Abstract
Schistosomiasis, a parasite-borne disease, is highly prevalent in Africa and Asia; it is estimated that close to 20 million people worldwide have a severe form of the disease. The chronic form can affect the gastrointestinal system and lead to hepatosplenic disease, and it may cause cardiopulmonary complications, including pulmonary hypertension. The exact pathogenesis of schistosomiasis-associated pulmonary hypertension (Sch-PH) remains unclear, although several mechanisms, including parasitic arterial embolization, pulmonary arteriopathy, and portopulmonary hypertension–like pathophysiology, have been suggested. The immunopathology of the disease is also unclear, although there are similarities with the immunology of idiopathic pulmonary arterial hypertension (PAH). Finally, the treatment of Sch-PH has not been well studied. There is some evidence on treating the underlying infection, with unclear effect on Sch-PH, and advanced PAH therapies are now being suggested, but more studies are needed to confirm their efficacy.
Schistosomiasis was first described in the nineteenth century by Theodor Bilharz. Initially known as bilharzia, the disease was eventually attributed to parasitic trematode worms (flukes) referred to as schistosomes, and it is now the second-most-prevalent endemic parasitic disease in the world. 1
Acute schistosomiasis is generally self-limited and benign. The chronic form of the disease, on the other hand, can cause severe health impairment. Hepatosplenic disease accounts for approximately 25% of chronic schistosomiasis and is of particular clinical interest, since it may result in liver fibrosis, portal hypertension, and migration of the parasite eggs to the lungs, which is associated with the development of pulmonary hypertension (PH) and ultimately right ventricular (RV) failure. Schistosomiasis-associated PH (Sch-PH) is recognized as the leading cause of PH in countries where schistosomiasis is endemic and, presumably, worldwide. 2 In North America and Western Europe, this disease is mostly encountered in the form of previously infected travelers or immigrants from regions of endemic disease and may be overlooked. The importance of Sch-PH becomes clearer in the context of advances in pulmonary arterial hypertension (PAH) therapy within the past 20 years, resulting in significant improvements in prognosis, life expectancy, and quality of life.
In this review, we describe the etiology, epidemiology, pathophysiology, and clinical manifestations of schistosomiasis, with particular attention to the cardiopulmonary consequences of chronic infection. We also summarize the data on treatment strategies.
SCHISTOSOMA LIFE CYCLE
Twenty-four Schistosoma species are currently recognized. These are categorized into 4 distinct groups, but only a few of these are known to be pathogenic to humans. The most prevalent human pathogens are S. mansoni and S. japonicum, primarily causing gastrointestinal disease, and S. haematobium, causing mainly genitourinary disease. S. mekongi and S. intercalatum are less prevalent but are also known to cause infections in humans. The larval forms of schistosomes, known as cercariae, live in standing or slowly moving bodies of fresh water. After direct contact with the host, these free-swimming forms penetrate through the skin, shed their bifurcated tails, and enter capillaries and lymphatic vessels. The tailless forms are termed schistosomula and migrate to the lungs and subsequently to the portal venous system via the systemic circulation. 