Abstract
Dynamic contrast–enhanced (DCE) time-resolved magnetic resonance (MR) imaging is a technique whereby the passage of an intravenous contrast bolus can be tracked through the pulmonary vascular system. The aim of this study was to investigate the prognostic significance of DCE-MR pulmonary blood transit times in patients with pulmonary arterial hypertension (PAH). Seventy-nine patients diagnosed with PAH underwent pulmonary DCE imaging at 1.5 T using a time-resolved three-dimensional spoiled gradient echo sequence. The prognostic significance of two DCE parameters, full width at half maximum (FWHM) of the first-pass clearance curve and pulmonary transit time (PTT), along with demographic and invasive catheter measurements, was evaluated by univariate and bivariate Cox proportional hazards regression and Kaplan-Meier analysis. DCE-MR transit times were most closely correlated with cardiac index (CI) and pulmonary vascular resistance index (PVRI) and were both found to be accurate for detecting reduced CI (FWHM area under the curve [AUC] at receiver operating characteristic analysis = 0.91 and PTT AUC = 0.92, respectively) and for detecting elevated PVRI (FWHM AUC = 0.88 and PTT AUC = 0.84, respectively). During the follow-up period, 25 patients died. Patients with longer measurements of FWHM (P = 0.0014) and PTT (P = 0.004) were associated with poor outcome at Kaplan-Meier analysis, and both parameters were strong predictors of adverse outcome from Cox proportional hazards analysis (P = 0.013 and 0.010, respectively). At bivariate analysis, DCE measurements predicted mortality independent of age, gender, and World Health Organization functional class; however, invasive hemodynamic indexes CI, PVRI, and DCE measurements were not independent of one another. In conclusion, DCE-MR transit times predict mortality in patients with PAH and are closely associated with clinical gold standards CI and PVRI.
Keywords
INTRODUCTION
Pulmonary arterial hypertension (PAH) is a condition associated with high morbidity and mortality.1,2 The increasing recognition of the need for rationalized therapy in patients with PAH has highlighted the need to develop prognostic markers that can aid the clinician in the assessment of disease severity at baseline and be used to follow up response to therapy. Hemodynamic and physiological measurements made at right heart catheter (RHC), such as mean right atrial pressure (mRAP), pulmonary vascular resistance index (PVRI), mixed venous oxygen saturation (mVO2), and cardiac index (CI) are the key prognostic indicators in PAH.3,4
Dynamic contrast–enhanced (DCE) time-resolved magnetic resonance (MR) imaging is a technique whereby an intravenous contrast bolus can be tracked through the cardiopulmonary vascular system in three dimensions with time.5–12 Several previous studies have assessed DCE-MR in patients with pulmonary hypertension,8,9,13–15 and significant correlations have been identified between time-resolved DCE-MR angiography and invasive prognostic indicators. However, the role of DCE-MR pulmonary vascular transit time (TT) measurements as prognostic markers in PAH has not yet been determined. The aim of this study was to investigate the utility of DCE-MR pulmonary vascular TTs for the prognostic assessment of patients with PAH.
METHODS
Consecutive patients between September 2009 and March 2011 diagnosed with PAH who underwent DCE-MR time-resolved imaging were included in this retrospective study. Fifteen age-matched patients with suspected pulmonary vascular disease found not to have pulmonary hypertension (mean pulmonary arterial pressure [mPAP] <25 mmHg) were also included. Patients with PAH were followed up to death or census on April 12, 2012. Patients were diagnosed following comprehensive assessment by a multi-disciplinary team. Treatment was standardized based on a National Commissioning Policy, with a phosphodiesterase type 5 inhibitor used as first-line monotherapy in World Health Organization (WHO) functional class III (unless a contraindication when an oral endothelin receptor antagonist is used) and intravenous prostanoid considered in WHO functional class IV. Patients showing no improvement on monotherapy were switched to an alternative monotherapy or escalated to a combination treatment with either a phosphodiesterase inhibitor and endothelin receptor antagonist or a phosphodiesterase inhibitor and prostanoid as recommended. This study was conducted following institutional review board approval; written informed consent was waived.
MR imaging
MR imaging was performed on a 1.5 T whole-body system (HDx, GE Healthcare, Milwaukee, WI) using a time-resolved three-dimensional (3D) spoiled gradient echo sequence with view sharing. 17 An 8-channel cardiac receiver array coil was used. The sequence parameters were as follows: echo time 1.1 ms, repetition time 2.5 ms, flip angle 30°, field of view 48 cm × 48 cm, in-plane parallel imaging ×2, in-plane resolution 200 × 80, bandwidth 250 kHz, slice thickness 10 mm, ~32 slices, 48 time points with an overall effective 3D frame rate of ~0.5 s. Images were acquired in a coronal orientation. Contrast injection of a 0.05 mL/kg patient weight dose of Gd-BT-D30A (Gadovist, Schering, Berlin) was injected at a rate of 4 mL/s, with the injection rate controlled using an activated pump injector (Spectris, MedRad) via the antecubital vein using an 18-G cannula, followed by a 20-mL saline flush.
