Abstract
The effects of the heptapeptide angiotensin-(1–7) (Ang-(1–7)), via its receptor Mas, oppose many of the effects of the classic angiotensin II signaling pathway, and pharmacological exploitation of this effect is currently actively pursued for a wide range of cardiovascular, neoplastic, or immunological disorders. On the basis of its vasodilatory and antiproliferative properties, Ang-(1–7) has consequentially also been proposed as a novel therapeutic strategy for the treatment of pulmonary arterial hypertension (PAH). In this study, we tested the effectiveness of Ang-(1–7) and its stable, cyclic analog cAng-(1–7) over a range of doses for their therapeutic potential in experimental PAH. In the monocrotaline (MCT) rat model of PAH, Ang-(1–7) or cAng-(1–7) were injected in doses of 30, 100, 300, or 900
Pulmonary arterial hypertension (PAH) is a rare but ultimately fatal disease defined by an increase in mean pulmonary arterial pressure to >25 mmHg at rest or >30 mmHg during exercise. 1 The pathogenesis of PAH is very diverse, in that PAH may be heritable or idiopathic or develop in association with a number of diseases, such as systemic sclerosis or HIV infection. The pathology of PAH is characterized by endothelial dysfunction, which becomes apparent as an imbalance in the release of vasoconstrictive and vasodilatory factors, such as endothelin and nitric oxide (NO), leading to pulmonary vasoconstriction and ultimately promoting structural remodeling of the pulmonary vasculature, resulting in a persistent elevation in pulmonary vascular resistance.2,3 A series of therapeutic drugs have been approved for the treatment of PAH that target three different pathways: prostacyclin analogs, endothelin receptor antagonists, and phosphodiesterase type 5 inhibitors. 4 While these drugs have helped to improve life expectancy and quality of life in PAH patients in the past 2 decades, mortality remains high, as one-third of PAH patients die within 3 years of diagnosis; this emphasizes the need for further research and improved therapeutic options. 5
The renin-angiotensin system (RAS) is a critical regulator of cardiovascular homeostasis, and cardiovascular diseases are accordingly frequently associated with an imbalance of the RAS.
6
Its main effector, angiotensin II (AngII), is formed from AngI by the endopepdidase angiotensin-converting enzyme (ACE) and exerts its effects mostly by acting through the AngII receptor subtype 1 (AT1). Activation of the ACE/AngII/AT1 axis mediates vasoconstrictive, proliferative, fibrotic, and inflammatory effects and therefore is a key player in the onset of many cardiovascular diseases.
7
In vivo, this pathway is endogenously opposed by the ACE2/Ang-(1–7)/Mas axis, which counteracts many of the effects of AngII.
8
The heptapeptide Ang-(1–7) is predominantly formed by ACE2-mediated cleavage of the octapeptide AngII
7
and exerts its effects through the G-protein-coupled receptor Mas.
9
Ang-(1–7) contributes importantly to cardiovascular homeostasis by triggering a variety of downstream targets, most notably by activation of endothelial NO synthase (eNOS) and thus the release of NO,10,11 causing vasodilation and—in broadest terms—endothelial protection. Ang-(1–7) also blocks mitogen-activated protein kinase signaling, thereby limiting smooth muscle cell proliferation, and can in parallel attenuate cardiac myocyte hypertrophy by reducing transforming growth factor
However, the translation of Ang-(1–7) as a novel drug therapy for PAH to the clinical setting is somewhat hampered by its rapid metabolism and degradation in plasma and tissue, predominantly, again, by ACE. 17 To improve its pharmacokinetic properties, a cyclic form of Ang-(1–7), cAng-(1–7), which is linked with a thioether bridge, was developed with the intention of mitigating its rapid degradation and increasing its half-life. Indeed, during continuous infusion, cAng-1–7 displays a 34-fold higher presence than conventional Ang-(1–7) in the circulating blood of Sprague-Dawley (SD) rats, 18 making it a promising candidate for clinical use in cardiovascular therapy.
In preparation for the translation of Ang-(1–7) therapy into first clinical PAH trials, we designed this study to (1) test a wide range of drug concentrations for their effectiveness in experimental PAH and (2) compare the efficacy of Ang-(1–7) with that of its stable, cyclic analog cAng-(1–7). To this end, we adopted a preventive drug administration approach, as the primary goal was to establish dose-response relationships for each individual compound before proceeding to therapeutic-intervention models.
