenafil and erythropoietin on hypoxia-induced pulmonary hypertension in mice. Mice were randomized into 5 groups and exposed to either hypoxia (10% oxygen) or normoxia for a total of 5 weeks. Hypoxic mice were treated with saline solution, erythropoietin (500 IU/kg 3 times weekly), sildenafil (10 mg/kg daily), or a combination of the two drugs for the last 2 weeks of hypoxic exposure. We measured right ventricular pressures using right heart catheterization, and the ventilatory response to hypoxia was recorded via whole-body plethysmography. Histological analyses were performed to elucidate changes in pulmonary morphology and appearance of right heart hypertrophy. Plasma levels of cardiotrophin-1 and atrial natriuretic peptide were quantified. Treatment with either erythropoietin or sildenafil alone lowered the hypoxia-induced increase of pulmonary pressure and reduced pulmonary edema formation, pulmonary vascular remodeling, and right ventricular hypertrophy. Notably, the combination of the two drugs had the most prominent effect. Changes in cardiotrophin-1 and atrial natriuretic protein levels confirmed these observations. The combination treatment with erythropoietin and sildenafil demonstrated an attenuation of the development of hypoxia-induced PH in mice that was superior to that observed for either drug when given alone.
INTRODUCTION
Exposure to chronic hypoxia occurs not only in high-altitude residents but also in patients suffering from chronic diseases associated with a reduced oxygen transport capacity during wakefulness and/or sleep, such as chronic heart failure, chronic obstructive pulmonary disease, and sleep apnea.1–4 A frequently observed comorbidity in these patients is pulmonary hypertension (PH), a progressive and so far incurable chronic disease characterized by increased pulmonary vascular resistance, high pulmonary pressure, and remodeling of the pulmonary vessels. 5 Although PH is a disease of various origins,6,7 chronic hypoxia is considered a major trigger. 8
It is well accepted that phosphodiesterase-5 inhibitors, such as sildenafil, are able to improve hemodynamic parameters and clinical outcome in patients suffering from PH.5,9,10 The monotherapeutic onsets are still insufficient, however, especially for pulmonary vessel remodeling and long-term outcomes; as a result, combined treatment strategies have been adopted, with heterogeneous success.5,11,12 Erythropoietin (Epo) is a pleiotropic cytokine that is upregulated during prolonged alveolar hypoxia and causes increased erythropoiesis. 13 Ultimately, Epo improves the blood's oxygen-carrying capacity, helping to cope with reduced oxygen uptake. 14 Of note, Epo also has several non-erythropoietic functions 15 and has been shown to generate tissue-protective effects in various tissues, such as skin, 16 retina, 17 neuronal, 18 and heart tissue. 19 In addition, protective effects of Epo in hypoxia-induced PH have been indicated in Epo-overexpressing (tg6) mice 20 and in mice deficient in the Epo receptor, 21 but not in two rat models.22,23
In this study, we tested the hypothesis that combined treatment with the vasodilator sildenafil and recombinant human Epo may have superior effects on hypoxia-induced PH, as compared to treatment with each drug alone. The primary endpoint of the study was right ventricular pressure (RVP), and secondary endpoints were morphology of the right heart ventricle and remodeling of the pulmonary arterial vessels. Using this approach, we were able to demonstrate synergistic effects of the two drugs in combination, as both the primary and secondary endpoints were significantly improved upon cotreatment with Epo and sildenafil, compared to results with single treatments.
MATERIAL AND METHODS
Animals and hypoxic treatment
Wild-type mice (C57BL/6) were randomized, first into normoxic and hypoxic groups and second to receive saline, Epo, sildenafil, or a combined (Epo and sildenafil) treatment via subcutaneous injections (n = 6). Humanz recombinant Epo (Eprex, Janssen-Cilag) was injected 3 times per week (500 IU/kg of body weight [BW]), and sildenafil (a kind gift from Pfizer) was injected daily (10 mg/kg of BW). The animals were exposed to 3 weeks of either hypoxia (10% oxygen) or normoxia (room air), after which the animals received the different drugs for additional 2 weeks under the same environmental conditions. This hypoxic mouse model for PH is well established, has been described by several groups,20,24–26 and is known to result in PH after 3 weeks of hypoxic exposure.
The normobaric hypoxic chamber was opened for 1 hour once per week to replace bedding, water bottles, and chow. Mice of both sexes (10–12 weeks old) were used, and all experiments were homogeneous regarding sex. In total, 96 animals were used. Experiments were approved by the Kantonales Veterinαr Amt in Zurich, Switzerland.
Behavioral observations and spontaneous activity
The animals were exposed to hypoxia for 3 weeks without treatment, followed by an additional 2 weeks of hypoxia with treatment. Three days before the end of the treatment period, a video camera was installed to record the spontaneous activity of each group of animals. The camera was recording from 9 PM to 1 AM, i.e., the most active phase; later, the behavior and movement of the groups were compared. An explanatory video is available online.