Images were acquired at breath hold, with the same automated breathing instructions given to each patient. The subjects were coached on the breathing and breath-hold maneuver and followed the instructions “breathe in, breathe out, breathe in, and hold your breath” prior to image acquisition.
Image analysis
All image processing was performed on a GE advanced workstation using the proprietary “functool” software. Time-resolved DCE-MR images of the cardiopulmonary system were derived by the subtraction of the baseline (precontrast arrival) images from each of the subsequent 3D image time frames acquired during the contrast passage. Signal enhancement versus time curves were generated by the placement of regions of interest (ROIs) at the pulmonary artery (PA) and left atrium (LA; Fig. 1). These were matched to the size of the vessel. Image analysis was performed by a radiologist with 6 years' experience of clinical imaging (observer 1). A second observer analyzed 30 randomly selected cases for assessment of inter-observer variability. Observer 1 reanalyzed these 30 cases after an interval of 1 month to determine the intraob-server variability of DCE-MR indexes. The observers were both blinded to the clinical details and RHC data for image analysis.
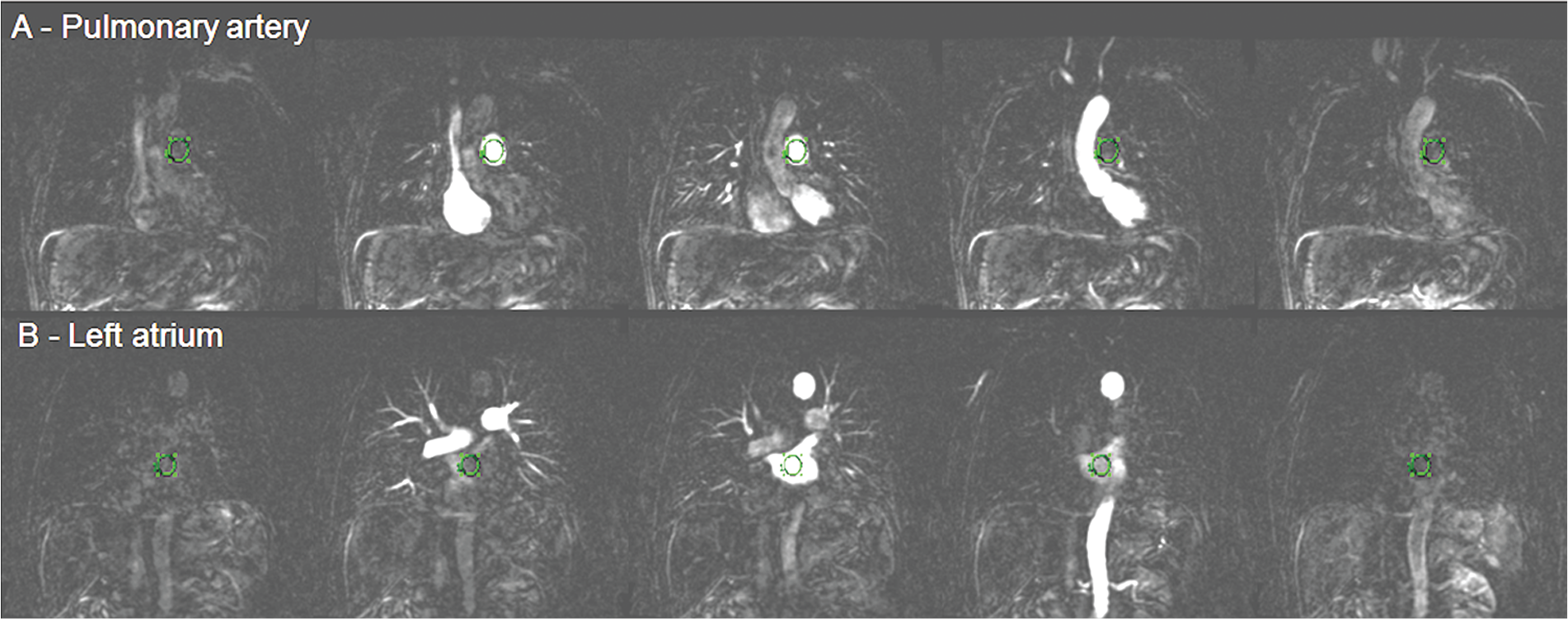
Dynamic contrast–enhanced subtracted time-resolved images showing the passage of contrast through the cardiopulmonary system. Coronal images with region of interests placed in the pulmonary artery (A) and in the left atrium (B) are shown.