METHODS
Animals and experimental design
Male SD rats of 200–220 g body weight (bw) were obtained from Charles River Laboratories (St. Constant, QC, Canada). All animals received care in accordance with the Guide for the Care and Use of Laboratory Animals (published in 1996 by the Institute of Laboratory Animal Resources, National Research Council). The study was approved by the Animal Care Committee of St. Michael's Hospital (protocol 397). All animals were allowed to acclimate for 5 days before start of the Ang-(1–7)/cAng-(1–7) injections. Ang-(1–7) and cAng-(1–7) were kindly provided by Tarix Pharmaceuticals (Cambridge, MA). At the inception of the study, rats were enrolled in a staggered manner, such that equal numbers of animals were enrolled per study group per day. PAH was induced by a single intraperitoneal injection of monocrotaline (MCT; 40 mg/kg), and 24 hours later once-daily subcutaneous administration of Ang-(1–7), cAng-(1–7) (30, 100, 300, or 900
Assessment of pulmonary hemodynamics and right ventricular hypertrophy (RVH)
Hemodynamic and RVH measurements were performed as previously reported. 19 In brief, 5 weeks after MCT treatment, rats were anesthetized, tracheotomized, and mechanically ventilated with a tidal volume of 6 mL/kg bw and a positive end-expiratory pressure of 3 mmHg at 80 breaths/min, as described elsewhere. 20 A 2F microtip Millar catheter (Millar, Houston, TX) was introduced via the right jugular vein into the right ventricle for real-time monitoring of right ventricular systolic pressure (RVSP; DasyLab, Norton, MA). After recording of RVSP curves, animals were exsanguinated, and the left lung was excised and fixed for histology in 10% neutral buffered paraformaldehyde. The right lung was snap-frozen in liquid nitrogen for postmortem protein analyses. For assessment of RVH, the Fulton index was calculated as the ratio of the right ventricular weight to the left-ventricular-plus-septal weight (RV/[LV + S]).
