Abstract
Pulmonary hypertension (PH) development remains a significant cardiovascular complication of haemoglobinopathies, severely affecting the morbidity and mortality of such patients. According to the 5th World Symposium on PH, PH related with chronic haemolytic anaemias is classified in group 5, mainly due to the multifactorial pathophysiology of PH in this patient population. There are no clear guidelines regarding the management of PH in patients with haemoglobinopathies; the use of specific pulmonary arterial hypertension (PAH) therapy in patients with β-thalassaemia and PH is based on data derived from other forms of PH, expert opinion and small series or case reports. The existing knowledge on the use of specific-PAH therapy in β-thalassaemia patients with PH is limited, and in most cases the diagnosis of PH is based on echocardiographic findings only. We herein report two patients with β-thalassaemia intermedia (TI) and PH, who got same initial approach but different outcome, to highlight the wide spectrum of TI-induced PH, the importance of optimal disease-directed therapy and the possible role of specific-PAH therapy. We also emphasize the central role of right heart catheterization in the diagnosis and follow-up of PH, since this information does facilitate the suitable use or withdrawal of specific PAH drugs in these patients.
Keywords
Case 1 description
A 43-year-old female suffering from β-thalassaemia intermedia (TI), with a history of splenectomy at the age of 40 and occasional blood transfusions, had been visiting the pulmonary hypertension (PH) clinic, due to shortness of breath during exercise, for the last six months.
Upon admission to the hospital, she was in New York Heart Association functional class (NYHA-FC) III, the haemoglobin level was 9.4 g/dL and her transthoracic echocardiography revealed a tricuspid regurgitant jet velocity (TRV) of 3.7 m/s, and left ventricular ejection fraction (LVEF) of 60%. Pulmonary function testing and gas exchange were normal. The high-resolution computerized tomography (CT) of the lungs did not show lung lesions and the CT pulmonary angiography revealed enlarged main, right and left pulmonary arteries. There was no evidence of intraluminal thrombosis. A perfusion lung scan was also free of chronic thromboembolic pulmonary hypertension signs. A cardiac magnetic resonance imaging (MRI) revealed a D-shaped left ventricle without evidence of iron overload. The patient underwent a right heart catheterization (RHC) that confirmed the presence of pre-capillary PH (Table 1, baseline).
