Abstract
For pediatric pulmonary arterial hypertension (PAH) patients treated with parenteral prostanoids, response predictors, and the dose–effect relationship are ill defined. We determined the following: (1) which pulmonary vascular hemodynamic variable, after initiating prostanoids, best correlates with a significant clinical response; (2) the time interval after treatment when if no pulmonary hemodynamic improvement has occurred, none is ever likely to; and (3) the relationship between the prostanoid dose and its hemodynamic effects. This is a retrospective cohort study of 31 pediatric patients with Group 1 PAH treated with parenteral prostanoids. We found the following: (1) A fall in mean pulmonary arterial pressure (mPAP) of ≥25% predicted freedom from adverse clinical events with 80.7% accuracy and was also associated with improved functional class. (2) Thirty-three percent of patients who avoided an adverse clinical event demonstrated a ≥25% reduction in mPAP after 1 year of treatment, and 65% by 2 years. (3) Lower mPAP was seldom seen with doses of epoprostenol >60 ng/kg/min (100 ng/kg/min for treprostinil). Cardiac index was positively correlated with the dose of epoprostenol but not treprostinil; cardiac index >4 l/min/m2 was seen at modest as well as high doses. We conclude that a ≥25% fall in mPAP on prostanoids indicates a positive clinical response which, if validated in other studies, may be useful for patient management or clinical trials. Some patients take more than 2 years for this change. Exceptionally high doses were generally not more effective than lower, although we could not determine whether lower doses would have been as effective.
Introduction
Prostacyclin analogues (prostanoids), as used to treat pulmonary hypertension (PH), are unusual drugs. Most medications are given in fixed doses, or dose adjustments are dictated by plasma levels or easily measured effects (e.g. intravenous pressors). Efficacy (or lack thereof) is often quickly apparent. Parenteral prostanoids, on the other hand, have a wide dosing range and adjustments are driven by relatively soft criteria. Pediatric PH guidelines suggest epoprostenol and treprostinil doses of ∼50–80 ng/kg/min, 1 but published reports and anecdotal observations indicate that a much wider range is actually used. Mean doses as low as 24.7 ng/kg/min, 2 even after 3 years of treatment, and dose ranges as wide as 49–195 ng/kg/min 3 are reported for epoprostenol. The same is true for treprostinil (15–350 ng/kg/min). 4 Reported prostanoid doses in adults range from 54 to 190 ng/kg/min. 5
Prostanoid dosing is typically increased over time due to the development of tolerance, or hoping to attain a greater therapeutic effect, but the latter can be difficult to precisely determine. Since relatively few patients ever achieve normalization of pulmonary hemodynamics, dosing may be advanced beyond what is actually needed for the maximum response in (the vain) hope of garnering further improvement. The consequences of using higher than optimal doses are poorly defined, though excessive dosing can cause counterproductively high cardiac output. Indeed, Rich and McLaughlin found that 12 of 55 patients treated with parenteral prostanoids developed high cardiac output and benefited from a dose reduction. 6 A greater risk of bleeding may also attend supra-optimal doses due to the inhibitory effects of prostanoids on platelet function.7–9 Furthermore, prostanoid infusion is often accompanied by side effects such as headaches, jaw pain, diarrhea, and vomiting, which may be more likely as the dose increases.3,4,10–12
Prostanoids are also unusual because their hemodynamic and clinical effects take much longer to develop than most medications; many months may be needed to observe a clinically significant effect, and some patients never have an adequate response.3,10,13–17 Identifying those patients destined to have an inadequate response as soon as possible after initiation of prostanoids would be useful for decision-making with respect to the need for additional therapies, surgical interventions, or listing for lung transplant. How hemodynamic and clinical responses to chronic prostanoids relate to survival in adults has been investigated,13,18 but we are unaware of similar data in pediatric patients.
We reviewed our experience treating children with Group 1 pulmonary (arterial) hypertension (PAH) with parenteral epoprostenol and/or treprostinil to answer the following questions: (1) Is there an interval of time after prostanoid initiation after which if no clinically significant pulmonary hemodynamic improvement has occurred, none is likely to? (2) To answer the preceding question, we needed to define a hemodynamic response to prostanoid therapy that is associated with significant clinical improvement. We therefore asked: what change in pulmonary vascular hemodynamics after initiation of chronic prostanoids best correlates with a significant clinical response? (3) Can we uncover a dose–response effect of these medications with respect to pulmonary hemodynamics? Are higher doses of prostanoid associated with excessively high cardiac output in our patients?
Methods
Design
This is an observational retrospective study of pediatric patients with Group 1 PAH treated with a chronic continuous infusion, by intravenous or subcutaneous route, of epoprostenol and/or treprostinil. PAH was defined as mean pulmonary artery pressure ≥25 mmHg, PVRi > 3 Wood units·m2, and mean pulmonary capillary wedge pressure ≤15 mmHg. “Chronic continuous infusion” means that the patient was discharged to home on the medication. Thirty-five patients treated at Boston Children’s Hospital between April 1999 and April 2019 met these criteria; 31 patients had baseline and follow-up hemodynamic data and form the basis for this report. Data were collected from hospital and Department of Cardiology electronic medical records. The end of follow-up was defined as date of death or lung transplant for patients who experienced these events, or as the date of the most recent clinic follow-up (as of April 2019) for all others. This study was conducted in accordance with the amended declaration of Helsinki. Approval for this study was obtained from Boston Children’s Hospital Institutional Review Board (IRB-P00030647), which exempted the need for informed consent from the subjects.
