Abstract
Right ventricular (RV) function is a predictor of outcomes in pulmonary arterial hypertension (PAH). The 6-min walk test (6MWT) is likely an indirect measure of RV function during exercise, but changes in absolute walk distance can also be influenced by factors like effort and musculoskeletal disease. Paired 6MWT with continuous electrocardiogram monitoring was performed in stable PAH patients, patients adding PAH therapies, and healthy controls. Heart rate expenditure (HRE) was calculated (integrating pulse during 6MWT) and then divided by walk distance (HRE/d). We also evaluated changes in peak heart rate, time above age-adjusted maximum predicted heart rate, and heart rate at 6 min. HRE/d was compared to invasive hemodynamic measures in patients who had right heart catheterization performed within seven days, WHO functional class assessment, and Emphasis 10 questionnaire. We measured two 6MWT in 15 stable PAH patients, 13 treatment intensification patients, and 8 healthy controls. HRE/d was reproducible in the stable PAH group (median difference, −0.79%), while it decreased (median difference, 23%, p = 0.0001) after adding vasodilator therapy. In 11 patients with right heart catheterization, HRE/d correlated strongly with stroke volume, r = −0.72, p = 0.01. Peak heart rate decreased after adding vasodilator therapy. HRE/d also correlated with WHO functional class and Emphasis 10 score. Continuous heart rate monitoring during 6MWT provides valuable physiologic data accounting for effort. HRE/d appears to enhance test reproducibility in stable patients while detecting change after adding therapy as compared to walk distance alone.
Introduction
Pulmonary arterial hypertension (PAH) is a lung vascular disease in which the right ventricle (RV) fails because of progressive increases in afterload. RV function is the strongest predictor of morbidity 1 and mortality, 2 and can be used as a marker of treatment response; therefore, quantifying RV function is critical. The complex RV geometry renders echocardiography problematic for precise and quantifiable measures, especially in the obese. Resting cardiac MRI is the gold standard for imaging and quantifying the RV, but it is expensive, less available, and sometimes difficult for patients to tolerate. 3 Right heart catheterization (RHC) also provides direct measurement of RV function but is invasive and costly. The 6-min walk test (6MWT) is a core component in the evaluation of PAH which appears to reflect RV performance during stress, and it can be serially measured in the clinic. While 6MWT offers a simple, non-invasive and inexpensive measurement, absolute distance walked is likely confounded by several non-cardiopulmonary factors including age, height, weight, 4 effort, and musculoskeletal disease.
There is little published data documenting heart rate changes during 6MWT. In a previous study using photoplethysmography (PPG), we proposed that the integral of the heart rate divided by the distance walked might be a useful surrogate of RV function. We hypothesized that the strategy would index walk distance for cardiac effort. We labeled this value “HRE/d” for “heart rate expenditure/walk distance” and found that HRE/d correlated strongly with resting RV function measured on same day nuclear (D-SPECT) imaging. 5 Furthermore, HRE/d decreased after adding vasodilator therapy but was stable on different days for those not changing therapy. This preliminary work suggested that measuring continuous heart rate during 6MWT could “uncover” meaningful cardiovascular improvement even when walk distance was not significantly different and offered better reproducibility for those not changing. Although this approach was interesting, we had more than 30% data loss (completely unusable recording) because PPG signals often deteriorated during 6MWT; in addition, most of the recordings required editing/imputation to eliminate noise in a usable recording. Therefore, we investigated a telemetric ECG measure of heart rate to avoid data loss for the current study.
We hypothesized (1) HRE/d measured by electrocardiogram would have less data loss than PPG, (2) HRE/d would provide a more reproducible measurement than 6-min walk distance (6MWD) alone, (3) HRE/d would decrease with therapy intensification, (4) HRE/d would correlate with invasively measured stroke volume, and (5) HRE/d would correlate with quality of life metrics. To test the hypothesis, we utilized MC10 Biostamp nPoint sensors, which are capable of recording continuous ECG through a patch worn on the skin.
Methods
Study population
All study procedures were approved by the Research Subjects Review Board, and each participant provided written consent prior to any research testing. Participants were recruited from the Pulmonary Hypertension Clinic and studied between December 2018 and February 2020. To be eligible for participation, adult patients (age >18 years old) must have had a diagnosis of WHO Group 1 PAH. 6 We enrolled treatment naïve patients or those with clinical indications (persistent breathlessness or exercise intolerance thought to be responsive to additional therapy) for additional PAH therapy into the “treatment intensification” (TI) group (Fig. 1). Patients without indication to change therapies (or those on maximal therapy) were assigned to the stable group (Fig. 1). We also recruited healthy controls (Fig. 1). We collected clinical information including N-terminal pro b-type natriuretic peptide (NT-pro BNP) measurement, WHO functional class assessment, and REVEAL 2.0 risk assessment 7 at baseline and follow-up in PAH participants only.