RVP, blood analysis
The RVP and venous blood gases were measured in anesthetized mice (2%–2.5% isoflurane in 100% oxygen) at the end of the 5 weeks of exposure. A small skin incision was made in the neck of the mice, and the right external jugular vein was isolated to insert a polyethylene (PE 10) catheter and forward it to the right ventricle of the heart. In each animal, the blood pressure within the right ventricle was continuously recorded at a 1-kHz sampling frequency for at least 30 seconds with a piezoelectric pressure transducer and the PowerLab system (ADInstruments). The correct anatomical position of the catheter tip was controlled by continuously monitoring the pressure signal curve. After recording, a 100-αL venous blood sample was drawn from the right ventricle via the catheter for blood gas analysis (AVL700 Radiometer, Copenhagen, Denmark). After blood collection, the animals were euthanized by cervical dislocation.
Histology
Organs (hearts and lungs) were removed and placed in 4% formalin for 24 hours before being embedded in paraffin blocks. Slices 5 αm thick were cut and stained with eosin and hematoxylin (HE).
Pulmonary vessel wall thickness was measured with image-analyzing software (MCID 7.0, Image Research, St. Catharines, Ontario, Canada). Pulmonary vessels near the alveoli were evaluated, and the diameters of approximately 20 vessels per slide were determined. From each animal, 5 slides were evaluated. Simultaneously, we assessed the number of occluded vessels per field (microscope magnification: 40 α 10 = 400-fold). From each HE-stained slide approximately 20 peribronchial pulmonary artery vessels were evaluated (microscope magnification: α400). The degree of occlusion was determined as the ratio between the outer circle and inner (i.e., luminal) circles of each vessel.
Right ventricular cardiac myocytes displaying central nuclei were selected for measurement of the cross-sectional area via the MCID image-analyzing system. For each heart 4 slides were obtained, and for each slide approximately 20 cardiomyocytes were measured. Right ventricular hypertrophy was also quantified with the MCID software. To this end, sections were acquired with a densitometry camera (CoolSNAP cf, Roper Scientific [Photometrics], Ottobrunn, Germany) after the image size of the chosen magnification had been calibrated against the image of a ruler acquired with the identical camera settings. With the drawing tool of the image analyzer, the left and right ventricles were separated manually with a thin line in the same way for all sections by a blinded investigator. Then the areas of the right and the left ventricles could be detected and measured automatically by the image-analyzing system on the basis of the differences in optical density between background and stained tissue. The results are presented as a ratio, RV/(LV + S), where RV is right ventricle, LV is left ventricle, and S is septum. From each animal, 5 slides were evaluated.
Plasma Epo levels
The plasma Epo levels in adult mice were measured by a specific enzyme-linked immunosorbent assay (ELISA) assay (mouse erythropoietin immunoassay; R & D Systems Europe, Abingdon, UK) according to the manufacturer's recommendations. The mean detection limits of Epo were 7.8 and 1,000 pg/mL. All samples were assayed in duplicates and normalized to protein concentration, which was determined by the bicinchoninic acid (BCA) protein assay reagent (Pierce Biological, Rockford, IL). Epo was measured at the end of the 5-week experiment.
Hematocrit
Hematocrit was determined by a routine capillary centrifugation, which allows for determination of the volume fraction of erythrocytes in blood (Haematokrit 24, Hettich, Tuttlingen, Germany). Values are expressed as the percentage of the volume of erythrocytes to the total blood volume. Blood for hematocrit measurements was collected at the end of the experiments.
Determination of ventilation
Ventilation parameters were monitored by whole-body flow-through plethysmography (Emka Technologies, Paris), as previously described.27,28 Briefly, mice were placed in a 600-mL chamber continuously supplied with a 0.7–0.8 L/min airflow controlled by flow restrictors. The ventilation of mice was recorded twice, before the interventions (exposure to hypoxia/drug treatment) and thereafter according to a standardized protocol: at least 5 minutes of recording at rest under normoxic conditions before the oxygen content within the chamber was gradually decreased (within 5 minutes) from room air to 10% oxygen, after which ventilation was recorded for another 10 minutes. The respiratory data were recorded and analyzed by use of IOX-2 software (Emka Technologies). The minute ventilation (VE) was calculated as the product of the breathing frequency (fB) and tidal volume (VT) and normalized to 100 g BW (mL/min/100 g BW).
Pulmonary edema
Lungs were excised from the animals and separated into individual lobes. The weight of a lung lobe was determined before and after heat drying at 70αC for 72 hours. Results are expressed as a ratio of wet weight to dry weight.
Cardiotrophin-1 and atrial natriuretic protein (ANP)
Cardiotrophin-1 and ANP were measured in serum by ELISA kits (ELM-Cardiothrophin-1/EIA-ANP-1, RayBiotech, Norcross, GA), according to the manufacturer's instructions.
Data management and statistics
Data are expressed as means ± SD of the indicated number of observations. Statistical comparisons between groups were performed via one-way ANOVA with a Newman-Keul's posttest. For the ventilatory data, the percentage change in normoxic and hypoxic VE from before until after 5 weeks of intervention was calculated for different groups and plotted (Fig. S1, available online). The percentage changes of VE under normoxic and hypoxic conditions were compared between groups via non-parametric statistical tests (Kruskal-Wallis rank-sum test, Mann-Whitney U test). Differences were considered significant when P < 0.05.
