Abstract
Although precapillary pulmonary hypertension is a rare but severe complication of patients with neurofibromatosis type 1 (NF1), its association with NF2 remains unknown. Herein, we report a case of a 44-year-old woman who was initially diagnosed with idiopathic pulmonary arterial hypertension and treated with pulmonary arterial hypertension-specific combination therapy. However, a careful assessment for a relevant family history of the disease and genetic testing reveal that this patient had a mutation in the NF2 gene. Using immunofluorescence and Western blotting, we demonstrated a decrease in endothelial NF2 protein in lungs from idiopathic pulmonary arterial hypertension patients compared to control lungs, suggesting a potential role of NF2 in pulmonary arterial hypertension development. To our knowledge, this is the first time that precapillary pulmonary hypertension has been described in a patient with NF2. The altered endothelial NF2 expression pattern in pulmonary arterial hypertension lungs should stimulate work to better understand how NF2 is contributing to the pulmonary vascular remodelling associated to these severe life-threatening conditions.
Keywords
Introduction
Neurofibromatosis type 1 (NF1, also called Recklinghausen disease) and type 2 (NF2, also known as bilateral acoustic neurofibromatosis) are two different genetic disorders that are characterised by multiple neurocutaneous tumours and vasculitis. While typical clinical features of NF1 are multiple café-au-lait-spots, axillary and inguinal freckling, cutaneous neurofibromas, and Lisch-nodules, the hallmark feature of the NF2 is the development of bilateral vestibular schwannomas (VS).1 Located on chromosome 22, the NF2 gene contains 17 exons that direct the synthesis of a 70-kDa protein named NF2, also called Merlin.
Precapillary pulmonary hypertension (PH) is one of the most serious complication of NF1 and causes significant morbidity and mortality.2 PH-NF1 is defined as a resting mean pulmonary arterial pressure (mPAP) >20 mmHg in association with normal pulmonary arterial wedge pressure (PAWP) ≤15 mm Hg and pulmonary vascular resistance >3 Wood unit measured by right heart catheterisation (RHC).3 Many of the symptoms of PH are nonspecific and include breathlessness on exertion or fatigue during routine activity. Since the aetiology and pathogenesis remain obscure, PH-NF1 is classified in group 5 of the PH clinical classification within the group entitled ‘PH with unclear and/or multifactorial mechanisms’.3 However, to our knowledge, there are currently no reported cases of precapillary PH in NF2 patients.
Case report