Lung histology and analysis of vascular remodeling
In each experimental group, 5 lungs from 5 different rats were fixed by intratracheal instillation of 10% paraformaldehyde for 30 minutes, and tissue sections of 1 cm3 were excised and fixed for another 24 hours. Dehydrated tissues were embedded in paraffin, and 5-
Western blot analysis
Snap-frozen lungs were homogenized in lysis buffer containing 100 mM NaCl, 30 mM HEPES, 1 mM EGTA (ethylene glycol tetraacetic acid), 1% Triton X-100, protease inhibitor mixture, and PhosSTOP (Roche, Mississauga, ON, Canada). Total protein concentration was determined with a bicinchoninic acid protein assay kit (Thermo Scientific, Waltham, MA). Equal amounts of protein (60
Statistical analysis
Data are given as means ± SEM. Different groups were compared by ANOVA followed by the Dunnett multiple-comparison test (Prism, ver. 5.0, GraphPad Software, La Jolla, CA). Statistical significance was assumed at
RESULTS
To probe for the effectiveness and potential dose-response relationships of Ang-(1–7)-based therapies in experimental PAH, we first focused on the effects of linear Ang-(1–7) on pulmonary hemodynamics and lung vascular remodeling. RVSP values showed a significant increase in MCT-treated animals compared to nontreated controls, consistent with the induction of PAH. In groups treated with 100, 300, or 900
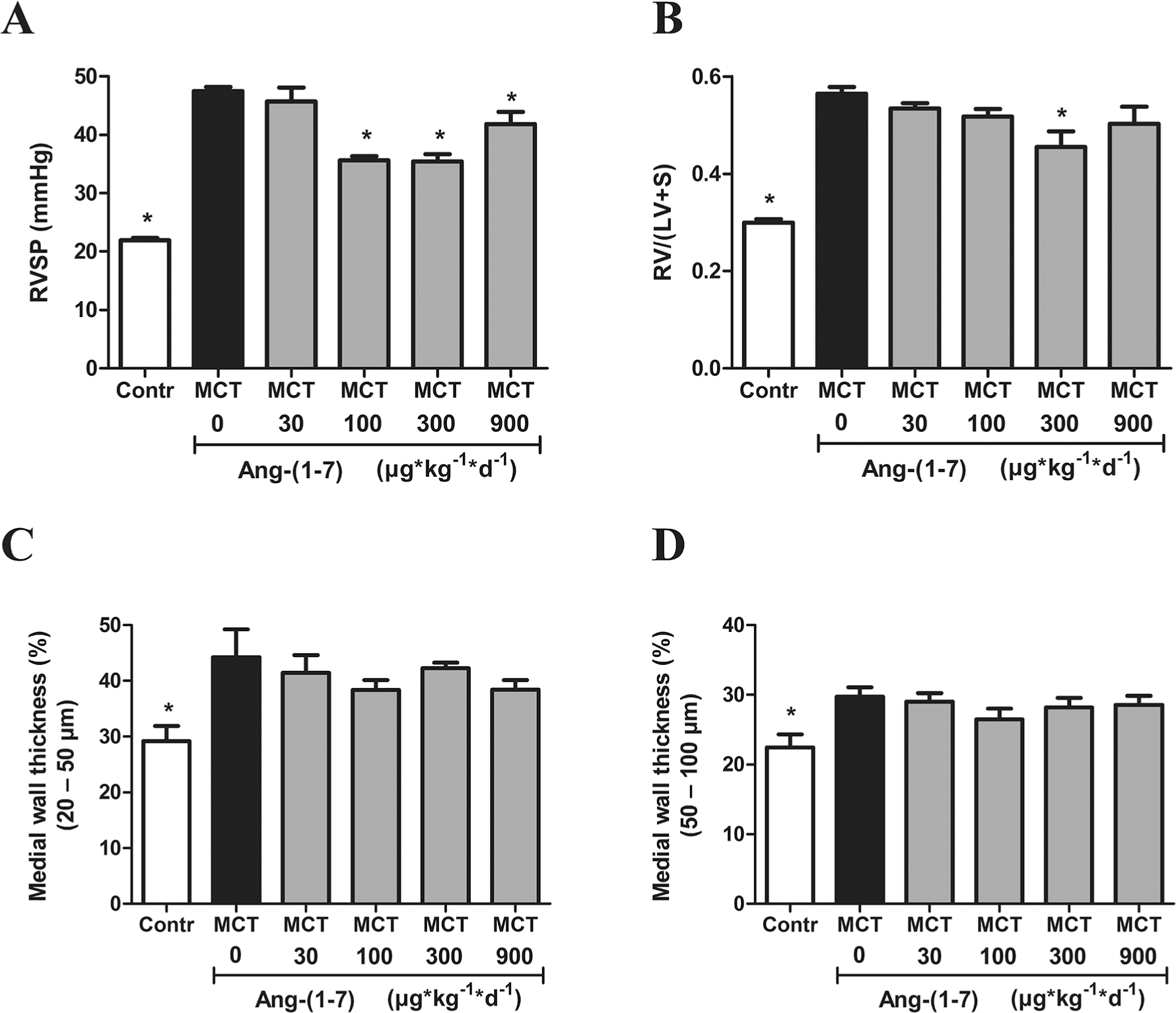
Dose-dependent effects of angiotensin-(1–7) (Ang-(1–7)) on pulmonary arterial hypertension, lung vascular remodeling, and right ventricular hypertrophy in the monocrotaline (MCT) model. Rats were injected with either vehicle (Contr), MCT only (MCT 0), or MCT plus Ang-(1–7) in doses of 30, 100, 300, or 900
