Abstract
Several animal studies have shown that regional lung perfusion could be effectively estimated by the hypertonic saline contrast electrical impedance tomography method. Here, we reported an application of this method to dynamically assess regional pulmonary perfusion defect in a patient with acute massive pulmonary embolism. A 68-year-old man experienced sudden dyspnea and cardiac arrest during out-of-bed physical activity on the first day after partial mediastinal tumor resection. Acute pulmonary embolism was suspected due to acute enlargement of right heart and fixed inferior venous cava measured with bedside ultrasound. The computed tomography pulmonary angiography further confirmed large embolism in both left and right main pulmonary arteries and branches. The regional time impedance curves, which were obtained by a bolus of 10 ml 10% NaCl through the central venous catheter, were then analyzed to quantitatively assess regional perfusion. Normal ventilation distribution with massive defects in regional perfusion in both lungs was observed, leading to a ventilation–perfusion mismatch and low oxygenation index (PaO2/FiO2 = 86 mmHg) at the first day of pulmonary embolism. The anticoagulation was performed with heparin, and the patient’s condition (such as shock, dyspnea, hypoxemia, etc.), regional lung perfusion defect, and ventilation–perfusion mismatch continuously improved in the following days. In conclusion, this case implies that electrical impedance tomography might have the potential to assess and monitor regional perfusion for rapid diagnosis of fatal pulmonary embolism in clinical practice.
Case report
A 68-year-old man experienced sudden dyspnea and cardiac arrest during out-of-bed physical activity on the first day after partial mediastinal tumor resection. Patient regained consciousness after 3 min of cardiopulmonary resuscitation. Norepinephrine was infused to maintain blood pressure, and oxygen was provided through a non-rebreather oxygen face mask (10 L/min). Acute pulmonary embolism (PE) was suspected due to acute enlargement of right heart and fixed inferior venous cava measured with bedside ultrasound. The computed tomography pulmonary angiography (CTPA) confirmed large embolism in both left and right main pulmonary arteries and branches (Fig. 1a and 1b). The electrical impedance tomography (EIT) was used to examine regional perfusion at the bedside as soon as the patient was admitted to the ICU. During the protocol of