Signal-time curves were generated from ROIs placed in the PA and LA (Fig. 2). The full width at half maximum (FWHM) of the bolus passage was defined as the width of the PA enhancement curve at half its maximum signal intensity. Pulmonary transit time (PTT) was defined as the time difference between the peak signal at the PA and the peak signal at the LA (PTT = time at peakLA – time at peakPA).5,15
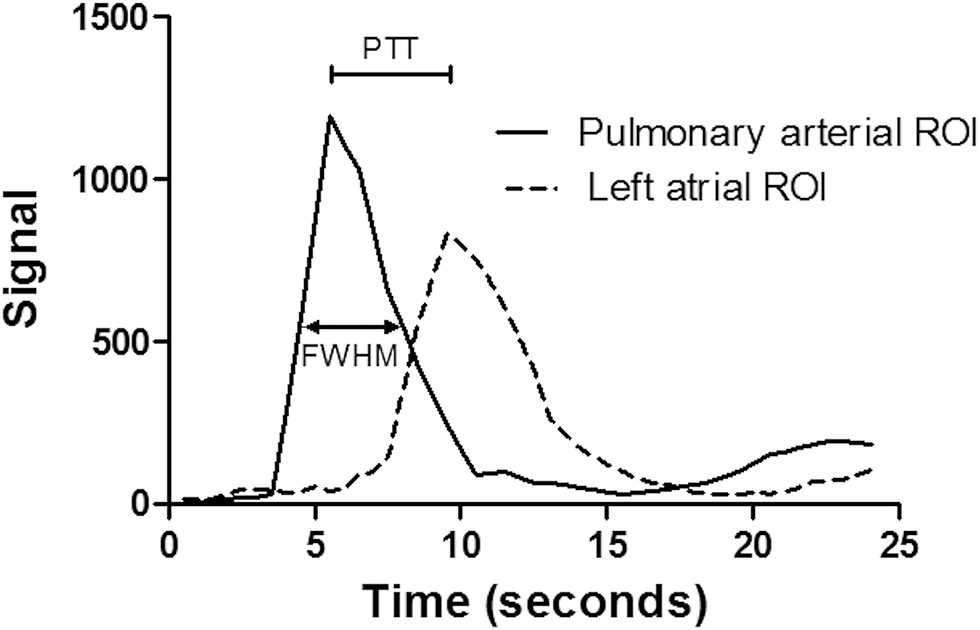
Signal-time curves are generated from regions of interest (ROIs) placed in the pulmonary artery and the left atrium. Pulmonary transit time (PTT) is calculated by subtracting the time of peak signal intensity on the pulmonary artery curve from the time of peak signal intensity on the left atrium. Full width at half maximum (FWHM) of the pulmonary artery is calculated by measuring the width of the pulmonary artery signal-time curve at half its maximum signal intensity.
Right heart catheter
RHC was performed via the internal jugular vein using a Swan-Ganz catheter. PAH was defined as mPAP ≥25 mmHg and pulmonary capillary wedge pressure (PCWP) <15 mmHg. Cardiac output (CO) was determined using the thermodilution technique. Pulmonary vascular resistance (PVR) was determined as follows: PVR = (mPAP – PCWP)/CO. CI is defined as CI = CO/body surface area. PVRI is defined as (mPAP – PCWP)/CI.
Statistics
Statistical comparisons of demographic information, catheter hemodynamics, and DCE-MR parameters were made between survivors and nonsurvivors using the unpaired t test for continuous data and the Fisher exact test for categorical data. Regression curve fitting was used to determine the best-fit relationship using linear, quadratic, exponential, logarithmic, and inverse-linear models. Hazard ratios and 95% confidence intervals were calculated using the Cox proportional hazards regression for potential predictors of mortality: age, male:female ratio, WHO functional class, mRAP, mPAP, mVO2, CO, CI, PVR, PVRI, PTT, and FWHM. Univariate parameters that returned a P value <0.2 were entered into the bivariate model. Kaplan-Meier survival curves were constructed with analysis performed using the log-rank test. Continuous variables were divided into two groups by the median value. Follow-up occurred at 6 months and overall mortality of the group was tested. A P value <0.05 was considered statistically significant. To perform and display the statistics, SPSS 19 (SPSS, Chicago, IL) and GraphPad Prism 5.03 (GraphPad, San Diego, CA) software were used.