RESULTS
Observations in hypoxic mice
With decreasing oxygen concentration inside the hypoxic chamber, mice became progressively lethargic within 1 hour. At 10% O2, the animals were resting motionless (e.g., paused eating and drinking and stopped grooming), using their efforts mainly for deep breathing, and the skin color of their paws and nose turned cyanotic. However, the animals' behavior improved over the next hours, and after 24 hours it was normalized, although the animals were still cyanotic and showed decreased climbing activity. At normoxia, the control animals gained approximately 5%–10% of their BW over the 5 weeks the experiment lasted (Table 1), with the group receiving sildenafil gaining significantly more than the other normoxic groups. The hypoxic animals lost almost 20% of their initial BW, and this was also the case for the animals receiving single therapy. For the hypoxic animals receiving the combination treatment, however, this weight loss was to a great extent omitted. Furthermore, the heart weight-to-BW ratio was increased in hypoxic animals compared to that in the normoxic groups, except for the animals receiving Epo and sildenafil simultaneously (Table 1).
Gained body weight (BW) and BW to heart weight (HW) ratio
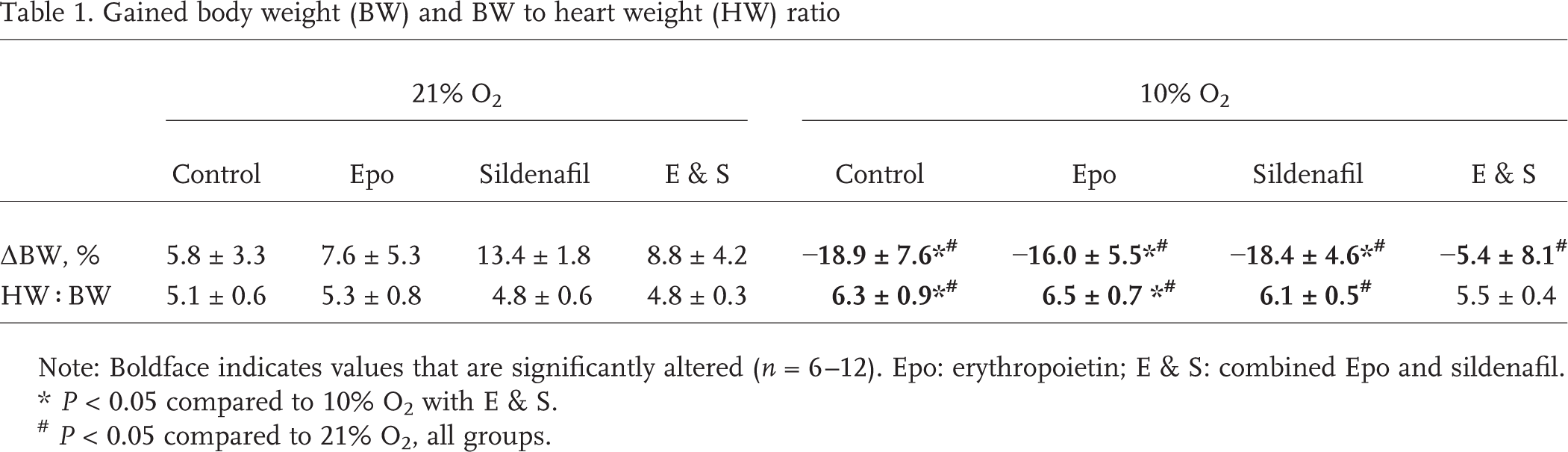
Note: Boldface indicates values that are significantly altered (n = 6–12). Epo: erythropoietin; E & S: combined Epo and sildenafil.
P < 0.05 compared to 10% O2 with E & S.
P < 0.05 compared to 21% O2, all groups.
Epo, hemoglobin, and hematocrit
Endogenous mouse Epo measured at the end of the experiment was elevated approximately 4-fold in plasma from hypoxic control animals, compared to that in normoxic mice (Table 2). As expected, hemoglobin levels were elevated after hypoxic exposure, and this was slightly, but significantly, attenuated by sildenafil as well as by the combination treatment In addition, hematocrit was increased in response to hypoxia but was unchanged with treatments, except for a small increase in the Epo-treated group. For all groups, there was a clear and significant sex difference (data not shown), with the hematological values for the female mice being approximately 25% lower than those for their male counterparts.
Erythropoietin (Epo) and hematocrit

Note: All parameters were measured in plasma. For hemoglobin, all normoxic values were significantly different from all hypoxic values (n = 6–12). Boldface indicates values that are significantly altered (n = 6–12). Epo(m): endogenous mouse Epo (n = 3).
E & S: combined Epo and sildenafil.
P < 0.05 compared to hypoxic control.
P < 0.05 compared to 10% O2 with E & S.
P < 0.05 compared to 10% O2 with sildenafil.
Venous blood analysis
Most of the measured plasma electrolytes were altered in response to hypoxia and returned to baseline with the combination treatment (Table S1, available online). Hypoxia caused a reduction in pH, which returned to normal values with the combination treatment. Lactate levels were increased under hypoxia; this was gradually attenuated by the single treatments, and levels returned to baseline with the combination treatment. With regard to base excess (BE) and bicarbonate (
Ventilation
The acclimatization to chronic hypoxia involved an elevation of ventilation (Fig. S1a). The percentage change in hypoxic VE (before vs. after 5 weeks of intervention; Fig. S1b) was increased in the Epo-treated animals, as compared to the hypoxic control group and the sildenafil group. The increase observed for the cotreated group was significant only when compared to that of the sildenafil group.