EIT measurements, a silicone EIT belt with 16 surface electrodes was placed around the patient’s thorax in one transverse plane corresponding to the fourth intercostal parasternal space and then connected to the EIT monitor (Dräger Medical, Lübeck, Germany). A bolus of 10 ml 10% NaCl was injected through the central venous catheter during a respiratory pause. The resulting regional time impedance curves were then analyzed to quantitatively assess regional perfusion.1–4 No adverse effect was found during the hypertonic saline bolus examination. Normal ventilation distribution (Fig. 1c) with massive defects in regional perfusion in both lungs (Fig. 1d) was observed, leading to a ventilation–perfusion mismatch and low oxygenation index (arterial oxygen partial pressure to fractional inspired oxygen ratio, PaO2/FiO2 = 86 mmHg). Due to a bleeding complication, only anticoagulation with heparin but no thrombolytic therapy was performed. Regional lung perfusion continuously improved in the following days as assessed with EIT and saline injection (Fig. 1f and h). The patient was transferred to regular ward on day 7 of his ICU stay. A follow-up CTPA re-examination demonstrated the significantly reduced emboli in both main pulmonary arteries (Fig. 1i and j). Clinical data during the treatment of PE are summarized in Table 1. EIT-based regional ventilation/perfusion ratio was calculated, which became more homogeneous during the treatment, as assessed by coefficient of variation (4.0 on day 0 → 0.5 on day 3). The patient was discharged from hospital after recovery at day 25. Data of this patient were partially presented in a recent publication with different focuses. 5
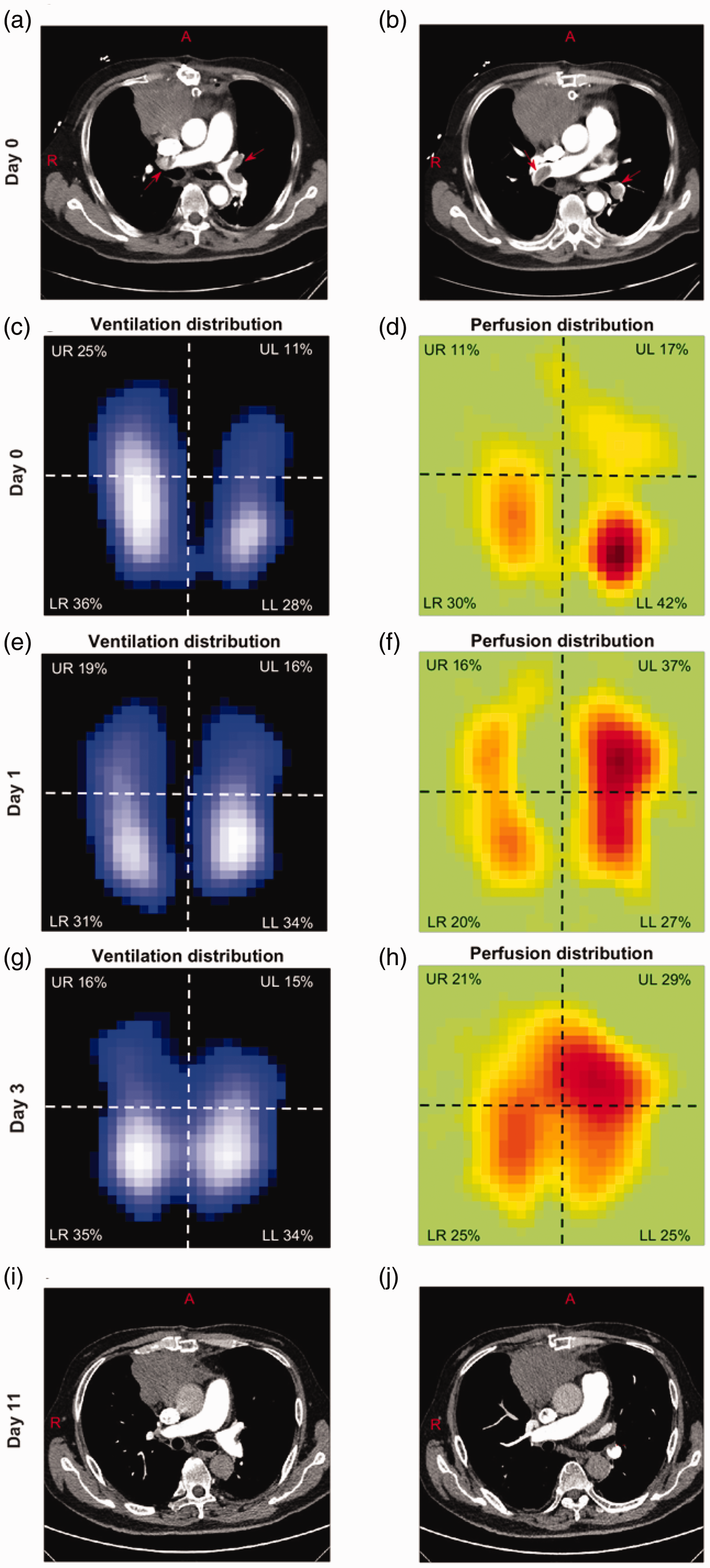
a and b: Computed tomography pulmonary angiography demonstrated large embolism in both left and right main pulmonary arteries and branches (day 0). Red arrows highlight the emboli in the pulmonary arteries. c, e and g: Functional EIT images of ventilation distribution on days 0, 1, and 3, respectively. Low ventilated regions are marked in dark blue and high ventilated regions in white. d, f, and h: Functional EIT images of perfusion distribution measured with saline bolus injection during respiratory hold on days 0, 1, and 3, respectively. Regions with high perfusion are marked in red. i and j: Computed tomography pulmonary angiography demonstrated the improvement after treatment (day 11).
Summary of clinical data during the treatment in the PE patient.
