Abstract
Pulmonary hypertension(PH)is a kind of hemodynamic and pathophysiological state, in which the pulmonary artery pressure (PAP) rises above a certain threshold. The main pathological manifestation is pulmonary vasoconstriction and remodelling progressively. More and more studies have found that ions play a major role in the pathogenesis of PH. Many vasoactive substances, inflammatory mediators, transcription-inducing factors, apoptosis mediators, redox substances and translation modifiers can control the concentration of ions inside and outside the cell by regulating the activity of ion channels, which can regulate vascular contraction, cell proliferation, migration, apoptosis, inflammation and other functions. We all know that there are no effective drugs to treat PH. Ions are involved in the occurrence and development of PH, so it is necessary to clarify the mechanism of ions in PH as a therapeutic target for PH. The main ions involved in PH are calcium ion (Ca2+), potassium ion (K+), sodium ion (Na+) and chloride ion (Cl–). Here, we mainly discuss the distribution of these ions and their channels in pulmonary arteries and their role in the pathogenesis of PH.
Keywords
Introduction
Considering that the increase of cardiac output and pulmonary artery wedge pressure will also affect the mean pulmonary artery pressure (mPAP), the sixth World Health Organization newly defined pulmonary hypertension (PH) as the mPAP ≥20 mmHg detected by right cardiac catheterization and pulmonary vascular resistance ≥3 Wood Units (WU) with precapillary morphology.1,2 At present, according to pathology, hemodynamics and clinical diagnosis and treatment, PH is divided into five categories: (1) arterial PH, (2) PH caused by left heart disease, (3) PH caused by lung disease or/and hypoxia, (4) chronic thromboembolism PH, (5) PH caused by unidentified multifactorial mechanism. At the Sixth World Health Organization Symposium, it was proposed that long-term responses to calcium channel blockers should be included in the first group of PH because these patients had a special prognosis and treatment, and a continuous relationship between arterial, capillary and pulmonary capillary angiomatosis was found. 1 The main clinical manifestations of PH are progressive shortness of breath, decreased exercise endurance, chest tightness, palpitation, and related manifestations caused by right heart failure, etc. The main mechanism of PH is pulmonary artery contraction and remodelling, which leads to the increase of pulmonary artery resistance. At present, the treatment of PH is based primarily on vascular tension and pulmonary vascular remodelling.2,3 From the past to the present research results, it can be known that ions play an important role in the pathogenesis of PH. The regulation of ion concentration inside and outside the cell mainly depends on the opening or closing of ion channels. The pathogenesis of PH involves a variety of substances, and finally take the ion channel as the target to achieve the corresponding function by controlling the ion concentration. It can be observed that the ions are at the center of different signal pathways. As a common and frequently occurring disease, there is no effective treatment for PH, and the disability rate and fatality rate are very high. From the point of view of ions, it is necessary to clarify the distribution and mechanism of ions and their corresponding ion channels in PH, so as to develop more effective drugs for the treatment of PH.
Ca2+
As one of the indispensable ions in many physiological functions of human body, Ca2+ participates in contraction and relaxation of muscle tissue, cell proliferation, stability of biological potential inside and outside the cell membrane, neuromuscular signal transduction, coagulation, regulation of enzyme activity, etc. 4 Free Ca2+ is mainly distributed outside the cell, and a small part of it is distributed in the cell. About 90% of the intracellular calcium ions are stored in the endoplasmic reticulum (ER) and mitochondria, also known as stored calcium. Calcium channels regulate the distribution of calcium ions inside and outside the cell and coordinate various physiological functions. Ca2+ plays an important role in the pathogenesis of PH. Different types of calcium channels are distributed in pulmonary vessels. These calcium channels regulate pulmonary vasoconstriction and remodelling alone or interacting with each other, thus taking part in the formation of PH. The main calcium channels are store-operated calcium channels (SOCCs), receptor-operated calcium channels (ROCCs) and voltage-dependent calcium channels (VDCCs). The following describes the mechanism of Ca2+ release-activated Ca2+ channel(CRAC), inositol 1,4,5-triphate receptors (IP3Rs), transient receptor potential channel (TRPCs), L-type calcium channels (LTCCs) in PH.
CRAC
CRAC is the highest channel in SOCCs, which is found in the electrophysiological study of mast cells and T lymphocytes.5,6 CRAC regulates the store-operated calcium entry (SOCE). Stromal interaction molecule (STIM) and Orai cationic channels consist of the CRAC. STIM is an ER Ca2+ receptor. In mammals, STIM subtypes can be separated into STIM1 and STIM2. Orai is the storage operation channel, which is divided into three subtypes of Orai1, Orai2, Orai3. 7 Here, we equate SOCCs with CRAC. In the inactivated state, STIM is dispersed in the ER and ORAIL is dispersed in the plasma membrane (PM). When G protein-coupled receptor(GPCR)activates phospholipase C (PLC), PLC decomposes phosphorylated lipid phosphatidylinositol 4,5-bisphosphate (PIP2) into diacylglycerol (DAG) and inositol 1,4,5-trisphosphate (IP3), IP3 acts on the corresponding target on ER, triggering the release of Ca2+ in ER, resulting in a decrease in the concentration of stored calcium ions. When STIM perceives Ca2+ depletion in ER, it aggregates between ER and PM, binds Orai and activates Orai pore-forming channels to promote Ca2+ into ER filling calcium pool (Fig. 1). Through the feedback mechanism, after dozens of milliseconds of Ca2+, STIM and ORAI are separated, and then CRAC is inactivated.8–11 SOCC exists widely in different cells, regulating vascular tension, blood pressure, immunity, nerve, muscle, secretion, gene transcription and translation, etc.12,13 SOCC plays an important role in cardiovascular regulation. It was found that inhibition of SOCC could inhibit the increase of intracellular Ca2+ concentration ([Ca2+]i) induced by endothelin-1 (ET-1) and angiotensin II (Ang II), reduce systemic blood pressure (including systolic and diastolic blood pressure) and inhibit the proliferation of vascular smooth muscle cells (VSMCs). 13 Conversely, ET-1 can also regulate SOCC. It had been found that in the model of coronary artery ischemia-reperfusion injury, ET-1 induced vasoconstriction requires activation of both SOCC and LTCCs. 14 In the pathogenesis of PH, it had been observed that the expression of STIM2 was up-regulated in pulmonary artery smooth muscle cells (PASMC) of patients with PH. STIM2 could up-regulate [Ca2+]i, and promote the proliferation of PASMC by up-regulating cell proliferation-related signal molecules such as CREB, STAT3, AKT, NFAT, Ki67, etc., and up-regulate the expression of Bcl-2 (an anti-apoptotic protein) to inhibit PASMCs apoptosis. Using siRNA to down-regulate the expression of STIM2 in PASMCs can reduce the expression of the above-mentioned cell proliferation signal molecules and anti-apoptosis proteins. 15 SOCC not only promotes the contraction and proliferation of PASMC, but also inhibits its apoptosis. SOCC causes pulmonary artery contraction and remodelling in many ways, which is an important mechanism for the formation of PH.