Hemodynamic measurements
Measurements of the RVP revealed that hypoxic animals had developed a severe increase in RVP, approximately 65%, after being exposed to hypoxia for 5 weeks (Fig. 1). This increase was reduced significantly by treatment with Epo and even more so with sildenafil. The combination treatment attenuated the pressure even further, thereby returning it almost, though not significantly, to baseline normoxic values.
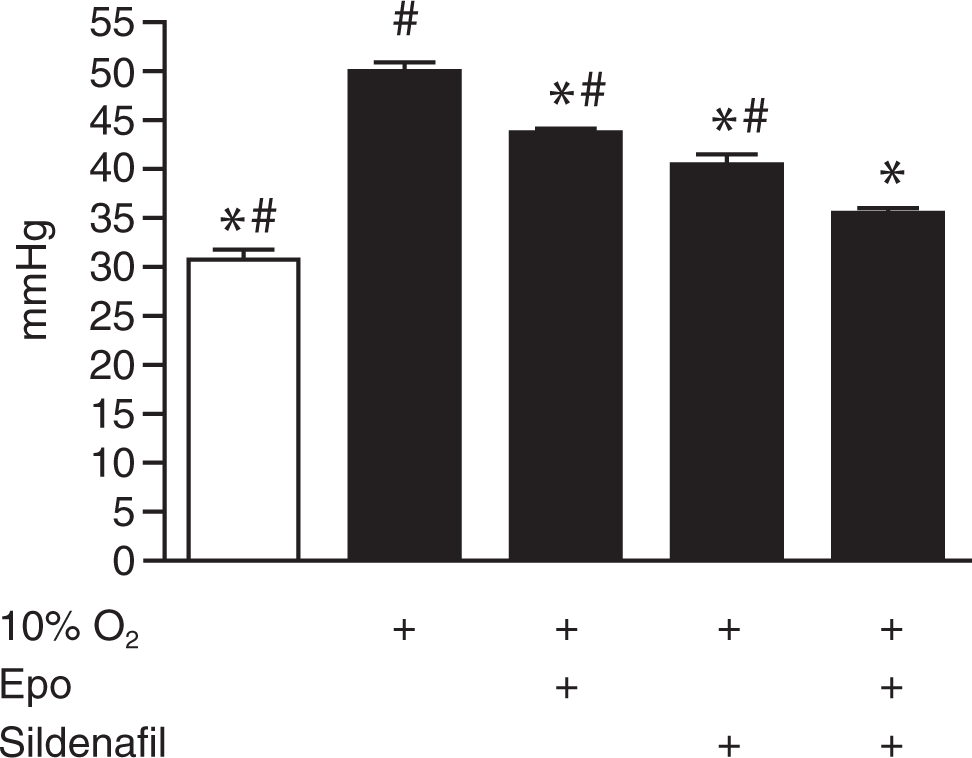
Hemodynamic measurements in anesthetized mice. Right ventricle pressure of different groups after 5 weeks of chronic 10% hypoxic exposure with or without treatment, compared to that of normoxic, untreated control mice (open bar); treatment groups, from left to right: hypoxia only, hypoxia and erythropoietin (Epo) alone, hypoxia and sildenafil alone, and hypoxia and combination of Epo and sildenafil (n = 5). An asterisk indicates P < 0.05 versus hypoxic control, and a pound sign indicates P < 0.05 versus hypoxia and cotreatment with Epo and sildenafil.
Pulmonary vascular remodeling
The histological stainings (Fig. 2A) clearly illustrate the increased arterial vessel wall diameters in lungs from animals exposed to hypoxia and also the modulating effect of different drug treatments. Pulmonary vessel wall thickness was evaluated (Fig. 2B). In normoxic animals (control and treatment groups), the vessel wall diameter was approximately 0.7 ± 0.07 αm. This rose to 1.86 ± 0.11 αm in the hypoxic control group but was gradually reduced with both Epo and sildenafil treatment and was reduced considerably more with the combination therapy. The percentage of occluded vessels (Fig. 2C) in lungs of normoxic animals was approximately 8 times that in the untreated hypoxic animals. Administering Epo, sildenafil, or both drugs simultaneously gradually lowered the number of occluded vessels.