3 After maturing into adults and mating within the portal venous system, the parasites subsequently migrate to the superior mesenteric veins (S. mansoni), the inferior mesenteric and superior hemorrhoidal veins (S. japonicum), or the vesical plexus and ureter-draining veins (S. haematobium). Approximately 6 weeks after infection, egg production commences, and it continues throughout the entire life of the adult worm (up to 5 years). Eggs pass through vessel walls into tissues and into the intestinal lumen or the bladder, resulting in shedding with feces or urine, respectively. Once the eggs hatch, miracidia are released and infect different types of freshwater snails, which act as the intermediate host of the parasite. After 2 generations of sporocysts have been produced within the snail, cercariae are released in freshwater, completing the life cycle of the schistosome (Fig. 1). 4
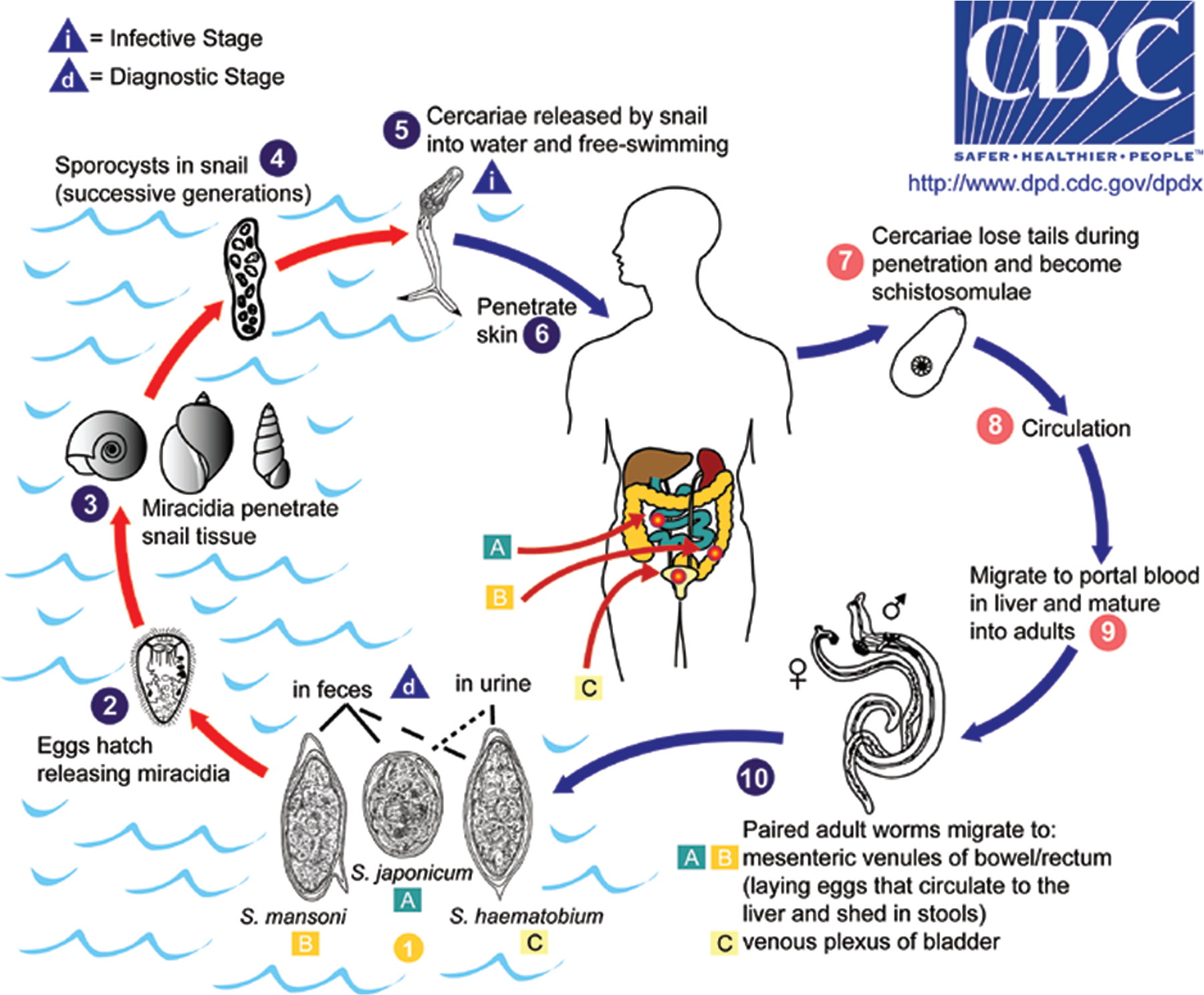
Schistosoma life cycle. Reproduced with permission from the Centers for Disease Control and Prevention (CDC: http://www.cdc.gov/dpdx/schistosomiasis/index.html).
CLINICAL MANIFESTATIONS
Schistosomiasis infection can be divided into immediate, acute, and chronic forms. The immediate form is typically characterized by an acute, pruritic, maculopapular eruption at the site of cercarial skin penetration within the first 24 hours of exposure. This may last up to 2 days, may occur even with Schistosoma species that do not usually affect humans, and is also known as Schistosoma dermatitis or swimmers itch.
The acute form, also known as Katayama fever, is associated with primary exposure and therefore is more common in areas with high transmission rates. It is a toxemic syndrome occurring 6–8 weeks after inoculation and is commonly observed in individuals traveling through areas of endemic disease; it is uncommon among endogenous populations. It can be asymptomatic, or it can present with nonspecific symptoms, such as headaches, myalgias, or arthralgias, right-upper-quadrant abdominal pain, bloody diarrhea, fever, malaise, eosinophilia, and, in the case of
S. mansoni, respiratory symptoms. 5 The latter can include cough, dyspnea, or wheezing and constitute the acute form of pulmonary complications associated with schistosomiasis. The severity of symptoms may vary, and although radiographic findings can be present, such as a nodular or reticulonodular pattern on chest radiograph or computed tomography, these do not necessarily correlate with symptomatology. 6
Acute schistosomiasis can be severe and may be complicated bymeningoencephalitis 7 or other neurological complications, 8 such as brain infarcts. 9 Cardiopulmonary complications, such as endomyocardial fibrosis, 9 pericarditis, 10 and myocarditis,11,12 can also occur. Nevertheless, acute disease is usually self-limited, resolves spontaneously within a few weeks once the parasites reach their target organ, and is not associated with significant long-term sequelae.
Clinical manifestations of chronic schistosomiasis vary with the characteristics of established infection, such as the egg burden and the duration of infection. 4 Manifestations also differ with Schistosoma species. Chronic intestinal (also referred as hepatointestinal) schistosomiasis, defined as the presence of parasites in the stool without portal hypertension, as well as chronic hepatosplenic schistosomiasis, defined as the presence of parasites in the stool with portal hypertension and usually periportal fibrosis (with or without splenomegaly), are mainly caused by S. japonicum and S. mansoni.13–15 S. haematobium, on the other hand, can cause chronic genitourinary disease. Extrapulmonary clinical manifestations of the disease are summarized in Table 1. Of note, chronic schistosomiasis–associated lung pathology can lead to pulmonary vascular remodeling and pulmonary hypertension, with subsequent RV dysfunction and failure. This may lead to shortness of breath on exertion or rest, as well as volume retention evidenced by lower-extremity edema, and ascites.
