Abstract
While the prognosis of idiopathic pulmonary arterial hypertension has improved significantly due to newer medications, lung transplantation remains a critical therapeutic option for severe pulmonary arterial hypertension. Hence, it is essential for patients awaiting lung transplantation to avoid complications, including thrombocytopenia, which may affect their surgical outcomes. Herein we present the case of a 21-year-old woman diagnosed with idiopathic pulmonary arterial hypertension at the age of 15. She developed thrombocytopenia while awaiting lung transplantation. Her medication was switched from epoprostenol to treprostinil, suspecting possible drug-induced thrombocytopenia. Furthermore, she was administered thrombopoietin receptor agonists in view of the possibility of idiopathic thrombocytopenic purpura, along with maximum support for right heart failure. Subsequently, her platelet count increased to >70,000/µL, enabling her to successfully undergo bilateral lung transplantation. Post-bilateral lung transplantation, pulmonary arterial hypertension as well as thrombocytopenia appeared to have resolved. In this case, we suspected that thrombocytopenia could have resulted owing to a combination of pulmonary arterial hypertension, right heart failure, drug interactions, and idiopathic thrombocytopenic purpura. Thrombocytopenia is a very critical condition in patients with pulmonary arterial hypertension, especially those awaiting lung transplantation. Several approaches are known to improve intractable thrombocytopenia in patients with pulmonary arterial hypertension.
Keywords
Case description
A 21-year-old woman was diagnosed with idiopathic pulmonary arterial hypertension (IPAH) at the age of 15 following abnormal electrocardiography findings detected during a routine checkup (Fig. 1a and d). Right heart catheterization revealed the mean pulmonary arterial pressure, right atrial pressure, cardiac index, and pulmonary vascular resistance to be 65 mmHg, 4 mmHg, 2.74 L/min/m2, and 13 Wood units, respectively. Her platelet count then was 216,000/µL. She was prescribed tadalafil, followed by ambrisentan and beraprost. Two years later, beraprost was replaced by continuous intravenous epoprostenol therapy.
Chest radiograph and computed tomography on admission (a, d); just before lung transplantation (b, e); and after lung transplantation (c, f) demonstrating improvement in the cardiac silhouette and pulmonary congestion following lung transplantation. Histopathology of the resected lung. Hematoxylin and eosin stain: low magnification (g); Elastica-Masson stain: low and high magnification (h, i, j), showing dilated vessels (black arrow), the media wall thickness of the pulmonary artery (white arrow), and cellular intimal proliferation (thin black arrow). There are no findings that suggest microangiopathic thrombocytopenia. The clinical course from the first visit to lung transplantation (k): As