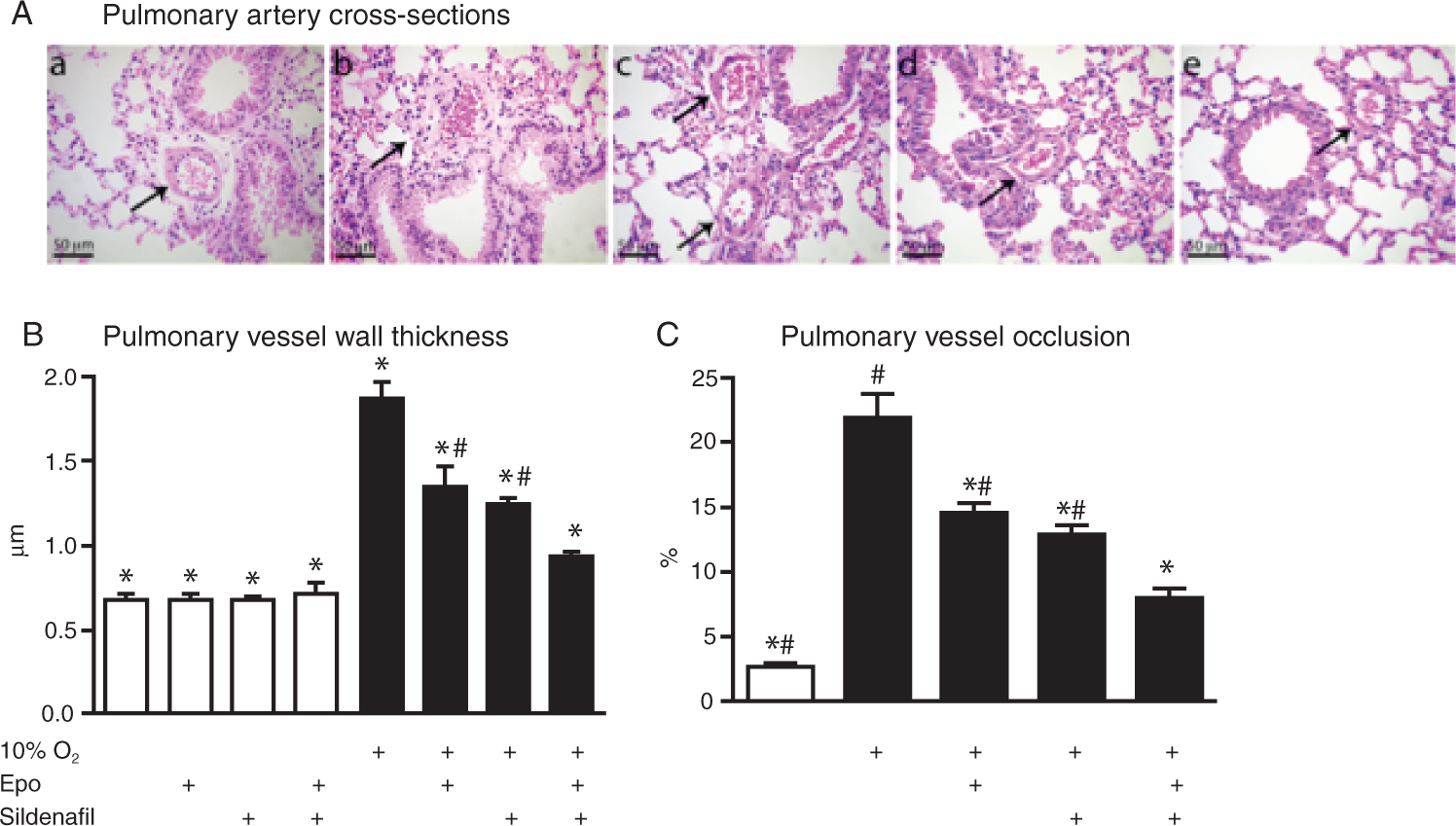
A, Representative images of small pulmonary arteries (50–200 αm) stained with eosin and hematoxylin (arrows), showing reduced hypoxia-induced remodeling of intimal and medial vessel layer upon treatment: a, normoxic control; b, hypoxic control; c, hypoxia and erythropoietin (Epo) alone; d, hypoxia and sildenafil alone; e, hypoxia and cotreatment with Epo and sildenafil. B, Pulmonary vessel wall thickness (n = 8); open bars for normoxic mice, filled bars for hypoxic mice. C, Pulmonary lung vessel occlusion (n = 8). Treatment groups in B and C are indicated below the bars, as in Figure 1. An asterisk indicates P < 0.05 versus hypoxic control, and a pound sign indicates P < 0.05 versus hypoxia and cotreatment with Epo and sildenafil.
Pulmonary edema
Pulmonary edema was estimated by the wet-to-dry lung weight ratio (Fig. 3). Under normoxic conditions there was no impact of the treatments on the intact lung. Hypoxic exposure for 5 weeks increased the wet-to-dry weight ratio of the untreated lung by about 28%. Edema formation was, however, reduced by both single treatments and returned to normoxic control levels under the combination of sildenafil and Epo.