Extrapulmonary clinical manifestations of chronic schistosomiasis infection

EPIDEMIOLOGY OF SCHISTOSOMIASIS, ITS HEPATOSPLENIC FORM, AND SCH-PH
Africa is the geographic area most affected by schistosomiasis, and the infection is also common in South America, the Caribbean, the Middle East, and parts of Asia. 1 It is estimated that 200 million people are infected globally, of whom 120 million have symptoms. Close to 20 million have severe chronic disease, which is particularly prevalent in sub-Saharan Africa (?85% of those infected with Schistosoma). In areas of endemic disease, the chronic form of the disease due to repeated infections is the most prevalent form. 16 Persons aged 5–20 years seem to be the most affected, with decreased prevalence and intensity in older populations, except for higher-risk groups.16,17 In addition, there is now increased recognition of higher disease frequency in younger children and infants than previously thought. 18 The elevated morbidity in a younger population, with potential lifelong negative repercussions, and the large number of people with residual morbidity from previous infections without active disease significantly increase the burden of schistosomiasis worldwide, to more than double the number of estimated infections. 16 Based on the above, there is a clear global interest in better control of the disease and improved treatment goals, mostly through mass drug treatment and promotion of proper sewage disposal, in hopes of reducing disease burden. 15
Among patients with chronic disease, the prevalence of hepatosplenic disease may be as high as 25%, although more recent studies estimate the prevalence at closer to 10%.2,19,20 Approximately 4%–8% of patients with hepatosplenic disease develop liver fibrosis. 4 The prevalence of pulmonary hypertension in patients with hepatosplenic chronic schistosomiasis has been estimated at 8%–25% by various studies based on differing definitions and diagnostic modalities.8,21–24 Thus, schistosomiasis-induced pulmonary hypertension is encountered commonly in areas of high endemicity (Fig. 2). 25
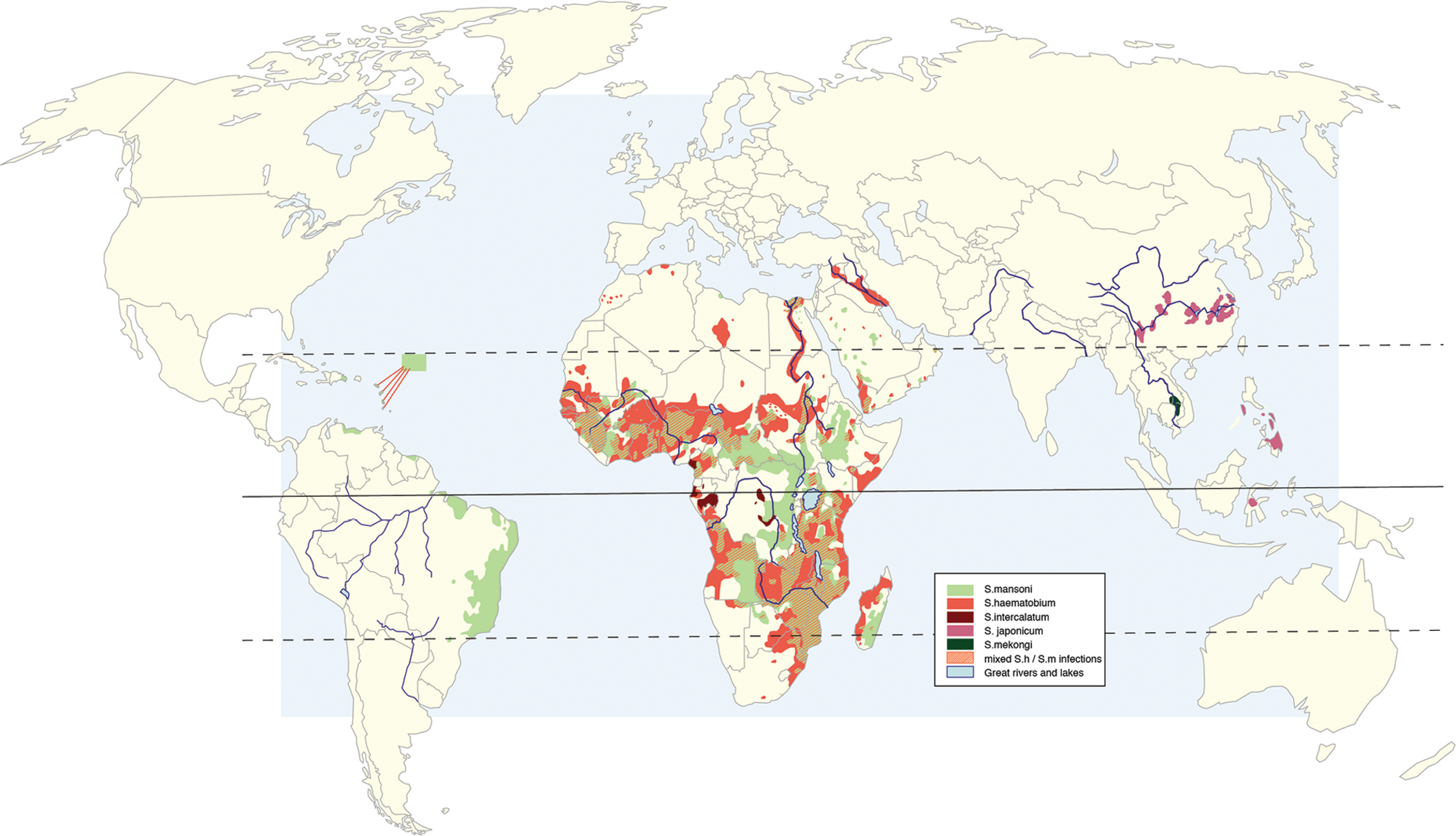
Global distribution of schistosomiasis infection. Copyright Institute of Tropical Medicine (ITM), Antwerp, Belgium; reproduced with permission; previous version published in Lancet. 25
The diagnostic criteria of Sch-PH have not been clearly defined. Moreover, this can be complicated by the fact that patients with chronic schistosomiasis in areas of endemic disease could also have non-Schistosoma-related PH. Nevertheless, the most commonly used definition of Sch-PH, and the one we would recommend, is based on the presence of the following: PH, as per the established definition (mean pulmonary artery [PA] pressure [mPAP] higher than 25 mmHg and PA wedge pressure lower than 15 mmHg); 26 Schistosoma parasite eggs in the stool or rectal biopsy, prior treatment for schistosomiasis, or exposure to a region where schistosomiasis is endemic; and ultra-sonographic evidence of liver disease consistent with hepatosplenic schistosomiasis. 27 It could be argued that sometimes, only one of the two latter findings is necessary, since PH could, in theory, develop in non-hepatosplenic chronic schistosomiasis cases. This is based on the premise that a parasite-induced inflammatory cascade can cause pulmonary vasculopathy leading to PH,23,24,28 although it is still believed that hepatosplenic disease is a necessary prerequisite for PH development in schistosomiasis.8,29 Moreover, a definition that includes both of these criteria can help exclude possible patients with PAH and concomitant Schistosoma infection, because Sch-PH without hepatosplenic disease is extremely rare.