idiopathic pulmonary arterial hypertension (IPAH) progressed, there was an exacerbation of thrombocytopenia. Switching from epoprostenol to treprostinil, the intensification of PAH treatments, and the administration of thrombopoietin receptor agonists improved thrombocytopenia to some extent, enabling lung transplantation. (BNP: brain natriuretic peptide, CI: cardiac index, mPAP: mean pulmonary artery pressure, PVR: pulmonary vascular resistance, RAP: right artery pressure, TRPG: transtricuspid pressure gradient).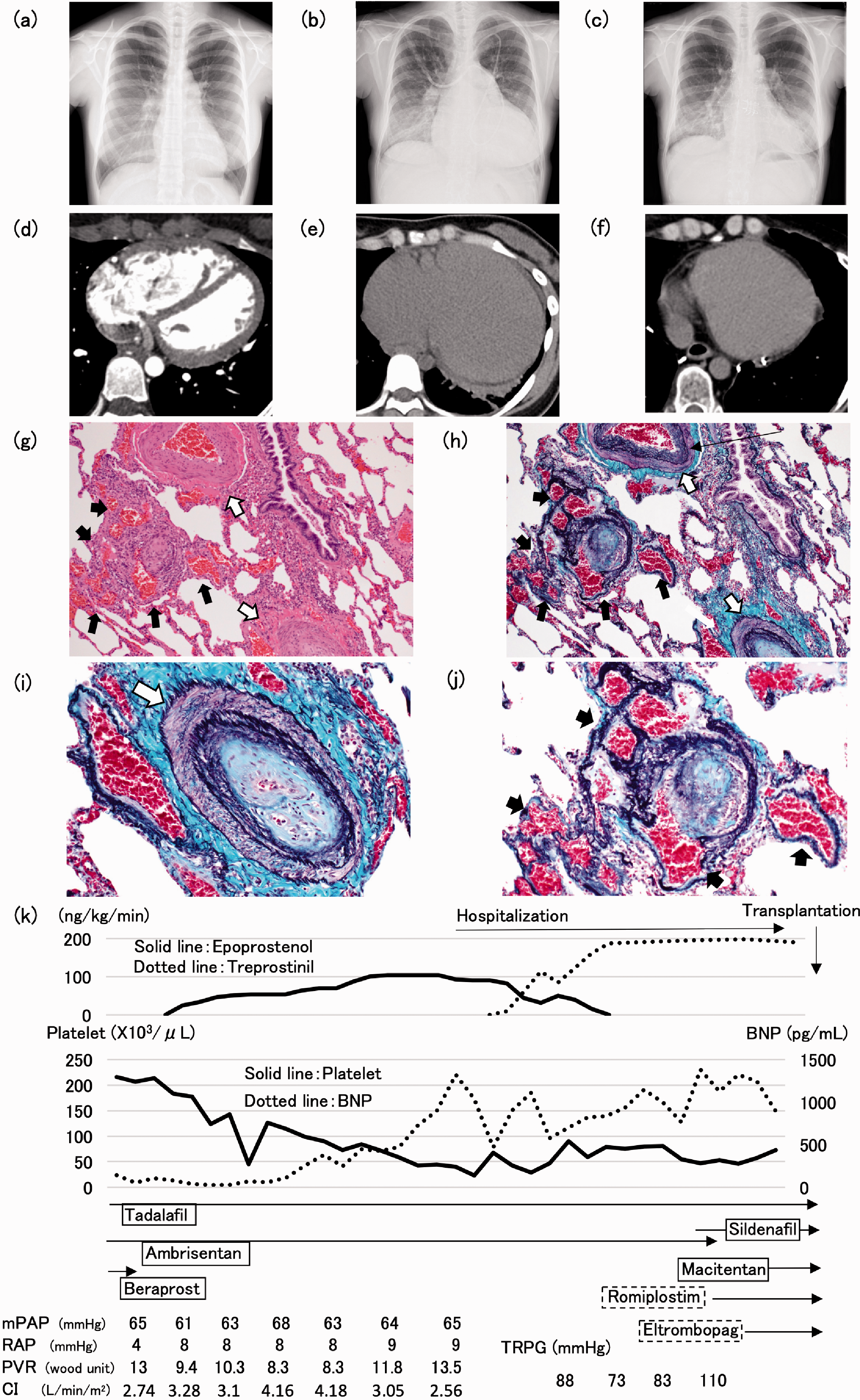
She was listed for bilateral lung transplantation from a brain-dead donor at the age of 19; subsequently, she developed mild thrombocytopenia; within a year, her platelet count decreased to 20,000/µL. We attempted the following additional strategies to ensure maximum support for right heart failure as a therapeutic intervention for thrombocytopenia until lung transplantation (Fig. 1b, e, and k):
Reduction of the epoprostenol dose secondary to a suspicion of epoprostenol-induced thrombocytopenia, following which her platelet count improved to 60,000/µL. Administration of prednisolone to treat possible idiopathic thrombocytopenic purpura (ITP) because of her elevated platelet-associated IgG (PAIgG) levels and increased megakaryocyte count on bone marrow biopsy. However, this was ineffective as her platelet count decreased again. Transition from epoprostenol 90.7 ng/kg/min to treprostinil 195 ng/kg/min; initially, her platelet count improved from 20,000 to 90,000/µL. However, three months after the transition was completed, her platelet count and pulmonary hemodynamics worsened. Administration of thrombopoietin receptor agonists (TRAs) for suspected ITP. Other therapeutic modifications: the addition of sildenafil; changing ambrisentan to macitentan; and increasing the treprostinil dose to a maximum of 204 ng/kg/min to counter pulmonary hemodynamic deterioration. She underwent successful bilateral lung transplantation once her platelet count improved to >70,000/µL and pulmonary hemodynamics stabilized. On histopathology, the diseased lungs showed Heath-Edwards grades up to 4, without any evidence of plexiform lesions or fibrin deposition in the pulmonary vessels (Fig. 1g–j).
Following lung transplantation, she did not require any further pulmonary vasodilators (Fig. 1c and f). She was maintained on prednisolone and immunosuppressants as required for post-transplantation therapy. Her pulmonary hemodynamics and platelet counts progressively normalized. She is continuing to show good progress at one year following surgery.
Discussion
We presented a case of severe IPAH and thrombocytopenia wherein we attempted to prepare the patient for lung transplantation by several measures including the transition from epoprostenol to treprostinil, intensification of PAH drugs, and administration of TRAs. Post-transplantation, pulmonary hypertension and thrombocytopenia resolved.