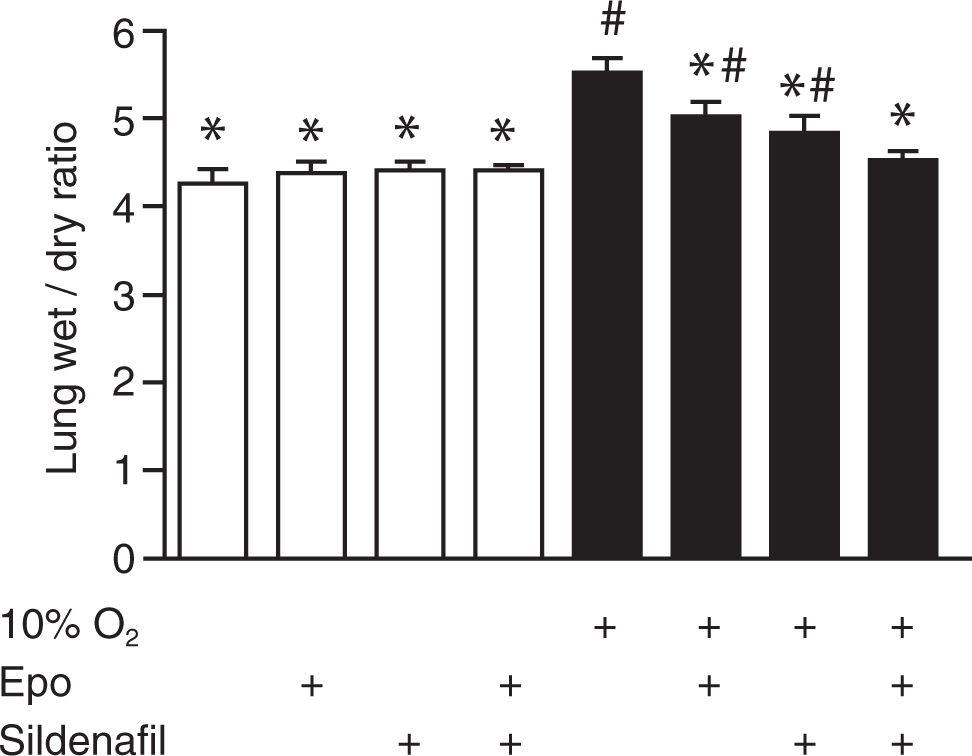
Quantification of pulmonary edema formation via estimation of lung wet-to-dry weight ratio, showing increased edema formation in mice chronically exposed to 10% oxygen for 5 weeks when compared to normoxic mice (open bars) with or without treatment and hypoxic mice with treatment (n = 3). Treatment groups are indicated below the bars, as in Figure 1. Epo: erythropoietin. An asterisk indicates P < 0.05 versus hypoxic control, and a pound sign indicates P < 0.05 versus hypoxia and cotreatment with Epo and sildenafil.
Right ventricular hypertrophy
Right ventricular hypertrophy (Fig. 4A) was assessed by histology. For the normoxic groups, control versus treatment, no change was detected in the RV/(LV + S) ratio, whereas an apparent enlargement of the RV after 5 weeks of hypoxia was observed (Fig. 4B). This enlargement was attenuated with both single treatments but significantly more with the combination of the two drugs.
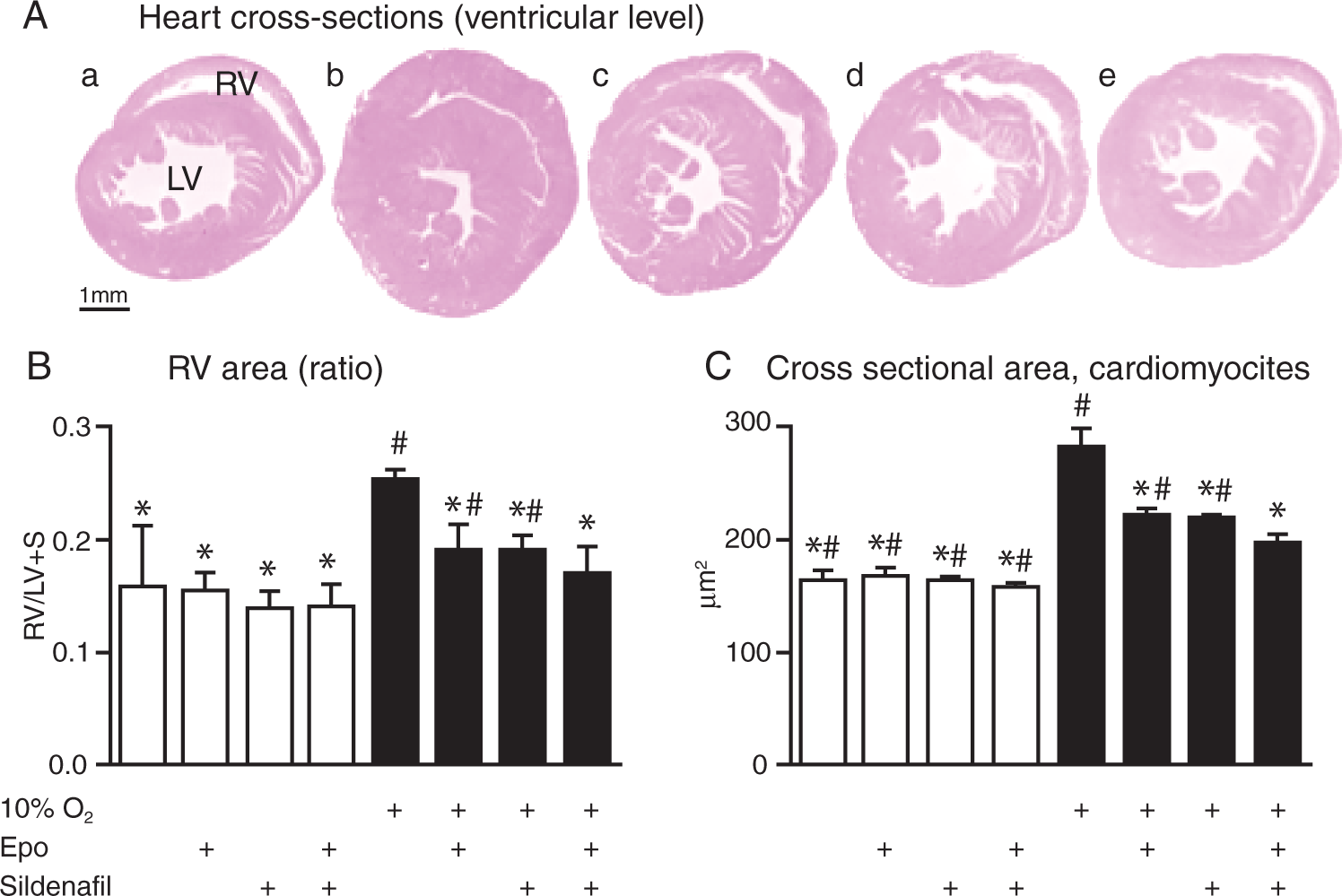
Different treatments prevent ventricular remodeling of the heart. A, Representative images of cross-sectional slices of the heart (horizontal cut through the mouse heart at ventricular level), showing reduced hypoxia-induced hypertrophic transformation of the right ventricle (RV) due to the treatments; LV = left ventricle: a, normoxic control; b, hypoxic control; c, hypoxia and erythropoietin (Epo) alone; d, hypoxia and sildenafil alone; e, hypoxia and cotreatment with Epo and sildenafil. B, Quantification of right ventricular hypertrophy via estimation of the ratio of RV area to (LV + septum) area (n = 4–6). C, Hypoxia-induced hypertrophy of the cardiomyocytes was affected significantly by the treatments (n = 4–6). Treatment groups in B and C are indicated below the bars, as in Figure 1. An asterisk indicates P < 0.05 vs. hypoxic control, and a pound sign indicates P < 0.05 vs. hypoxia and cotreatment with Epo and sildenafil.
The cross-sectional areas of the right ventricular cardiomyocytes were also determined (Fig. 4C), and again no change was observed in the normoxic groups, whereas almost a doubling was detected after hypoxic exposure. Epo or sildenafil alone could diminish the hypoxia-induced enlargement, and a further reduction was observed when both drugs were administered simultaneously.
Cardiotrophin-1 and ANP
The cytokines cardiotrophin-1 and ANP were measured in plasma obtained from all mouse groups at the end of the experiment (Table 3). Plasma levels of both factors were unaltered during normoxia but highly augmented under hypoxia. This augmentation was lowered with both single treatments and was lowered significantly more when Epo and sildenafil were given in combination.
Plasma values for cardiotrophin-1 (CT-1) and atrial natriuretic protein (ANP)

Note: Bold numbers represent values that are significantly altered. All normoxic values were significantly different from all hypoxic values (n = 5). Epo: erythropoietin; E & S: combined Epo and sildenafil.