The reported prevalence of PH in chronic hepatosplenic schistosomiasis is not well defined because of variance in diagnostic methodology and the definition of PH, and it has been estimated to be as low as 7.7% and as high as 25%.8,21–24 Specifically, when echocardiography was used to screen PH (defined as PA systolic pressure higher than 40 mmHg) in chronic hepatosplenic schistosomiasis, the prevalence was calculated at 18.5%. 22 This decreased to 7.7% when right heart catheterization was used to confirm the presence of PH. 22 Another echocardiographic study found a 10.7% prevalence of PH in patients with liver fibrosis and chronic hepatosplenic schistosomiasis. 24 A prior study that also used echocardiography for PH diagnosis (but with a criterion of calculated mPAP of at least 20 mmHg) found a prevalence of 25%. 21 In an autopsy study of 313 patients diagnosed with chronic schistosomiasis in a Brazilian area of endemic disease, the prevalence of hepatosplenic schistosomiasis with PH was reported at 7.7%. 8 In addition, when 34 patients with portal hypertension and esophageal varices due to hepatosplenic schistosomiasis were studied using right heart catheterization, ?12% were found to have mPAP higher than 25 mmHg. 23 Conversely, when a series of PH patients were evaluated for the etiology of their disease in an outpatient referral clinic in Bahia, Brazil, 25% were found to have schistosomiasis as the underlying pathology. 30
NATURAL HISTORY OF HEPATOSPLENIC SCHISTOSOMIASIS AND SCH-PH
As per the life cycle of the parasite, the produced eggs (Fig. 3) are primarily released into the environment through the gastrointestinal tract (for S. japonicum and S. mansoni) of the host. Nevertheless, there are a number of ova that are retained within the portal venous system. These eggs become targets for the host immune system and result in localized inflammatory lesions, which most often progress to microabscesses and subsequently to granulomas. Eventually, collagen deposition ensues, and granulomas are replaced by fibrotic lesions. Depending on the topography of these fibrotic lesions, periportal fibrosis occurs in 4%–8% of chronically infected patients 4 and leads to portal venous system destruction and portal hypertension. The resulting portosystemic shunts, in the setting of elevated portal pressures, are thought to be a key component of the less common but severe complications of chronic hepatosplenic schistosomiasis. More specifically, parasite eggs can migrate to the lungs28,31 and the central nervous system21,33 and result in grave illness. Although the migration of Schistosoma eggs to the lungs and resultant mechanical pulmonary arterial obstruction were believed to constitute a key component of eventual development of PH in patients with chronic schistosomiasis and portal hypertension,28,34 this may not be the only mechanism. There is evidence of Sch-PH and corresponding histopathologic changes of the lung without a significant parasite burden.27,35,36 Interestingly, there are rare reports of pulmonary schistosomiasis without the presence of portal or pulmonary hypertension. 37 Although it is speculated that there may be cases of Sch-PH without concomitant portal hypertension, 2 we could not find any published reports to that effect.
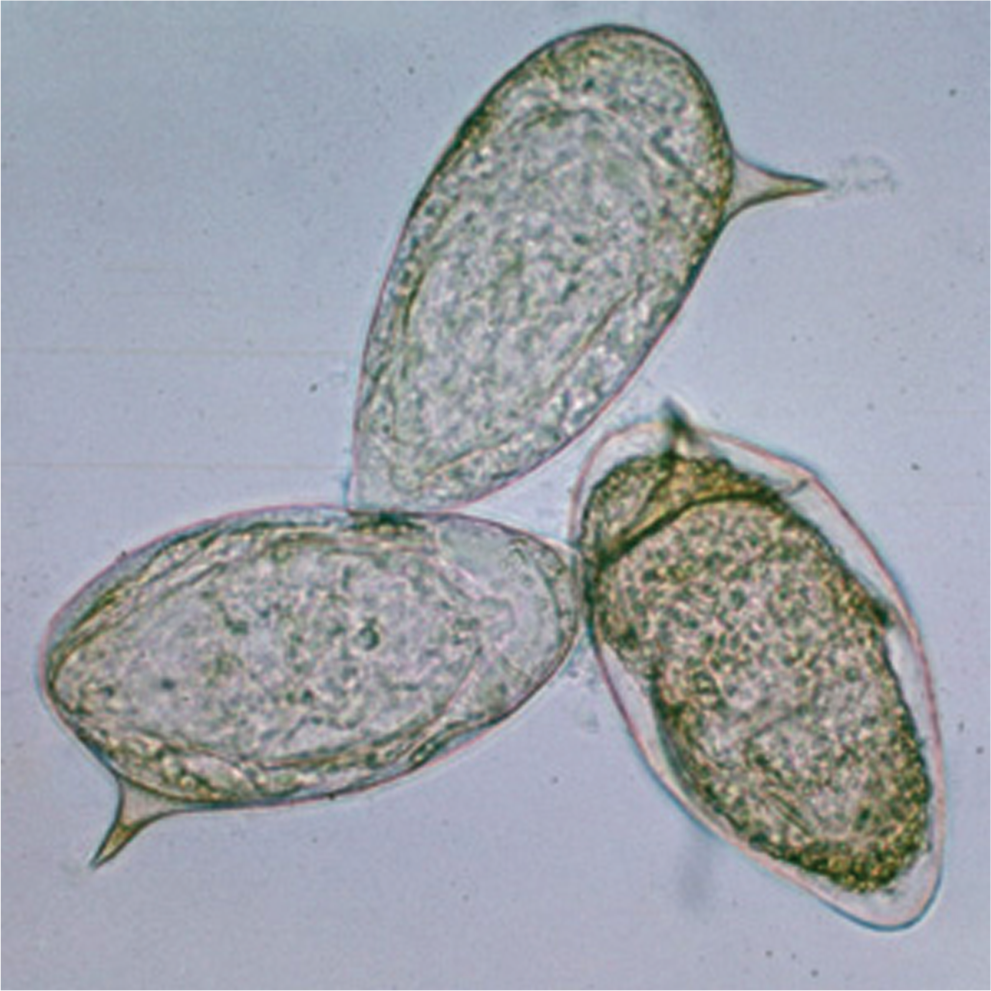
Schistosoma mansoni eggs (unstained wet mount). Reproduced with permission from Centers for Disease Control and Prevention (http://www.cdc.gov/dpdx/schistosomiasis/index.html).