Hemodynamic measurement
Hemodynamic data were obtained from cardiac catheterizations (cardiac cath) that were performed as clinically indicated; no tests or procedures were performed for research purposes. Patients underwent cardiac catheterization under either conscious sedation or general anesthesia. Baseline hemodynamics from the cardiac cath performed closest to but prior to the initiation of prostanoids were used in analysis. One patient’s baseline cath was done approximately 4 years prior to prostanoids, but echocardiograms performed at that cath and prior to prostanoid initiation suggest suprasystemic right ventricular pressures and there had been no relevant clinical events (see next section) in the interim. Hemodynamic variables measured include mean pulmonary arterial pressure (mPAP), right atrial pressure, pulmonary capillary wedge pressure, and cardiac index (C.I.). C.I. was measured via thermodilution or Fick’s principle with assumed oxygen consumption. Body surface area-indexed pulmonary vascular resistance (PVRi), indexed systemic vascular resistance (SVRi), and the ratio of PVRi to SVRi (Rp/Rs) were calculated.
Endpoints
We wished to determine (1) time for significant hemodynamic response after initiation of therapy and (2) the percentage of patients with a significant hemodynamic response. To do this, we first defined a significant clinical response as absence of any of the following adverse events (AEs) during follow-up after initiating prostanoid therapy: balloon atrial septostomy (BAS), rescue using an extracorporeal membrane oxygenator, reverse Potts shunt, listing for lung transplant, and death without lung transplant. By using Classification and Regression Tree Analysis (CART), we identified the maximum change, relative to baseline, of each of three pulmonary hemodynamic variables: mPAP, PVRi, Rp/Rs that best discriminated patients with an AE from those with the absence of any AE. We used this hemodynamic indicator to define the outcome of “clinically useful” prostanoid therapy. An additional endpoint was World Health Organization assessments of functional class (FC), made at the time of baseline and at the most recent follow-up clinic visit.
Statistical methods
Patient characteristics and targeted pulmonary hypertension medications.
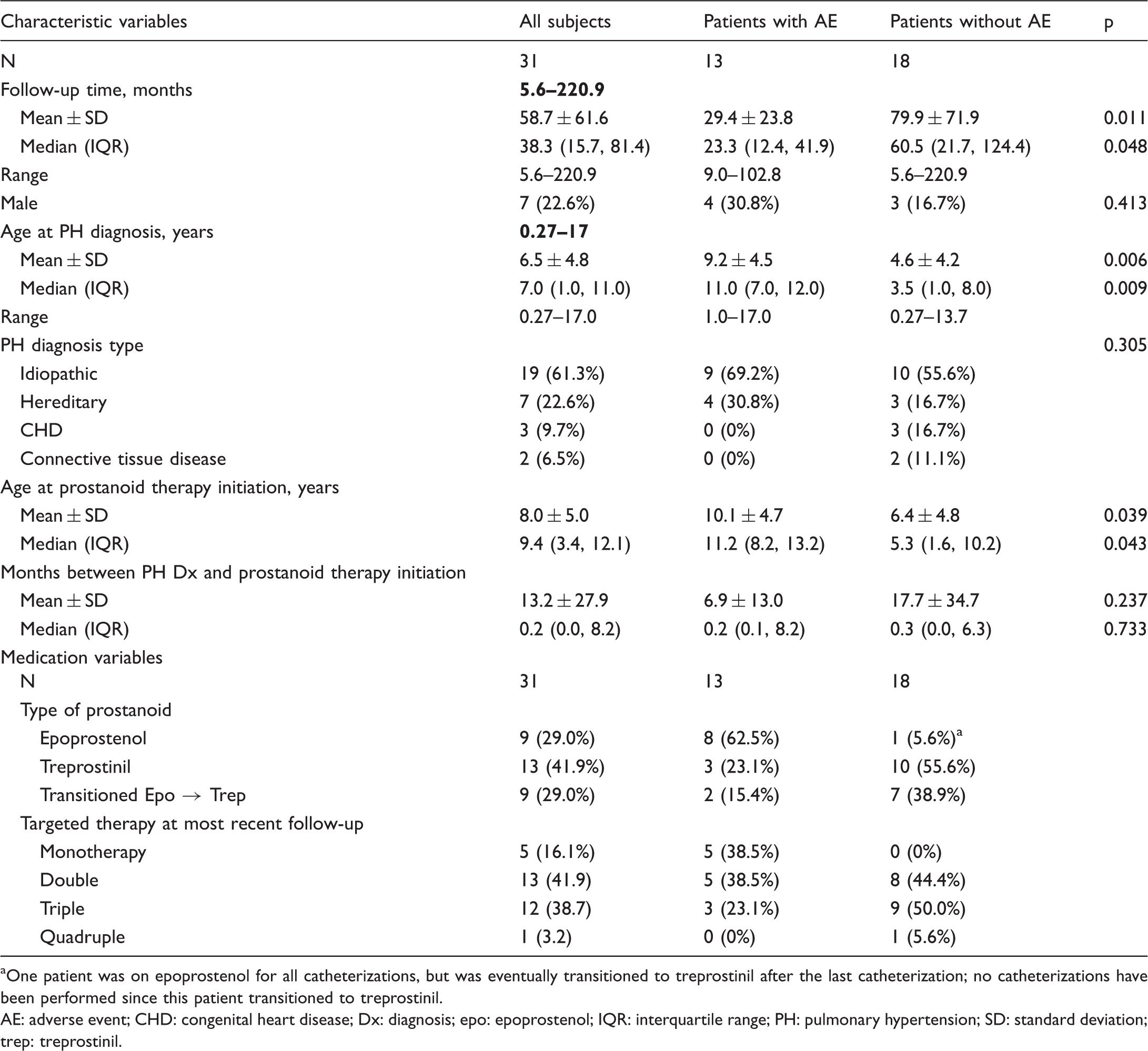
One patient was on epoprostenol for all catheterizations, but was eventually transitioned to treprostinil after the last catheterization; no catheterizations have been performed since this patient transitioned to treprostinil.
AE: adverse event; CHD: congenital heart disease; Dx: diagnosis; epo: epoprostenol; IQR: interquartile range; PH: pulmonary hypertension; SD: standard deviation; trep: treprostinil.
