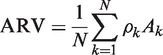Abstract
Objective
To investigate the possible use of electrical impedance tomography (EIT) in monitoring focal cerebral infarction in a rabbit model.
Methods
A model of focal cerebral infarction was established in eight New Zealand rabbits using a photochemical method without craniectomy. Focal cerebral infarction was confirmed by histopathological examination. Intracranial impedance variation was measured using 16 electrodes placed in a circle on the scalp. EIT images were obtained using a damped least-squares reconstruction algorithm. The average resistivity value (ARV) of the infarct region on EIT images was calculated to quantify relative resistivity changes. A symmetry index was calculated to evaluate the relative difference in resistivity between the two sides of the cerebrum.
Results
EIT images and ARV curves showed that impedance changes caused by cerebral infarction increased linearly with irradiation time. A difference in ARV was found between measurements taken before and after infarct induction.
Conclusions
Focal cerebral infarction can be monitored by EIT in the proposed animal model. The results are sufficiently encouraging that the authors plan to extend this study to humans, after further technical improvements.
Introduction
A cerebral infarction is an ischaemic stroke resulting from a disturbance in the vessels supplying blood to the brain, and may be caused by atherothrombosis or embolism. 1 Approximately 1.5 million people die of stroke each year in China; 62.4% of these deaths are attributed to cerebral infarction. 2 Current medical imaging modalities, such as magnetic resonance imaging (MRI) and computed tomography (CT) scanning of the brain, have high sensitivity and specificity for detecting cerebral infarction. 3 However, MRI and CT cannot be used to monitor continuously the exacerbation of infarct volume or repetition of infarction in patients after initial stroke onset. Consequently, such conditions cannot be detected until they become severe and result in overt symptoms that are dangerous to patients. Thus, there is a need for a noninvasive and radiation-free means of continuously monitoring patients who are at risk of further deterioration, following cerebral infarction.
As a functional imaging technique, electrical impedance tomography (EIT) can generate cross-sectional images of electrical properties inside the body that are related to functional changes in tissues or organs. EIT has the potential to provide a uniquely useful new imaging method in clinical or experimental neuroscience, 4 enabling the imaging of epileptic seizures 5 and blood-flow changes during cortical evoked activity 6 in humans, and of cortical spreading depression7,8 and fast neuronal depolarization9,10 in anaesthetized animals.
Furthermore, its noninvasiveness and the fact that it does not involve ionizing radiation make EIT a potential method of continuous monitoring. In previous studies, EIT has been proposed as a research technique for monitoring intracranial bleeding. Detection of intracerebral haemorrhage 11 and subarachnoid haemorrhage 12 by EIT has been demonstrated in anaesthetized neonatal piglets. A clinical study showed that the intracranial resistivity changes in humans (caused by the influx and efflux of irrigating fluid) could be detected and imaged repeatedly by EIT during the twist-drill drainage operation, in patients with subdural haematoma. 13
In rats with global cerebral ischaemia induced by diathermy of both vertebral arteries and reversible occlusion of the carotid arteries, impedance measurement and preliminary impedance imaging with scalp or cortical electrodes were performed, to detect the ischaemia.14,15 However, no studies on imaging impedance changes related to infarct by EIT have been conducted for cerebral infarction.
During cerebral ischaemia, brain impedance increases by tens or hundreds of per cent at frequencies <100 kHz, 16 because tissue impedance is largely determined by the extracellular space at such frequencies. This increase occurs as ions and water move from the extracellular to the intracellular compartment as a result of ischaemia. 17 Therefore, as in previous work,14,15 the change in intracranial impedance resulting from global cerebral ischaemia can be imaged by the EIT technique, which enables the internal distribution of the impedance of an object to be imaged by means of a ring of external electrodes. 18 However, the monitoring of impedance variation caused by focal ischaemia with infarction of brain tissue (such as embolic occlusion of distal vessels) remains a challenge for brain EIT, because the change in impedance due to focal ischaemia is smaller than that due to global cerebral ischaemia. Given the high resistance of the skull, the signal arising from changes in the brain can be substantially attenuated, and much of the applied current may pass directly between the electrodes in the scalp without entering the brain.