RESULTS
Eighty-five patients were identified with PAH and had DCE-MR imaging and RHC within 48 hours. Two patients were excluded due to the nondiagnostic quality of images; this was due to significant breathing motion artifact. Four patients were excluded due to the presence of an intra-cardiac shunt identified at multidisciplinary assessment. Thus, 79 patients in total were included, comprising 35 patients with idiopathic PAH, 37 patients with PAH associated with connective tissue disease, and 7 patients with PAH associated with congenital heart disease. During the follow-up period, 25 patients with PAH died.
Group comparisons
The demographic RHC and DCE TT data are presented in Table 1. As expected, patients with PAH had significantly higher WHO functional class, mPAP, CI, PVRI, and mVO2 than control patients (mPAP < 25 mmHg). DCE measurements, FWHM (P = 0.001), and PTT (P = 0.001) were also significantly higher in patients with PAH when compared to the control subjects. Control subjects were not significantly different in terms of age or gender when compared to patients with PAH (P = 0.832 and 0.401, respectively).
Patient demographics, hemodynamics, and time-resolved three-dimensional magnetic resonance angiography contrast transit times for patients with pulmonary arterial hypertension (PAH), survivors, nonsurvivors, and control subjects (mean pulmonary arterial pressure <25 mmHg)
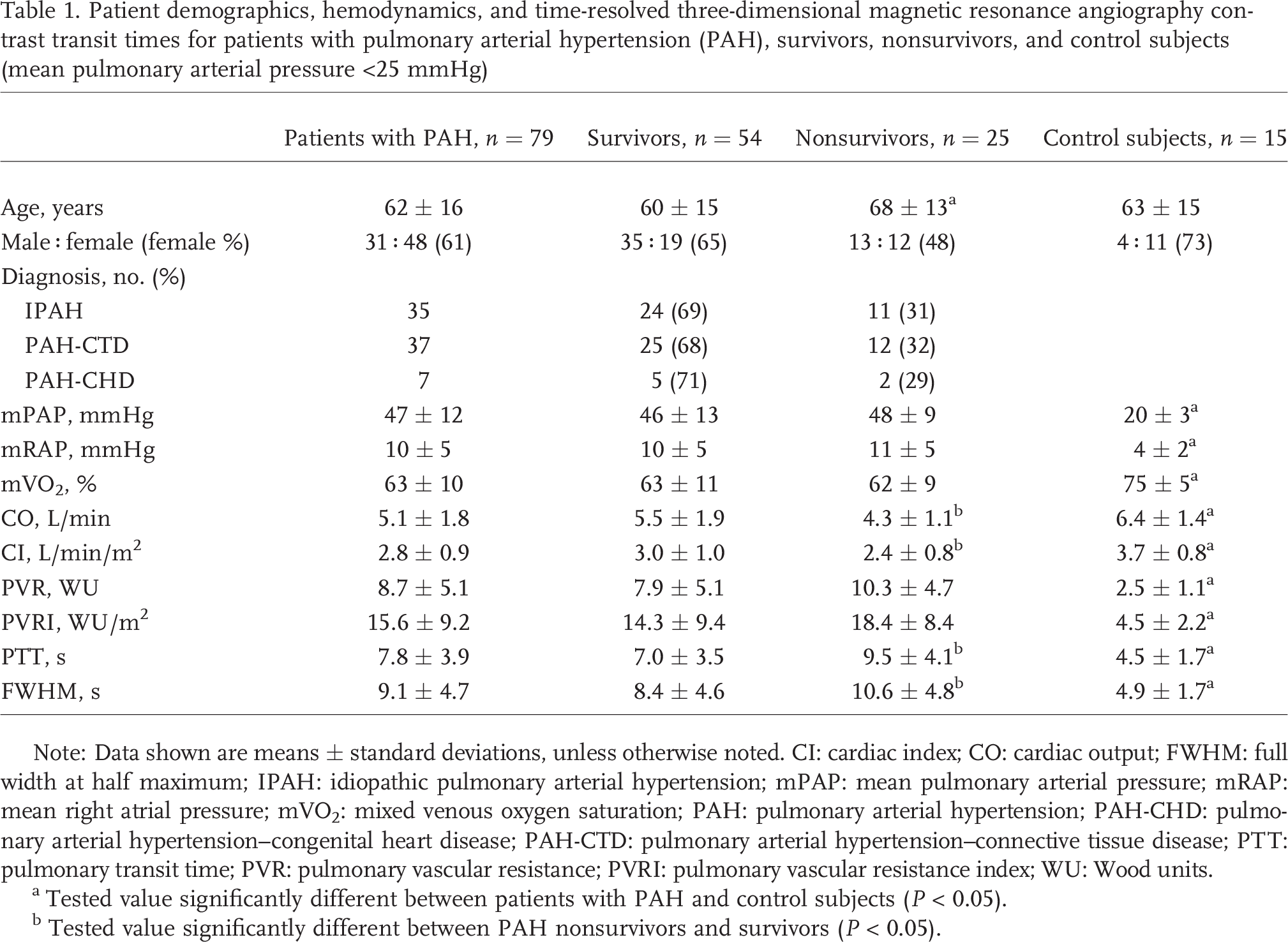
Note: Data shown are means ± standard deviations, unless otherwise noted. CI: cardiac index; CO: cardiac output; FWHM: full width at half maximum; IPAH: idiopathic pulmonary arterial hypertension; mPAP: mean pulmonary arterial pressure; mRAP: mean right atrial pressure; mVO2: mixed venous oxygen saturation; PAH: pulmonary arterial hypertension; PAH-CHD: pulmonary arterial hypertension-congenital heart disease; PAH-CTD: pulmonary arterial hypertension-connective tissue disease; PTT: pulmonary transit time; PVR: pulmonary vascular resistance; PVRI: pulmonary vascular resistance index; WU: Wood units.