Despite moderate reductions in RVSP, the overall therapeutic effectiveness of Ang-(1–7) was thus rather limited throughout all groups. In a second set of experiments, we therefore tested its chemically modified, more stable cyclic analog cAng-(1–7) in the identical model and dosing regime. For all groups treated with cAng-(1–7) (30, 100, 300, or 900
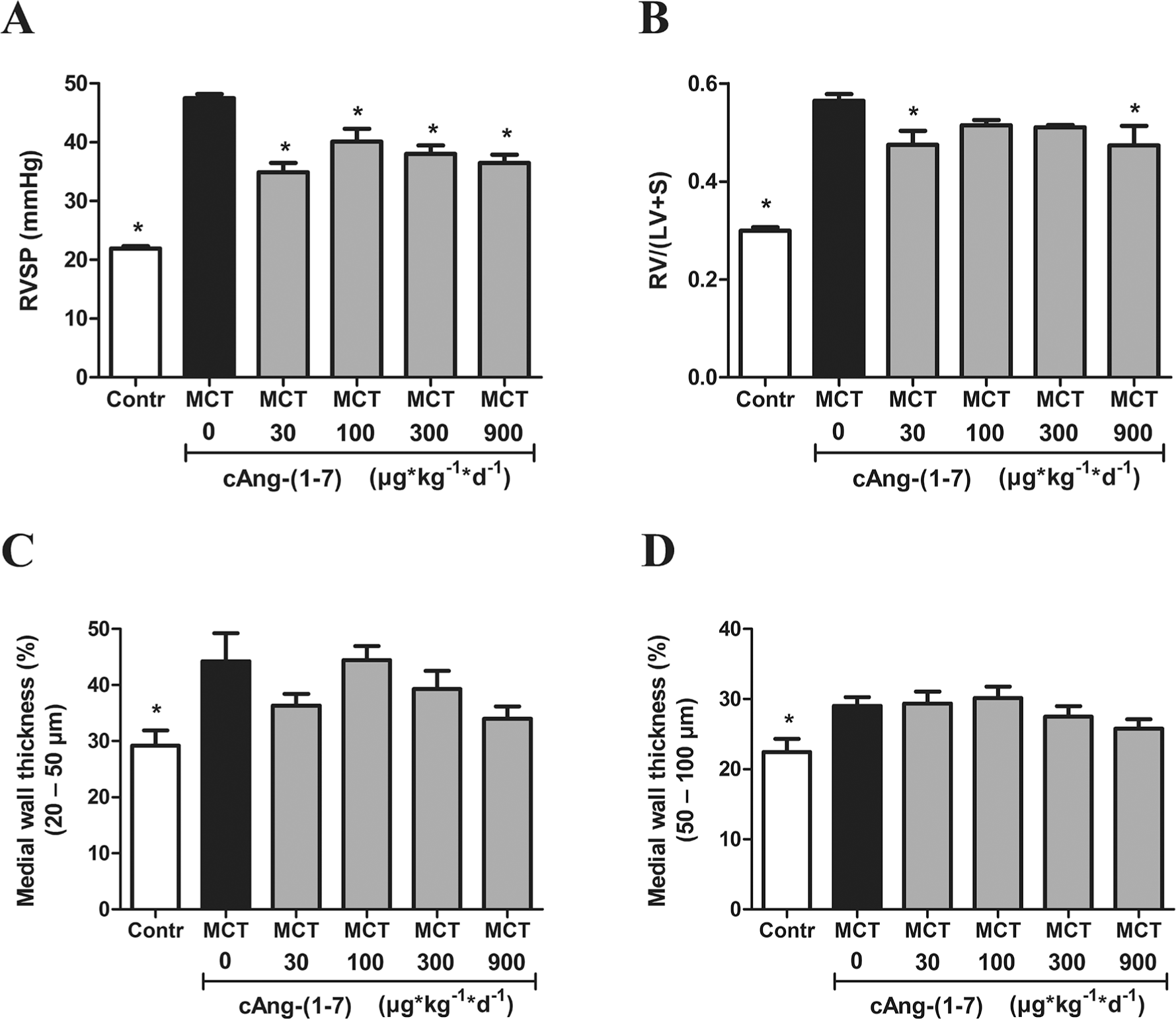
Dose-dependent effects of cyclic angiotensin-(1–7) (cAng-(1–7)) on pulmonary arterial hypertension, lung vascular remodeling, and right ventricular hypertrophy in the monocrotaline (MCT) model. Rats were injected with either vehicle (Contr), MCT only (MCT 0), or MCT plus cAng-(1–7) in doses of 30, 100, 300, or 900
Ambrisentan is a specific endothelin receptor A antagonist that is clinically approved for the treatment of PAH.
22
Since the effectiveness of both Ang-(1–7) and cAng(1–7) to attenuate PAH and lung vascular remodeling in MCT rats was modest overall, we probed for a potential systematic error by using ambrisentan as a positive treatment control. MCT-treated animals receiving ambrisentan at a dose of 90
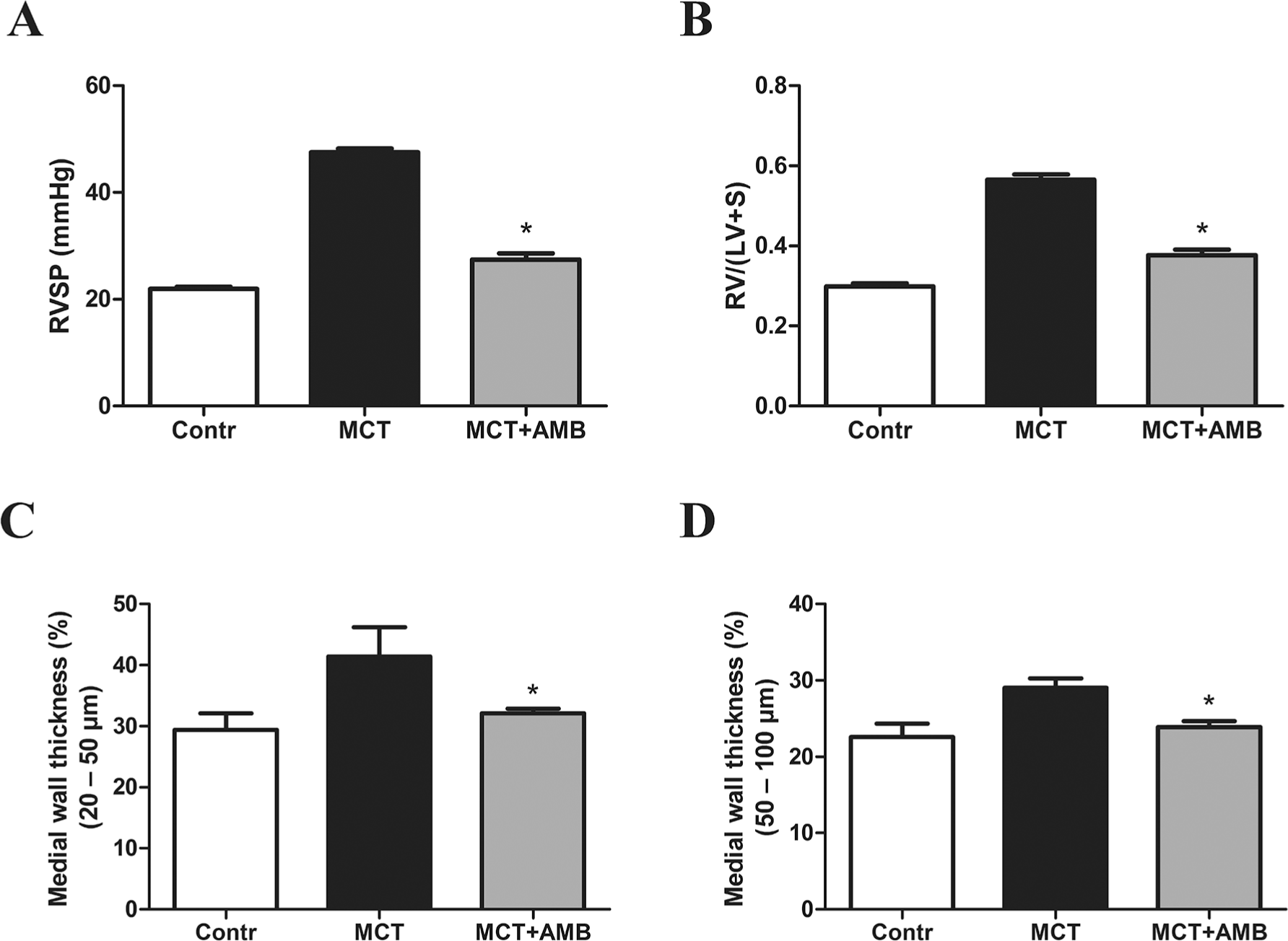
Effects of ambrisentan (AMB) on pulmonary arterial hypertension, lung vascular remodeling, and right ventricular hypertrophy in the monocrotaline (MCT) model. Rats were injected with either vehicle (Contr), MCT only (MCT 0), or MCT with subsequent administration of 90
In order to probe whether the limited effectiveness of Ang-(1–7) and cAng-(1–7) in the MCT model may have been the result of a loss of target, we next probed for expression levels of the Ang-(1–7) receptor Mas by Western blot. Mas receptor expression was comparable between lungs collected from control and MCT animals. Moreover, in all lungs from animals treated with Ang-(1–7), Mas expression tended to be higher, as compared to the control groups, especially in groups treated with doses of 30, 100, or 300
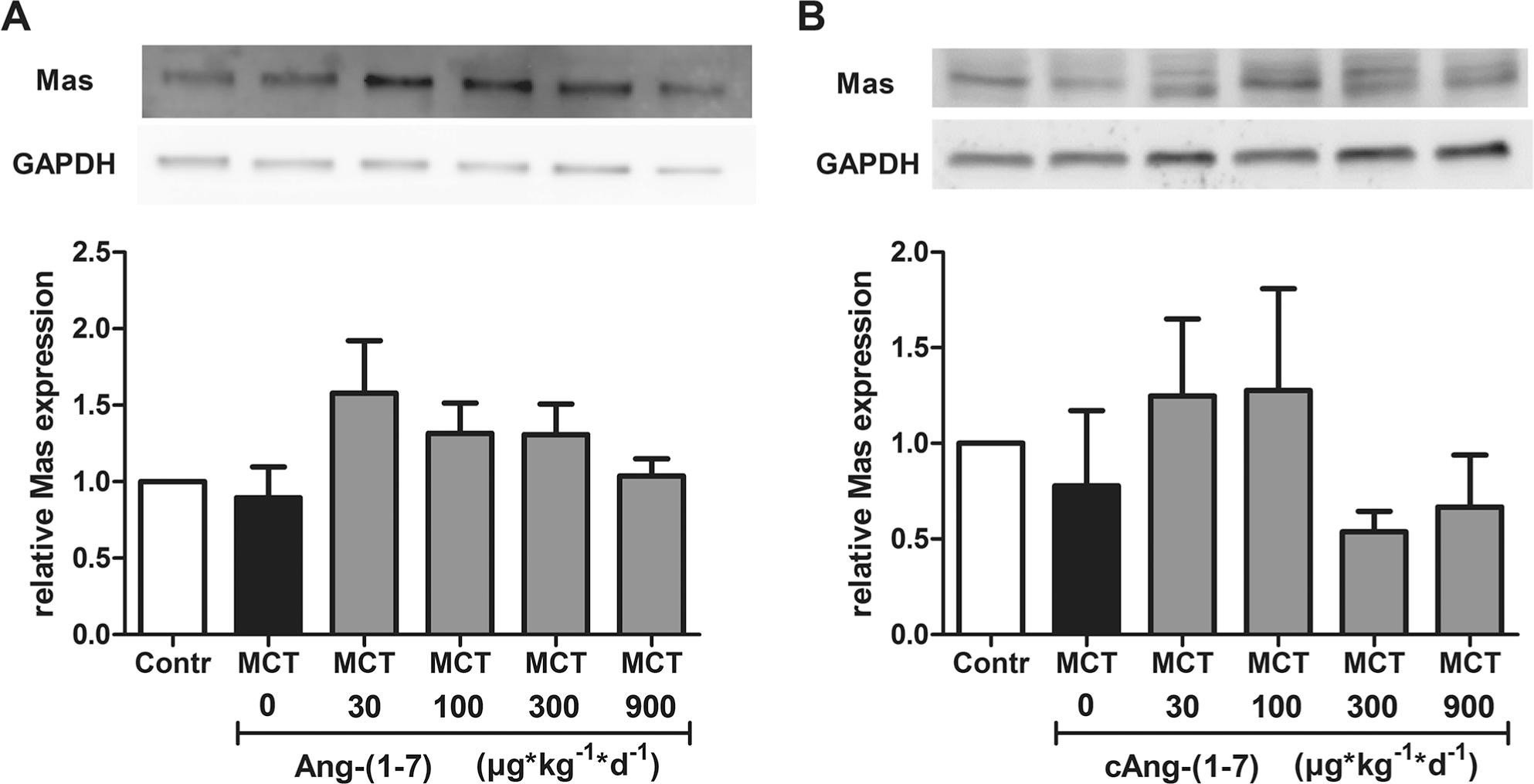
Dose-dependent effect of angiotensin-(1–7) (Ang-(1–7);