The present study aimed to establish a rabbit model of focal cerebral infarction induced by photochemical injury without craniectomy, and to investigate the possible use of EIT in monitoring the progression of cerebral infarction. The animal model chosen was similar to infarction in humans caused by thrombosis of the terminal artery.
Materials and methods
Ethics
The study was approved by the Ethics Committee for Animal Research of the Fourth Military Medical University and adhered to this institution’s guidelines on animal experimentation.
Animal model
Eight New Zealand rabbits weighing 2.4 ± 0.52 kg (mean ± SD) were anaesthetized by intravenous injection of 3 g/l pentobarbital (1 ml/kg) into the marginal vein of the ear. After 3–5 min, each animal’s head was immobilized, the hair was removed from an area of ∼14 cm2 and the skin was disinfected with surgical spirit. The skin and periosteum were exposed and separated, and a blind hole 5 mm in diameter was drilled 5 mm to the left of the sagittal suture and 5 mm anterior to the coronal suture. The hole penetrated both the outer plate and the diploe layer of the frontal bone, leaving the inner plate integrally intact. A gelatin sponge was used for haemostasis. Next, the photosensitive dye tetrachloro-tetraiodofluorescein disodium (35 g/l) (Rose Bengal, Sigma, St Louis, MO, USA) was slowly injected into the marginal vein of the ear at 1 ml/kg. Approximately 3 min later, an LG150B type cold light source (Photo-machine Technological Exploration Corp., Shaanxi, China) was used to direct light (wavelength 540 mm, intensity 600 mW/cm2) directly onto the blind hole for an uninterrupted period of 30 min. Subsequently, the incision was sutured and disinfected.
EIT procedure
An EIT system designed by our group was used to perform impedance measurements. 19 This system can operate within a frequency range of 1–190 kHz and the output of the current source is at ∼0–1.25 mAp-p, with better than 0.01% measured precision compared with the full-scale range of the input voltage. Data acquisition speed was up to 3 frames/s.
Before any surgical procedure was performed, 16 flake copper electrodes, each with two holes, were arranged on each rabbit’s head in a circle with equal spacing, in a clockwise direction (Figure 1). A self-drilling screw, 2 mm in diameter, was used to fix a copper electrode onto the rabbit’s skull through one of the two holes. The remaining hole was used to connect the sterile-hooked electrode to the EIT system. Good sensitivity and signal-to-noise ratio in the EIT system were obtained by the use of the opposite-drive adjacent-measurement protocol for data acquisition.
20
A sinusoid current of 1 mAp-p and 50 kHz was applied between pairs of opposite electrodes; the resulting potential differences were measured by the remaining adjacent electrode pairs at a rate of 1 frame/s.