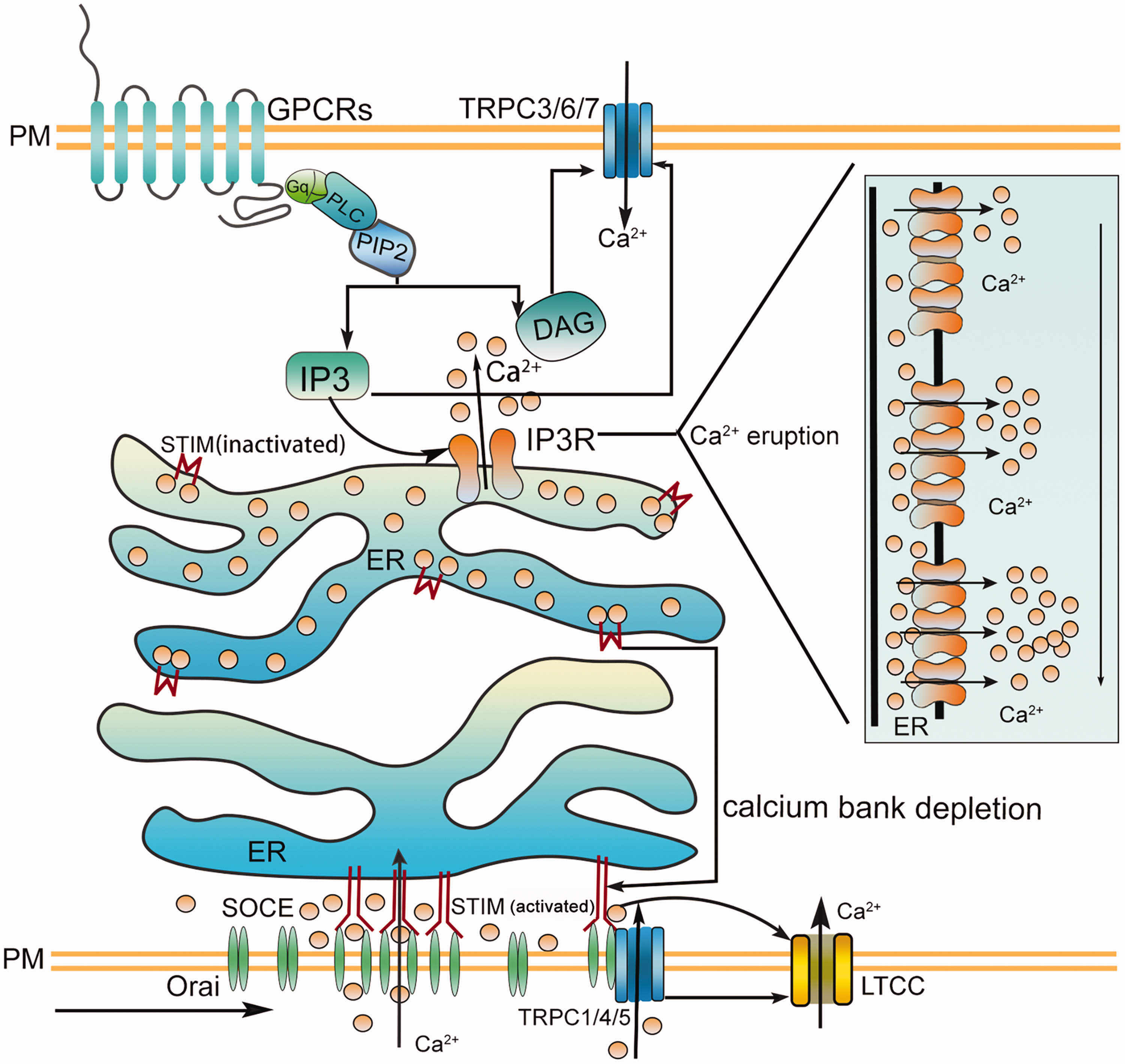
Regulation of CRAC, IP3R, TRPC, LTCC channels. In the inactivated state, STIM is dispersed in the ER and ORAIL is dispersed in the plasma membrane (PM). When GPCR activates PLC, it then decompresses phosphorylated PIP2 into DAG and IP3. IP3 acts on IP3R of the endoplasmic reticulum, triggering the release of Ca2+ from the endoplasmic reticulum, leading to a decrease in the concentration of stored Ca2+. When STIM senses endoplasmic reticulum Ca2+ depletion, it accumulates between the endoplasmic reticulum and plasma membrane, binds ORAI, activates ORAI pore forming channels, and promotes Ca2+ entry. In general, DAG directly activates TRPC3/6/7, but IP3 can also directly activate TRPC channels in some specific cells. STIM and Orai combine with TRPC1/4/5 to activate TRPC. TRPC openness promotes Ca2+ influx and depolarization of cells, which in turn activates LTCCs hyperpolarization. Under the stimulation of IP3 and Ca2+, from the activation of a single IP3R channel to the cooperative opening of IP3R organized into clusters, the final Ca2+ eruption is produced.
IP3R
IP3R is a channel made up of subunits of 4-260 kDa, which belongs to ROCC. 16 IP3R is mainly distributed in ER and releases stored calcium in a short oscillatory manner, which is the main channel of calcium release in VSMCs. 17 IP3R has IP3R1, IP3R2 and IP3R3 phenotypes, among which IP3R1 plays a leading role in human pulmonary arterial smooth muscle cells (HPSMCs). Under the regulation of some stimuli (hypoxia, inflammation) or substances (Ang II, norepinephrine, ET-1, etc.), IP3R expression is up-regulated and increases [Ca2+]i, which promotes vasoconstriction and remodelling and leads to increased vascular resistance. However, when Ca2+ concentration rises to a certain level, IP3R will in turn be inactivated.18–20 IP3 binds with IP3R in PASMCs to activate the channel. PKG, PKC, CaMKII, and so on, can further promote the binding of IP3 and IP3R through phosphorylation, thus strengthening the opening of the channel. Channel activation promotes the release of stored calcium, up-regulates [Ca2+]i, and regulates the contraction and proliferation of PASMCs.20–22 IP3Rs is associated with a number of different signal pathways to regulate diverse functions in different cells. 23 It is activated or inhibited by different substances in vivo, such as calcium binding protein with EF-hand structure, proto-oncogene, tumour suppressor gene, protein kinase, phosphatase, microRNAs, cellular redox products and so on.24–28 In pulmonary arteries, vasoactive substances and signal pathway molecules such as ET-1, 5-HT, NE, Ach and CaSR, CN/NFAT pathway can regulate pulmonary vascular tension by up-regulating the activity of IP3Rs, promoting the release of stored calcium and regulating pulmonary vascular tension.29,30 The released Ca2+ can further regulate the activity of IP3R and give positive feedback to the activity of IP3R, thus further promoting the increase of [Ca2+]i. However, the increase of Ca2+ to a certain extent gives negative feedback on the activity of IP3R.31–33 The mutual regulation mechanism between IP3Rs and calcium ion depends in part on its spatial distribution in the cell. IP3R can be organized into clusters and open in coordination to form calcium eruptions (Fig. 1). 33 However, inhibition occurs in some spatial distributions. It was found that the endothelial cell (EC) were flattened by a certain pressure on the artery, and the influx of Ca2+ mediated by IP3 decreased with the increase of pressure. Considering the change of endothelial shape, the geometry of the diffusion space between IP3R and nearby Ca2+ was changed, which limited the IP3-mediated Ca2+ signal. 34 However, it does not rule out that there are differences in the regulatory mechanism of calcium in different cells by IP3R. Interestingly, the same channel phenotype may have opposite function in different blood vessel cells. Mountian et al. found that the expression of IP3R1 in EC of spontaneously hypertensive rats was less than that of the control group, while the expression of IP3R2 was higher. 35 To further clarify the role and possible mechanism of IP3R1 in blood pressure regulation, recently, Yuan et al. knocked out the IP3R1 gene in mouse EC and found that mice were the most likely to develop hypertension. After IP3R1 gene knockout, the reactivity of EC to Ach decreased, and the expression of nitric oxide synthase (NOS) and NFAT decreased. 36 Because NOS is involved in the synthesis of nitric oxide (NO), it is speculated that IP3R1 can promote the synthesis of NO, and IP3R1 can maintain the vasodilation function in EC. However, different phenotypes in the same cells may have opposite functions. It was found that in PASMC, PAP increased in PH animal model after knockout of IP3R2 gene. The specific mechanism may be that IP3R2 promotes the apoptosis of PASMC and inhibits intracellular calcium ion concentration. 37 IP3R can also interact with other channels to regulate calcium concentration. The main function of SOCC is to sense the change of calcium concentration in calcium store. When the IP3R channel is opened, the calcium ion in ER is released, which reduces the calcium concentration in the calcium store. Calcium depletion activates SOCC to promote the influx of extracellular calcium ions, thus increasing [Ca2+]i. IP3R and SOCC cooperated to regulate intracellular calcium concentration. 38 Release of Ca2+ by IP3Rs can also activate channels such as VDCC and TRP to further increase Ca2+ influx. 18 IP3Rs is regulated by a variety of substances or signal pathways, which is the principal channel for calcium storage and release, and can be used as an important therapeutic target for PH. However, it is worth noting that different or the same IP3R subtypes have different effects on blood vessels in different cells and different distribution spaces.
TRPC
TRPC was first discovered in the study of light mutants in Drosophila melanogaster. 39 TRPC is a receptor-type cationic channel with six transmembrane regions (S1–S6) and the ends of NH2 and COOH. 40 TRPC is widely distributed in mammalian tissues and cells and has a wide range of functions. In blood vessels, it can participate in the mechanical stimulation of blood vessels and GPCR-related signal pathways, EC-related vasodilation, vascular permeability, neovascularization, vasoconstriction, remodelling and so on. The above functions are mainly realized by regulating [Ca2+]i. It plays an important role in the formation of cardiovascular disease.41,42 TRPC can be separated into multiple subtypes of TRPC1-7. All subtypes (except TRPC2, which does not combine with any TRPC protein) can combine with each other to form homotetramers or heterotetramers to perform their corresponding functions. 43 TRPC regulates Ca2+ mainly through receptor-operated channel(ROC)and store-operated channel (SOC).44,45 ROC is mainly TRPC3/6/7, SOC is mainly TRPC1/4/5. TRPC3/6/7 activation is closely linked to GPCRs/PLC. PLC decomposes PIP2 to produce IP3 and DAG, DAG directly activates TRPC3/6/7. IP3 can also directly activate TRPC channels in some specific cells (Fig. 1).46–49 SOC can direct the transfer of TRPC1 to the PM through Orai1-mediated Ca2+, and then the interaction between STIM1 and TRPC1 triggers the Gq/PLC-related signal pathway to regulate calcium concentration (Fig. 1).50,51 TRPC1, TRPC4 and/or TRPC5 can also participate in SOCC directly without the mediation of STIM1 and Orai1. 52 There is also a certain relationship between TRPC and other ion channel proteins. The regulation of TRPC on Ca2+ depolarizes cells and then activates LTCCs hyperpolarization (Fig. 1). TRPC can also promote RyRs-mediated Ca2+ release, activate BK channels, cause potassium ion outflow and regulate smooth muscle cell membrane contraction.41,53 It had been found that in the model of PH, activation of TRPC not only regulated the contraction and proliferation of PASMC, but also mediated the proliferation and migration of pulmonary adventitia fibroblasts. TRPC mediated the synthesis of extracellular matrix proteins by up-regulating the expression of type I collagen and fibronectin, thus participating in pulmonary adventitia remodelling. 54 TRPC has a direct or indirect effect on the regulation of calcium ions. GPCRs and various angiogenic substances (5-HT, Ang II, ET-1, etc.) can activate TRPC to regulate intracellular calcium concentration, mediate vascular contraction and remodelling. The role of TRPC in the pathogenesis of PH cannot be ignored.