Endothelial NO synthase (eNOS), which becomes activated when it is phosphorylated at its Ser1177 residue (p-eNOS), represents a potential downstream target of the ACE2/Ang-(1–7)/Mas axis and serves at the same time as a marker of intact endothelial function in PAH.3,10 Expression of eNOS was similar between control and MCT-treated animals. A trend to a gradual increase in eNOS levels was evident with increasing doses of Ang-(1–7) (Fig. 5A) or cAng-(1–7) (Fig. 5D), with the notable exception of the highest cAng-(1–7) dose. The p-eNOS levels were distinctly lower in lungs from MCT animals than in controls, in line with reduced eNOS activity, as previously reported in this model.
23
In contrast, p-eNOS levels were reversed almost to control levels in rats receiving higher Ang-(1–7) doses (100, 300, or 900
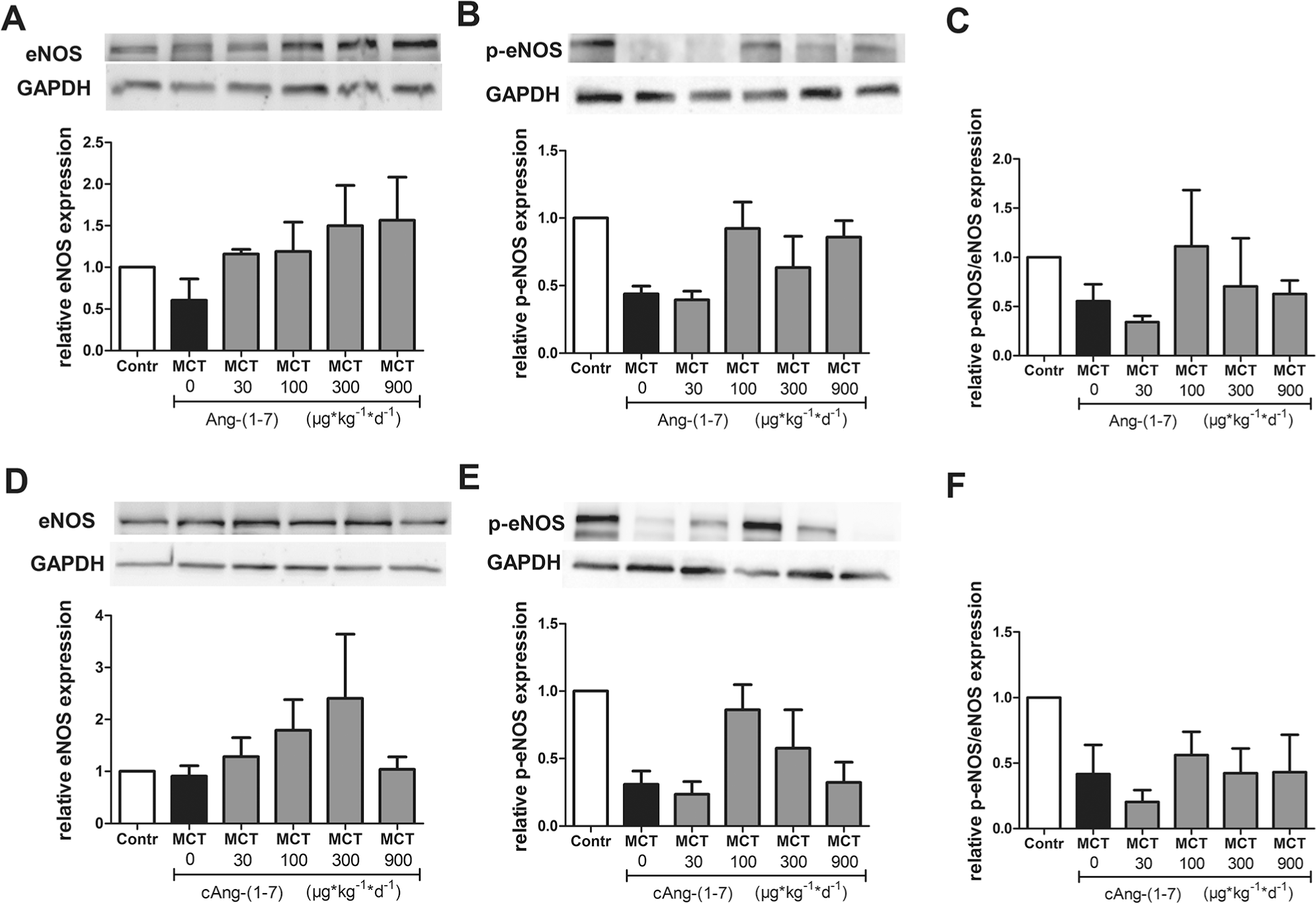
Dose-dependent effect of angiotensin-(1–7) (Ang-(1–7);
DISCUSSION
In this study, we aimed to identify the optimal dosing strategy for Ang-(1–7), which has recently evolved as a promising therapeutic strategy for PAH, and to compare the effectiveness of the linear Ang-(1–7) peptide to that of its more stable cyclic derivative, cAng-(1–7). While medium doses of Ang-(1–7) (100 or 300
The results from this study can therefore not unequivocally confirm the exciting promise for the therapeutic potential of Ang-(1–7) in experimental PAH suggested in two previous studies. Both Shenoy and colleagues
16
and Chen and coworkers
15
had reported significant, striking reductions in RVSP, medial-wall thickness, and RVH in an MCT rat model after, respectively, overexpression (by lenti-Ang-(1–7) viral particles) or continuous infusion (24
To specifically examine the effects of Ang-(1–7) and cAng-(1–7) on endothelial function, we probed for eNOS and p-eNOS expression in lung tissue. Interestingly, for groups treated with Ang-(1–7), the combined ratio of p-eNOS to eNOS was inversely related to the effects on RVSP, in that the two treatment groups with the most prominent decrease in RVSP (100 and 300