Targeted medication use
The targeted PH medications used, and their doses, varied according to era (e.g. early-on phosphodiesterase inhibitors were not available), severity of illness, practice in the PH community (e.g. the recent increased use of multiple medications), and other factors. Prostanoids were generally reserved for patients having insufficient response to oral medications, or patients presenting with FC III or IV symptoms, significant right ventricular dysfunction, and/or right ventricular pressure ≥ systemic. Generally, the dose was slowly increased with a goal of ∼75–100 ng/kg/min for epoprostenol and ∼125–150 ng/kg/min for treprostinil, with systemic hypotension and side-effects limiting the rate of escalation and maximum dose. After reaching a plateau dose, the dose was increased with worsening of PH-related symptoms and to account for somatic growth. The prostanoid dose was generally not decreased unless it was thought that the patient could be transitioned off that medication. Parenteral prostanoids were started as: sole initial therapy (n = 5); initial therapy along with other targeted medications (n = 8); add-on to oral targeted monotherapy (n = 16); add-on to two or more oral targeted therapies (n = 2).
Results
Baseline hemodynamics and functional class. a .
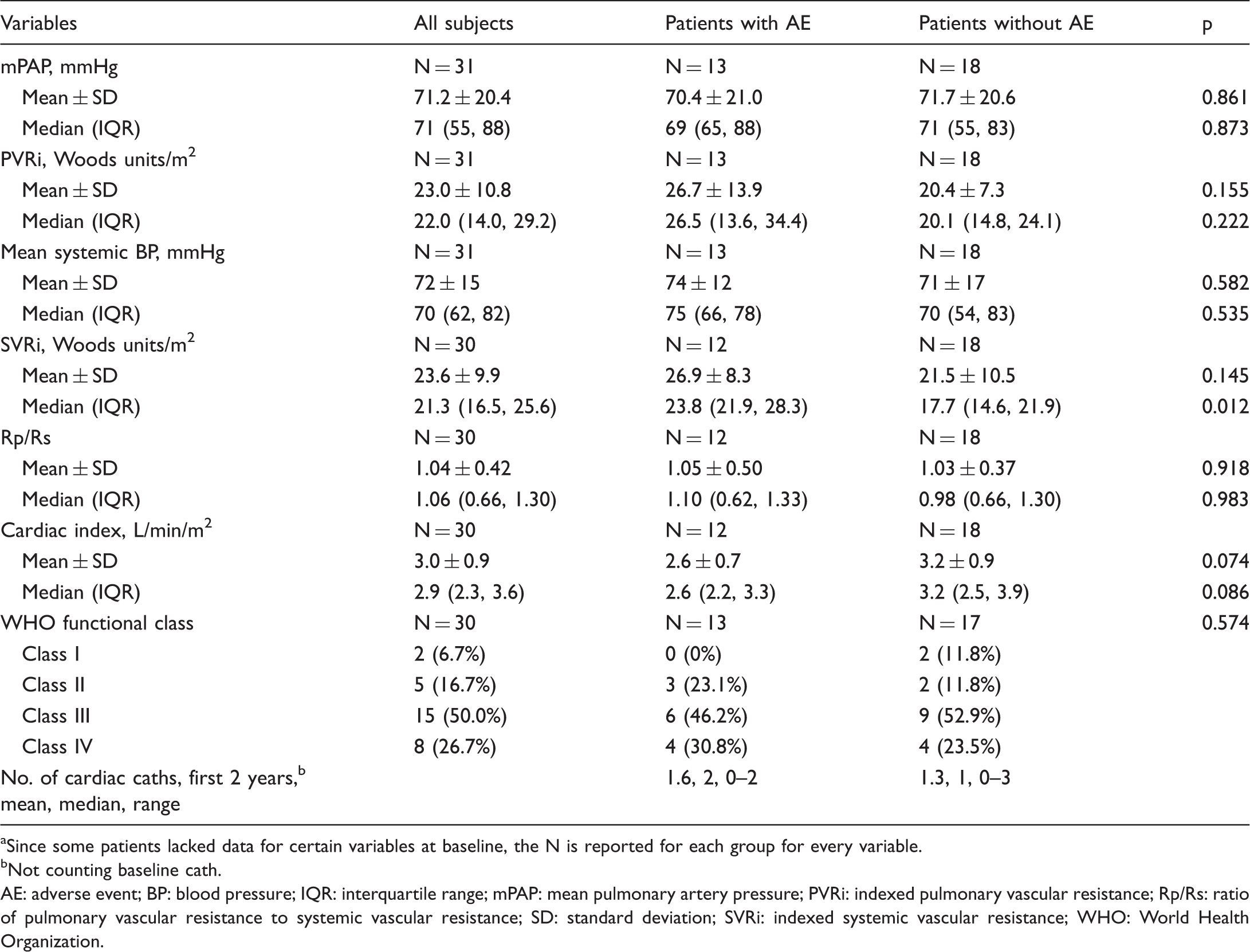
Since some patients lacked data for certain variables at baseline, the N is reported for each group for every variable.
Not counting baseline cath.
AE: adverse event; BP: blood pressure; IQR: interquartile range; mPAP: mean pulmonary artery pressure; PVRi: indexed pulmonary vascular resistance; Rp/Rs: ratio of pulmonary vascular resistance to systemic vascular resistance; SD: standard deviation; SVRi: indexed systemic vascular resistance; WHO: World Health Organization.
CART analysis of changes in pulmonary hemodynamic variables associated with the absence of an AE (N = 31 patients, 131 caths).