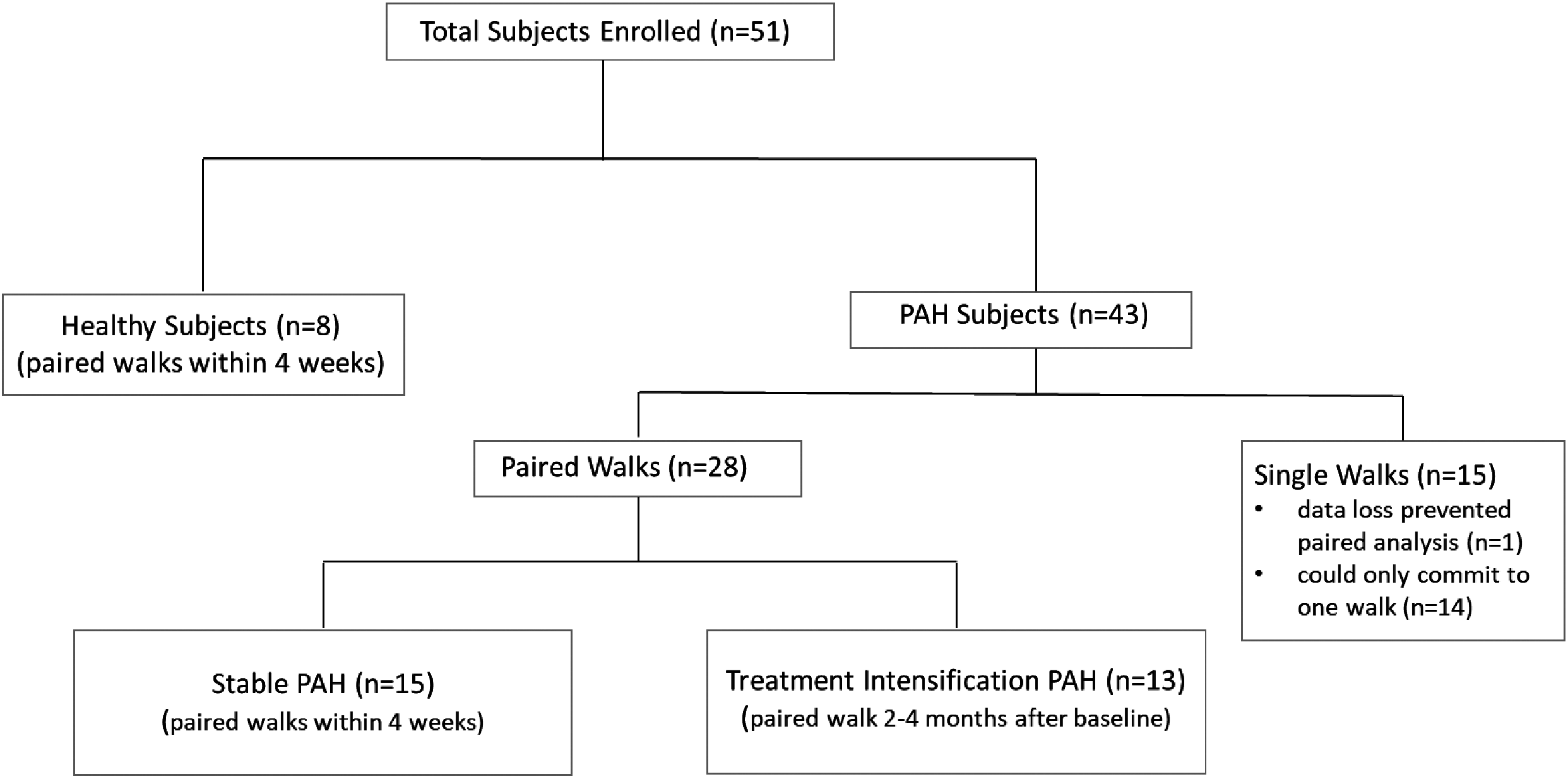
Participants who completed 6MW with continuous heart rate monitoring. All PAH patients had Emphasis 10 questionnaire and WHO functional class assessment at each walk.
6-Min walk with heart rate monitoring
We used the MC10 Biostamp nPoint to measure continuous heart rate during 6MWT; the device has FDA 510k clearance, and the cloud storage is fully Health Insurance Portability and Accountability Act of 1996 compliant. In the stable groups without planned treatment changes (PAH and healthy control), paired walks were completed within four weeks to measure variability in the test (Fig. 1). The TI group had testing at baseline and again 2–4 months later (Fig. 1). The rest of the participants could not commit to completing paired walks in the pre specified time frame. The 6MWT was performed according to American Thoracic Society guidelines. 8 Blood pressure, oxyhemoglobin saturation, and heart rate were monitored using a CARESCAPE V100 Monitor before and after the walk with the participant seated. Participants completed an Emphasis 10 questionnaire 9 at the time of each walk. Treatment decisions were made at the time of clinical visit by the PH physician (DJL or RJW). Risk assessment was performed using REVEAL 2.0. 7
Two sensors were placed on the chest (one on the 2nd left intercostal space and one at the cardiac apex) to minimize data loss (some participants have best signal at apex and others at sternal border). Heart rate recording started after the two MC10 Biostamp nPoint sensors were placed on the patient’s chest and continued for the duration of the 6MWT plus an additional 3 min after the walk was completed. Once the sensors were removed and placed back on the docking station, the data were uploaded to a secure cloud. Raw ECG tracings were analyzed using LabChart (ADInstruments) software. Heart rate was averaged to an integer every 4 s before graphing and analysis. Heart rate parameters evaluated included baseline heart rate, total heart beats, heart rate expenditure (‘HRE/d’, calculated by integrating pulse during 6MWT and dividing by walk distance in meters), peak heart rate, heart rate at 6 min, heart rate at 7 min, heart rate reserve (peak heart rate − baseline heart rate), and heart rate recovery (heart rate at 6 min − heart rate at 7 min). Of note, we documented the difference between ECG heart rate at 6 min (precise end of walk) and the pulse captured and recorded with pulse oximetry on the CARESCAPE V100.