LTCC
LTCC is a Cav1 channel, which belongs to VDCC, subtypes including Cav1.1, Cav1.2, Cav1.3 and Cav1.4. Different phenotypes have different biophysical characteristics. CaV1.2 and CaV1.3 are widely distributed and expressed in most excitable cells (smooth muscle, myocardium, etc.) with high sequence homology. The distribution of CaV1.1 and CaV1.4 is relatively limited. CaV1.1 is mainly distributed in skeletal muscle and CaV1.4 is mainly distributed in the retina. LTCC is a voltage-dependent non-specific calcium channel, which can maintain a long-term activation state and form a long-term inward current.39,55,56 LTCC is the primary channel involved in the influx of extracellular calcium ions into cells. When endovascular pressure increases, vascular smooth muscle membrane depolarizes, activates LTCC channels, and allows a large amount of calcium ions to flow into the cytoplasm, causing vasoconstriction. Cav1.2 is the main subtype of LTCC channels to regulate vascular smooth muscle contraction. 57 It has been observed that knockout of Cav1.2 gene can reduce the frequency of calcium discharge by about 70%.58,59 The calcium influx varies with different channel activity. When the channel activity is low, a small amount of isolated LTCC randomly opens to produce a small amount of Ca2+ influx. When associated with some substances, such as protein kinase C α, a large number of clustered LTCC channels increase activity and cooperatively open, resulting in persistent and sizable amount of Ca2+ influx. PKCα is essential for LTCC to produce persistent substantial amounts of calcium current.60–62 LTCC-mediated Ca2+ flow into the cytoplasm can activate RyR2 channels and produce calcium sparks through the mechanism of calcium-induced calcium release. 63 It has been found that the β subunit of LTCC structure plays an important role in the up-regulation of LTCC activity in VSMCs, and participates in the increase of blood pressure induced by Ang II. 63 LTCCs plays an important role in the vasoconstriction of PH. The expression of LTCCs is up-regulated in the model of hypoxic PH, which leads to PASMC contraction. The use of inhibitors of LTCCs can significantly reduce the absolute contraction of pulmonary artery. 64 A variety of vasoconstrictor substances can regulate vasoconstriction by mediating LTCCs. LTCCs inhibitors can significantly inhibit arterial constriction induced by ET-1, U46619 and 5-HT. It is worth noting that there are species (rat and mouse) differences in vascular responses to LTCC and vasoconstrictors. 65 It is speculated that there may be differences in the expression of corresponding receptors or phenotypes of certain substances in different species. LTCC does not function independently and is associated with other ion channels to mediate vasoconstriction. LTCC channels can interact with RyR, TRPC, BK and other channels to regulate vascular tension. LTCC can couple with RyR to produce calcium sparks. The specific mechanism is that LTCC regulates [Ca2+]i, which affects the concentration of calcium in sarcoplasmic reticulum, and finally triggers the release of calcium sparks from RyR. 59 The increase of [Ca2+]i induced by opening LTCC can activate the potassium channel, which leads to the outflow of K+, and then the cell membrane is hyperpolarized. Hyperpolarization of cell membrane leads to the decrease of LTCC activity, then the decrease of Ca2+ influx and the decrease of [Ca2+]i, and finally leads to contractile vasodilation. It can be used as a feedback regulation mechanism. 66 It has been found that CaV1.2, Orai1 and TRPC1 channels can form macromolecular complexes in close distribution areas, and functional crosstalk exists to regulate [Ca2+]i. After 5-HT and Thapsigargin (TG) stimulation, crosstalking between the three channels was enhanced. 57 Weigand et al. found that hypoxia-induced pulmonary vasoconstriction could be achieved by up-regulation of [Ca2+]i by SOCE and VDCC. SOCE or VDCC alone could not provide sufficient calcium concentration to trigger PASMC contraction, and SOCE activation could mediate the secondary activation of VDCC. 67 On the contrary, some studies had found that after STIM1 perception calcium store was depleted and activated, the activated domain binds to the C-terminal of CaV1.2 channel, which markedly inhibits CaV1.2 channel.68,69 It is possible that SOCE has different regulatory effects on calcium channels of different phenotypes. LTCCs not only regulate Ca2+, but also couple with potassium channels and mediate K+ currents. It has been found that CaV1.3 channels can activate large and small conductance potassium channels activated by calcium, which can weaken K+ currents in the absence of CaV1.3. 55 Since dihydropyridine cannot completely block this channel at present, there is no highly selective LTCC inhibitor at present. 56 The effects of different phenotypes of LTCCs are different, and the effects are also different in different species. It is necessary to further clarify the sequence differences between subtypes and strictly grasp the differences between human and animal models to achieve accurate medical care.
Ca2+ plays an important role in the occurrence and development of PH. Many signal molecules and vasoactive substances eventually affect the concentration of Ca2+, resulting in vasoconstriction and remodelling. At the same time, Ca2+ can also activate other ion channels. Calcium channels are widely distributed and have different expressions and functions in different cells, tissues and species. Therefore, it is necessary to clarify the differences and develop highly selective targeted drugs to achieve personalized treatment.
K+
K+ is the main cation of intracellular fluid, most of which exist in cells, and play an important role in human circulation, endocrine, immune inflammation, nerve, etc. In the pathogenesis of PH, K+ participates in the change of membrane potential, cell proliferation, apoptosis, inflammation, regulation of vascular tension, etc. K+ plays an important role in the pathogenesis and treatment of PH. There are mainly five different functional types of potassium channels: voltage-gated potassium channels (Kv), calcium-activated potassium channels (KCa), ATP-sensitive potassium channels (KATP) and double-pore domain K+ channels (K2P), inward rectifier potassium channels(Kir). 70 The following mainly discusses the role of Kv, KCa, KATP and K2P channels in the pathogenesis of PH.
Kv
Kv is separated into Kv1-12 subfamily members, which are expressed in almost all VSMC, involved in VSMC proliferation, migration and secretion, mediating vasoconstriction and remodelling. 71 Kv channel plays an important role in the pathogenesis of PH. It has been found that Kv channel function is inhibited in secondary (chronic hypoxia) or primary PH, but the inhibition degree of primary PH is higher. Inhibition of Kv channel can cause cell membrane depolarization, induce [Ca2+]i increase and eventually lead to pulmonary vasoconstriction and PASMCs proliferation.72,73 However, later studies found that there were differences in the expression and function of different subtypes of Kv channel. Among the subtypes of Kv channel, Kv1.3 is mainly involved in cell proliferation. In the proliferation model, it was found that the expression of Kv1.3 and its subsidiary subunit Kvβ2 was up-regulated, and inhibition of Kv1.3 would inhibit cell proliferation (Fig. 2).74–76 There was a negative correlation between Kv1.5 and Kv1.3 expressions, and the up-regulation of Kv1.5 was accompanied by the down-regulation of Kv1.3 (Fig. 2).76–78 Other factors that regulate cell proliferation, such as microRNA and mitogen, also regulate cell proliferation by down-regulating the expression of Kv1.5.79,80 Kv channel can not only promote cell proliferation, but also inhibit apoptosis. It has been found that after inhibiting the anti-apoptotic protein Survivin, the expression of Kv1.5 and Kv2.1 was up-regulated, and the apoptosis of PASMCs was promoted in the model of PH.81,82 Kv channel plays a central role in cell proliferation, and its expression and activity are the key to VSMCs dedifferentiation and transformation into proliferation. Kv1.3 is an important subtype involved in cell dedifferentiation. PAP-1(a Kv1.3 channel blocker) can down-regulate the expression of αSMA, FGFR-1, PDGFR-b and EGRF-1(cell cycle regulatory factor) to inhibit the proliferation of SMC and effectively improve the degree of vascular stenosis.83,84 To sum up, highly selective inhibitors can effectively inhibit pulmonary vascular remodelling and prevent the further progress of the pulmonary artery. One of the main pathological mechanisms of PH is the continuous progression of pulmonary vascular remodelling, so targeting Kv channel will be a potential and effective treatment direction.