Tested value significantly different between patients with PAH and control subjects (P < 0.05).
Tested value significantly different between PAH nonsurvivors and survivors (P < 0.05).
Correlations and receiver operating characteristic (ROC) analysis
The strongest bivariate relationships were between 1/FWHM and CI (inverse-linear: r2 = 0.61, P < 0.0001) and 1/PTT and CI (inverse-linear: r2 = 0.58, P < 0.0001). The next-strongest bivariate relationships were found between FWHM and PVRI (linear: r2 = 0.56, P < 0.0001) and PTT and PVRI (linear: r2 = 0.52, P < 0.0001). FWHM (linear: r2 = 0.17, P = 0.0001) and PTT (linear: r2 = 0.14, P = 0.001) demonstrated relatively weaker correlations with mPAP (Table 2).
Relationships between invasive right heart catheter measurements and dynamic contrast–enhanced time-resolved magnetic resonance data
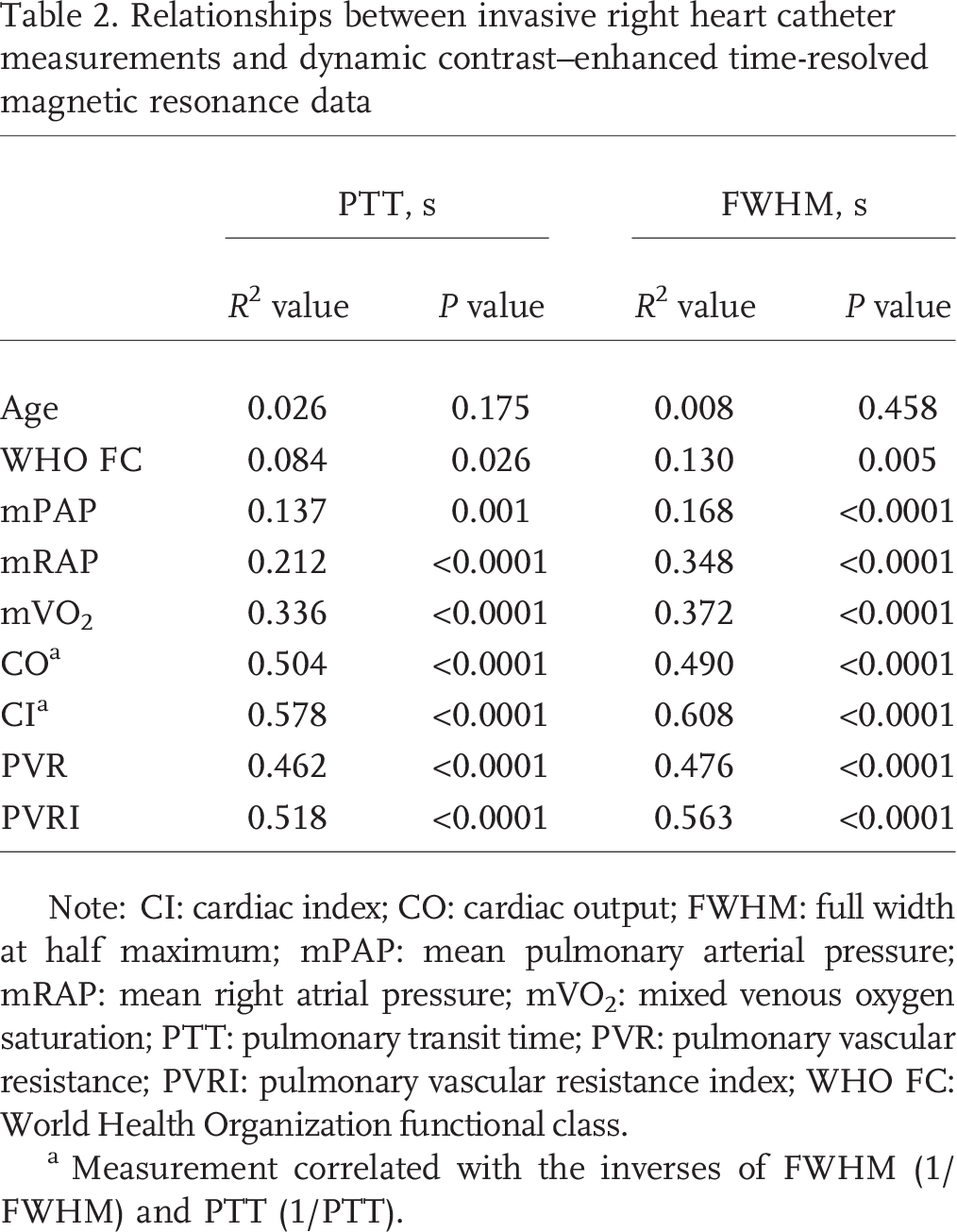
Note: CI: cardiac index; CO: cardiac output; FWHM: full width at half maximum; mPAP: mean pulmonary arterial pressure; mRAP: mean right atrial pressure; mVO2: mixed venous oxygen saturation; PTT: pulmonary transit time; PVR: pulmonary vascular resistance; PVRI: pulmonary vascular resistance index; WHO FC: World Health Organization functional class.