Statistical analysis
We compared baseline and follow-up data using paired t-test for continuous variables; we used one-way ANOVA to make between group comparisons, and Pearson correlation to correlate continuous pulse parameters with other clinical variables. Bland–Altman plots were used for comparisons. The calculations were performed using Prism 7.0 (GraphPad Software Inc.).
Results
Participant characteristics
We enrolled a total of 51 participants; 43 with PAH and 8 healthy controls. Among the 51 enrolled, 28 PAH participants (15 stable controls, 13 TI) and 8 healthy controls had paired walk data as planned. Of the remaining 15 participants, 1 stable participant had data loss preventing paired analysis and only 1 walk was able to be analyzed; 14 had a single walk because they could not commit to follow-up. Out of 87 total walks (28 paired PAH, 8 paired healthy control, and 15 single PAH walks) only one (1.1%) was unable to be analyzed because of data loss. The majority of patients were female with idiopathic disease whose baseline demographics and therapies are listed in Table 1.
Baseline demographics.
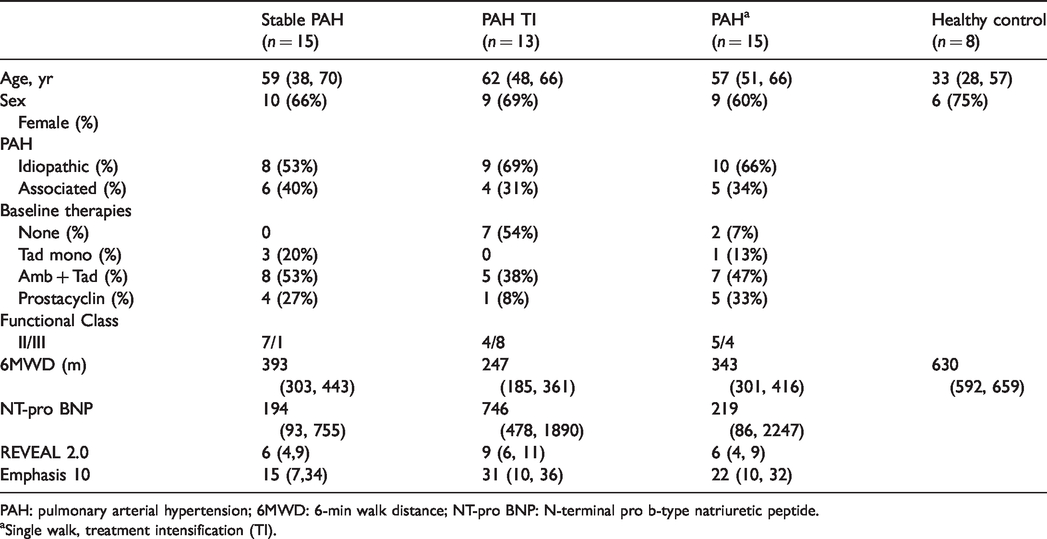
PAH: pulmonary arterial hypertension; 6MWD: 6-min walk distance; NT-pro BNP: N-terminal pro b-type natriuretic peptide.
aSingle walk, treatment intensification (TI).
HRE/d improves 6MWT reproducibility in stable participants
In stable PAH patients and healthy controls, HRE/d was highly reproducible between walks (Fig. 2a and c). Of note, stable PAH participants had a wide range of RV size and function. In these stable patients, the median difference in HRE/d was −0.79% (95% confidence interval, −4.0%, 3.6%) while the median difference in 6MWD was numerically higher with more variability at −1.0% (95% confidence interval, −9.0%, 2.8%; Fig. 2e). Bland–Altman plots suggested that HRE/d has less variability than 6MWD in stable PAH and healthy control participants (Fig. 3). In patients who added therapy (TI), HRE/d decreased by a median of 23% (95% confidence interval, −11%, −36%) while 6MWD increased by 27% (95% confidence interval, 1.5%, 46%; Fig. 2b and f). There was marked variability in the absolute 6MWD change, especially because some treatment naïve individuals had very low baseline 6MWD (Fig. 2h).