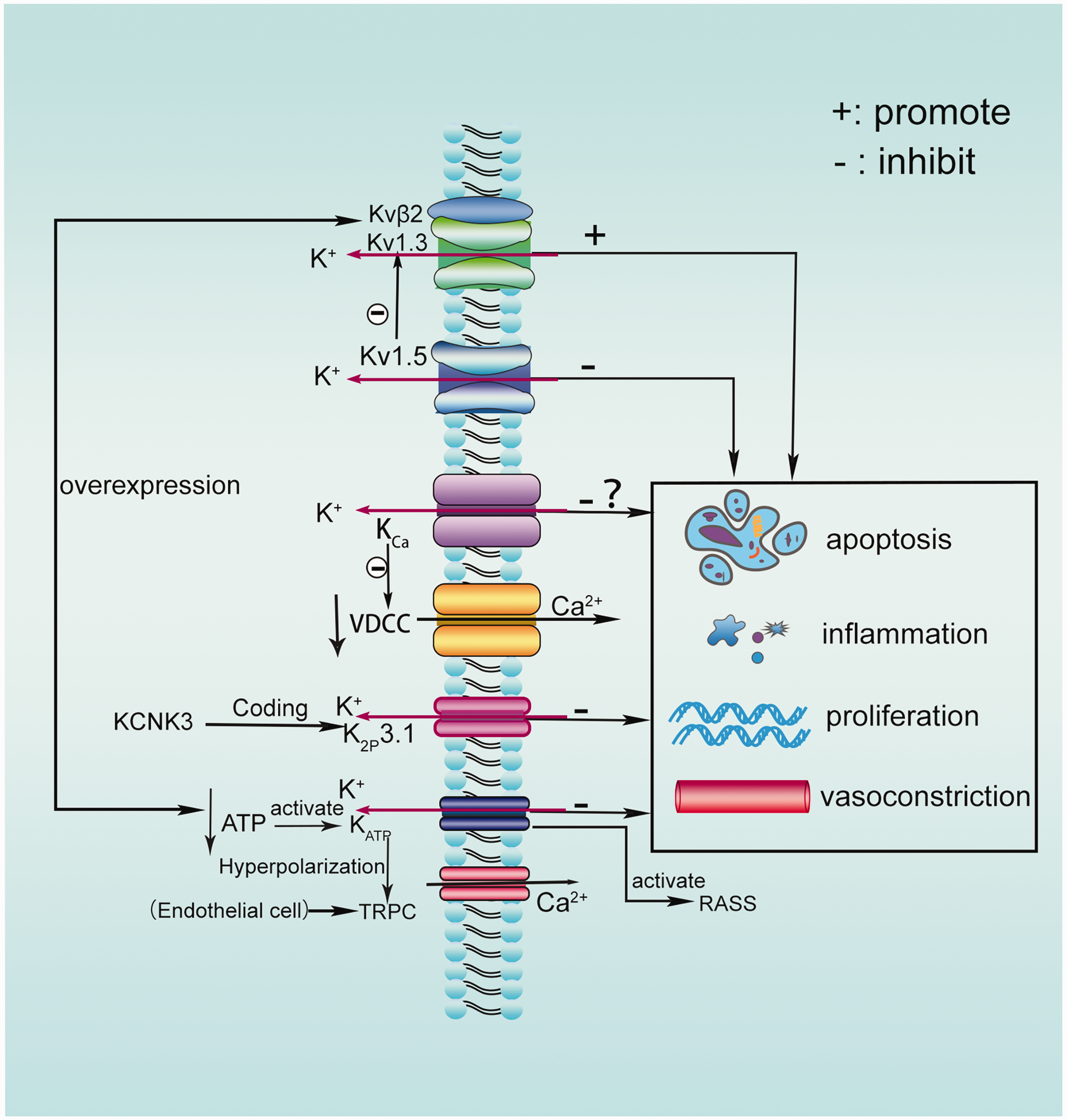
Regulatory effects of overexpression of different potassium channels on vasoconstriction and remodelling. In general, activation of potassium channels inhibits inflammation, cell proliferation and vasoconstriction to be involved in the pathogenesis of PH. However, individual phenotypes showed the opposite effect. The expression of Kv1.3 and its subunit Kvβ2 is up-regulated to promote cell proliferation. Up-regulation of Kv1.5 is accompanied by down-regulation of Kv1.3, which can inhibit cell proliferation mediated by Kv1.3 channel. Activation of KCa channel can promote K+ outflow, make membrane hyperpolarization, inactivate VDCC, and reduce [Ca2+]i, thus effectively relaxing blood vessels and inhibiting vascular remodelling. However, inhibition of KCa3.1 in some laboratories can alleviate vasoconstriction and remodelling. KCNK3 encodes K2P3.1 channel, and k2P3.1 overexpression can inhibit vasoconstriction and remodelling. The decrease of ATP activates KATP channel, hyperpolarizes the membrane, relaxes blood vessels, lowers systemic blood pressure and activates RASS system. In vascular endothelial cells, KATP activation can activate TRPC channel to make Ca2+ influx.
KCa
KCa was first found in red blood cells. 85 KCa can be divided into three subtypes: large conductance Ca2+-activated potassium channels(BK), small-conductance Ca2+-activated potassium channels(SK), intermediate Ca2+-activated potassium channels (IK). SK (KCa2.1, KCa2.2, KCa2.3) and IK (KCa3.1) channels are not widely distributed in smooth muscle cells. SK is mainly distributed in the nervous system. At present, BK channels are widely studied in vascular diseases.86,87 BK is a channel of seven transmembrane domains (S0–S6), which is composed of several subunits (α, β, γ). 88 The tertiary structure of BK channel is composed of transmembrane region and cytoplasmic region. Pore domain and voltage sensor constitute transmembrane region. Calcium sensor is distributed in cytoplasmic region, which is composed of RCK1 and RCK2. RCK1 and RCK2 are potassium conductance structures. 89 When the intracellular calcium concentration increases, the calcium ion binds to the calcium sensor, and then opens the BK channel, which causes potassium ion outflow, leading to membrane hyperpolarization and vasodilation. As a negative feedback regulation of vasoconstriction and calcium influx, 90 the presence of β1 subunit in the transmembrane region can increase channel calcium sensitivity to regulate the contraction and relaxation of smooth muscle cells. In SMC, calcium sparks released from sarcoplasmic reticulum activate BK channels, and the concentration of calcium ions can reach micromoles. The specific mechanism is that calcium ions in calcium sparks gather at the edge of α and β1 subunits in the structure of BK channels, and the channels are metamorphosed and opened. It was found that β-1 subunit gene knockout could eliminate the outward potassium current produced by calcium spark activated BK channel.91,92 CAMP, PKC and H2S can also promote the opening of BK channel in rat PASMC. BK is regulated by many different substances. 92 KCa channels are voltage- and calcium-dependent, and can mediate vasoconstriction or relaxation by regulating the activity of signal molecules such as PKG, PKA, NO, CO, ROS, ET-1, Ang II and H2O2.66,93–96 After the activation of the channel, the outflow of K+ was increased, which led to the hyperpolarization of the cell membrane. Hyperpolarization of cell membrane leads to the inactivation of VDCC, which in turn leads to the decrease of [Ca2+]i, which plays an important role in the regulation of arterial blood pressure. 66 This channel plays an important part in the regulation of pulmonary artery tension, and many substances achieve pulmonary artery relaxation by opening KCa channels.97,98 Through gene knockout of BK-Beta1 subunit, it was found that blood pressure in gene knockout group was significantly higher than that in control group. 99 Si et al found that EC and SMC in blood vessels of KCa3.1−/− mice showed a decreased response to Ach hyperpolarization and endothelium-derived hyperpolarizing factor (EDHF)-mediated vasodilation, resulting in an increase in arterial blood pressure. 100 Minami et al. found that ANG II and ET-1 can contract arterial vessels by inhibiting KCa channels. 101 Interestingly, Peng et al. found that under normoxic conditions, ET-1 plays a dual role in PASMCs under different concentrations. When ET-1 is 5 nM, BK is activated by up-regulating [Ca2+]i. When the concentration of ET-1 increased to 10 nM, ET-1 inhibited BK. However, under hypoxia, ET-1 in HPSMCs markedly inhibited the opening of BK channel at 5 nM. It is suggested that there may be differences in the regulation of BK channels by upstream molecules in different concentrations and different environments. 102 It is worth noting that recently, Guo et al found that the expression of KCa3.1 in PASMCs induced by chronic hypoxia was up-regulated than that in the control group. By injecting TRAM-34(a Kca3.1 highly selective inhibitor) into the abdominal cavity of male SD rats with intermittent hypoxia, it was found that it could reduce pulmonary artery contraction or remodelling and right ventricular pressure, inhibit ERK/p38MAPK signal transduction in PASMC, and reduce the proliferation or differentiation of PASMC. It is suggested that the function of different subtypes of KCa channel may be different. 103 From the above contradictory experimental results, it is suggested that we should pay attention to the drug concentration and action conditions of KCa channels in the treatment of PH in the future, and clarify the differences of action mechanisms of different families of channels.