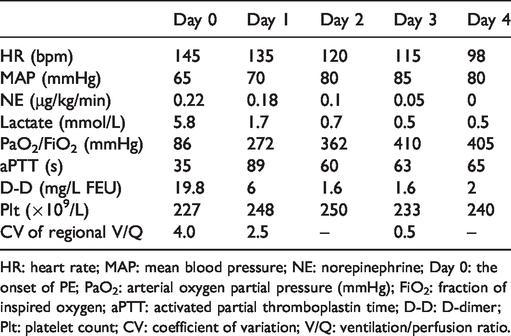
HR: heart rate; MAP: mean blood pressure; NE: norepinephrine; Day 0: the onset of PE; PaO2: arterial oxygen partial pressure (mmHg); FiO2: fraction of inspired oxygen; aPTT: activated partial thromboplastin time; D-D: D-dimer; Plt: platelet count; CV: coefficient of variation; V/Q: ventilation/perfusion ratio.
Discussion
To date, bedside ultrasound has been commonly used as a screening tool for the massive PE in the suspected PE patients in the emergency condition. On one hand, direct and visual assessment of regional lung perfusion could not be obtained by ultrasound. On the other hand, the ultrasound examination was operator-dependent. Diagnosis of PE still relies mainly on CTPA examination, even moving the patients to the CT room was a high risk and a difficult procedure. Hence, CTPA might be improper for the unstable critically ill patients. Rapid bedside diagnostic tool for acute PE would be of great interest in clinical practice. This case provides a unique proof of concept to use EIT as an additional bedside tool for rapid massive PE diagnosis.
Compared to previously published EIT-based methods using pulsatility signal to estimate perfusion, the presented method is clinically feasible and more accurate. 6 It was performed with a bolus of indicator (10 ml 10% NaCl) during a respiratory hold, thereby producing a dilution curve that follows typical first-pass kinetics. The potential advantages of saline contrast method should be emphasized in clinical practice: (1) Bedside evaluation without patient transportation to the radiology department and interruption of the full intensive care therapy. In the present case, the medical staffs spent about 50 min to transfer the patient from regular ward to CT room and finished the examination at a high-risk condition. As comparison, a total of 30 min was taken to identify the defect of regional lung perfusion by EIT and saline injection at the bedside. (2) As a bedside tool, EIT could be easily repeated and provide dynamic data to guide therapy. This case revealed that the perfusion findings determined by EIT were consistent with the CTPA scans acquired before and after the ICU stay. Further study is required to validate the relevance of EIT to diagnosis and guide clinical decisions for fatal PE at the bedside.
Despite the potential benefits of low-molecular-weight heparin (LMWHs) over unfractionated heparin, unfractionated heparin is still used in some cases of PE. The first two indications for iv unfractionated heparin treatment are a creatinine clearance of ≤30 ml/min, 6 and severe obesity (BMI > 40 kg/m2). 7 This patient’s renal function was normal, but he had a relative obesity and a high risk of bleed for the anticoagulation. Importantly, the unfractionated heparin (UFH) could be easily antagonized by the protamine in the condition of bleeding after anticoagulation. However, it might be difficult to correct the coagulation dysfunction caused by LMWHs. Hence, we chose unfractionated heparin in the patient.
Conclusions
EIT might have the potential to assess and monitor regional perfusion for rapid diagnosis of fatal PE and therapeutic effect in clinical practice.
Footnotes
Authors’ contributions
H.H. and Z.Z. prepared the final copy of the manuscript and EIT images. H.H. and S.Y. performed the EIT examination and obtained patient’s consent. H.H., S.Y., Y.L., I.F., and Z.Z. drafted the manuscript for important intellectual content and interpreted the data.
Consent for publication
All authors read and approved the final manuscript.
Ethical approval and consent to participate
The Institutional Research and Ethics Committee of the Peking Union Medical College Hospital approved this study on human subjects. Written informed consent was obtained from the patients’ next of kin.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Zhanqi Zhao received consultant fee from Draeger Medical. Inéz Frerichs reports funding from the European Union’s Framework Programme for Research and Innovation Horizon2020 (CRADL, Grant No. 668259; WELMO, Grant No. 825572) and reimbursement of speaking fees, congress, and travel costs by Dräger Medical. Other authors declare no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CAMS Innovation Fund for Medical Sciences (No. 2020-I2M-C&T-B-042). Capital’s Funds for Health Improvement and Research (NO. 2020-2-40111) and Peking Union Medical College Hospital of Medical Novel Medical Technology Project (No. XJS20190210).