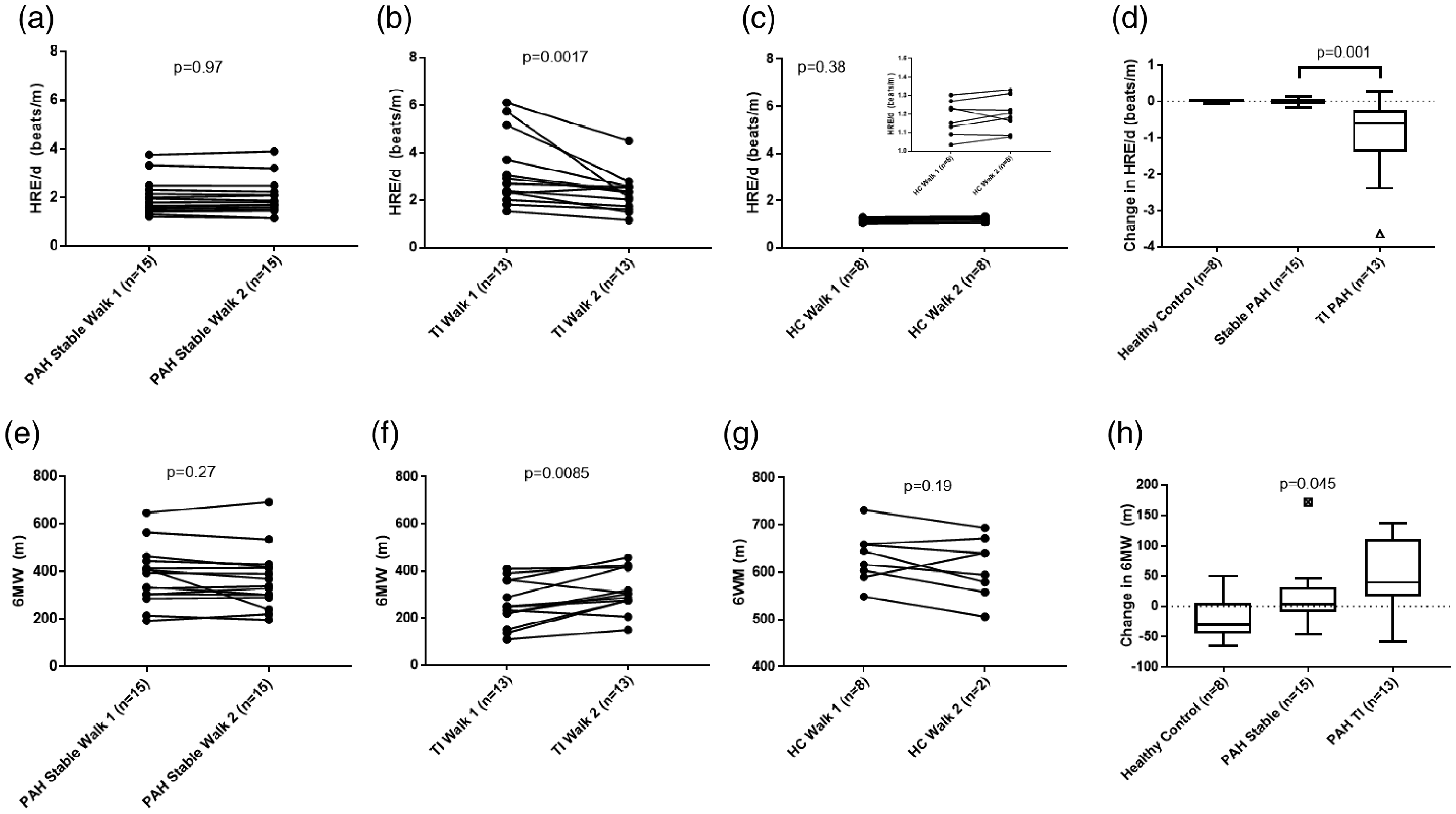
Paired HRE/d and 6MWT in PAH and healthy controls. (a) HRE/d is very reproducible in paired 6MWT in stable PAH patients. (b) HRE/d decreases significantly after adding vasodilator therapy. (c) HRE/d was strongly reproducible in eight healthy controls. (d) Absolute change in HRE/d was significantly increased after adding therapy in PAH compared to stable PAH patients. (e) There is more variability in 6MWD in stable PAH patients. (f) The 6MWD increased after adding vasodilator therapy. (g) There is more variability between 6MWD in healthy controls compared to HRE/d. (h) Although there was a significant increase in walk distance compared to stable PAH patients there is more variability than what was seen with HRE/d.
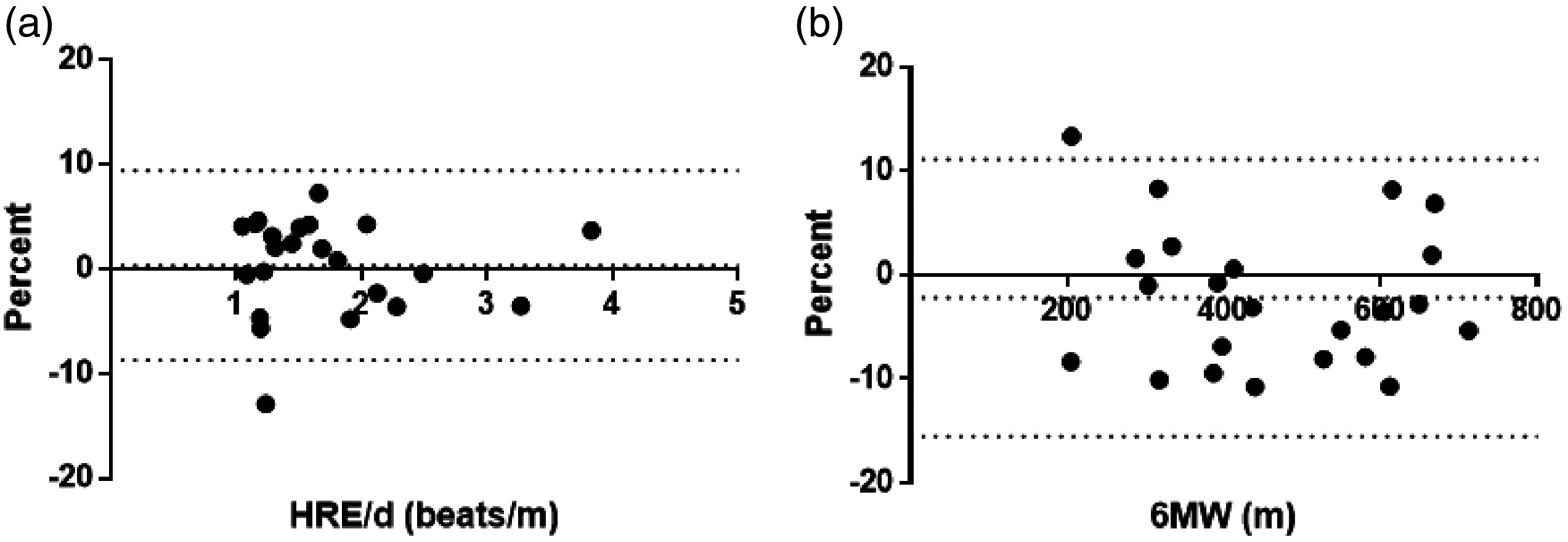
Bland–Altman plots show (a) HRE/d has less variability than (b) 6MWD in stable PAH and healthy controls.