PAH is often reported to be complicated by thrombocytopenia, due to several reasons.1–4 Some conditions responsible for the etiology of PAH may induce platelet aggregation and destruction, resulting in thrombocytopenia. First, congestive splenomegaly may cause thrombocytopenia. Our patient was found to have mild splenomegaly on CT scan and abdominal ultrasound. The spleen size did not show any change following lung transplantation. Platelet counts may decrease with the deterioration of pulmonary hemodynamics as estimated by brain natriuretic peptide (BNP) and transtricuspid pressure gradient (TRPG), although this association is not consistent. It is possible that congestive splenomegaly induced by worsening pulmonary hemodynamics may be one of the critical factors related to thrombocytopenia. Second, one study reported the role of thrombin in the activation and binding of protease-activated receptor-1 on platelets in patients with IPAH. This binding leads to platelet degranulation and adenosine diphosphate release, resulting in platelet aggregation. Moreover, IPAH decreases nitric oxide and soluble guanylyl cyclase production, thus inducing platelet activation and aggregation. 5 This may be true in our case as the platelet count increased with a decrease in pulmonary arterial pressure following lung transplantation. Third, microangiopathic thrombocytopenia has been reported to be related to PAH. 4 Platelets circulating through the numerous plexiform lesions are often found to be damaged. Fibrin deposition in plexiform lesions on lung pathology and the absence of fibrin deposition on other organs indicate the presence of microangiopathic thrombocytopenia in PAH. 4 In our case, lung pathology did not reveal plexiform lesions and fibrin deposition in the pulmonary vessels, suggesting that reasons other than platelet destruction in the microvessels may have been responsible for the condition.
Drug-induced thrombocytopenia, especially with prostacyclin, has been reported in PAH patients.2,3 Epoprostenol is widely known to be associated with thrombocytopenia. 2 Although treprostinil may also induce thrombocytopenia, transitioning from intravenous epoprostenol to treprostinil reportedly improves thrombocytopenia. 3 Epoprostenol has a much higher affinity than treprostinil for the EP3 receptor, which promotes platelet aggregation.6,7 Thrombocytopenia may be further exacerbated through the platelet EP3 receptor. In our case, thrombocytopenia improved temporarily on switching from epoprostenol to treprostinil and resolved after prostacyclin termination, indicating that drug-induced platelet aggregation may also have contributed to thrombocytopenia.
One study reported ITP-induced thrombocytopenia in some cases with PAH. 1 Oral prednisolone, intravenous immunoglobulin, or splenectomy were found to yield unsatisfactory results. 1 In our case, we made a diagnosis of ITP based on elevated PAIgG levels and bone marrow pathology. As described previously, prednisolone administration was not practical. However, TRA increased the platelet count. Our patient is currently on immunomodulators and prednisolone as a part of post-lung transplantation therapy. Hence, it is impossible to rule out ITP as a reason for her thrombocytopenia.
In conclusion, several factors can induce thrombocytopenia in patients with PAH. In our case, PAH etiology, right heart failure, drugs, and ITP may have contributed to severe thrombocytopenia. Considering that the patient’s platelet count returned to normal following lung transplantation and the normalization of pulmonary hemodynamics, it may be inferred that thrombocytopenia may mostly resolve with a new pair of lungs with normal vasculature and reduced RV afterload. Improving thrombocytopenia, even temporarily, is crucial as a bridge to transplantation. A comprehensive approach to cover all possible etiologies for thrombocytopenia may be required in patients with PAH awaiting transplantation.
Footnotes
Acknowledgements
The authors cordially thank all members of the lung transplantation team of Okayama University. Additionally, we also thank Dr. Seiichiro Sugimoto and Mrs. Tomoyo Okawa to support us for her surgical aftercare, Dr. Shun Sato to accompany her when she moved to Okayama University for lung transplantation and Mr. Takeshi Inagaki for dedicated rehabilitation in the pre and post-operative periods.
Authors’ contribution
TK, AS, JT, and NT constructed an idea for the manuscript. TK and AS took responsibility in the construction of the whole manuscript. Kohei T took responsibility in the pathological part. JT, NT, TS, SS, TO and Koichiro T reviewed the article before submission for its intellectual content.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Ethical approval
We got written informed consent for patient information and images to be published which was approved by the Ethical Committee of Chiba university Hospital (No. 826).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Guarantor
Takayuki Kobayashi.
ORCID iD
Ayako Shigeta https://orcid.org/0000-0002-9289-2901 Nobuhiro Tanabe https://orcid.org/0000-0001-5593-9128 Seiichiro Sakao ![]()