To test whether loss of the Ang-(1–7) receptor Mas may have limited the effectiveness of Ang-(1–7) or cAng-(1–7) treatment in our study, we probed lung samples for Mas receptor expression. Except for the two highest-dose groups of cAng-(1–7), all treatment groups showed strong Mas receptor expression above the level of both the control and the untreated MCT groups, indicating that the modest effectiveness of Ang-(1–7) or cAng-(1–7) was not attributable to a loss of target.
As a positive control, we treated rats with the endothelin receptor antagonist ambrisentan, an established drug for the treatment of PAH, which received approval by the Food and Drug Administration in 2007 24 and has been proven effective in PAH treatment in various studies.22,25,26 In line with these reports, treatment with ambrisentan significantly reduced RVSP, RVH, and vascular remodeling in our MCT rat model, therefore ruling out systematic errors in the experimental model.
It should be considered that this study tested the effects of Ang-(1–7) and cAng-(1–7) in only a single animal model of PAH. While the classic MCT model remains the most frequently utilized PAH model, other approaches, such as the Sugen-hypoxia model, have been proposed to yield a more accurate reflection of human PAH disease because of a lesser initial inflammatory component and the development of plexiform lesions. 27 While we cannot exclude the possibility that Ang-(1–7) therapy may prove more beneficial in such models (and, ultimately, in patients), as compared to the MCT model, this notion seems unlikely, because (1) we are, at present, not aware of any treatment approach that has provided striking therapeutic benefit in the Sugen-hypoxia model yet not in the MCT model of PAH, and (2) Ang-(1–7) would, on the basis of its well-documented anti-inflammatory effects,28,29 be expected to yield false-positive rather than false-negative results in the MCT model.