K2P
K2P channel was found in yeast in 1995. 104 K2P is a K+ channel with the characteristics of four transmembrane domains and two P domains in series, so it is named K2P. 105 K2P is encoded by KCNK1-18 gene and has 15 members. According to its physiological and pharmacological characteristics, K2P is separated into five families: (1) TWIK (Tandem of P domains in Weak Inward rectifier K+ channel), (2) TREK/TRAAK (TWIK-Related K+ channel/TWIK Related Arachidonic Acid-Stimulated K+ channel), (3) THIK (Tandem pore domain Halothane-Inhibited K+ channel), (4) TALK (TWIK-related Alkaline pH-activated K+ channel) and (5) TASK (TWIK-related Acid-Sensitive K+ channel). K2P is involved in many physiological and pathological processes, including PH, endocrine, pH homeostasis, nerve excitability, heart rhythm, temperature, etc.70,106,107 The electrophysiological characteristics of K2P channel are different from other K+ ion channels. K2P channel has no voltage sensor and is not subject to the change of membrane potential. K2P channel produces a kind of instantaneous inactive current, which is called K+ leakage current. Studies have shown that it is almost or weakly sensitive to clinical K+ channel blockers, but can be regulated by different stimuli, such as GPCR, PKC, PH, NO, oxides, anaesthetics, arachidonic acid, mechanical force, Ca2+, etc.106,108,109 K2P channel plays an important role in the occurrence and development of PH. It has been observed that in the model of PH, K2P is involved in pulmonary artery relaxation and endothelial hyperpolarization to maintain arterial blood pressure. 110 At present, the most widely studied gene in PH is KCNK3, which encodes K2P3.1 channel. It was noted that the KCNK3 gene encoding K2P channel was mutated and down-regulated in some non-hereditary or hereditary PH patients. In the experimental model of PH, it was found that KCNK3 plays an important role in regulating vascular tension and can inhibit distal neovascularization and some hemodynamic changes in the early stage of PH. Up-regulation of KCNK3 can inhibit vasoconstriction, proliferation of PSAMC/EC/fibroblasts, inflammatory response, and improve ventricular hypertrophy and dysfunction associated with PH (Fig. 2).111,112 It was found that the expressions of ERK1/2, AKT, SrC, HIF1- α, Survivin and vWF were up-regulated in the tissues of KCNK3 gene mutant rats, thus promoting cell proliferation or inhibiting apoptosis, while the expression of vasodilators eNOS and EDHF was down-regulated. The efficacy of sildenafil was significantly decreased in KCNK3 gene mutant PH rats. 113 In PH, RELM-β, PLC and IP3 can regulate KCNK3 to achieve vasoconstriction, cell proliferation, inflammation and other functions. 114 The study of KCNK3 gene in PH shows that KCNK3 gene can be used as an index to detect the susceptibility of PH patients and can be used as a target for gene therapy.
KATP
The KATP channel is an octameric complex composed of four subunits with pore-forming inward rectifier K+ channels (Kir6.1 or Kir6.2) and four regulatory sulfonylurea receptors (SUR1 or SUR2). The genes ABCC8 and KCNJ11 encode SUR-1 and Kir6.2 subunits respectively, which are situated on human chromosome 11. ABCC9 and KCNJ8 encode SUR-2 and Kir6.1 subunits, respectively, which are situated on human chromosome 12. ABCC9 (SUR2) and KCNJ8 (Kir6.1) are mainly expressed in smooth muscle and ECs, while ABCC8 (SUR1) and KCNJ11 (Kir6.2) are mainly expressed in pancreas and neurons. However, there were also reports of ABCC8 mutation and loss of function in children with idiopathic pulmonary arterial hypertension (IPAH).115–117 As the name implies, KATP channels can be opened due to the decrease of cell ATP, which hyperpolarizes the membrane and connects metabolism with electrical excitation. In the endocrine system, insulin release can be regulated to regulate blood sugar. KATP channels are widely expressed in the vascular system and play an important role in vascular tension regulation and systemic blood pressure maintenance. KATP participates in the formation of PH and hypertension. 118 In PASMC, Kir can control calcium influx through VDCCs, and it may regulate extracellular potassium concentration to regulate PASMC membrane potential to mediate vasoconstriction or relaxation. It is worth noting that in vascular ECs, KATP activation can increase [Ca2+]i through receptor- or store-operated channels, such as activating TRPC channels to make Ca2+ inflow (Fig. 2).119,120 Vasoconstrictor substance (ET-1,Ang II,NE,NPY1), vasodilator substance (NO,H2S,A2, EET), translational modifier substance(PKC,PI3-AKT,PKA),mediating immune inflammatory substance (LPS,TLR3,TNF,IFN), redox substance (ROS,H2O2,GSH) and transcription inducing factor (HIF-1α, NF- κB, FOXO1) can regulate vascular tension, cell proliferation and inflammation by mediating KATP activity.121,122 It was found that the expression of SUR-2 and Kir6.1 was down-regulated in hypertensive rat model.123–125 The use of KATP channel opener iptakalim can effectively reduce PAP, right ventricular systolic pressure and pulmonary arteriole wall remodelling in the model of hypoxic PH. KATP can decrease the expression of IL-β (IL-10) in peripheral blood and lung tissue, enhance the expression of platelet EC adhesion molecule-1 and eNOS, and finally reduce the inflammatory reaction and EC damage.126,127 Interestingly, long-term use of some K+ channel openers (diazoxide, minoxidil) could cause some side effects: abnormally aggravating PAP, promoting myocardial hypertrophy, increasing vascular volume, etc. The specific mechanism was that channel openers can activate renin-angiotensin-aldosterone system to promote water and sodium retention due to significant reduction of systemic pressure (Fig. 2).128,129 Therefore, in the treatment of PH with KATP channel opener, we should pay attention to its side effects. We can cooperate with diuretics to improve the side effects caused by the feedback mechanism.
Na+
Na+ is the principal extracellular cation, which is closely related to the excitability of cells. In human body, Na+ participates in water electrolysis, blood vessel, muscle, nerve function, etc. Ca2+, K+, H+ participate in Na+ entry and exit under different chemical conditions. Na+ is widely disseminated in different cells. In blood vessels, it is allocated in epithelial cells, SMC, EC, etc. Under certain conditions, Na+ affects cell proliferation, migration and contraction, which mediate vasoconstriction and remodelling, resulting in increased vascular resistance. The following is mainly about the pathogenesis of epithelial Na+ channels (ENaCs), Sodium-calcium exchanger (NCX) and Na+-H+ exchanger (NHE) in PH.
ENaC
ENaC is a mechanically sensitive ion channel, which can fall into three subtypes: α, β and γ. ENaC belongs to the degenerin protein family and participates in mechanically related myogenic responses.130,131 Angiotensin and aldosterone can act on heart, artery and kidney by regulating ENaC activity. Short-term regulation can maintain electrolyte, volume and blood pressure stability, but promote mechanical myogenic occurrence, vascular sclerosis, vascular remodelling, increased vascular resistance and systemic hypertension in the long term (Fig. 3). 132 ENaC is widely distributed. ENaC is expressed in SMC, baroreceptor nerve endings and nodular ganglion (inferior ganglion of vagus nerve), cardiovascular regulatory area of brain (outside hypothalamic nucleus), kidney, taste buds and EC. ENaC regulates humoral circulation and blood pressure through nerves, heart, kidney, blood vessels, biological receptors, etc.39,133–137 Vascular sclerosis may occur when ENaC is over-expressed for a long time. At present, EC sclerosis is mainly considered, and the degree of sclerosis is positively correlated with ENaC density. The specific mechanism of sclerosis may be through PI3K/Akt signal pathway to inhibit the release of NO from EC, thus regulating vasoconstriction and inflammation. High sodium and aldosterone can aggravate vascular sclerosis, and the use of aldosterone or ENaC inhibitors can alleviate the degree of atherosclerosis.90,130,138–141 Under the action of mechanical stimulation (pressure, traction, shear force, etc.), ENaC can promote the myogenic occurrence of blood vessels, contraction and proliferation of VSMC. Inhibition of ENaC could eliminate pressure-induced contraction of VSMC and increase of intracellular Ca2+ and Na+ in mouse kidney. 142 Kim et al. found that inhibition of ENaC in the posterior cerebral artery could inhibit the increase of Ca2+ concentration, myogenic response and myosin light chain phosphorylation induced by pressure.143,144 Recently, Choi et al. have confirmed that the expression of ENaC is up-regulated in the posterior cerebral artery of spontaneous PH rats, which significantly increases the myogenic response and myogenesis. 145 In the ENaC study, we know that ENaC can be regulated by angiotensin, aldosterone, blood pressure, shear force, baroreceptor and so on to harden blood vessels. When vascular sclerosis occurs, vascular pressure, shear force of blood flow, expression of substances regulating vascular tension and renal sodium excretion change, and baroreceptor is stimulated, which finally increases the expression of ENaC.146–148 Over and over again, a positive feedback circulatory mechanism is formed between ENaC and arteriosclerosis, which finally leads to the gradual increase of blood pressure. However, the specific mechanism of up-regulation of ENaC expression in specific sclerotic vessels has not been clarified, which needs further study. The study of ENaC is mainly in cerebral vessels and renal vessels, but virtually not in pulmonary vessels. ENaC is not excluded in the formation of PH, so it can be utilized as a research direction for the pathogenesis of PH.