HRE/d correlates with invasive hemodynamics
Eleven PAH patients had a RHC performed ±7 days of HRE/d measurement. There were no clinical or medication changes between the RHC and 6MWT. HRE/d correlated strongly with hemodynamic parameters including stroke volume, cardiac output, and pulmonary arterial compliance (stroke volume/pulse pressure; Fig. 4). The correlations were very similar when indexed for body surface area.
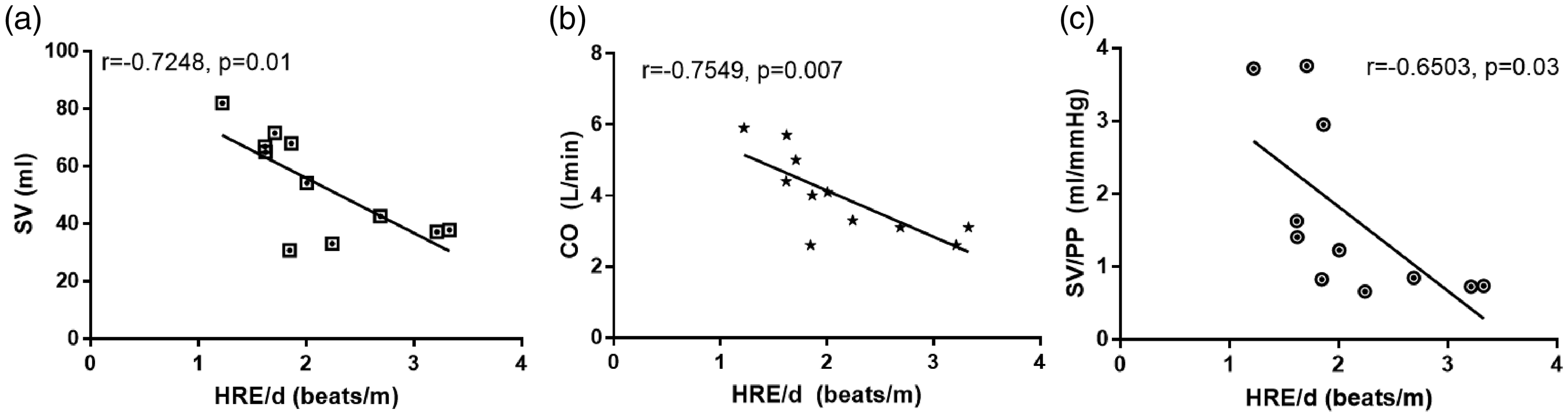
HRE/d correlates with invasively measured (a) SV, (b) CO, and (c) SV/PP hemodynamics.
Heart rate changes during 6MWT
In stable PAH participants and healthy controls, there was no change in peak heart rate, heart rate at 6 min, or heart rate at 7 min (Table 2). In the TI group, there was a decrease in peak heart rate after therapy was added (Table 2). There was no difference in heart rate at baseline, 6 min, 7 min, nor was there a difference in heart rate recovery after adding therapy. None of our healthy controls achieved a heart rate >80% of their age-adjusted maximum, while 24/43 (56%) PAH participants had a heart rate >80% of their age-adjusted maximum during 6MWT. Age correlated stronger with 1/6MWD (r = 0.38, p = 0.0008) than with HRE/d (r = 0.24, p = 0.04). We did identify a significant difference, 22 beats/min (95% confidence interval, 20, 25 beats/min) between the heart rate measured at 6 min using MC10 Biostamp nPoint compared to the value captured with the clinical pulse oximeter at the usual time (once patients are seated and the value registers). For one patient, we identified a supra-ventricular tachycardia during 6MWT with the MC10 sensor.
Heart rate changes during 6MWT.