Patients’ data at baseline and follow-up.
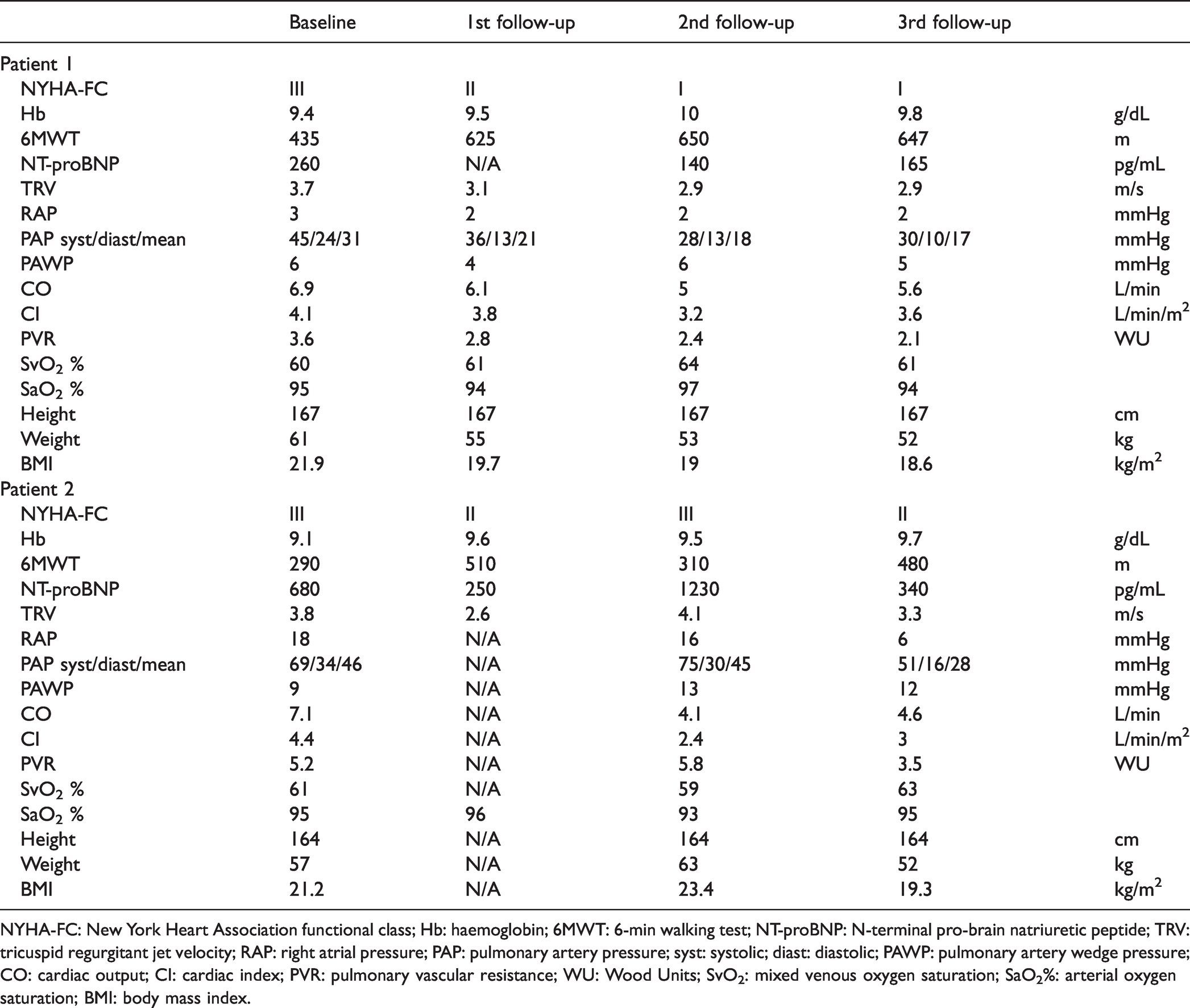
NYHA-FC: New York Heart Association functional class; Hb: haemoglobin; 6MWT: 6-min walking test; NT-proBNP: N-terminal pro-brain natriuretic peptide; TRV: tricuspid regurgitant jet velocity; RAP: right atrial pressure; PAP: pulmonary artery pressure; syst: systolic; diast: diastolic; PAWP: pulmonary artery wedge pressure; CO: cardiac output; CI: cardiac index; PVR: pulmonary vascular resistance; WU: Wood Units; SvO2: mixed venous oxygen saturation; SaO2%: arterial oxygen saturation; BMI: body mass index.
We treated the patient with intensification of haemoglobinopathy-directed therapy (regular blood transfusions, chelation, hydroxyurea) to maintain the Hb level near 10 g/dL, together with acenocoumarol targeting an international normalization ratio (INR) between 2.5 and 3 and initiation of pulmonary arterial hypertension (PAH) specific therapy. She was started on oral bosentan 62.5 mg twice daily up-titrated to 125 mg twice daily one month later; the therapy was well tolerated. The patient presented significant clinical and haemodynamic improvement (Table 1, 1st follow-up). Three years later, after the 2nd follow-up, including a RHC, and the confirmation of sustained improvement, we decided to discontinue bosentan, and carry on only with the haemoglobinopathy-directed therapy and anticoagulants. Two years following bosentan discontinuation, she remains in NYHA-FC I and has normal haemodynamic parameters (Table 1, 3rd follow-up).
Case 2 description
A 31-year-old female suffering from TI, presented to the PH clinic with a one-year history of progressive dyspnoea on exertion and leg oedema. She underwent splenectomy 14 years ago and since then she has been receiving occasional blood transfusions.
Upon admission to the hospital, she was in NYHA-FC III, and her transthoracic echocardiography revealed a TRV of 3.8 m/s, and LVEF of 60%. As with the first case, the CT pulmonary angiography of the patient revealed dilatation of the main, right and left pulmonary arteries, the perfusion lung scan showed no signs of segmental or subsegmental defects and the cardiac MRI showed a D-shaped left ventricle without evidence of iron overload; the remainder of the diagnostic work-up was unremarkable. The patient underwent a RHC that confirmed the presence of severe pre-capillary PH (Table 1, baseline).