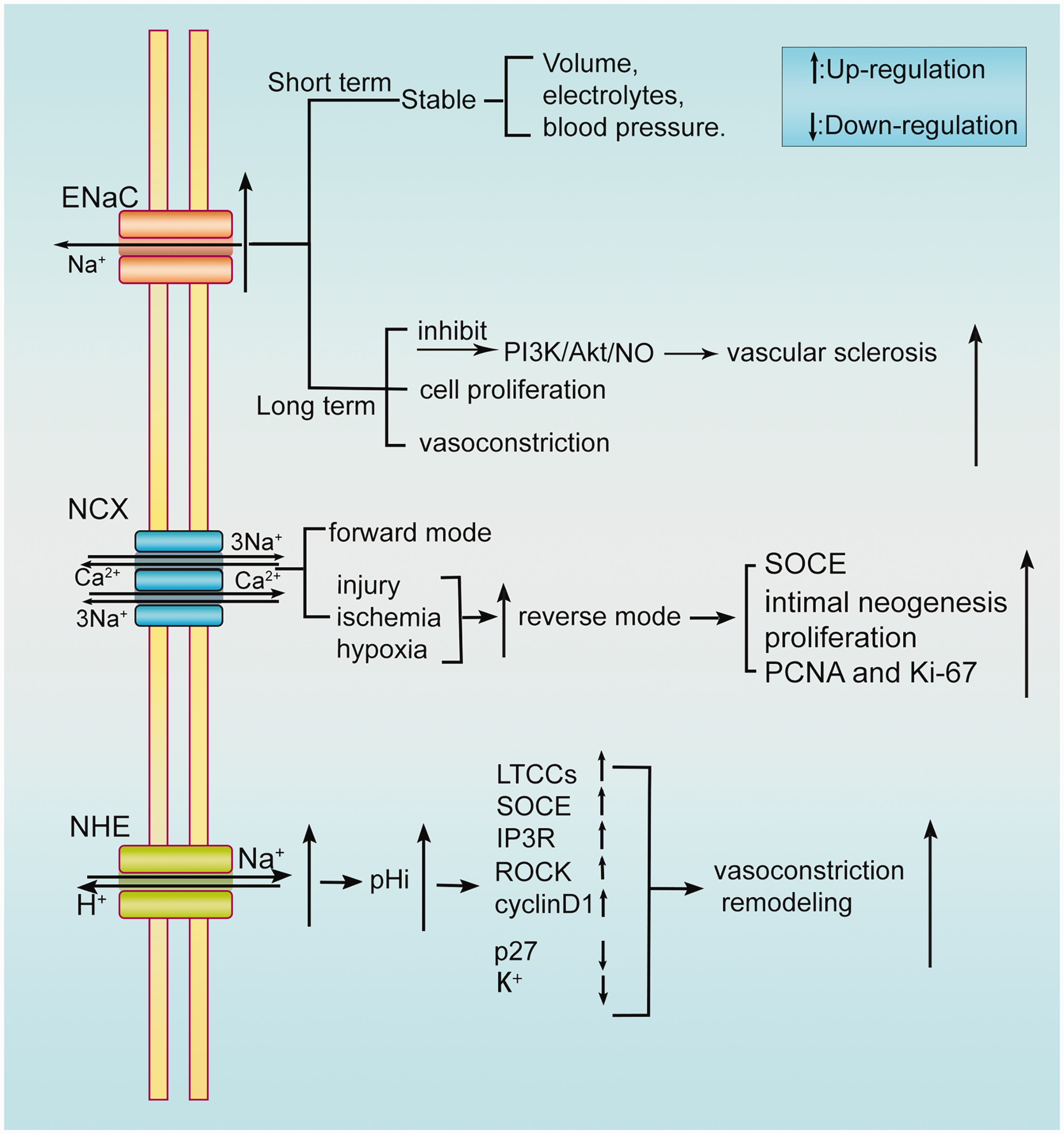
Regulation of sodium channel in vasoconstriction and remodelling. When ENaC is overexpressed, electrolyte, volume and blood pressure can be retained in the short term. However, when ENaC is overexpressed for a long time, it may inhibit the release of nitric oxide by endothelial cells through PI3K/Akt signal pathway, which leads to vascular hardening. NCX is divided into forward mode and reverse mode. In cardiomyocytes, forward mode is important for diastolic calcium excretion. Under some conditions (injury, ischemia, hypoxia, etc.), activation of reverse mode can mediate SOCE, to promote calcium influx. NHE activation can increase intracellular pH and up-regulate calcium channels (LTCC, SOCE and IP3R), cyclinD1, ROCK, down-regulate p27, resulting in increased intracellular calcium concentration, cell proliferation, vasoconstriction, etc.
NCX
NCX is encoded by the SLC8 gene family and has three subtypes: NCX1, NCX2 and NCX3. NCX2 and NCX3 were noted in the brain and skeletal muscles. NCX1 is widespread in mammalian cells. 149 NCX consists of 938 amino acid residues. In NCX, three Na+ and one Ca2+ bind to the centre of the protein. NCX is symmetrical and can produce two-way ion exchange reaction. 150 NCX is divided into forward mode and reverse mode. In the forward mode, three Na+ are passed into the cell and one Ca2+ is transferred out of the cell at the same time. In cardiomyocytes, this function is very important for the timely excretion of diastolic calcium ions. Under some conditions (injury, ischemia, hypoxia, etc.), NCX can be reversed, namely reverse mode. In the reverse mode, three Na+ were transferred out of the cell, while one Ca2+ was transferred into the cell, which increased ([Ca2+]i), mediated vascular contraction and remodelling, and finally led to the increase of vascular resistance.151,152 At present, NCX1 is widely studied in blood vessels. In order to study the role of the reverse mode of NCX in the proliferation of VSMC, Liu et al. found that the expression of NCX1 was up-regulated and neointima was formed in the model of vascular injury with balloon. Inhibition or knockout of NCX1 could inhibit neointimal formation, VSMC proliferation, SOCE, proliferating cell nuclear antigen (PCNA) and Ki-67 expression (Fig. 3). 153 Reverse mode of NCX participates in the regulation of calcium concentration in PASMC of patients with IPAH. Zhang et al found that the expression of NCX1 in PASMC of IPAH patients was up-regulated, the reverse pattern of NCX was enhanced, and [Ca2+]i was increased, thus promoting the contraction and proliferation of PASMC. 152 NCX can mediate inflammation related to macrophages, dendritic cells and B cells. NCX is also engaged in insulin secretion, calcium reabsorption in the kidney, and learning and memory related to the hippocampus.151,154 We will not elaborate on it here.
NHE
The NHE family is composed of at least 10 known isomers, and the expression is tissue-specific.NHE-1 phenotype is mainly expressed in vascular tissues, while other subtypes can be expressed in gastrointestinal tract, kidney, brain tissue, germ cells, etc.155–158 Na+-H+ exchange channels play a major role in vasoconstriction and remodelling. It was noted that the activity of NHEs in aortic smooth muscle cells of spontaneously hypertensive rats was significantly higher than that in the control group. 157 In the hypoxia-induced model, it was found that the right ventricular systolic pressure, right ventricular weight and the inner wall thickness of pulmonary arterioles in NHE1 knockout mice were significantly higher than those in the control group. 159 In the pathogenesis of PH, it is considered that NHE-1 affects intracellular pH (pHi). Some previous studies have shown that NHE-1 activity increases and pHi shows alkaline shift in PASMC in chronic hypoxia-induced PH model. 160 When the cells are alkalized, IP3R, LTCC and SOCE can be activated, thus increasing [Ca2+]i and inhibiting potassium influx, which leads to vasoconstriction and remodelling (Fig. 3).161–163 It was found that after inhibiting the effect of buffer pair (CO2/HCO3–) on pH, NHE-1 gene knockout led to cell acidification, and then decreased arterial tension, arterial media remodelling, blood pressure and Rho-mediated VSMC sensitivity to calcium ions. 164 In PASMC, HIF-1 could up-regulate NHE-1, and thus up-regulate pHi, cause alkaline drift of cells, promote cell proliferation and mediate vascular remodelling. 165 In view of the fact that ET-1 can up-regulate the expression of HIF-1, it is speculated that ET-1 can further enhance the activity of NHE-1. 166 NHE1 (–/–) mice down-regulate Rho kinases (ROCK1 and ROCK2) and cyclinD1 and up-regulate p27 expression (Fig. 3). 159 Interestingly, Huetsch et al. recently used the SuHx model (the model constructed by vascular endothelial growth factor receptor inhibitor SU5416 combined with chronic hypoxia exposure, which is more suitable for the pathological development of PH). The results showed that the activity of NHE in PASMC increased in SuHx model, and PASMC still proliferated and migrated obviously when pHi was unchanged. The degree of proliferation and migration reduced after the pharmacological inhibition of NHE, which is contradictory to the fact that NHE regulates cell contraction and proliferation by changing pHi. 167 Considering that NHE is not the only substance that regulates intracellular acid and base, there are other buffer pairs. Therefore, the result of constant pHi in the laboratory does not rule out the interference of other regulatory substances, which can be further verified after the strict control of other ion buffer substances. In short, the regulatory mechanism of NHE in the formation of PH cannot be ignored and can be used as a new target for the treatment of PH in the future.