Finally, we considered whether the moderate therapeutic effects in our study may have been attributable to the dosing regimen or route of delivery. Importantly, the bioavailability of Ang-(1–7) after subcutaneous injection is reported as 98%, suggesting that almost all injected Ang-(1–7) enters the bloodstream, which makes this route of delivery highly effective. 30 However, the half-life of Ang-(1–7) in plasma is relatively short 17 and may therefore have limited the therapeutic effectiveness in our study, as the drug was administered only once daily. As a consequence, effective Ang-(1–7) therapy may require more frequent administration of the drug, which, however, limits the feasibility of such an approach in the clinical scenario. In line with this view, ambrisentan, which was presumably taken up in a more continuous fashion with the drinking water, showed clear beneficial effects in our MCT model. Recently, studies by Magalhães and colleagues 31 in which Ang-(1–7) was delivered continuously via implanted osmotic minipumps showed beneficial effects on lung vascular remodeling and RVH in a murine model of ovalbumin-induced chronic allergic lung disease. While we cannot exclude that continuous delivery via minipumps might have yielded more advantageous effects of Ang-(1–7) or cAng-(1–7) in our study, it is important to note that Magalhães and coworkers 31 studied the effects of Ang-(1–7) in a model of chronic lung inflammation. In view of the potent anti-inflammatory action of Ang-(1–7),28,29 it may not be surprising that Ang-(1–7) was more potent in alleviating pathological changes in these chronically inflamed lungs independent of the actual mode of drug delivery.