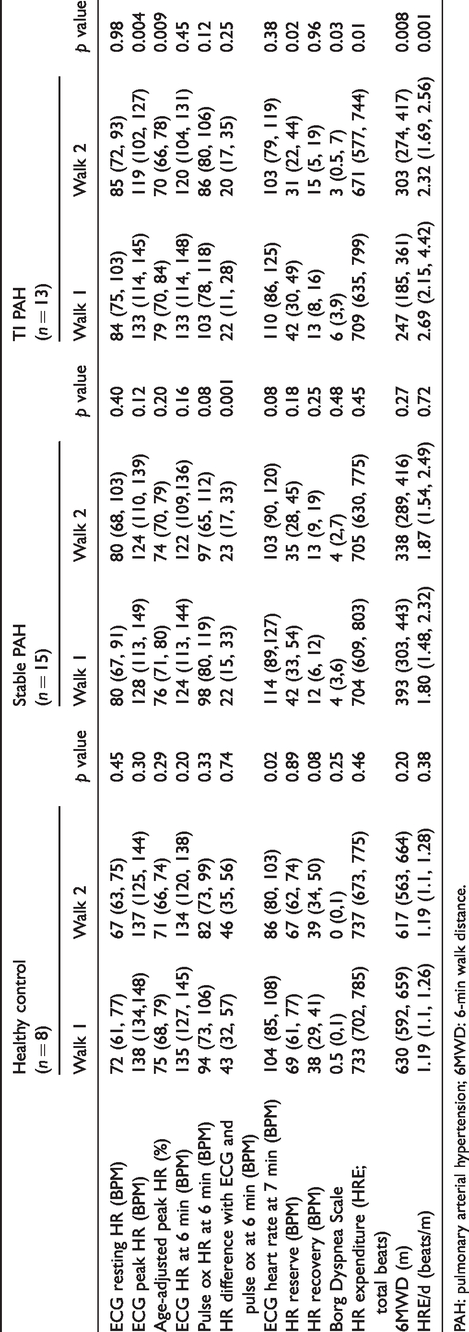
PAH: pulmonary arterial hypertension; 6MWD: 6-min walk distance.
HRE/d and clinical variables in PAH
We analyzed the calculated HRE/d in all PAH walks against REVEAL 2.0, Emphasis 10, functional class, and NT-pro BNP. HRE/d in PAH participants correlated strongly with REVEAL 2.0 and Emphasis 10 (Fig. 5a and b). HRE/d was also distinct for different REVEAL 2.0 risk categories and WHO Functional Classification (Fig. 5c and d). Interestingly, for this group, HRE/d did not correlate with NT-pro BNP; this is different than our previous study 5 and was likely influenced by outliers with severe renal dysfunction or obesity. In PAH participants with paired walks, the changes in HRE/D correlated strongly with changes in REVEAL 2.0 and Emphasis 10 (Fig. 6).
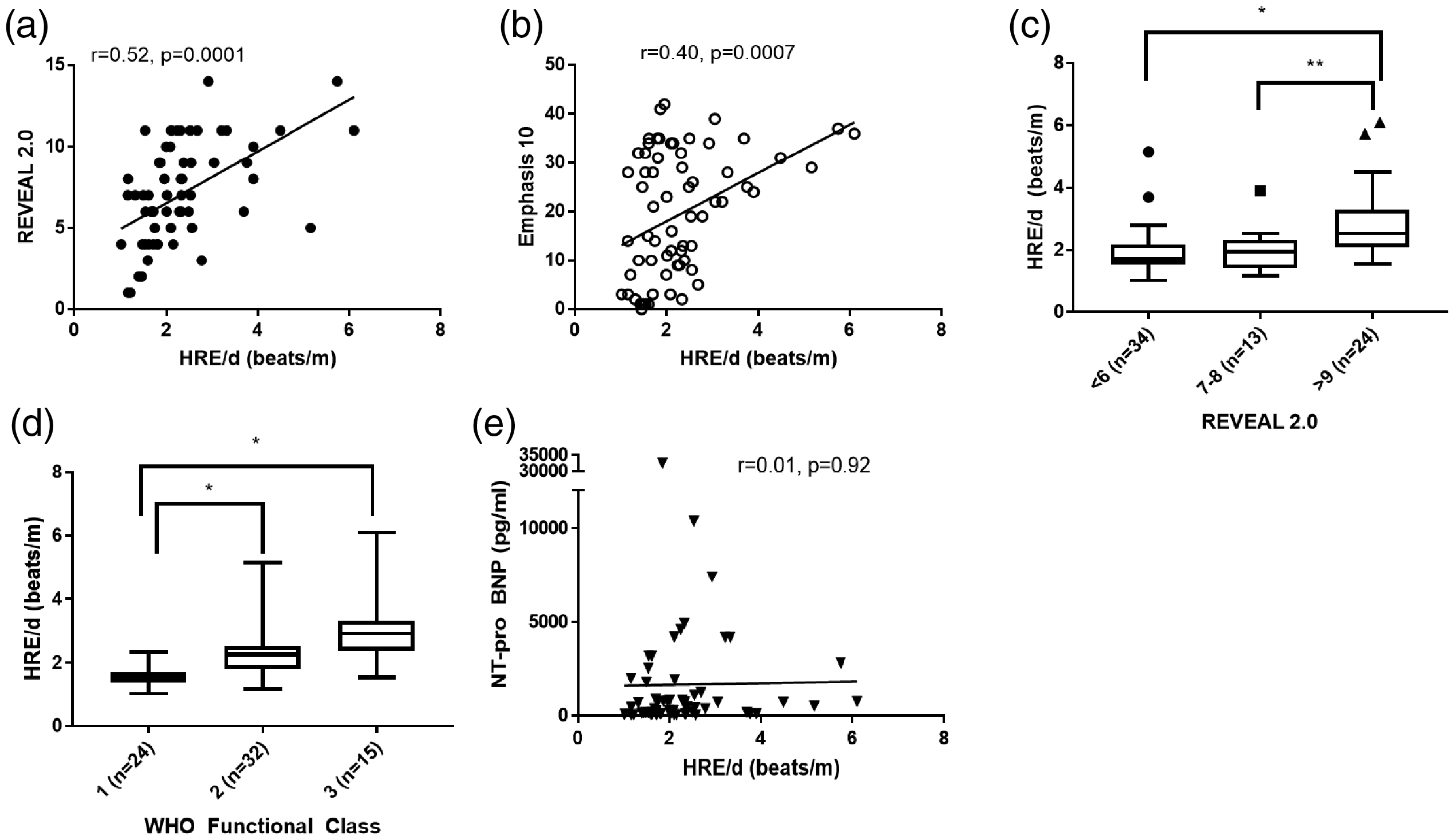
HRE/d correlates with functional status and quality of life. (a) HRE/d correlates with REVEAL 2.0 Risk Assessment and (b) Emphasis 10 questionnaire. (c) HRE/d was associated with REVEAL 2.0 low risk (<6), intermediate risk (7–8), or high risk category (>9). (d) HRE/d was associated with WHO Functional Class assessment. (e) HRE/d did not correlate with NT-pro BNP. *p = 0.0001 and **p = 0.01.