The histopathology of pulmonary vascular complications related to chronic schistosomiasis was first described in the 1930s. 38 In 1958, 2 cases of pulmonary schistosomiasis were described, one of which was consistent with PH and produced right heart failure. 39 This was thought to be acute heart failure due to pulmonary hypertension from pulmonary arterial egg embolization, although it can be argued this was most likely a case of chronic PH and eventual end-stage RV failure. The pulmonary histopathology of chronic schistosomiasis was revisited in the 1970s, 28 at which point large and acute egg burdens with related granulomas and evidence of obliterative arteritis were seen.
Three distinct pathophysiologic mechanisms that may be causing PH in the setting of chronic hepatosplenic schistosomiasis have been described. Moreover, the actual pathogenesis likely includes a combination of these 3 mechanisms. The first, earliest described, and most commonly cited mechanism is based on parasite egg embolization of the pulmonary arteries. This occurs in the setting of portal hypertension, mainly because of periportal fibrosis from parasite-induced granulomas, usually without cirrhosis. The resulting portosystemic shunts lead to parasite egg migration to the lungs, which can cause pulmonary arterial egg embolization, leading in turn to pulmonary arterial obstruction, destruction, and plexiform lesion formation.28,31 The latter seems to be related to the formation of periovular granulomas, which affect both the lung parenchyma and the pulmonary vasculature and can have a course similar to that of the granulomas of the liver, resulting in some element of fibrosis and tissue destruction.28,34 Most likely this can also cause a certain extent of distal pulmonary vascular bed loss28,31,34 and elevation in pulmonary vascular resistance (PVR).
Although this mechanism was initially considered the main explanation for pulmonary hypertension, it is now thought to be compounded by a PA vasculopathy or pulmonary arteriopathy, most likely associated with an inflammatory response.23,24,28 More specifically, it has been suggested that the mechanical obstruction of the pulmonary arteries by the eggs may not significantly increase PVR and that Sch-PH is due to the aforementioned vasculopathy. 40
This second theorized mechanism of Sch-PH is a diffuse and heterogeneous vasculopathy, often described as an obliterative arteritis,24,28,34 that may28,41 or may not35,36 have a direct relation to parasite egg burden. It may be associated with an immune-mediated inflammatory cascade, 41 and it results in significant pulmonary vascular changes, including inflammation, medial thickening, and intimal remodeling.28,42 The most interesting aspect of the resulting lesions in Sch-PH is the striking similarity to idiopathic PAH (iPAH),41,43 which may have implications for treatment of the disease.
One recent study 36 indicated that S. mansoni eggs, as well as soluble egg antigen, were not found in postautopsy lungs of patients who died from PAH believed to be related to chronic schistosomiasis infection. This could indicate that once the immunologic cascade from eggs and egg antigens has begun, it may eventually result in pulmonary vasculopathy, leading to PH, regardless of whether the parasite has been cleared from the lungs. 35 This implies that even if the parasite is treated and the parasite eggs and related antigens are eradicated, pulmonary vasculopathy can persist. The latter can continue to progress, leading to worsening PH. Of note, this theory is also concordant with the “2-hit hypothesis” that has been proposed as a possible basis of PAH development. 44 More specifically, chronic schistosomiasis infection would be a second hit, or environmental factor, precipitating the pulmonary vasculopathy in a select number of patients with the appropriate and as-yet-unspecified predisposition.
To complicate matters, another study examining a mouse model of chronic Schistosoma infection and PH noted a correlation between lung parasite egg burden and both RV index (RV mass/(left ventricular + septal mass)) and pulmonary vascular remodeling, with plexiform lesions also being present. 41 Furthermore, in another chronic schistosomiasis mouse model, pulmonary vascular remodeling was noted only in close proximity to granulomas. 45 Taken together, these studies indicate that although a higher parasite egg burden may correlate with worse disease (also noted in prior studies), 28 its presence is not necessary for severe PH to be present.35,36
The third hypothesized mechanism is conceptually different from the ones described above. It is based on the premise that Sch-PH may be a variant of portopulmonary hypertension with similar pathophysiology, 46 that is, related to high-flow states of the pulmonary arteries. This process would include the development of hepatosplenic disease due to Schistosoma, followed by portal hypertension and eventually pulmonary hypertension, providing an explanation for PH in chronic hepatosplenic schistosomiasis without any evidence of pulmonary vascular granulomas or parasite egg embolization, as previously described in autopsy case reports. 36 This raises the question whether PH associated with Schistosoma infections is really due to parasite egg–induced pulmonary vascular granulomas, antigen-associated vasculopathy and ensuing cascade, the underlying liver disease and ensuing portal hypertension emulating portopulmonary hypertension pathophysiology, or a combination of these mechanisms.
The recent data 36 indicating few or no eggs or egg antigens in autopsied lungs of patients with Sch-PH suggest that this may actually be either a late-stage vasculopathy (possibly induced by early granulomas that subsequently resolved or an inflammatory trigger from the Schistosoma antigens) or a variant of portopulmonary hypertension. 47 The latter view is also supported by the low prevalence of vasoreactivity noted in Sch-PH, similar to portopulmonary hypertension. 27 Of note, the same autopsy findings did not confirm the presence of old granulomas, which again argues against the initial theory of parasite egg embolization and granuloma formation as critical. On the other hand, it has been suggested that initial granulomas within the pulmonary vasculature could lead to arteriolar fibrin deposition, endothelial hyperplasia, congestion, revascularization, and eventually plexiform lesions,34,48 with the granulomas likely losing their characteristic appearance with time and becoming unrecognizable pathologically.