P < 0.05 compared to hypoxic control.
P < 0.05 compared to 10% O2 with E & S.
DISCUSSION
In this study, we found convincing evidence for additive effects when combining the two drugs sildenafil and Epo for the treatment of hypoxia-induced PH in a mouse model. All endpoints improved after 2 weeks of single treatments and to an even higher extent by simultaneous application of both drugs.
Observations in hypoxic mice
Mice recovered within the first 24 hours of hypoxic exposure, so that they restarted eating, drinking, and interacting socially. Clear differences were observed, however, in overall movement and the intensity thereof. In general, untreated control animals appeared fatigued and moved in the hypoxic chamber only in order to acquire food or water. In the groups receiving treatment, however, an apparent increase (not quantified) in the ability to move and interact was observed, and this was especially clear for the group receiving combined treatment (see the video available online). On the basis of these observations, our initial conclusion was that treatment with sildenafil alone improved the overall health status and that this effect was augmented in combination with Epo. This was further supported by the fact that hypoxia-induced weight loss was alleviated by the combination treatment.
Effects on blood parameters
Hematocrit, as expected, increased with hypoxic exposure. Endogenous Epo also exhibited a tendency for a hypoxia-induced augmentation. Sildenafil treatment, alone as well as in combination with exogenous human Epo, was able to attenuate this increase. We suggest that the reason for this was an overall improved ventilation-perfusion match, leading to better systemic and tissue oxygenation that resulted in reduced endogenous Epo release and ultimately in lowering of hemoglobin and hematocrit levels.
Lactate levels were higher under hypoxia, because impaired cellular respiration leads to a switch to anaerobic glucose utilization, thereby increasing the production of lactate. 29 This phenomenon is known as lactic acidosis and will also contribute to a lower pH value. Indeed, we did observe that pH and lactate levels mirrored each other, with pH being lower in hypoxic control animals and lactate higher. Both values normalized in all hypoxic groups rethe pH level was even further increased. The rather low pH value in normoxic animals is most likely due to isoflurane-induced respiratory acidosis. 30
The concentration of several blood electrolytes was altered in hypoxic animals, and with treatments, especially the cotreatment, these values returned toward baseline levels. Electrolytes play a crucial part in maintaining homeostasis within the body. They are critical for the generation of energy, maintenance of cell wall stability, contraction of muscles, movement of water and fluids, and numerous other functions. Thus, preserving the electrolyte balance is of utmost importance.