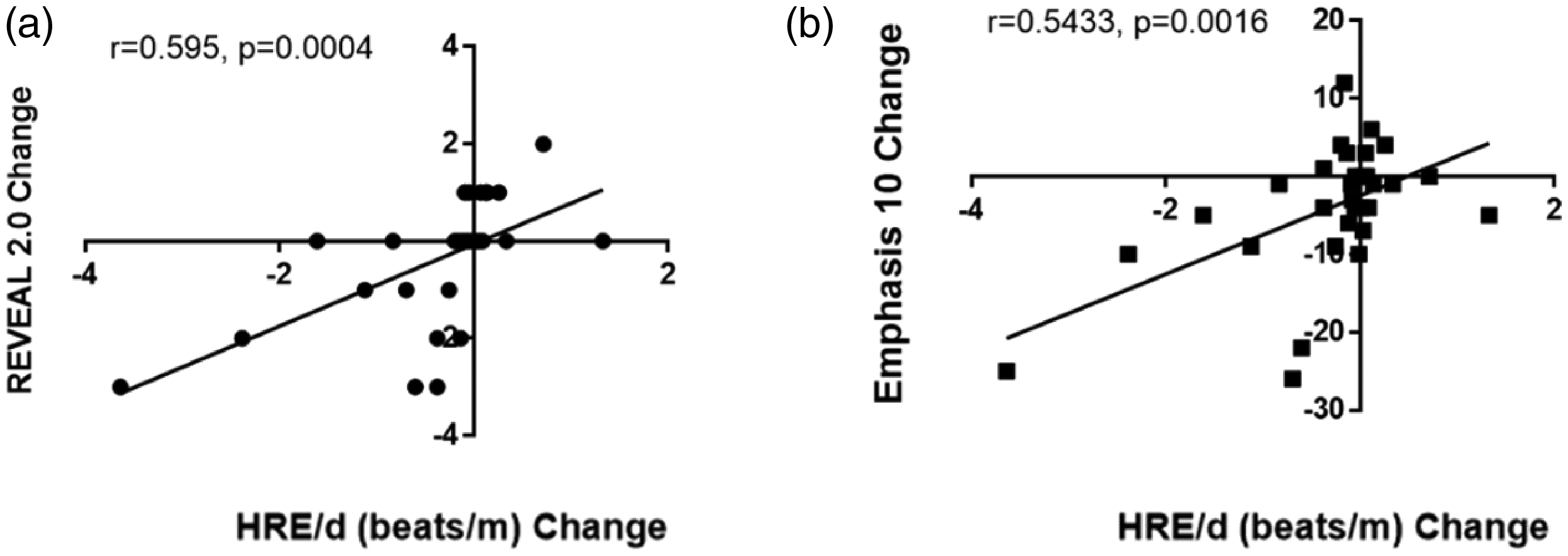
Changes in HRE/d correlates with changes in (a) REVEAL 2.0 risk assessment and (b) Emphasis 10 questionnaire.
Discussion
Our findings strengthen our initial report 5 and demonstrate the added value of continuous electrocardiogram monitoring during 6MWT over that of PPG. Specifically, only one walk was excluded because of poor data collection, and we did not impute any data. HRE/d appears more reproducible in stable PAH patients than 6MWD and correlates with invasive hemodynamics and validated clinical assessments. Electrocardiogram monitoring provides physiologic data that increases the utility of the 6MWT, especially when evaluating longer walk distances where greater variability between walks’ distances exists.