A 44-year-old woman, with symptoms of progressive shortness of breath that last more than one year, was referred to our hospital due to clinical suspicion of PH. The patient was evaluated through RHC to assure the diagnosis of PH (Fig. 1a). At the initial evaluation, the mean PAP was of 38 mmHg, the mean PAWP was 10 mmHg, and the pulmonary vascular resistance was 7.02 WU. Because she had no evidence of embolism or obstruction of her pulmonary arteries (Fig. 1b) or any other comorbidity that could have explained these elevated pulmonary artery pressures, the initial diagnosis was idiopathic pulmonary arterial hypertension (IPAH). She was treated accordingly with a combination of macitentan, tadalafil and selexipag, and her mPAP improved to 26 mmHg, and she kept low-risk profiles for over four years.
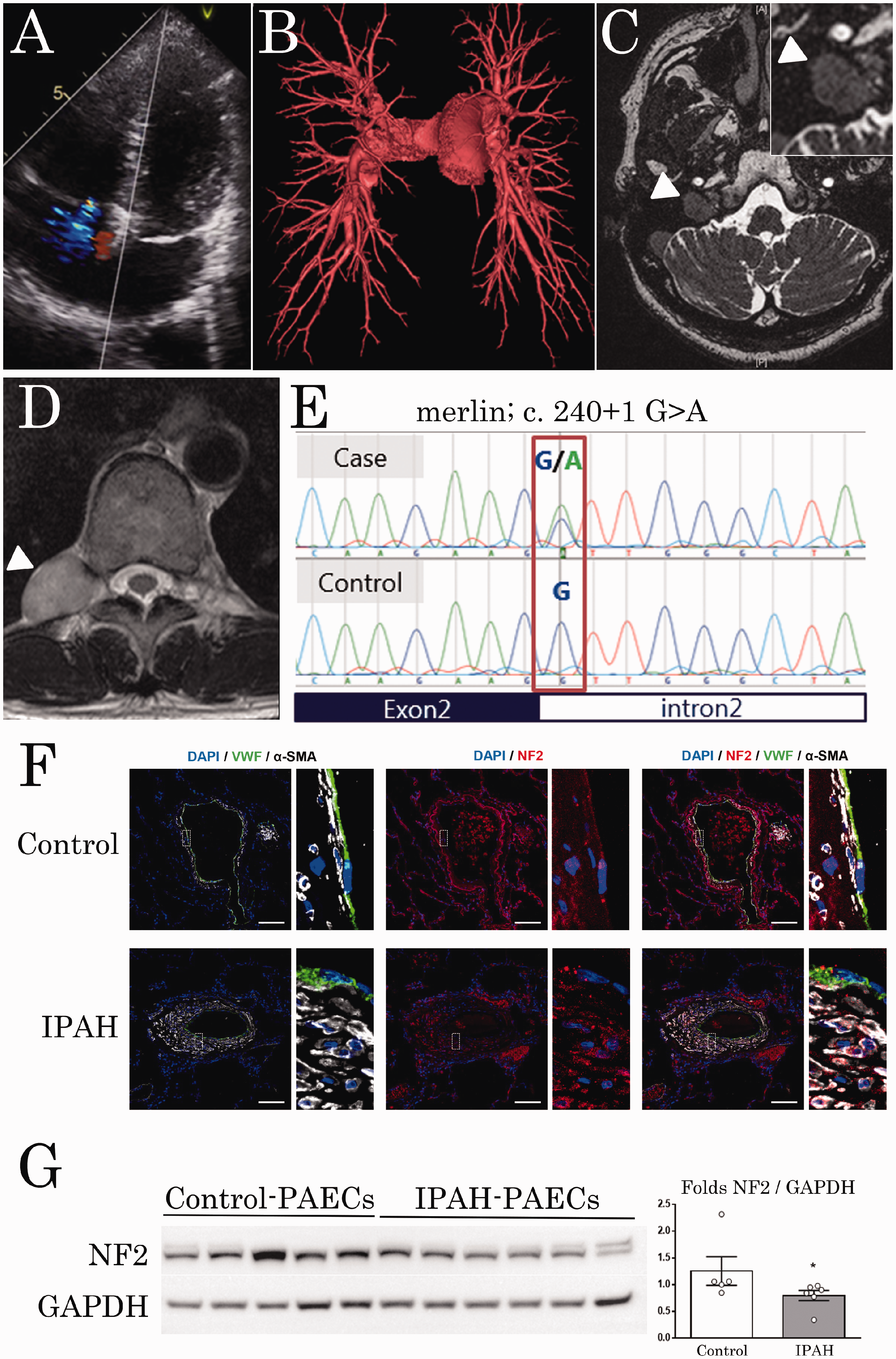
Phenotypes of neurofibromatosis type 2 (NF2) associated with pulmonary hypertension: (a) echocardiography (four-chamber view) showing enlarged right atrium and ventricle compressing the left atrium and ventricle and associated with high-velocity tricuspid valve regurgitation. (b) CT angiogram image demonstrating no evidence of thromboembolism or obstruction of the pulmonary arteries. (c) Magnetic resonance image in a true-FISP sequence showing a right acoustic schwannoma (white arrow). (d) Axial T2-weighted magnetic resonance image of the thoracic spine showing a spinal nerve sheath tumour between Th10-11 (white arrow). (e) Sanger sequencing revealed a splicing donor mutation in the single base downstream of exon 2 in the NF2 gene; c.240 + 1 G>A. (f) Double staining of NF2 with α-SMA, vWF and DAPI in pulmonary arteries from control subjects and IPAH patients. Scale bars: 100 μm. (g) Representative Western blots and quantification of NF2:GAPDH ratio in human pulmonary ECs derived from control and IPAH. Data are presented as the means ± standard error of the mean of n = 5–6 patients per group and were analysed using Student’s t-test.
However, detailed enquiries about her family history revealed that her father and father’s uncle had both been diagnosed as having acoustic schwannomas and NF2. Thereafter, magnetic resonance imaging revealed that she also had an acoustic schwannoma and a spinal nerve sheath tumour in Th10-11 (Fig. 1c and d). The results of the genetic testing revealed a splicing donor mutation in the downstream of exon 2 in NF2 gene (Fig. 1e). Of note, no mutation in the Bone Morphogenetic Protein Receptor type 2 (BMPR2) gene were found. To our knowledge, this is the first time that precapillary PH has been described in a patient with NF2. Interestingly, the NF2-PH patient was responsive to approved PAH therapies.