The differing pathophysiologic mechanisms that are likely contributing to the development of Sch-PH may also provide an explanation for its bimodal age distribution. 49 Specifically, the younger age peak of PH in schistosomiasis patients may coincide with a large burden of egg embolization in the lung, resulting in granulomas and vascular-bed destruction. The older age peak (after the third decade of life) may coincide with a chronic vasculopathy associated with an immune-mediated inflammatory response and chronic, subacute infection, or pathophysiology similar to that of portopulmonary hypertension in the setting of long-standing liver disease. 49
Apart from Sch-PH, there also have been reports of hepatopulmonary syndrome (HPS) associated with schistosomiasis infection. The pathogenesis, although unknown, is suspected to be similar to that of HPS without the presence of Schistosoma infection, 50 that is, the formation of intrapulmonary vascular dilatations resulting in intrapulmonary shunting and pronounced hypoxia without evidence of PH.51–53 The prevalence of HPS is estimated to be approximately 6% in patients with schistosomiasis-associated liver fibrosis or as high as 11% in all patients with hepatosplenic schistosomiasis, versus 5.3%–16% in cirrhosis of various etiologies in patients awaiting liver transplantation in Brazil. 54
One of the interesting questions posed with regard to Sch-PH is which patient will develop it and why. Although this is clearly a very difficult question and probably has a multifactorial explanation based on both genetic predisposition and environmental factors, an interesting theory published in the 1970s is worth mentioning. Based on the concept that host immunosuppression is integral to parasite survival and to ensure “successful parasitism,” Dr. Warren 55 hypothesized that the person-to-person variability of PH development in patients with chronic schistosomiasis is due to an immune-modulatory defect or variance present in these individuals. This results in an unwelcome response for both the parasite and the host, in this case pulmonary arterial vasculopathy and related PH, eventually resulting in host disease and death.
IMMUNOLOGY OF SCH-PH
The pathologic changes of the pulmonary vasculature in Sch-PH are similar to those found in iPAH.41,43 It is therefore hypothesized that similar cytokine cascades and pathobiological pathways may be implicated in pulmonary vascular histopathology of both these types of PH. 2 The prevailing hypothesis is that regardless of the actual Sch-PH pathophysiology, whether pulmonary vascular granulomas, immune-mediated vasculopathy, or poorly described humoral-associated portosystemic shunting, the end result is cytokine release and the ensuing cascade, similar to the one found in iPAH, ultimately leading to pulmonary vascular remodeling and plexiform-lesion formation.
There has been extensive study of the immunologic cascade characterizing the acute as well as the chronic reaction to schistosomes,56–59 in part due to interest in the immunomodulatory effect of chronic infection. 60 As noted above, the initial immune host reaction is driven by antigens released by schistosomules during the acute infection. This is T helper 1 cell mediated with the release of multiple cytokines, including tumor necrosis factor α, interleukin (IL)-1, IL-6, IL-12, interferon γ, and transforming growth factor (TGF-)β.42,56,57,61–64 It is a strongly proinflammatory phase that slowly evolves into a secondary immune response. The latter, which is due to the parasite eggs and their antigens, is integral to the granuloma formation and is mediated by T helper 2 cells65,66 while mostly modulated by a separate group of cytokines, including IL-4, IL-5, IL-10, and IL-13.42,63,67–69 The importance of the above series of responses is based on the long-term presence of the infectious stimulus, necessitating the formation of granulomas in an attempt to isolate the pathogen and avoid further tissue damage. 42 Unfortunately, these granulomas, although initially protective, can lead to local fibrosis, which can become excessive in the presence of predisposing factors, such as variations of the connective tissue growth factor gene, as previously described by Dessein and colleagues. 70 Such excessive and pathologic fibrosis can lead to subsequent organ damage and eventual host health decline. Furthermore, in addition to genetic predispositions that are not well defined in Sch-PH, the inflammatory component itself may be important in PH development, as there is some evidence of inflammation playing a role in iPAH pathophysiology, in addition to S. mansoni infection. 71
To this extent, various inflammatory cytokines known to be present in Schistosoma infection have also been associated with PAH. Kolosionek et al., 42 in a recent review, reported on cytokines known to be associated with Schistosoma infection (such as IL-1, IL-4, IL-5, IL-6, IL-10, IL-13, TGF-β, and IFN-γ) and their possible role in iPAH pathophysiology. For example IL-1, a well-known proinflammatory cytokine that plays a role in schistosomiasis,57,72 has also been linked to pulmonary vasoconstriction, 73 and its antagonism may lower PA pressures. 74 IL-4 has clearly been shown to have an association with Schistosoma infection and granuloma formation 68 and is now also being linked to Sch-PH 64 and pulmonary vascular remodeling in general, 75 a hallmark of iPAH. Another example is IL-10, which has anti-inflammatory and antifibrotic effects, limiting immune response, and has been shown to increase with ongoing Schistosoma infection.41,60 Blockade of the IL-10 receptor (IL-10R) may worsen Schistosoma disease progression and increase the likelihood of lung pathology development. 76 Moreover, IL-10 has been shown to have antiproliferative effects in relation to the pulmonary vasculature and to lower PA pressures in a monocrotaline-induced PAH murine model. 77
IL-13 is another cytokine that seems to play an integral role in Schistosoma infection and has been studied extensively in this capacity. 78 Multiple investigators have demonstrated an association between disease progression, fibrosis, and IL-13 function.61,79–81 Other publications have shown that the presence of IL-13Ra2, an IL-13 receptor, is integral to down-modulation of granuloma formation and attenuation of inflammatory disease activity.82,83 This is most likely due to the nonsignaling nature of IL-13Ra2, which acts as a buffer to IL-13 signaling. Its blockade (or absence in deficient mice) results in functional IL-13 signaling upregulation, mainly through increased binding of IL-13 to IL-13Ra1, resulting in worse disease in an Sch-PH experimental model. 84 Although there is a clear association between IL-13 pulmonary vascular remodeling and PAH,75,85 the actual effect of IL-13 is a little less straightforward. In a mouse model of vascular remodeling induced by inhaled allergens, IL-13 inhibition resulted in significantly less pulmonary arterial remodeling than in untreated controls. 75 Conversely, there are data indicating that IL-13 inhibits PA smooth muscle cell proliferation as well as downregulates endothelin-1 gene expression, both of which would inhibit PAH development or progression. 85 The same study noted that IL-13Ra2 overexpression blunts STAT (signal transducer and activator of transcription)-mediated IL-13 signaling, pointing toward a “decoy molecule” IL-13Ra2 function, and also found that iPAH patients had higher IL-13Ra2 messenger RNA (mRNA) levels, likely resulting in IL-13 signaling attenuation. In animal models of iPAH, there was also an absolute (in mice with hypoxia-induced PH) or relative (in mice with monocrotaline-induced PH) IL-13Ra2 decrease, which can result in greater IL-13 activity and could explain the development of PH. 85 Moreover, there are data indicating that IL-13 production may be correlated with parasite egg burden (lung and liver) as well as pulmonary vascular remodeling and increased migration of PA smooth muscle cells. 41 Finally, a recent study suggests that TGF-β is a key upstream signal to the IL-13 (and IL-4) cytokine pathways in relation to Sch-PH. 64