The 6MWT is a submaximal exercise test that was first reported by Butland et al. in 1982 after comparing paired 2-, 6-, and 12-min walks’ tests in patients with chronic respiratory failure. 10 The different times produced internally consistent walk distances and 6 min was chosen as being a sensible compromise to assess exercise tolerance. 10 They acknowledged that longer walk distances were associated with greater variability which might reflect greater random variation. 10 This was recently confirmed in 56 healthy young adults completing paired 6MWD (median 6MWD difference was 32.5 ± 31.9 m). 4 The variability in 6MWD is a common problem typically encountered in routine clinical practice. The greater random variability among those patients walking longer distances may have contributed to the muted 6MWD benefits seen in combination studies.11–14 Random variability in longer walks is an alternate explanation for failure of 6MWT in combination studies as opposed to a “ceiling effect”. 15 The 6MWT has other limitations. Changes between walks can be influenced by non-PAH factors including effort or musculoskeletal disease (e.g., scleroderma). Age and anthropomorphic variables (height and weight) can also influence walk distance 4 so assuming all participants have the same chance of improving walk distance is not accurate. HRE/d was reproducible on two different days over a large variety of walk distances in healthy controls and stable PAH patients (up to 700 m); this reproducibility would likely make it more sensitive to clinical/physiologic change, especially if effort is a concern. If the relationship between HRE/d and stroke volume is confirmed in further studies, it could be particularly valuable in identifying inadequate treatment in younger patients who may be less symptomatic but still have lower stroke volumes and therefore remain higher risk. 16
Resting heart rate in PAH has significant prognostic implications and is one of the parameters in the REVEAL 2.0 risk assessment. 7 Provencher et al. measured heart rate during 6MWT using pulse oximeter (PPG) and observed that chronotropic response (peak heart rate − resting heart rate) correlated with walk distance in treatment naïve patients with PAH. They concluded that a lack of chronotropic response may reflect more severe PAH with loss of physiologic reserve. 17 They found no correlation between resting, invasively measured SV, and chronotropic response. After starting bosentan monotherapy, they measured no change in chronotropic response. In contrast, we found HRE/d correlated highly with resting SV during RHC. Our TI group had a decrease in chronotropic response and peak heart rate after adding therapy. This observation might be related to better heart rate monitoring using electrocardiogram or the use of combination therapy and (hypothetically) a larger improvement in SV; the Dutch group reported that combination therapy in treatment naïve patients produced larger improvements in SV than monotherapy. 18 Tonelli et al. measured continuous heart rate during a single 6MWT and found patients with PAH had more shallow HR increases (during 6MWT) compared to healthy controls; moreover, those with subsequent clinical worsening had a more shallow slope compared to stable patients. 19 They did not evaluate for changes after adding therapy or for reproducibility of the measurement. Tonelli et al. may have identified patients with higher resting HR and poor baseline RV function. Minai et al. evaluated HR recovery after 6MWT at a single time point and found a recovery of <16 beats/min was associated with more severe disease and clinical worsening. 20 When we examined serial walks after TI, we found that HR recovery was often unchanged despite impressive changes in several other HR measures. In young healthy adults, Halliday et al. found a correlation with resting HR and increase in HR with 6MWD. 4 We believe this observation by Halliday et al. may be related to effort during the 6MWT and is part of what HRE/d seeks to address. Importantly, in our cohort we found HR had already dropped substantially at the time that pulse oximetry measured the HR (after stopping walk) as compared to HR on telemetric ECG at 6 min in both our healthy control and PAH participants. Our findings reported here and previously 5 illustrate the limitations associated with pulse measured by PPG (especially when done in the conventional manner after stopping exercise) and the significant advantage of telemetric ECG. Furthermore, our data underscore the need for both baseline and follow-up measures to determine the performance of new endpoints (e.g., heart rate recovery), especially as they might relate to therapy intensification or prognosis.
Using continuous telemetric ECG measurement, we found a reduction in peak HR, HR expenditure, and HR expenditure adjusted for distance walked (HRE/d) after beginning or intensifying therapy. We hypothesize these changes reflect improved RV function and may be more sensitive to therapy intensification than 6MWD, especially for those walking longer distances (where random variation in walk distance may be more likely).4,10 Importantly, these HR parameters were unchanged in the stable PAH and healthy control group where we would not expect there to be any changes in RV function. In addition, these observations were made in patients with wide variation in baseline 6MWD and in RV function. We also found that HRE/d did not correlate as strongly with age as 6MWD and may partially correct 6MWD for age. Our hypothesis that the HR changes associated with therapy intensification reflect improvement in RV function is supported by data (invasive, Fig. 4, and in our previous nuclear study 5 ). In recently reported registries,21–24 reassessment of risk after TI predicted long-term outcomes better than baseline assessment. 25 The French group advocated hemodynamic testing to better stratify the large intermediate risk group, identifying stroke volume index as a helpful determinant. 23 While multi-center confirmation and longer term study is clearly necessary, we propose that this non-invasive, inexpensive test might substitute for imaging or hemodynamics and could be done more frequently.
There are limitations to our study. We had small numbers in both our stable and TI group. A large number of our TI patients were treatment naïve with short walk distances at baseline so that absolute improvements in 6MWD were larger than generally observed for patients already on therapy. Only 11 patients underwent RHCs; the hemodynamic correlations are therefore preliminary but do confirm a small number of observations with different methodology in a previous cohort. 5 HRE/d has only been compared to resting RV function and future work is needed to evaluate whether the relationship holds true with an exercising RV. We did not have any patients on AV nodal blocking therapy, and patients on beta-blockers especially will need to be studied separately.
In summary, electrocardiogram HR monitoring during 6MWT provided physiologic data that may allow clinicians to account for effort or random variability in the walk distance (especially for longer walks) and therefore detect clinical improvement more easily than using walk distance alone. HRE/d is a reproducible and relatively inexpensive measure that may be an indirect measure of RV function. The variability between serial measures of HRE/d was less compared to 6MWD alone, and HRE/d correlated with clinically meaningful outcomes. This combination of improved reproducibility, and if confirmed, its relationship to RV function might make HRE/d an optimal non-invasive risk assessment tool. Further studies evaluating HRE/d are warranted to determine its utility in predicting longer term outcomes and in detecting therapeutic response for those with longer walks.
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DJL received support from Entelligence Young Investigator Award which allowed this work to be completed. No other author received support.
Ethical approval
The RSRB approved this protocol before the research was completed.
Consent
Consent was obtained from each participant before data were collected.
Guarantor
DJL guarantees the accuracy and integrity of the work.
Contributorship
All authors were involved in study design and data collection/analysis. All authors approved the manuscript.