Similarly with the 1st case, the patient was treated with intensification of haemoglobinopathy-directed therapy (regular blood transfusions, chelation, hydroxyurea), along with anticoagulants (i.e., acenocoumarol targeting INR 2.5–3) and PAH specific therapy (bosentan up-titrated to a dose of 125 mg twice daily in one month). Additionally, diuretics were added to her treatment. The patient presented significant clinical improvement along with the improvement of her echocardiographic parameters (Table 1, 1st follow-up). However, two years later, she decided to discontinue bosentan on her own will, staying on the transfusion/chelation programme only. One year later, her clinical status was severely deteriorated as well as her RHC-derived haemodynamics (Table 1, 2nd follow-up). Consequently, oral double combination therapy comprised of bosentan (125 mg twice daily) and sildenafil (20 mg three times daily) was added, followed by clinical and haemodynamic improvement as confirmed by her last RHC (Table 1, 3rd follow-up).
Discussion
Over the last 20 years, the presence of PH in patients with β-thalassaemia has been an area of special interest. The reported prevalence of PH for both forms of the disease, β-thalassaemia major (TM) and β-thalassaemia intermedia (TI), varied greatly and was affected by factors such as advancing age, frequency of transfusions and history of splenectomy; however, these studies were mostly based on echocardiographic findings. 1
Recently, the prevalence of PAH confirmed by RHC in patients with β-thalassaemia was reported as 2.1% with an ≈5-fold higher prevalence in TI than TM (4.8% and 1.1% for TI and TM respectively). 2
According to the 5th World Symposium on PH statements, PH related with chronic haemolytic anaemias was moved from group 1 (PAH) to group 5 (unclear and/or multifactorial mechanisms), because of significant differences from other forms of PAH regarding: (i) the pathological findings, (ii) the haemodynamic characteristics (low pulmonary vascular resistance and high cardiac output) and (iii) the lack of evidence of response to PAH specific therapy. 3
Thus, there are no clear guidelines on the management of PH in this patient population; the reported data regarding the use of PAH specific therapy in patients with β-thalassaemia and PH are limited, and in most cases the diagnosis of PH is regrettably based on echocardiographic findings only.
Sildenafil has been used in some patients with beneficial effects on both the clinical status and echo measurements.4–7 A recent study revealed that tadalafil significantly decreased the pulmonary artery systolic pressure and improved the right ventricular systolic function in TI patients. 8 Bosentan has also been reported as safe and efficient treatment for patients with β-thalassaemia and PH; however, there was a lack of RHC in all cases except one. 9 , 10 In addition, epoprostenol has been successfully used in two patients with β-thalassaemia, to whom RHCs confirmed the diagnosis of severe PH and the response to treatment. 11 , 12
We treated both patients with intensification of haemoglobinopathy-directed therapy together with PAH specific therapy. However, it seems that the disease-directed therapy was effective enough for the 1st patient while it was not for the 2nd who required the use of PAH specific therapy. This variance could be related with the severity of PH at the time of diagnosis and shows the need to evaluate each case separately, given the multifactorial mechanisms of PH development in patients β-thalassaemia (i.e. haemolysis, anaemia, reduced nitric oxide bioavailability, iron overload, hypercoagulopathy). 1
By reporting these two cases we highlight the wide spectrum of PH in patients with TI, the importance of optimal chelation transfusion therapy, as well as the need of PAH specific therapy in some patients. In this regard, we also emphasize the importance of PH diagnosis and follow-up with RHC, since this information does facilitate the suitable use or withdrawal of specific PAH drugs in these patients.
In conclusion, we would like to highlight the need for national and multi-national long-term registries in this certain population that could aid diagnostic and treatment plan, considering that the conduction of randomized controlled clinical trials is not realistic given the small sample size.
Footnotes
Authors’ contributions
All authors contributed equally to this study.
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AA has received in the past honoraria and travel awards from Actelion/Janssen; DT has received in the past travel awards from Actelion/Janssen; PK has received in the past honoraria and travel awards from Actelion/Janssen; VV has no conflict of interests to declare; AA has received in the past honoraria and travel awards from Actelion/Janssen and Pfizer; SEO has received in the past honoraria, travel awards and grants from Actelion/Janssen and Pfizer.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Informed consent
The patients have agreed to anonymously report their data that were obtained during their regular follow-ups.