Of note, there is some evidence for Smad2 expression in pulmonary periovular granulomas in mice with chronic schistosomiasis. 84 This likely has an effect similar to that of depression of bone morphogenic protein receptor 2 (BMPR2), an upstream suppressor of Smad2, which occurs with loss-of-function mutations described in cases of PAH in humans.84,86 In the same mouse model, there were elevated levels of resistin-like molecule (RELM-)α (a hypoxia-induced mitogenic factor) in schistosomiasis-induced pulmonary granulomas, 84 consistent with prior reports underscoring the importance of RELM-α in chronic hypoxia–induced PH in various mouse models.87,88 Finally, some studies are starting to focus on hypoxia-inducible factor1-α as another connecting link between PAH and chronic schistosomiasis-associated lung disease. 89
TREATMENT OF SCH-PH
Schistosomiasis has been historically treated with praziquantel. An enterally well-absorbed pyrazinoisoquinoline derivative, it is metabolized in the liver, and its inactive metabolites are excreted primarily in the urine. It causes detachment of adult worms from the host vascular wall and ultimately their death, resulting in a cure rate of up to 90%. If the patient is not cured, the parasite and egg burden is significantly reduced with the first treatment, and a second treatment course usually is successful in eradicating the parasite. Moreover, there are data showing that praziquantel will suppress schistosomal egg granulomas within the lungs, reducing inflammatory cell and fibroblast numbers. 90 Of note, praziquantel is not active against schistosomulae and also has a short half-life, rendering it a poor choice for chemoprophylaxis (artemether is a better choice for the latter). 4
Initial attempts to assess whether antischistosomiasis medical therapy was curative for pulmonary forms of the disease were not very promising. A murine model with portal vein ligation–induced portal hypertension infected with S. mansoni and with confirmed parasites and related changes within the lungs, including granulomas and vasculopathy (noted on necropsy), was compared to a group of uninfected control mice. 91 Both chemotherapy-treated (eradicated parasite eggs in stool) and untreated infected mice had persisting pulmonary vascular changes not seen in the uninfected group. More specifically, although the lung parenchyma appeared to heal with resolution of granulomas in the treated groups, the pulmonary vessels underwent defective repair, resulting in abnormal anastomosing vessel aggregates. 91 A similar, angiomatoid pattern of repair was noted in the hepatic vasculature of a murine model of S. mansoni–induced hepatic fibrosis. 92
Conversely, a case report from 1990 noted an S. haematobium–associated PH patient being cured by the use of praziquantel. 93 A more recent study, looking specifically at RV systolic pressure (RVSP) and RV hypertrophy (RVH), had quite promising results. Using a murine model infected by S. mansoni, the investigators treated mice with either praziquantel or control at 17 weeks after infection and then euthanized them at 25 weeks. Untreated mice had significantly more elevated RVSP and RVH, compared to praziquantel-treated mice. Moreover, the treated mice also manifested reversal of pulmonary vascular remodeling. A positive association between the presence of parasite eggs in the lungs and changes in RVSP, RVH, and pulmonary vascular remodeling was also established, indicating that early egg clearance from the lungs either prevented or reversed the above changes. 94 In the same study, although serum levels of inflammatory cytokines were not affected by treatment, lung mRNA of IL-13 was lower in the praziquantel-treated mice. 94 Despite these promising results, whether these treatment effects would be manifested if treatment were initiated at a later stage of disease progression remains unanswered. It has been theorized that there may be a “point of no return” with regard to pulmonary vascular remodeling, after which treatment of the infection will not result in reversal of PAH, or will be associated with impaired healing, as described above.2,91,92,95
It is unclear whether Sch-PH should be treated solely with S. mansoni chemotherapy, eradicating the parasite and therefore eliminating the etiologic factor, or with additional treatments geared toward PAH (e.g., prostanoids, endothelin receptor antagonists, or phosphodiesterase-5 [PDE-5] inhibitors; Table 2). 96 There are very few data regarding the use of these therapies in the setting of Sch-PH. Nevertheless, because of the similarities between iPAH and Sch-PH, the latter has been included in group 1 of the WHO classification for PH, allowing for the use of PAH-targeted treatments based on extrapolation from PAH clinical trials unassociated with schistosomiasis. A more recent case report noted significant improvement of functional status and 6-minute walk test (6MWT) distance (from 154 to 484 m, 6 months after treatment) in a patient with hepatosplenic schistosomiasis and PH treated with a PDE-5 inhibitor. 97 Abstracts published in 2004 hinted toward similar results. A case series of 7 patients on oral PDE-5 inhibitors for 3 months, who underwent pre- and posttherapy magnetic resonance cardiac imaging, showed significant RVH reversal, cardiac index improvement, and clinical improvement (functional class and 6MWT distance) after treatment. 98 A larger series of 13 patients with Sch-PH studied by the same group of investigators showed significantly improved functional class, 6MWT distance (394 ± 115 vs. 122 ± 68 m), and mPAP (80 ± 9 vs. 97 ± 14 mmHg) after 6 months of treatment with a PDE-5 inhibitor. 99 In a retrospective review performed on Sch-PH patients treated with PAH-specific therapies between 2003 and 2010 in a single center in Sao Paulo, Brazil, functional and hemodynamic data were compared at baseline and after treatment. 100 The authors showed that in this series of 12 patients treated mostly with either PDE-5 inhibitor or endothelin receptor antagonist monotherapy (only 1 patient received combination therapy), functional class, 6MWT distance, and cardiac index improved after treatment (mean time between evaluations was 34.9 ± 15.5 months).