In a series of studies, Ang-(1–7) has been applied successfully as a single daily injection with distinct beneficial effects in various disease models other than PAH.32,33 Consistently, measurements of Ang-(1–7) levels in lung homogenate (Fig. S1) show a good dose-response relation, demonstrating effective delivery of the drug to the lung and its accumulation in the pulmonary parenchyma. Nevertheless, the argument that the limited therapeutic effect in our study may have resulted from the short half-life of Ang-(1–7) cannot be completely refuted. However, the same argument cannot be equally applied for cAng-(1–7), which is largely resistant to degradation by ACE
34
and, as a result, has a 34-fold longer half-life in vivo than Ang-(1–7).
18
However, the improved bioavailability of cAng-(1–7) did not increase its therapeutic effectiveness over Ang-(1–7) in our study. Finally, a wide range of Ang-(1–7) concentrations, from 30 to 900
In summary, both Ang-(1–7) and cAng-(1–7), when administered immediately after induction of PAH, showed moderate beneficial effects in the MCT rat model. Because of the overall limited therapeutic effectiveness of the approach, we refrained from further pursuing this strategy in a therapeutic regime where Ang-(1–7) or cAng-(1–7) therapy would be initiated 3 weeks after MCT treatment, at a time when PAH and vascular remodeling were already established. The optimal dose for Ang-(1–7) in our study seems to be around 100–300
Appendix
Supplementary Methods
Ang-(1–7) levels in rat lung homogenates were determined in duplicate with a rat Ang-(1–7) enzyme-linked immunosorbent assay kit (NeoScientific, Cambridge, MA) according to the manufacturer's instructions.
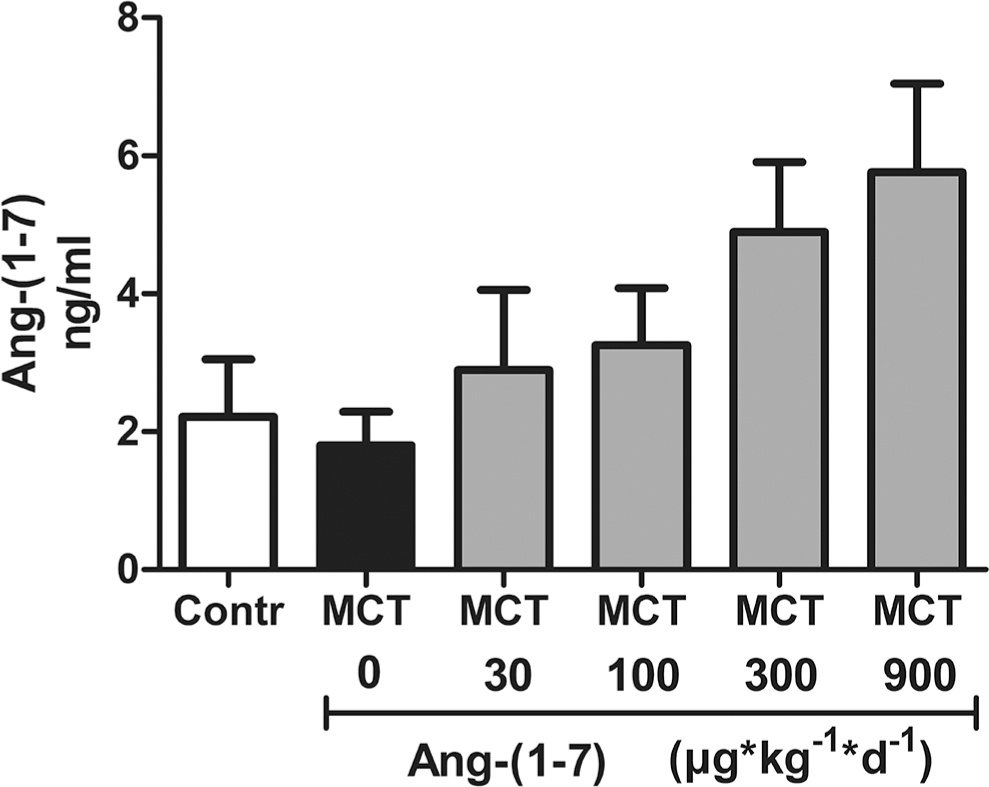
Angiotensin-(1–7) (Ang-(1–7)) levels in lung homogenate. Ang-(1–7) concentration was determined by enzyme-linked immunosorbent assay in lung homogenates of rats who had been injected with either vehicle (Contr), monocrotaline (MCT) only (MCT 0), or MCT plus Ang-(1–7) in doses of 30, 100, 300, or 900