Establishment of an animal model of focal cerebral infarction using a photochemical method without craniectomy. Location of the irradiated area and arrangement of electrodes on the rabbit’s head for electrical impedance tomography to monitor focal cerebral infarction.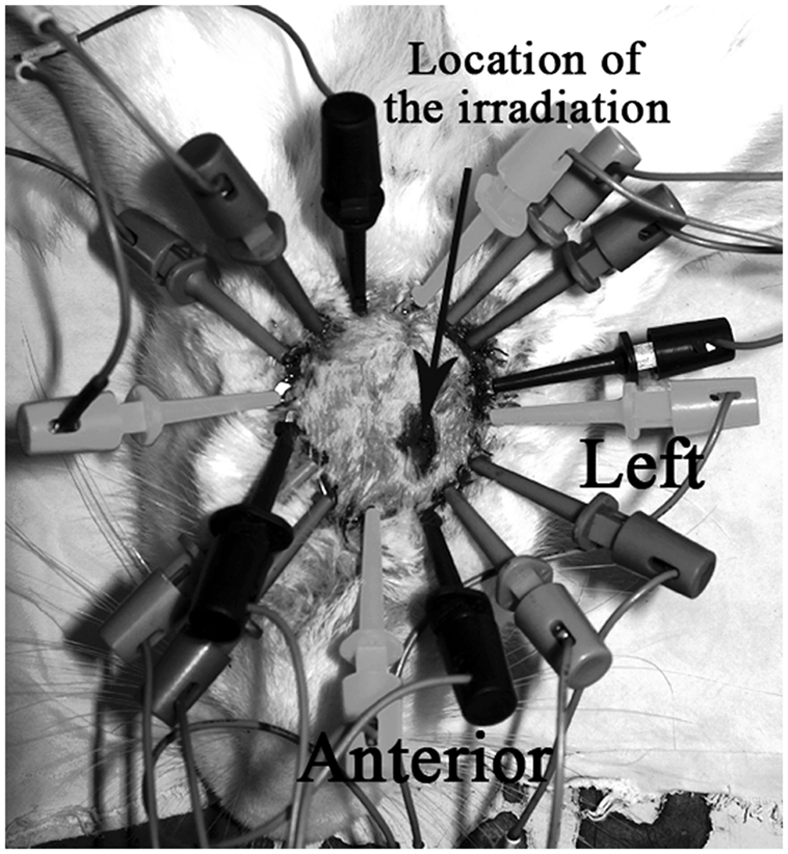
Before the irradiation stage, measurements were acquired for 10 min as reference data; thus, each animal served as its own control. Then, the irradiation and subsequent processes were conducted and monitored to obtain imaging data for 2 h.
The difference between the imaging and reference data was used for image reconstruction by means of a damped least-squares reconstruction algorithm, 21 based on a circular finite element model with homogeneous resistivity distribution. The reconstructed values were mapped into a cross-sectional EIT image to enable the relative impedance changes within a given period to be imaged dynamically. In EIT images, blue represents a regional increase in resistivity due to local ischaemia and green represents the baseline.
Data analyses
The total variation trend in the images was determined. The average resistivity values (ARV) in EIT images were used as an index to quantify the resistivity changes caused by the decrease in blood flow volume in the cerebrum, and to reduce the influence of artefacts on the other side of the cerebrum.
13
ARV was calculated as follows:
The relationship between ARV and cold light irradiation time was subjected to linear regression analysis. Variation in ARV was analysed by the two-tailed paired-samples t-test, using SPSS® 14.0 for Windows® (SPSS Inc., Chicago, IL, USA). P-values <0.01 were considered significant.
Validation of the animal model
Rabbits were killed via intravenous injection (75 g/l pentobarbital, 1 ml/kg) 48 h after model establishment and the brain was removed. An area 1.5 cm in diameter surrounding the infarct was removed and fixed in 0.1% formaldehyde for 24 h; the remainder of the brain was discarded. The length and width of each infarct were measured with a Vernier calliper. The infarcts were then cut into 3-mm thick slices for haematoxylin & eosin staining, and observation under an optical microscope.
Results
The EIT data on focal cerebral infarction were successfully collected from all eight rabbits. When each experiment ended, each rabbit remained motionless and alive.
Anatomical and pathological observations
As shown in Figure 2A and B, light-exposed tissue was slightly coloured and rounded. The infarction foci were mostly round with a clear boundary located at the cortex, and measured (mean ± SD) 3.52 ± 0.45 mm in diameter and 2.86 ± 0.12 mm in depth. Sites of oedema could be observed on the infarction surface and a minority were plane or irregular. The haematoxylin & eosin staining results in Figure 2C demonstrated morphological changes in nerve cells in the necrotic focus. Some cells displayed cytoplasmic tumefaction and vacuolization. Some cells were also shrunken and showed karyolysis. Fragmentation of cells and karyopyknosis were observed. All morphological changes observed were classical signs of infarction.