Updated World Health Organization pulmonary hypertension classification 96

Note: ALK1: activin-like receptor kinase-1; BMPR2: bone morphogenic protein receptor type 2; CAV1: caveolin-1; ENG: endoglin; HIV: human immunodeficiency virus; KCNK3: potassium channel superfamily K member 3.
It is apparent that there is a paucity of evidence regarding PAH-specific treatment use in Sch-PH (Table 3). The similarity in pathology and immunohistochemistry between these two entities supports the hypothesis that PAH medications may be effective in Sch-PH, with some small case series hinting toward this as well.99,100 There is a clear need for high-quality clinical trials to better assess this issue, but the financial barriers related to the geographic distribution of the disease present a major concern, making drug availability very sparse. Based on the evidence to date, it is our opinion that Sch-PH should always be treated, initially with eradication of the underlying parasitic infection with at least 1 course of praziquantel (or 2, if stool studies indicate the need). Once it has been confirmed that the patient is parasite free, Sch-PH should be treated with PAH-oriented advanced medical regimens, according to current PAH treatment guidelines. 101 Finally, if there is clear evidence of portal hypertension, with or without evidence of advanced liver disease, although an approach similar to treatment of Sch-PH should be used, additional consideration should be given, since portopulmonary hypertension could be the underlying pathophysiology. This would include closer monitoring of liver function and caution with endothelin receptor antagonist therapy, as well as consideration of liver transplantation based on underlying liver disease and cardiovascular hemodynamics. Of note, there is some evidence, albeit not very robust, on the use of bosentan,102,103 ambrisentan,104,105 iloprost,103,106 epoprostenol,107,108 treprostinil,109,110 and sildenafil110,111 in portopulmonary hypertension, and one could extrapolate their use to Sch-PH with portal hypertension.
Data on PAH-targeted therapies and antiparasitic agents in Sch-PH

Note: CI: cardiac index; ERA: endothelin receptor antagonist; FC: functional class; PAH: pulmonary arterial hypertension; PDE-5i: phosphodiesterase-5 inhibitor; PVR: pulmonary vascular resistance; RVH: right ventricular hypertrophy; RVSP: right ventricular systolic pressure; Sch-PH: schistosomiasis-associated pulmonary hypertension; 6MWD: 6-minute walk distance.
In addition to the above, there are other treatment considerations that further complicate the management of Sch-PH. Although there is some observational evidence supporting the use of anticoagulation in patients with PAH,112,113 many patients with chronic hepatosplenic schistosomiasis will have portal hypertension and will be at risk for variceal esophageal hemorrhage, making anticoagulation quite risky. For the esophageal varices, some patients with portal hypertension may be candidates for a TIPS (transjugular intrahepatic portosystemic shunt) procedure, but in the setting of PAH this can significantly increase blood flow to the right ventricle, with risk of acute RV failure. 114 Moreover, the formation of a large, artificial portosystemic shunt also provides an added conduit for further parasite migration to the lungs, thus increasing lung parasite burden (which correlates with worse disease, as noted above). 94 Another concern is the possibility of pulmonary emboli composed of dead and dying schistosomes after treatment; in the setting of PH, this can cause significant increase in PVR and acute-on-chronic RV failure. 15 Finally, there is evidence that patients with Sch-PH are even less likely to be vasoreactive to inhaled NO challenge (0%–3.5%)27,115 than patients with iPAH (10%–15%).116,117 Moreover, the data indicate that mPAP and mixed venous oxygen saturation correlate with Sch-PH severity and function. 115 The latter is an evenmore intriguing finding when coupled with the evidence that Sch-PH appears to have a more benign presentation from a hemodynamic perspective (lower mPAP, higher cardiac output, and lower PVR) as well as a trend toward a more favorable clinical course, compared to iPAH. 27
From the above, it is clear that more data are needed in order to better ascertain which therapies are truly effective in Sch-PH. Future trials should be focused on the use of specific PAH therapies in Sch-PH patients and related clinical outcomes.
SUMMARY AND CONCLUSIONS
In this review, we summarized the current knowledge regarding chronic Sch-PH. More specifically, we reviewed the current literature on Sch-PH and readdressed the prevailing hypotheses surrounding its pathophysiology and natural history. Finally, we also reported the current evidence on therapeutics, in the context of its immunopathologic links to iPAH.
Schistosomiasis and its associated chronic hepatosplenic form remain a very common and potentially lifethreatening disease in regions of endemic disease, and the sheer number of infected individuals makes this one of the most common etiologies of PH worldwide.2,118 Despite evidence linking Sch-PH to iPAH, our understanding of the pathogenesis of the former is still poor, with 3 different likely mechanisms at its core. Moreover, it is quite possible that Sch-PH has various phenotypes, with similar pathogenetic mechanisms but variant clinical courses, prognoses, and response to therapy. This is once again reminiscent of iPAH, with the classic example of the vasoreactive subgroup of patients.
Although there is a consensus on treating schistosomiasis itself, there is very little evidence that this can be effective for the underlying PH, with the more likely case being that the underlying pathologic changes are either permanent or defectively repaired, resulting in vascular remodeling similar to that noted in iPAH. Finally, although this is a clear indicator toward attempting to treat this type of PH with medical regimens widely used for PAH, the lack of clinical trials and the limited resources in areas of endemic disease for procurement and distribution of these very expensive medications do not allow for their widespread use.