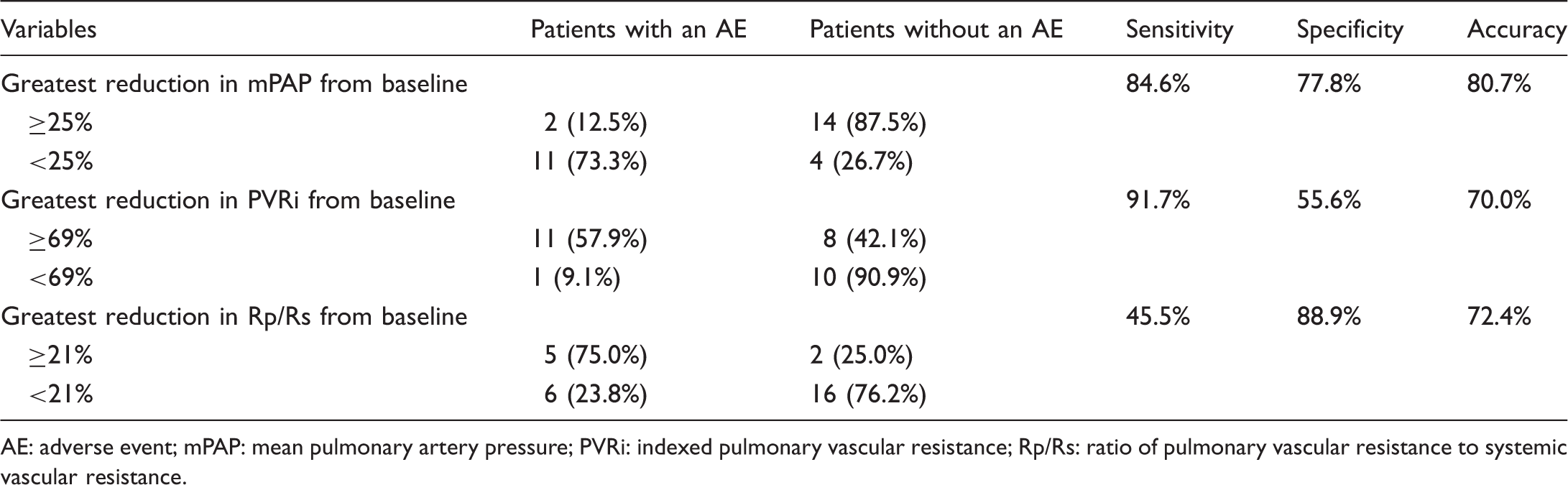
AE: adverse event; mPAP: mean pulmonary artery pressure; PVRi: indexed pulmonary vascular resistance; Rp/Rs: ratio of pulmonary vascular resistance to systemic vascular resistance.
CART analysis identifying the optimal threshold for mPAP reduction associated with achieving a functional class of I/II at the most recent follow-up (N = 29 a patients).
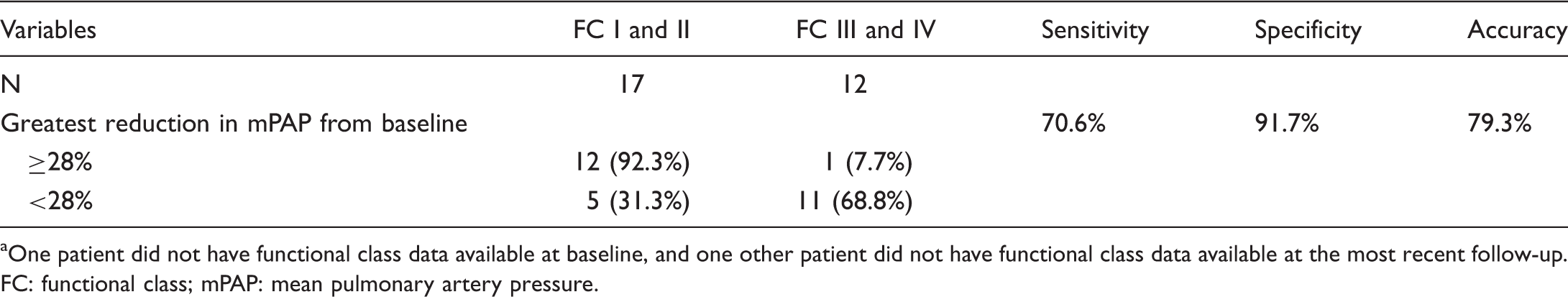
One patient did not have functional class data available at baseline, and one other patient did not have functional class data available at the most recent follow-up.
FC: functional class; mPAP: mean pulmonary artery pressure.
Fig. 1 displays the KM estimate of the time to first achieving an mPAP reduction of ≥25%. For those who did not achieve this level of reduction, time is censored at last cath date. Of the 18 patients without an AE, half had a reduction of mPAP by at least 25% by 17.2 months of treatment. By 2 years, 65% of patients (95% CI, 43%–87%) without an AE had a reduction of mPAP of at least 25%. Among the 18 patients with an AE, only 2 achieved a reduction in mPAP of at least 25%, with the KM estimate reaching only 20%. We also related this reduction in mPAP to doses of prostanoids. Table 5 displays the average dose level at the first cath indicating an mPAP reduction of ≥25%; this was 28.5 ± 10.0 ng/kg/min for epoprostenol and 88.1 ± 18.9 ng/kg/min for treprostinil. Among the 15 patients who never achieved a 25% reduction, the doses in Table 5 are the highest doses given during follow-up. Given the large difference between the epoprostenol and treprostinil doses, we sought to identify any differences between the times to first follow-up cath for patients receiving each form of prostanoid. The two groups were comparable: for epoprostenol, the mean time to first follow-up cath was 14.1 months (median 9 months, range 3.5–81.4 months); for treprostinil, the time was 11.1 months (median of 6.1 months; range 3–54.9 months).