Cl–
Cl– is the most abundant anion in vivo, and its extracellular concentration is higher than that of intracellular fluid. Cl– accounts for about 70% and 65% of the total anions in tissue fluid and plasma, respectively. 168 Cl– is involved in a variety of physiological functions, including cell volume regulation, proliferation, membrane potential changes, signal transduction, acidolysis balance, blood pressure regulation, etc. Cl– channels are widely distributed, which are classified as calcium-activated chloride channels (CaCCs), volume-or swelling- sensitive Cl– (Clswell), cysticfibrosis transmembrane conductance regulator, P64-related chloride channels and ligand-gated chloride channels. The chloride channels distributed in smooth muscle cells are mainly CaCCs and Clswell channels. 169
CaCC
CaCC includes at least two protein families, TMEM16 and bestrophins (corresponding to BEST1-4 in humans). TMEM16 is divided into TMEM16A and TMEM16B (corresponding to ANO1 and ANO2 in humans, respectively). Later studies have found that TMEM16A is a potential CaCC.170,171 CaCC is not only permeable to chloride ions, but also permeable to univalent anions such as I–, Br–, F–, SCN–, etc. 172 CaCC is a group of heterochannel proteins that are sensitive to anions and dependent on Ca2+. 173 The increase of [Ca2+]i can trigger CaCC activity, cause chloride ion outflow, form inward current and cause cell depolarization (Fig. 4). The factors regulating CaCC gating are [Ca2+]i, membrane depolarization, extracellular anions and intracellular protons. Among them, Ca2+ plays a key role in the activation of CaCC. TMEM16 is the pore-forming subunit of CaCC. There are conservative regulatory calcium binding sites in the pore region of the TMEM16A structure, in which the sixth transmembrane segment is the main gated part of the channel activated by Ca2+, and the sixth transmembrane segment has pore lining residues and Ca2+ binding sites. After binding with calcium ions, the transmembrane segment was activated after conformational change and maintained the stability of the current activated state. 174 It was found that the activation of TMEM16A was regulated by binding to at least two Ca2+ in a certain order and coupled with cell membrane voltage. 175 The coupling relationship between Ca2+ and membrane voltage changes at different concentrations, and the coupling intensity is higher at a low concentration, but when it is increased to a certain concentration, the coupling disappears. 176 With the increase of [Ca2+]i, the first Ca2+ binds to the corresponding binding site, and then the anion is allowed to enter the channel, and the channel structure rapidly undergoes a voltage-dependent conformational change, making the channel open, thus mediating the anion current, and then the second Ca2+ binds to the calcium binding site. The secondary binding of Ca2+ is mainly to completely activate the channel and maintain the stability of the current conformation. 174 The source of calcium ion can be released through VDCC, IP3R, Na+-Ca2+ reverse exchange, SOCE, etc.177–181 CaCC is widely expressed in different tissues, causing different physiological functions, such as cell contraction/proliferation, regulation of blood pressure, heart/neuron excitation, glandular secretion, olfactory signal transduction, etc.182–186 In pulmonary vessels, CaCC can cause SMC proliferation, contraction and increase of PAP. Through the study of 40 healthy donor lungs and 38 IPA donor lungs, Papp et al found that the expression of TMEM16A gene in PASMC of IPAH patients was significantly higher than that of the control group, and Cl– efflux increased, resulting in cell depolarization. Knockout or inhibition of TMEM16A can repolarize cells, inhibit cell proliferation and alleviate vascular remodeling. 187 TMEM16A could be used as a novel target for reversing PH. In PH, TMEM16A can up-regulate the expression of cyclin. Shang et al studied the mechanism of TMEM16A promoting cell proliferation in PH rat model, and found that the expression of cyclin E, D1 and TMEM16A were up-regulated in PH group. Knockout of TMEM16A gene could reverse the expression of cyclin. 186 However, there were some contradictions in the results of TMEM16A. Zeng et al. found that the overexpression of TMEM16A in basilar artery SMC could down-regulate the activity of TGF-β1/Smad3, TGF-β1/ERK and JNK1 pathway in Ang II-induced hypertension model. Tissue inhibitors of metalloproteinases (TIMP-1 and TIMP-2) were down-regulated when TMEM16A was overexpressed, and matrix metalloproteinases (MMP-2, MMP-9 and MMP-14) were down-regulated by inhibiting the activity of WNK1 (reduced Cl–-sensitive serine/threonine kinase). 188 It is speculated that the overexpression of TMEM16A can reduce the deposition of arterial matrix and play a protective role in vascular remodelling (Fig. 4). It is thought that there are differences in the role of TMEM16A in different types of arteries. It is worth noting that after specific knockout of TMEM16A in uterine SMC of mice, Qu et al. found that the calcium signal pattern, size and overall uterine contraction induced by agonists (potassium chloride, oxytocin and prostaglandin) in the gene knockout group had no significant change compared with the control group. It is speculated that TMEM16A may have no obvious effect on uterine SMC contraction, but it does not rule out the difference of experimental consequences caused by the difference of experimental detection methods. 189 Some studies have found that specific knockout of TMEM16A in VSMC does not cause complete ablation of TMEM16A protein, and TMEM16A protein still exists in VSMC in the form of truncation after gene knockout. This situation does not rule out the incomplete knockout of genes, but we still need to pay attention to it when doing experiments. 190 TMEM16A also regulates with some inflammatory factors (IL-4, IL-13, IL, etc.) and signal molecules (RhoA/ROCK, AKT, ERK1/2, SRC, CaMKII, EGFR, etc.), thus participating in inflammation, cell proliferation and signal transduction. 191 All in all, TMEM16A protein plays an important role in the pathogenesis of PH. The differences of experimental results suggest that TMEM16A may have distinct functions in different tissues, so its mechanism needs to be further clarified. At the same time, it should be noted that the expression of the protein should be detected after knocking out TMEM16A.
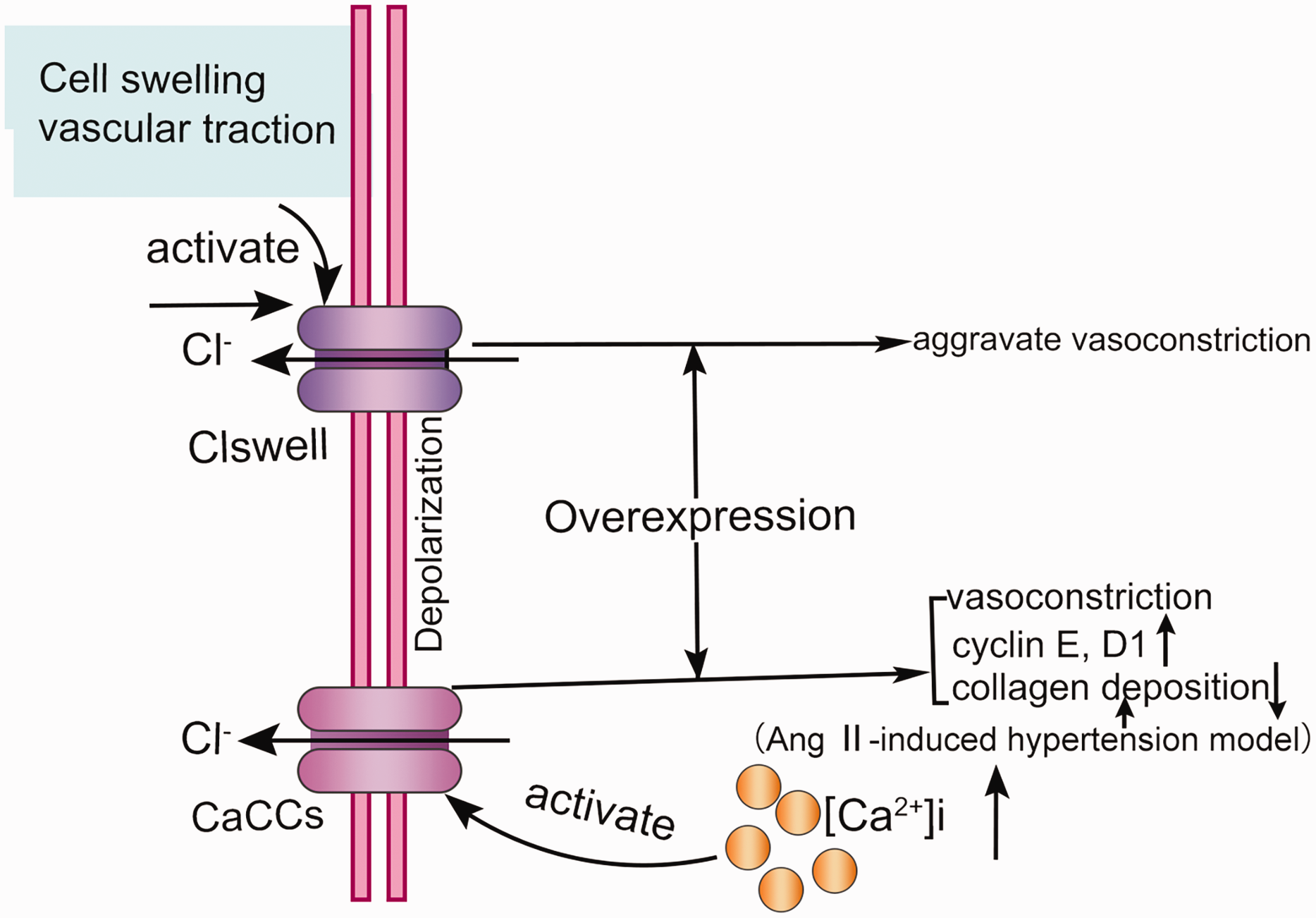
Regulation of chloride channel in vasoconstriction and remodelling. The increase of [Ca2+]i can activate calcium-activated chloride channels (CaCC), to cause Cl– outflow, form inward current, cause cell depolarization, up-regulate cyclin E and D1, thus promote vasoconstriction and remodelling. However, the overexpression of CaCC can reduce the deposition of vascular matrix in Ang II-induced hypertension model. Volume- or swelling-sensitive Cl– (Clswell) is activated when the cell volume increases and the blood vessel is pulled, which further aggravates the vasoconstriction.