Measurement correlated with the inverses of FWHM (1/FWHM) and PTT (1/PTT).
FWHM and PTT both demonstrated reasonable diagnostic accuracy for the detection of PAH (area under the curve [AUC] 0.80 and 0.79, respectively). Higher accuracy was demonstrated for the detection of low CI (defined as less than the median value; AUC at ROC analysis 0.91 and 0.92, respectively) and good accuracy for the detection of low PVRI (defined as less than the median value; AUC at ROC analysis 0.88). PTT was also accurate for detection of low PVRI (AUC 0.84). FWHM was less accurate for detecting elevated mRAP (AUC 0.77), mPAP (AUC 0.76), and lower mVO2 (AUC 0.83). See Figures 3 and 4.
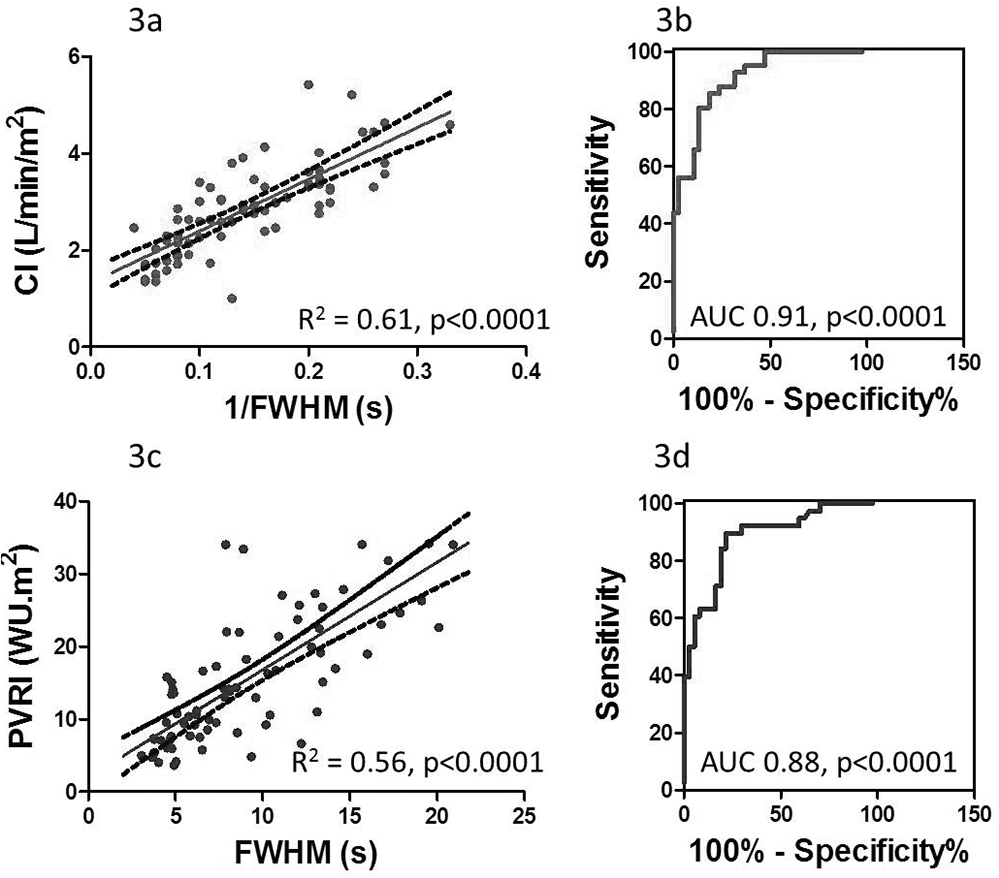
Scatterplots showing the relationship of 1/full width at half maximum (FWHM) versus cardiac index (CI; a) and FWHM versus pulmonary vascular resistance index (PVRI; c). Receiver operating characteristic curves: showing the accuracy of FWHM for the detection of CI ≤ median values (b) and PVRI ≥ median values (d). AUC: area under the curve; WU: Wood units. A color version of this figure is available online.