Time to first reduction in mPAP ≥ 25% (N = 31, 16 reductions; 131 caths). Dose of prostanoid at first mPAP drop ≥25% (ng/kg/min), and maximum dose of prostanoid for those without a ≥25% decrease. One patient did not have dose data available at the time of their first ≥ 25% reduction in mPAP. IQR: interquartile range; mPAP: mean pulmonary artery pressure; SD: standard deviation.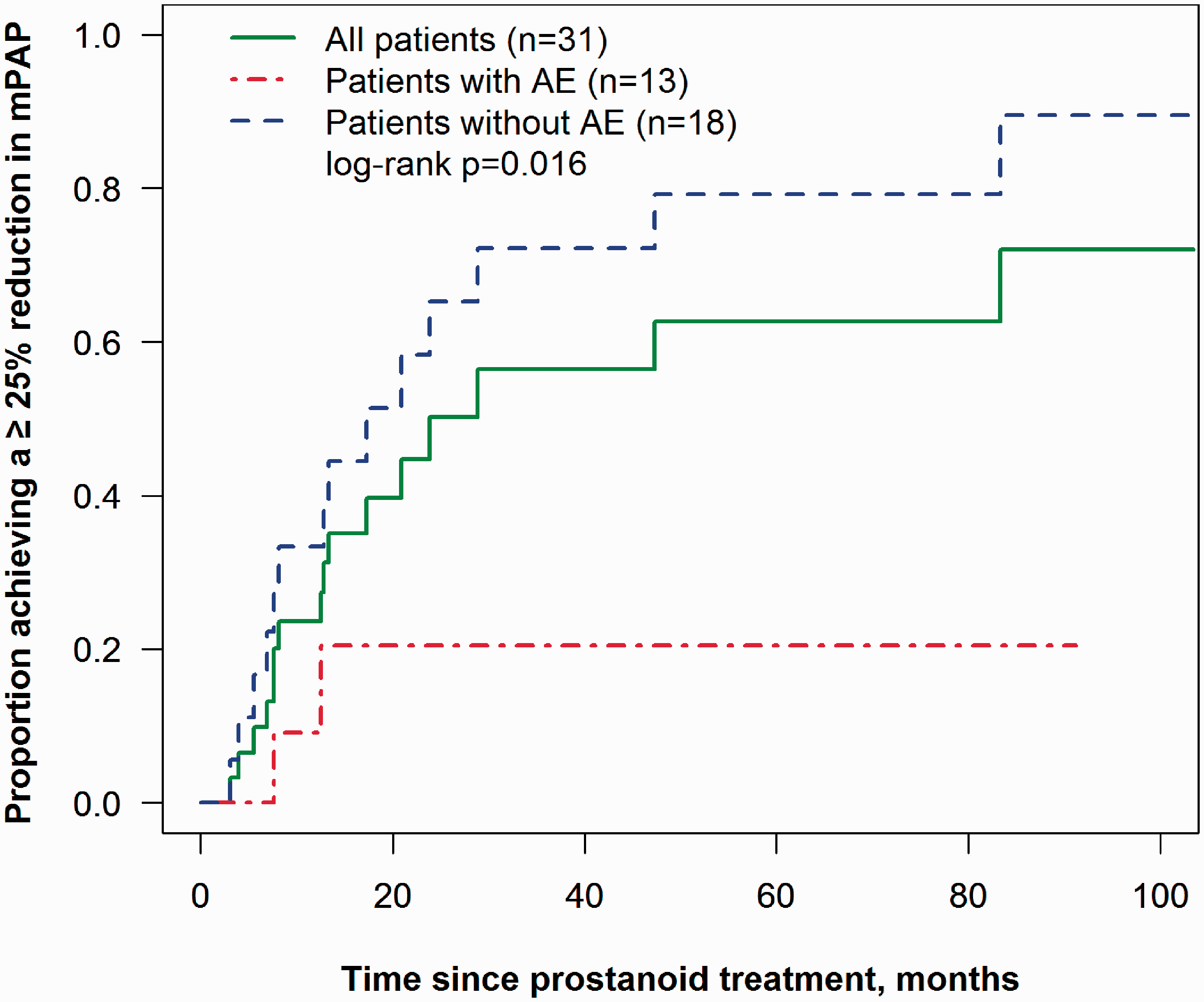
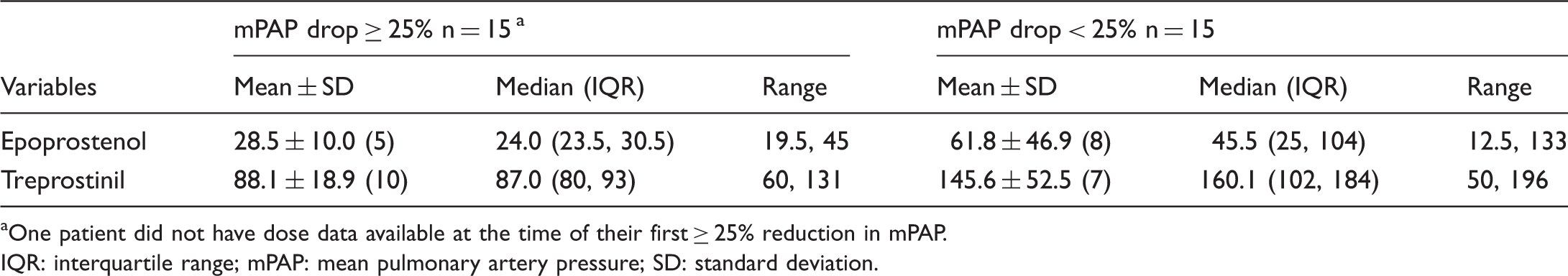
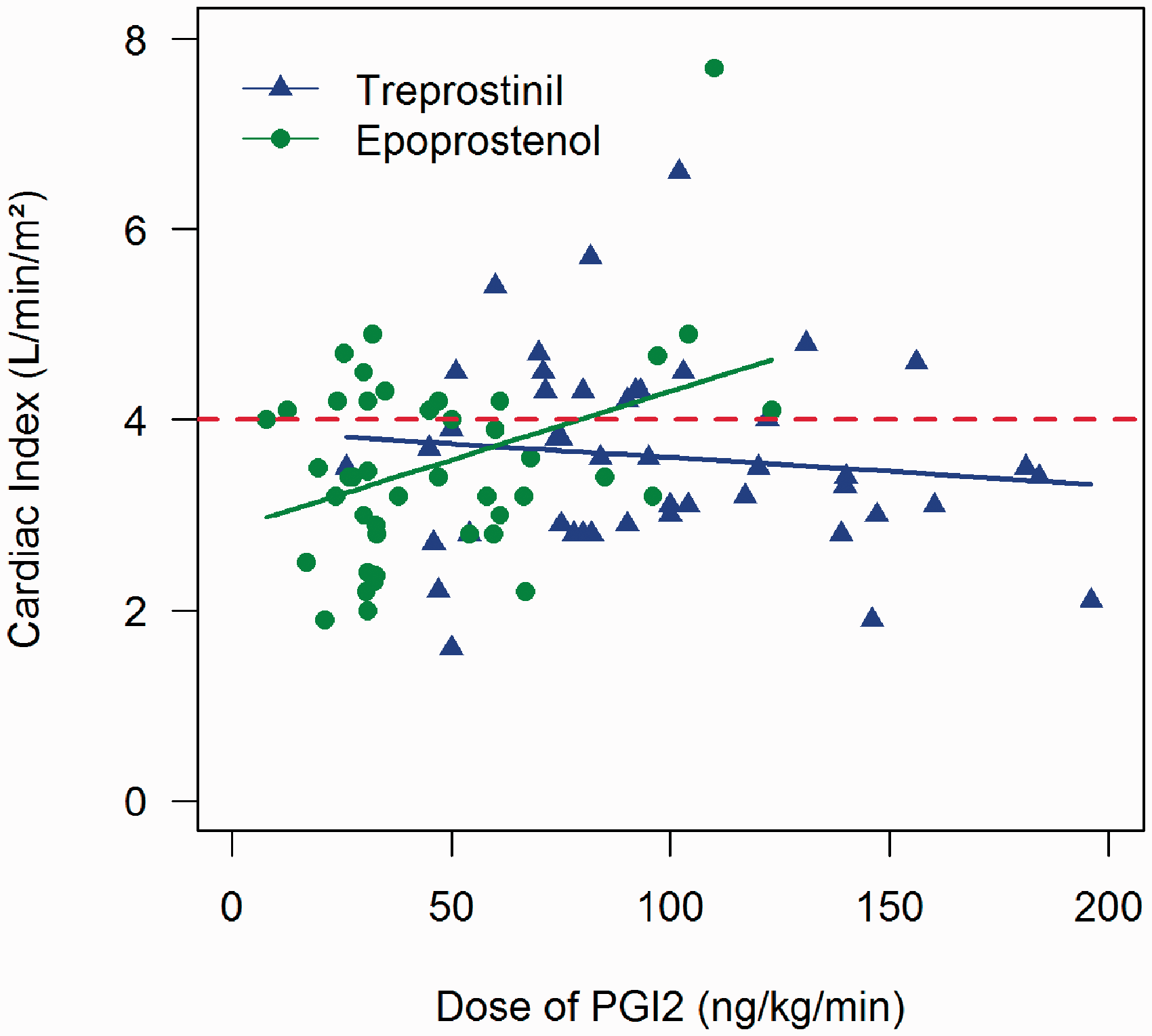
Fig. 2 displays the relationship between doses of prostanoid and the percentage fall in mPAP (relative to baseline) on that dose. Doses of epoprostenol greater than approximately 60 ng/kg/min (∼100 ng/kg/min for treprostinil) were generally not associated with lower mPAP. For C.I. versus prostanoid dose analysis, there were 29 patients with 88 observations; epoprostenol = 14 patients with 43 observations and treprostinil = 20 patients with 45 observations. (The discrepancy in patient numbers is because some patients transitioned type of prostanoid, and two patients had only one follow-up cath, without CI data.) Analyses of C.I. as a function of dose revealed a significant interaction (p = 0.037) between drug and dose: with treprostinil, there is no association between C.I. and dose (p = 0.431); with epoprostenol, there is an increase in C.I. of 0.14 L/min/m2 (95% confidence interval 0.02–0.26) with each additional 10 ng/kg/min (p = 0.011). But, as revealed in Fig. 3, C.I. >4 l/min/m2 was observed with modest as well as higher doses of prostanoid.