Clswell
As the name implies, Clswell channels are regulated by cell volume and are activated when the cell expands and the volume increases. It can participate in cell proliferation/apoptosis, angiogenesis, pH regulation, LTCC/SOCC activation, etc. 192 In SMC, Clswell is activated when sensing changes in cell volume caused by mechanical stretching and osmotic pressure. 193 By stimulating α1-adrenergic receptor to regulate the change of arteriole pressure, it was found that the chloride current induced by cell swelling (IClswell) was sensitive to the change of transmural pressure and mechanical traction. The greater the pressure, the higher the traction force and the gradual enhancement of IClswell, which depolarizes the cells and further contracts the blood vessels (Fig. 2). The use of Clswell blockers can inhibit the spontaneous myogenic tension of blood vessels. However, Ca2+-activated chloride current (IClCa2+) mainly maintained the continuous contraction of arteries, and its ability to maintain vasoconstriction decreased with the increase of transmural pressure. 194 It is assumed that Clswell further increases blood pressure in the process of rising blood pressure. Angiotensin and epinephrine can contract blood vessels by acting on Clswell. Clswell can be used as a research direction to control the further rise of PAP, but at present, there are few studies on its role in pulmonary vessels, which need to be further explored.
Conclusions
To sum up, we know that various ions play an important role in the pathogenesis of PH, such as pulmonary vasoconstriction or relaxation, cell proliferation and apoptosis, myogenic reaction, inflammation, maintenance of cell volume, collagen deposition, fibrosis and so on. The ions do not function independently, but interact with each other, mediating the opening or closing of the ion channels, thus regulating the ion concentration on both sides of the membrane. In addition to the interaction between ions, ion channels can be regulated with different chemicals, such as vasoactive substances, inflammatory mediators, transcription-inducing factors, apoptosis factors, translation modification factors, etc. (Fig. 5). Vascular mechanical traction stimulation can also regulate the activity of ion channels. Ions participate in vascular tension, vascular remodelling and systemic blood pressure regulation through the above interactions. In addition to the vascular system, ions can also regulate the nervous system, endocrine system, metabolism, acid–base balance and so on. In short, ion channels are widely distributed, participate in different functions of the human body, and play an important role in human physiology and pathology. After years of research, it has been found that many substances use ions as targets to achieve related functions, which show that ions are in the transportation hub of various signal pathways. At present, various laboratories have studied the pathogenesis of PH from diverse aspects, and developed related drugs. However, the efficacy of drugs is poor, considering that a sole drug only acts on one aspect, while PH is formed under the interaction of different stimulating factors, not a single factor. Therefore, the combination of drugs is imperative, or finding the common target of various stimulating factors, such as ions, will achieve twice the result with half the effort. Recently, it has been proposed that energy metabolism may be a universal feature in the pathogenesis of PH, and almost all functions of the human body cannot be achieved without energy supply, which should also be paid attention to. It is worth noting that there are different subtypes of ion channels, and there are differences in channel subtypes and their expression in different tissues. Distinct subtypes or the same subtypes may have different functions in different tissues and conditions. Therefore, in drug research and development, we should pay attention to the differences of ion channel subtypes, distribution tissue and function, so as to achieve accurate targeted therapy and avoid some side effects. At present, many drugs that can be effective in animal models are not applicable in human experiments, so we pay attention to the differences of target genes, ion channel subtypes and drug doses among different species in drug research and development. In a word, as the target of various substances involved in the pathogenesis of PH, ions can be used as an important direction in the treatment of PH.
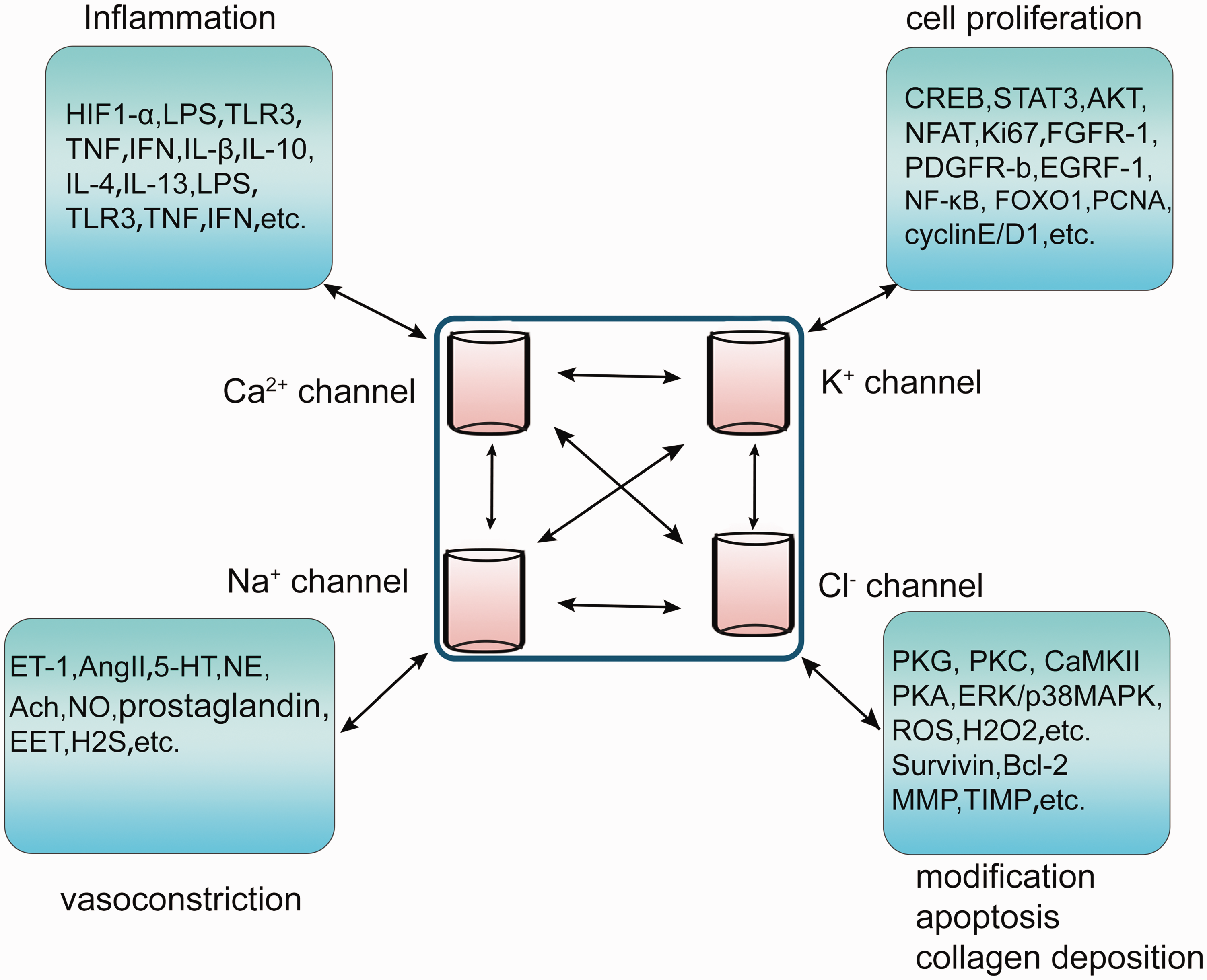
The interaction between ion channels and different substances is shown. Ions play a major role in the pathogenesis of pulmonary arterial hypertension (PH). Many different chemicals (vasoactive substances, inflammatory mediators, transcription inducing factors, apoptosis factors, translation modifiers, etc.) and mechanical stimulation mediate vascular tension and vascular remodelling through the activity of various ion channels. On the contrary, ions can also regulate the above diverse chemical substances and physical stimuli. The ions and their channels are not independent of each other, but influence each other.
At present, the treatment of PH is mainly focused on ET-1, NO, PDE5, guanylate cyclase, prostacyclin and so on. It is pointed out that the combination of drugs can improve the symptoms and delay the progress of PH, but the effect is still not as expected or cured.195–197 We need to find some new therapeutic targets in different directions. In our description of ions and their channels, the above-mentioned ions and their channels can be used as research targets for the treatment of PH. As we have previously described, in hereditary or non-hereditary PH patients, the gene KCNK3 encoding K2P channel is mutated or down-regulated, suggesting that gene targeting therapy can be undertaken. In addition to KCNK3 gene, there are also some studies on ion channel gene targeting therapy in CaV1.2, Kv1.5 and BK channels.92,198–200 However, at present, there are still some problems in the therapeutic drugs related to ion channels. Some ion channel inhibitors have different selectivity at different concentrations. However, there are still some problems in the therapeutic drugs related to ion channels. Some ion channel inhibitors have different selectivity at different concentrations; some are proved to be effective in vitro, but they cannot function effectively because of some reactions and interference in vivo; some ion channel blockers take longer time to spread, so the time to take effect is slower, increasing the concentration of the inhibitor can accelerate the time to take effect, but the drug appears non-targeted effect, which affects the curative effect or produces some side effects. Some showed inhibitory or excitatory effects at different concentrations, while some inhibitors had poor specificity and could inhibit different channels at the same time. 201 Therefore, at present, there are still some problems in the drug treatment of ion channels, and we are still not fully clear that under some conditions, the change in channel expression is the result, or the inducement, or just a compensatory change in the progression of the disease. The phenotypes of ion channels are diverse, and many different subtypes may be distributed in the same vessel, and there may be significant differences in the function of different subtypes. At the same time, in the whole between different ion channels, different cells can influence each other, which can significantly reduce our drug selectivity, accompanied by certain side effects, so it is difficult to achieve accurate and effective treatment. Therefore, it will be an interesting topic to take gene targeting therapy as the key research direction of PH therapy in the future.
Footnotes
Authors’ contributions
GL drafted the manuscript; DF and HT revised the manuscript; AD approved the final version of the manuscript.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This article is supported by the National Natural Research Foundation of China (81570052).
Guarantor
AD.