Establishment of an animal model to enable the use of electrical impedance tomography to monitor focal cerebral infarction using a photochemical method without craniectomy. (A) Top view of the brain 48 h after model establishment. (B) Cross-section of the infarct focus induced on the cortex. (C) Histopathological section of infarct focus (haematoxylin & eosin staining); original magnification ×100. The colour version of this figure is available at: http://imr.sagepub.com.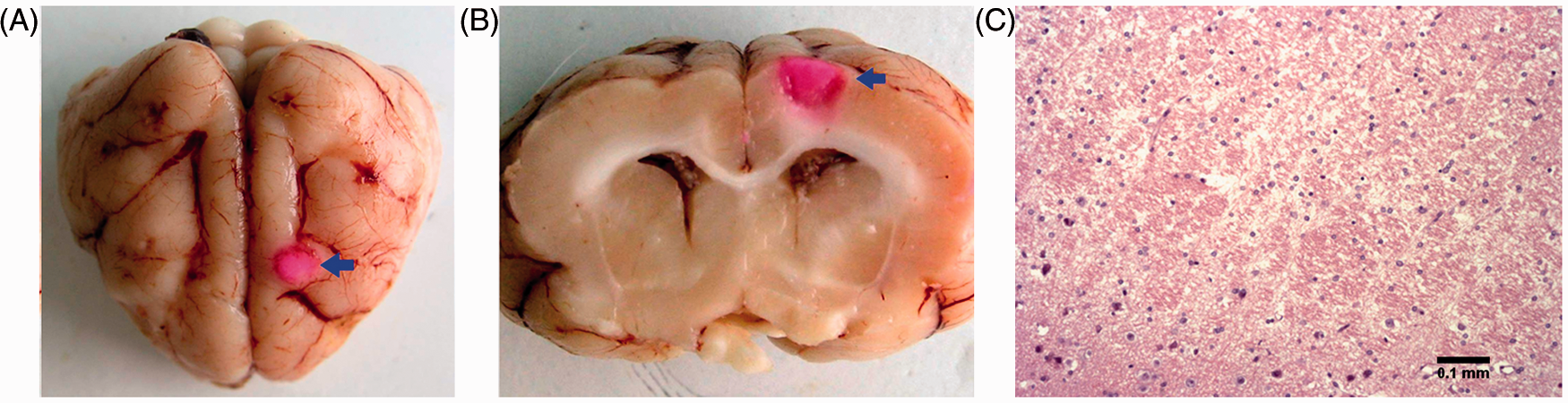
EIT imaging
Twenty EIT image frames were averaged to reduce random noise and improve the EIT signal-to-noise ratio. Figure 3 shows the reconstructed images for the 30-min irradiation period and the subsequent 60 min, showing the changes in resistivity occurring in the eight rabbits with focal cerebral infarction. During the 10 min before irradiation, no significant resistivity changes in the EIT images were observed. These images and data constituted the reference frame for later reconstruction and the control group for later comparison (Figure 3, 0 min). Between the beginning and end of irradiation, a region of interest gradually became visible (the blue areas in the upper left, shown on the right of the images in Figure 3). The finding implies that resistivity in the left frontal lobe of the brain began to increase. For the next hour, the region of resistivity gain (blue) remained relatively stable, with increments of resistivity and expansion in the area, although an evident annular region with increasing resistivity was observed in certain images; the authors considered that this could indicate physiological or reconstruction artefact.
Tomographic images of intracranial electrical impedance showing the onset and progression of focal cerebral infarction in eight rabbits, in a model induced by a photochemical method without craniectomy. R1 to R8 represent eight anaesthetized rabbits. Image orientation is indicated in the diagram at top right, in which A, L, P and R represent the anterior, left, posterior and right parts of the brain. The spectral bar at the bottom represents the possible range of colours from diminished resistivity (red) to normal baseline intensity (green) and increased resistivity (blue). The numbers 1.5 and −1.5 are the upper and lower limits of the resistivity values, respectively. The colour version of this figure is available at: http://imr.sagepub.com.