Dose of prostanoid vs. % reduction in mPAP (N = 31, 90 caths). Data points reflect only the up-titrating/stable doses; no caths where the prostanoid was being down-titrated were included. Negative values on the y-axis indicate an increase in mPAP. Dose of prostanoid vs. cardiac index. Circles = epoprostenol (p = 0.011); triangles = treprostinil (p = 0.431).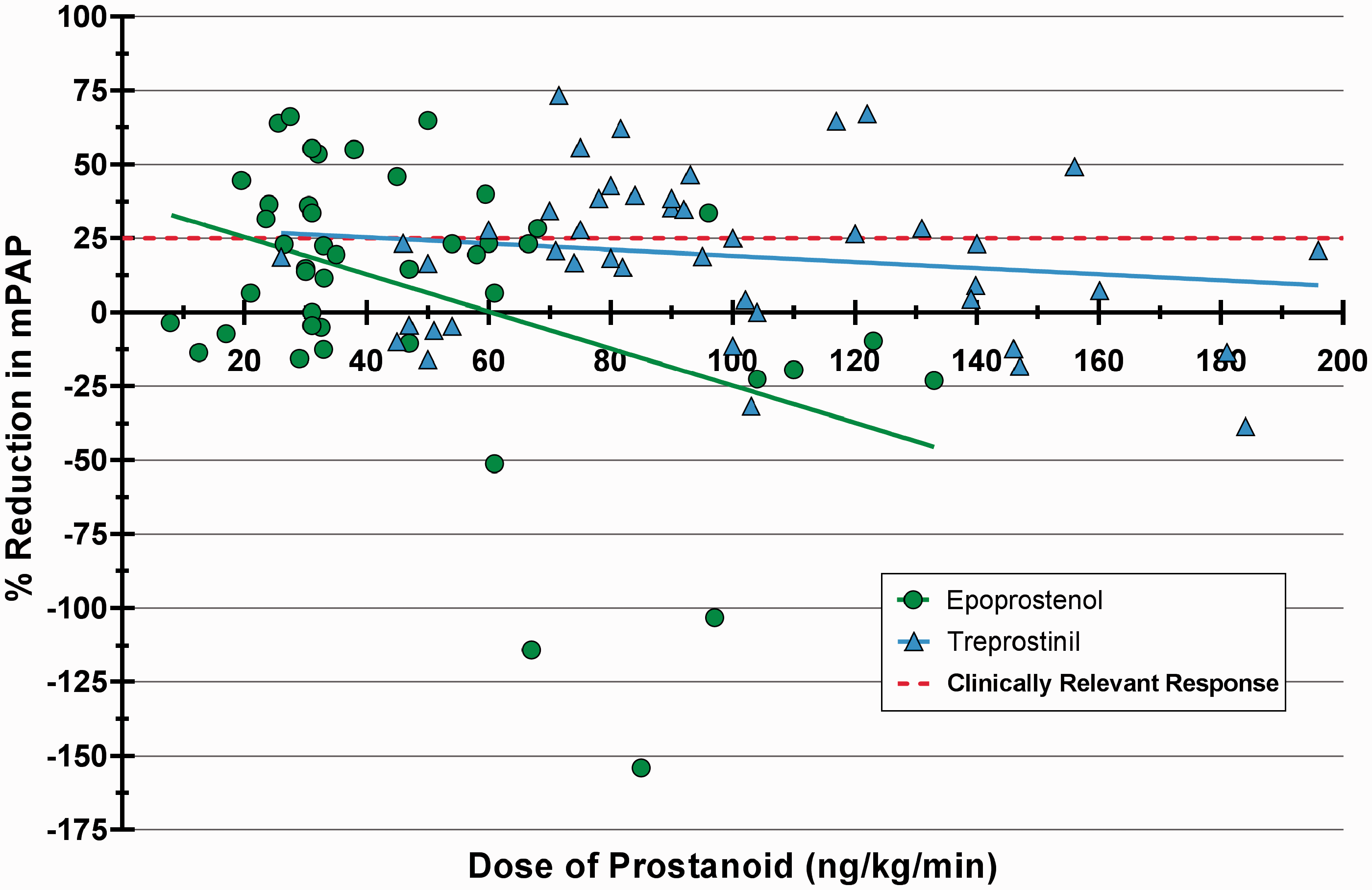
Discussion
Our goals were to better understand chronic parenteral prostanoid therapy with respect to time-to-effect and dose–response, and to determine what change in pulmonary vascular hemodynamic variables with chronic prostanoid therapy is best associated with a favorable clinical response. We could conceivably have chosen another variable, or variables, known to be associated with outcome (e.g. FC, brain natriuretic peptide, 6-minute walk distance, right ventricular failure) for our time-to-clinical effect determination. None, however, seemed suitable: e.g. we had no 6-minute walk data for our young patients, brain natriuretic peptide is quite variable, categorical variables would likely be less robust than continuous ones. We explored pulmonary hemodynamic variables because very high PVR and Rp/Rs, and low C.I. are known to associate with poor outcomes. 1 In short, we wanted to identify the variable that best discriminates patients with, versus those without, significant clinical improvement. Our major findings, starting with the last question and the most important finding, are as follows.
We determined which of three pulmonary vascular hemodynamic variables (maximum change, relative to baseline, in PVRi, mPAP, and Rp/Rs) and at what threshold, best correlated with the absence of AEs in our patients. A maximum fall in mPAP of ≥25% predicted freedom from AEs with 80.7% accuracy (84.6% sensitivity and 77.8% specificity), performance superior to maximum change in PVRi and Rp/Rs. Because FC is clinically important and a survival predictor, we also asked whether that fall in mPAP also correlates with good (I or II) versus poor (III and IV) FC. A very similar fall in mPAP (≥28%) correlated with FC I or II, with similar accuracy, better specificity, but lower sensitivity. While these data suggest that change in mPAP might serve as a useful prognosticator, their interpretation must be tempered by the following: There were relatively few follow-up cardiac caths (Table 2), and we may have missed maximal hemodynamic improvement in some patients, since hemodynamics may improve, but later deteriorate. But Siehr et al. 15 found that deterioration generally occurs after 2 years of prostanoid use. The number of caths in our AE patients in the first 2 years of life was, if anything, greater than the number for the non-AE patients (Table 2); if maximal hemodynamic improvement was missed, it was more likely to have been missed in those who showed the most improvement. Furthermore, one might expect that randomly not detecting maximum improvement in hemodynamics would introduce noise into the data, and lessen the likelihood of statistically significant findings. But the CART analysis showed a robust association between maximum improvement and clinical outcome, and in the direction that one would expect. Our data are consistent with studies of parenteral prostanoids in adults, where absence of a fall in PVR of >30% 13 or mPAP, 22 or persistence of NYHA class III or IV after prostanoid therapy was correlated with decreased survival.13,22 We are unaware of any similar reports of hemodynamic predictors in pediatric patients, although one showed that WHO-FC, NT-proBNP, and TAPSE predicted transplant-free survival in pediatric PAH patients. 23 Substantially more patients, at several centers, will be required to confirm this finding, but we suggest that the percentage change in mPAP is worth further exploration as it may prove useful for evaluating efficacy of pediatric parenteral prostanoid therapy, both for individuals and populations of patients.