To identify whether the NF2 expression pattern is altered in lungs of PAH patients, confocal microscopic analyses and double labelling for NF2 with alpha smooth muscle actin (α-SMA), von Willebrand factor (VWF) were used to localise NF2 in lung specimens from patients with IPAH and control subjects (Supplemental methods). Interestingly, we found strong NF2 expression within the endothelium of distal pulmonary arteries from control specimens as compared with a weak staining within the endothelium of IPAH patients (Fig. 1f). Consistent with these in situ observations, we found that primary early (<5) passage cultures of human endothelial cells (ECs) isolated from lung tissues obtained from IPAH patients displayed lower NF2 protein levels as compared to control cells (Fig. 1g).
Discussion
To the best of our knowledge, this is the first case of precapillary PH associated with NF2 (NF2-PH). On the one hand, the diagnosis of NF2 was established on clinical criteria with the identification of a heterozygous pathogenic variant in NF2, and on the other hand, the diagnosis of PH was confirmed by RHC.
NF2 is a rare autosomal dominant disorder characterised by the presence of bilateral nerve acanthosis, neurological lesions such as vestibular schwannoma, spinal nerve sheath tumour and meningioma are also frequently associated.1 The diagnosis of NF2 is often challenging if patients do not show hearing loss, first because the incidence is low (approximately 1 in 50,000), but also because the associated skin lesions are characteristically less severe than NF1.4 In comparison, NF1 occurs in 1 in 3000 people, and it relatively readily recognisable because of the characteristic skin findings, namely, café-au-lait spots and skin neurofibromas.
Even if NF1 (also known as neurofibromin) has been reported to promote pulmonary arterial smooth muscle cell (PA-SMC) proliferation in PAH via activation of the Ras-mitogen-activated protein kinase signal transduction pathway,5 little is known about the role of NF2 in vascular homeostasis and remodelling. Here, we obtained evidences that NF2 is physiologically expressed in pulmonary ECs and lost in the dysfunctional pulmonary endothelium in IPAH. The in situ findings were replicated in vitro, with cultured pulmonary ECs from IPAH patients exhibiting lower levels of NF2 compared with control pulmonary ECs. Since it has been suggested that NF2 is a central regulator of Ras and Rac activation after growth factor stimulation,6 the reduced endothelial NF2 might lead to endothelial dysfunction, a process central to vascular remodelling. In addition, NF2 is also a negative regulator of mechanistic target of rapamycin complex 1 (mTORC1)7 and nuclear factor-κB (NF-κB).8 Interestingly, activation of both mTORC and NF-κB pathways contribute to the pulmonary vascular remodelling in experimental and human PAH.9,10 Taken altogether, these data strongly support the notion that NF2 could act via multiple mechanisms to either promote or modulate pulmonary vascular remodelling.
In conclusion, the observation that the dysfunctional endothelium in remodelled PAH vessels is associated with a decrease in NF2 protein levels should stimulate work to better understand how NF2 is contributing to the pulmonary vascular remodelling associated to these severe life-threatening conditions. And we highlight the importance of carefully considering medical and family histories.
Supplemental Material
sj-pdf-1-pul-10.1177_20458940211029550 - Supplemental material for Pulmonary hypertension associated with neurofibromatosis type 2
Supplemental material, sj-pdf-1-pul-10.1177_20458940211029550 for Pulmonary hypertension associated with neurofibromatosis type 2 by Hirohisa Taniguchi, Tomoya Takashima, Ly Tu, Raphaël Thuillet, Asuka Furukawa, Yoshiko Furukawa, Akio Kawamura, Marc Humbert, Christophe Guignabert and Yuichi Tamura in Pulmonary Circulation
Footnotes
Acknowledgement
The authors would like to thank Rika Takeyasu for data collection.
Author contributions
Y. T. takes responsibility for the content of the manuscript, including the data and analysis. Y. T. and H. T. conceived and designed the study. T. T. performed DNA sequencing, bioinformatics analysis and primary data analysis. L. T. and R. T. performed cellular and molecular studies. A. K. and M. H. participated in data analysis and interpretation. A. F. and Y. F. contributed to sample collection. H. T. prepared the first draft of the manuscript. C. G. and Y. T. supervised the manuscript preparation process. All authors reviewed the content of the draft, approved the final manuscript and agreed to be accountable for all aspects of the work.
Conflict of interest
Over the last three years, C.G. reports grants from Acceleron and Janssen and grants and personal fees from Merck, outside the submitted work. M.H. reports grants and personal fees from Acceleron, Bayer, Merck, and Janssen and personal fees from GSK, outside the submitted work.
Ethical approval
This study was approved by a local institutional ethics committee at the International University of Health and Welfare (approval number 5-16-25).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Informed consent
Informed consent was obtained from the patient.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