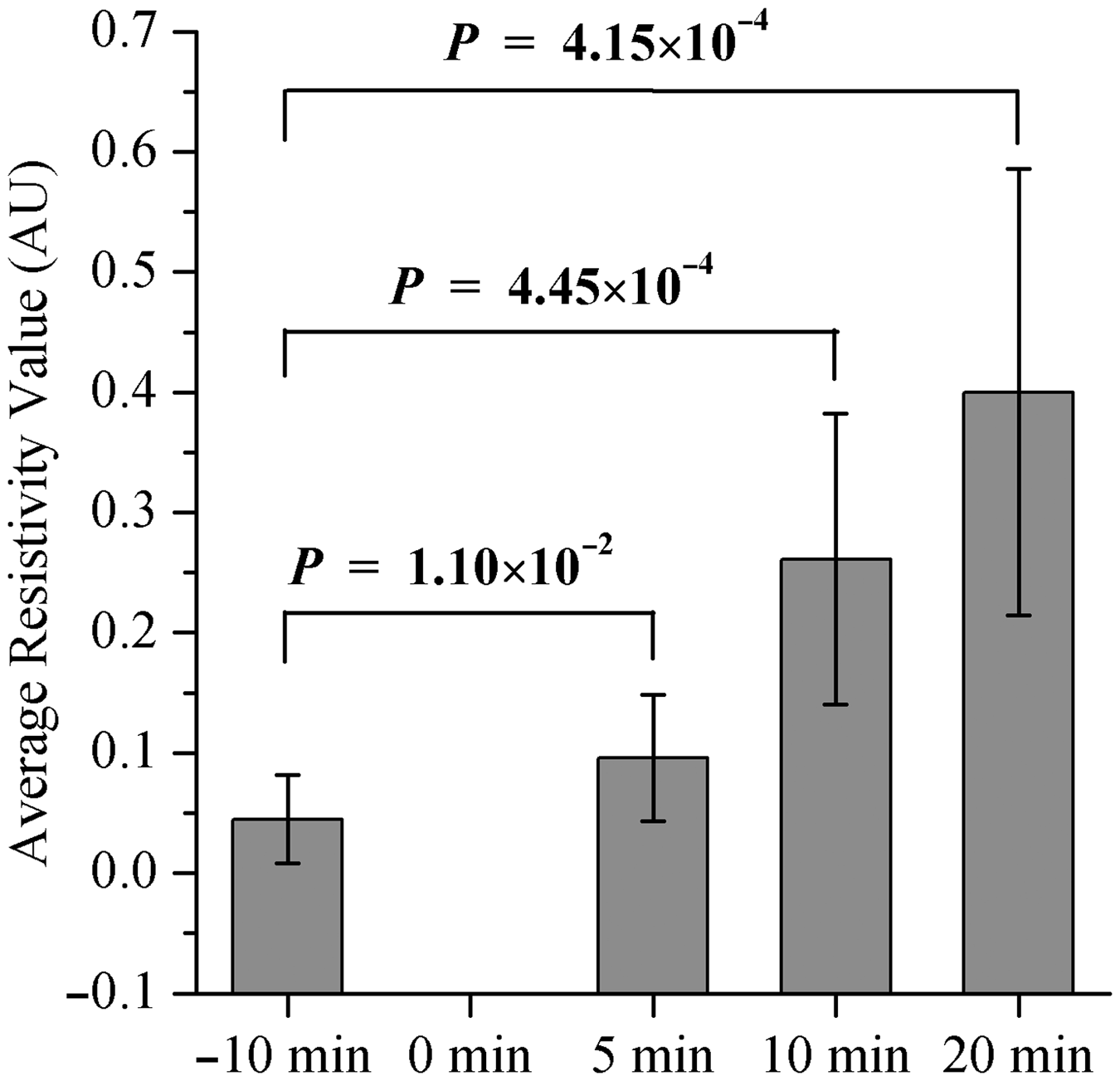
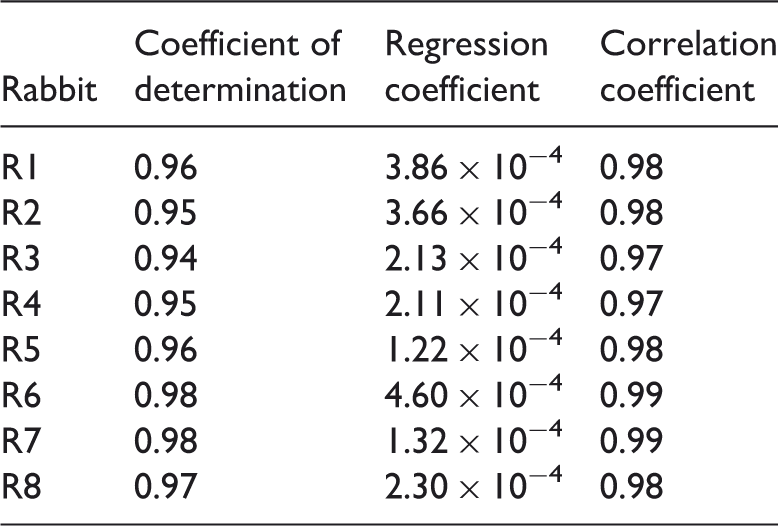
Figure 4 shows the changes in ARV with time for all animals. For each rabbit, the ARV curve showed two segments. One was a transitory flat segment with a constant ARV of nearly zero; the other showed an almost linear continuous increase. Table 1 provides the results of linear regression analysis of the relationship between ARV and cold light irradiation time. A significant linear correlation between ARV and irradiation time for each rabbit was observed (P < 0.01). The regression coefficient and the coefficient of determination for all animals were (2.65 ± 1.24) ×10−4 and 0.96 ± 0.01, respectively (mean ± SD). The differences between ARV for the 10-min period before the introduction of infarction and the ARV values for 5, 10 and 20 min after the beginning of irradiation are shown in Figure 5. A significant increase in impedance compared with the pre-irradiation (control) value was first observed 10 min after the beginning of irradiation (P < 0.01).
Average intracranial resistivity values plotted against time for eight rabbits (R1 to R8), in which a model of focal cerebral infarction was induced using a photochemical method without craniectomy. The x-axis shows time after the start of irradiation. AU, arbitrary units. Comparison of average intracranial resistivity values before and after the induction of a focal cerebral infarct in eight rabbits. The x-axis shows time after the start of irradiation. In the timescale on the x-axis, 0 min indicates beginning of irradiation. A significant increase in impedance compared with the pre-irradiation value was first observed 10 min after the beginning of irradiation (P < 0.01; two-tailed paired-sample t-test). AU, arbitrary units. Results of linear correlation analysis of average intracranial resistivity values against irradiation time after the start of irradiation in eight rabbits (R1 to R8), in which a model of focal cerebral infarction was induced using irradiation (600 mW/cm2) without craniectomy to enable the use of electrical impedance tomography to monitor focal cerebral infarction. A significant linear correlation between ARV and irradiation time for each rabbit was observed (P < 0.01).
The SI curves are shown in Figure 6. A monotonic increase was observed for every rabbit during the 50 min period after irradiation began. Subsequently, the curve showed a significant increase in animal R3 and a slow decrease in animals R6 and R7. These findings could be attributed to the region of reduced resistivity that appeared in the EIT images after the beginning of irradiation (Figure 3). This region was located on the same side as the region of interest in animal R3 and on the opposite side in animals R6 and R7 (Figure 3).