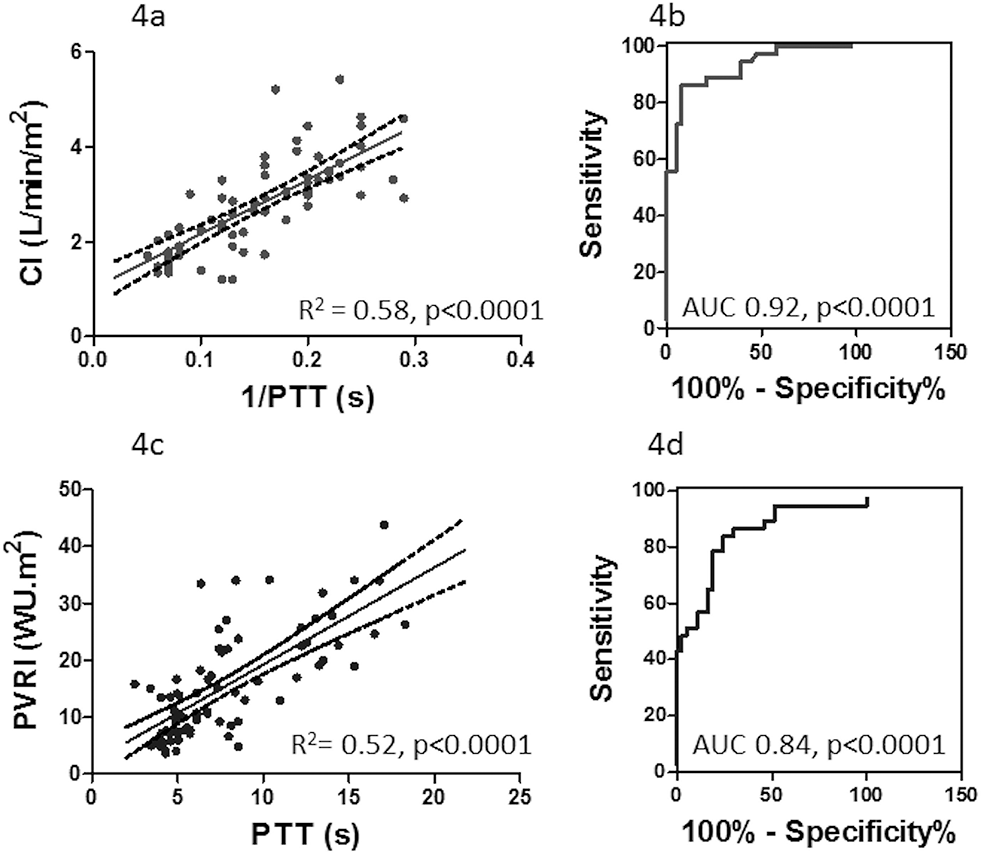
Scatterplots showing the relationship of 1/pulmonary transit time (PTT) versus cardiac index (CI; a) and PTT versus pulmonary vascular resistance index (PVRI; c). Receiver operating characteristic curves: showing the accuracy of PTT for the detection of CI ≤ median values (b) and PVRI ≥ median values (d). AUC: area under the curve; WU: Wood units. A color version of this figure is available online.
Survival analysis
Cox proportional hazards regression analysis. During the follow-up period to death or census, 25 patients had died; these were significantly older (P = 0.020) and had lower CO (P = 0.014) and prolonged PTT (P = 0.007) when compared to patients who survived to census. CO (P = 0.006) and PTT (P = 0.010) were the strongest predictors of mortality from Cox proportional hazards analysis. CI (P = 0.011), FWHM (P = 0.013), WHO functional class (P = 0.028), age (P = 0.034), and PVR (P = 0.043) were also significantly associated with adverse outcome at Cox regression analysis (Table 3).