Effects on ventilation
The VE of mice at rest was generally increased after chronic hypoxia for 5 weeks, a well-known mechanism to compensate for lower oxygen levels termed “ventilatory acclimatization to hypoxia.”31,32 The percentage change of hypoxic VE (before vs. after 5 weeks of hypoxia + treatment) was, in contrast to that in normoxic conditions, significantly altered within different groups. Of note, those animals receiving Epo (either alone or in combination with sildenafil) showed a significant increase in VE under hypoxic conditions, as compared to the untreated hypoxic control group and the sildenafil single-treatment group. Despite the fact that endogenous Epo is released under hypoxic conditions,33,34 this finding is in agreement with already published data demonstrating that exogenously applied Epo affects the peripheral cardiorespiratory network through the carotid bodies, by increasing hypoxic VE to ultimately improve oxygenation during hypoxia. 27 The observed stimulating effect of Epo treatment on VE was more evident when compared to results in the sildenafil single-treatment group, suggesting that Epo and sildenafil have counterbalancing effects on ventilation in hypoxia.
Effects on RVP
RVP is an indirect measure, but it represents a good estimate of the pulmonary artery pressure. 35 RVP was our primary endpoint, and as we hypothesized, the animals developed PH upon hypoxic exposure. RVP could be reduced by both Epo and, to a larger extent, sildenafil. Furthermore, we observed an additive effect of the two drugs when applied in combination. After only 2 weeks of treatment, while the animals were still exposed to hypoxia, RVP returned almost to normoxic baseline levels when the two drugs were administered in combination.
Effects on pulmonary vessels and lung edema formation
The remodeling of the pulmonary arterial vessels was assessed by wall thickness as well as by counting the number of occluded vessels. For both parameters, an increase was observed after 5 weeks of hypoxia. Either drug alone could diminish these alterations of the pulmonary vasculature, but the effect was significantly higher with the combination treatment. This was also the case for pulmonary edema formation, indicated by an increase in the wet-to-dry lung weight ratio upon hypoxia in the untreated control animals that was nearly abolished by the treatments.
Effects on RV morphology and parameters of heart function
RV hypertrophy was verified by histology, demonstrating attenuation in the hypoxia-induced increases in the RV/(LV + S) ratio and cardiomyocyte cross-sectional area with both single-drug treatments and a significantly higher effect with the combination treatment. To further evaluate heart function, we measured the plasma values of cardiotrophin-1 and ANP. The first cytokine is a cardiac hypertrophic factor associated with growth and dysfunction of the heart, 36 and the latter is a hormone released from the atria of the heart in response to high blood pressure that acts to reduce the water, sodium, and adipose loads on the circulatory system, thereby reducing blood pressure. 37 The levels of both factors were increased in plasma from hypoxic untreated animals and attenuated by each single treatment and significantly more by the cotreatment, supporting the finding of a reduction in RV hypertrophy and RVP.
Accordingly, we conclude that both sildenafil and Epo have beneficial effects on improving the pathophysiological changes induced by prolonged exposure to hypoxia and subsequent development of PH. Sildenafil is already approved for the treatment of PH, and in a randomized, double-blind, placebo-controlled crossover study, sildenafil was found to improve World Health Organization functional class and hemodynamics in patients suffering from PH. 38 So as a pulmonary vasodilator, sildenafil is an established therapeutic option, albeit one with limited effects. The effects of Epo in the setting of PH is less investigated, but using tg6 mice that constitutively overexpress human Epo in an oxygen-independent manner, Weissmann and coworkers 20 demonstrated that these transgenic mice display reduced vascular remodeling and/or media thickness, as well as blunted vascular resistance and vasoconstrictor response, both under control conditions and in a model of chronic normobaric hypoxia. This is in support of our results; what distinguishes our findings from theirs is that we used exogenous and not endogenously overproduced Epo and that we initiated the treatment only after the onset of PH. We also kept the hematocrit at a more physiological level, thereby avoiding possible side effects due to the excessive erythrocytosis occurring in tg6 animals. Other groups have reported, for instance, a blunted response to U46619 in isolated perfused lungs of tg6 animals 39 as well as in Epo-treated rats. 40 In the monocrotaline rat model of PH, both negative 22 and positive 41 effects of Epo have been reported, whereas in Hilltop and Madison rats exposed to hypoxia, no effects were observed. 23 Finally, an Epo receptor knockout model revealed a higher pulmonary vascular response after hypoxic exposure. 21 The effects of Epo in the setting of hypoxia and PH are therefore not clear, and we thus propose that the results from our study reveal an additive effect of combining sildenafil and Epo and that these findings can provide the basis for further investigations and the development of novel therapeutic strategies not only for the treatment of PH but also in other cardiovascular pathologies.