Curves of symmetrical index plotted against time for eight rabbits (R1 to R8), in which a model of focal cerebral infarction was induced using a photochemical method without craniectomy. The x-axis shows time after the start of irradiation. AU, arbitrary units.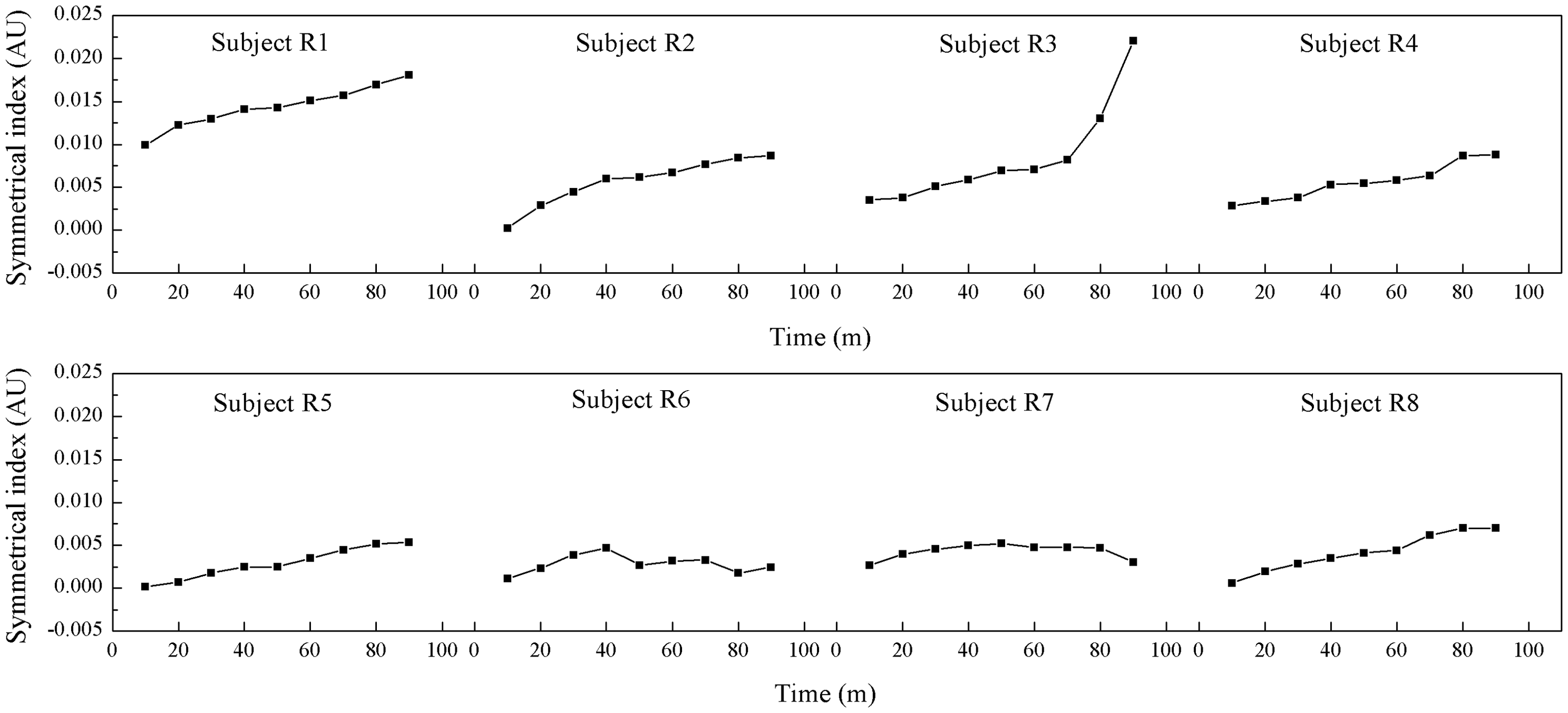
Discussion
In this work, animal studies were performed using EIT to monitor intracranial resistivity variation during focal cerebral infarction. An animal model of focal cerebral cortical infarction was established in rabbits by a photochemical method without craniectomy. The cortical region of cerebral infarction, as well as infarction size and depth, was controlled in this model. Pathological analysis showed that typical cerebral infarction was observed, with lamellar necrotic tissue, thus verifying this as a cerebral infarction model. Reproducible EIT results were observed during the onset of cerebral infarction and subsequent processes. Given that the cold light was directed directly into the blind hole, EIT images and ARV curves were able to reveal the resistivity increase located in the left anterior region of the brain. Data on the symmetry of resistivity (SI results) displayed unilateral resistivity changes in the cerebral cortex. These findings therefore indicate that the rabbit model used was suitable for studying EIT for the detection of microvascular obstruction, and demonstrate the feasibility and sensitivity of EIT for monitoring focal cerebral cortical infarction. Indeed, the present findings may have substantial implications for the imaging of focal cerebral infarction in humans by EIT, in future.