Regarding the time course of improvement with prostanoids, our estimates indicate that 33% of patients who will avoid an AE will demonstrate a ≥25% reduction in mPAP after 1 year of treatment, 50% by 1.5 years, and 65% by 2 years (Fig. 1). This is slower than that described by Barst et al., who found that 79% of their pediatric patients had a ≥25% fall in mPAP by the end of the first year of treatment with prostacyclin (their table 7). 10 But our caths were performed at infrequent intervals, and some patients had certainly achieved a 25% fall in mPAP well before being revealed by cath. It is also possible that Barst’s group increased the dose of prostanoid more rapidly than did we. Our findings are consistent with general clinical experience, and the (as yet unproven) notion that pulmonary vascular remodeling, rather than reduction in smooth muscle activation, largely accounts for prostanoids’ favorable effect. Our experience—though it presumably overestimates the time-to-effect—indicates that early prognostication with respect to ultimate outcomes of pediatric patients cannot be provided by change in mPAP, since 35% of patients had a significant hemodynamic response only after 2 years of therapy.
Finally, we found that the average dose of prostanoid required to achieve an initial clinically relevant response was 28.5 ± 10.0 ng/kg/min for epoprostenol and 88.1 ± 18.9 ng/kg/min for treprostinil. Although epoprostenol is more potent than treprostinil for PH therapy, this ratio of doses is greater than previously reported (treprostinil dosing generally being 130%–200% that of epoprostenol).24–26 The large dose differential could have been due to differences in the timing of follow-up catheterizations: if patients treated with epoprostenol had caths earlier than treprostinil patients, their 25% mPAP reduction could have been recorded earlier, before any further increase in the dose of epoprostenol. However, the average times to first follow-up cath were comparable for the two groups, making this unlikely. Also, this relatively large differential may be a reflection of the fact that the perceived need for dose adjustments is somewhat subjective, and adjustments are often made in response to relatively “soft” information. In any case, these data should not be interpreted as suggesting that treprostinil doses this much in excess of epoprostenol doses is typical or should be taken as a guide to dosing.
Increasing epoprostenol beyond ∼60 ng/kg/min and treprostinil beyond 100 ng/kg/min was only inconsistently associated with a further fall in mPAP (Fig. 2). Rich and McLaughlin reported 12 patients with idiopathic PH on chronic epoprostenol (mean 98 ± 61 ng/kg/min) with a high cardiac output (mean 10.1 ± 2.3 l/min). They weaned the epoprostenol to a mean dose of 60 ± 50 ng/kg/min, with a fall in cardiac output to 7.7 ± 1.4 l/min without change in mPAP; no patient required a dose adjustment for 3 months, their FC remained stable, and patients reported fewer prostanoid side effects. The authors speculated that reducing prostacyclin dose brought cardiac output back into normal range, favorably reducing neurohormonal activation and therefore potentially limiting tolerance development while emphasizing, in effect, that it is possible to have too much of a good thing. 6 Our data lend little support for the notion that relatively high doses are usually accompanied by greater effect, but we recognize that it is possible that lower doses would have been even less effective. This possibility deserves strong consideration given that prostanoid doses often need to be escalated because of the development of tolerance.6,27 In our patient cohort, C.I. was often > 4 l/min/m2, but these higher outputs occurred with moderate as well as high dose prostanoids (Fig. 3).
Limitations
1. Our patient population was small, and from a single center, and therefore our findings would benefit from validation using a multicenter cohort. 2. The relative infrequency and lack of uniformity with respect to the timing of follow-up cardiac caths places wide confidence limits around our data regarding timing of response. In fact, our design must overestimate the time-to-hemodynamic-effect of parental prostanoids. And, as noted above, our findings with respective to the possible predictive power of hemodynamic changes need to be interpreted with this in mind. 3. Our dose–response data were not from individual patients, serially measured (at different doses) and hence is not a true dose–response determination. Possibly, some patients on very high prostanoid doses would have even higher mPAP on lower doses. And because patients can experience prostanoid tachyphylaxis, requiring higher than previous doses, some patients may eventually require unusually high doses. 4. Because the dosing weights used to calculate the dose of prostanoid were only periodically updated, the dosages used for our analyses were likely overestimated at some time points. 5. We realize that the non-prostanoid targeted PH medications confound interpretation of the hemodynamic and clinical changes observed in our patients. However, given the limited number of patients and large number of non-prostanoid medication permutations, it is not possible to investigate these interactions.
Conclusions
1. The percentage fall in mPAP after initiation of prostanoid therapy may serve as an indicator of clinical efficacy (avoiding AEs) and function improvement in pediatric PAH patients treated with chronic parental prostanoids. We found that a decline of ≥25% is associated with a much lower risk of AEs than a lesser fall in mPAP. If validated by other studies, this may prove useful for patient management and clinical trials. 2. While about one-half of pediatric patients develop a clinically significant effect from parenteral prostanoids by 1.5 years of therapy, others may take 2 years or more to demonstrate such an effect. 3. Our data indicate that very high doses of parenteral prostanoids are not associated with a large favorable change in mPAP in most patients, but the utility of such doses remains unclear.
Footnotes
Authors’ contribution
JBT, TJK, LAS: study design, analysis, interpretation, and writing; JEM: study design, analysis, interpretation; ML: analysis; and MPM: study analysis and interpretation.
Acknowledgements
We acknowledge the expert assistance of Lynne Patkin in manuscript preparation.
Conflict of interest
MPM was site PI for industry sponsored trials from United Therapeutics, Actelion, Bayer, and GSK and a consultant for Actelion. The rest of the author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Guarantor
TJK takes responsibility for the content of the manuscript, including the data and analysis.