The principle of the photochemically induced cerebral infarction animal model is that Rose Bengal induces a photosensitivity reaction to certain wavelengths of light and gradually releases free radicals, resulting in injury to the endothelium and aggregation of platelets on the endothelium. Blood cells would release adenosine diphosphate and initiate intravascular coagulation and consequent thrombosis, leading to ischaemic brain injury and to consequent inflammation and necrosis. 22 This method was validated in the present study by the final histopathological examination, performed 48 h after model establishment. Infarction in this model occurs through a process similar to the formation of thrombi by microangiopathy in humans. This animal model has been shown to be suitable for the study of clinical cerebral infarctional disease. 23 Moreover, in the blind hole, the inner plate of the skull was preserved to ensure that the intracranial environment was comparatively stable, thereby decreasing injury and the probability of infection in the animal.
There is one aspect of our study design that warrants further explanation, namely the use of the reference (control) method that we adopted, which has also been applied in many other studies.11–15 In the present study, images generated in animals during the 10 min before irradiation commenced were used as control group data and served as the reference frame of the dynamic imaging in EIT of the brain. The validity of the infarction model and the accuracy of the EIT system, which was adjusted by calibration procedures to ensure the drift was <1%, indicated the correctness and suitability of the method that we selected.
Although cerebral infarction based on an animal model can be observed using EIT imaging, several factors should be considered before EIT is used to identify and locate regional resistivity changes during cerebral infarction in humans. In the present experiments on cerebral infarction, the electrodes were fixed onto each rabbit’s scalp. However, noninvasive scalp electrodes would be employed to record intracerebral impedance changes during cerebral infarction in a clinical situation. Thus, it is of great importance that the scalp electrodes should maintain good contact and provide the lowest measurement noise, in order to ensure adequate measurement accuracy. One possible means of improving measurement accuracy is to optimize the design and configuration of the electrode system for EIT of the brain. 24 For initial clinical studies, various types of Ag/AgCl and hydrogel bio-electrodes for brain EIT need to be evaluated in terms of contact impedance, uniformity, signal-to-noise ratio and stability in the frequency or time domain, in order to identify the optimum approach for the monitoring of cerebral infarction using EIT.
Since complicated influences will be at work in clinical cases, consideration must be given to the possibility of measurement data being invalid because of complete or partial disconnection of electrodes resulting from patient movement, perspiration, manipulations by clinical staff and defective electrode leads. 25 Before proceeding to clinical studies, it is essential to study preprocessing methods for identifying and deleting invalid data in order to reduce reconstruction failures in EIT studies.
The present animal study of EIT has simplified the head model as a homogeneous circle and the resulting inaccuracy of this model would cause significant imaging errors if used in the reconstruction of human data. An anatomically accurate head model with a realistic distribution of skull resistivity has been shown to improve the quality of reconstruction images in brain EIT. 4 Furthermore, according to our previous studies, the resistivity of the live human skull is inhomogeneous and there is an inverse relationship between the percentage thickness of diploe and skull resistivity of the three-layered skull.26,27 Thus, further work is needed to incorporate accurate geometry of the head and the inhomogeneous resistivity of the skull into the reconstruction algorithm, in order to improve the localization accuracy and image quality of brain EIT in patients.
In conclusion, the present study demonstrates that EIT is sensitive to impedance variations caused by focal cerebral cortical infarction in rabbits. Although more studies are needed, that focus on the design of an electrode system for the human head (and the development of data pre-processing and reconstruction methods for clinical practice), the present study results suggest the future possibility of using EIT for the monitoring of cerebral infarction.
Footnotes
Acknowledgements
The authors wish to express their gratitude to Professors Guodong Gao and Zhenwei Zhao of the Neurosurgery Department of Tangdu Hospital, Fourth Military Medical University, Xi’an, China, for providing valuable suggestions on the establishment of the animal model.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This work was partially supported by the National Natural Science Fund of China (grants 50937005 and 30870653) and the Scientific and Technologic Development Projects Fund of Shaanxi Province (grant S2011SF1363).