Univariate Cox proportional hazards regression analysis
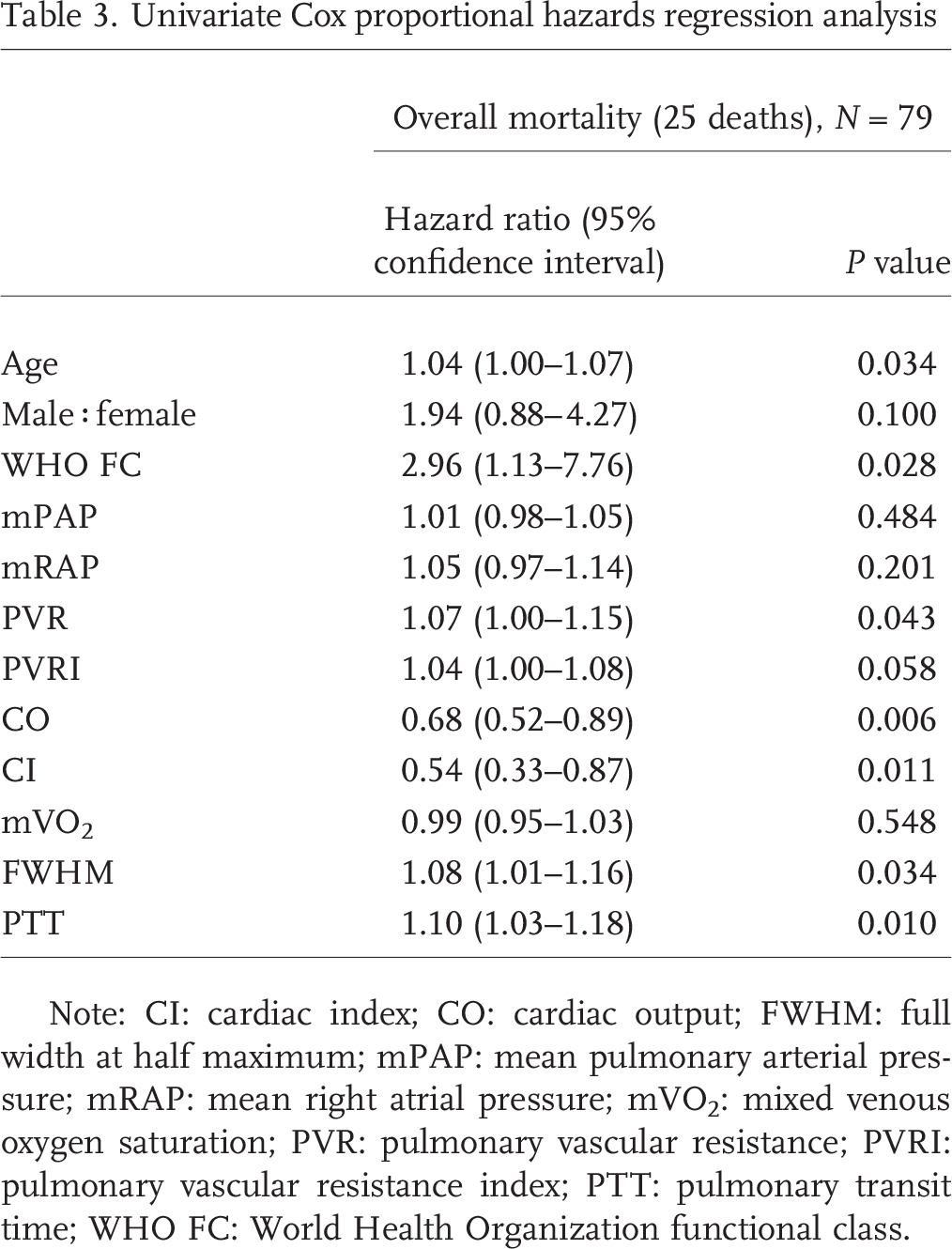
Note: CI: cardiac index; CO: cardiac output; FWHM: full width at half maximum; mPAP: mean pulmonary arterial pressure; mRAP: mean right atrial pressure; mVO2: mixed venous oxygen saturation; PVR: pulmonary vascular resistance; PVRI: pulmonary vascular resistance index; PTT: pulmonary transit time; WHO FC: World Health Organization functional class.
Tables 4 and 5 present the results of bivariate analysis of PTT and FWHM on covariate predictors of mortality. Both PTT and FWHM predicted mortality independent of age, gender, and WHO functional class. However, the prognostic value of DCE indexes was not independent of invasive hemodynamic measurements of PVR or CO.
Bivariate Cox proportional hazards regression analysis of mortality predictors versus full width at half maximum (FWHM)
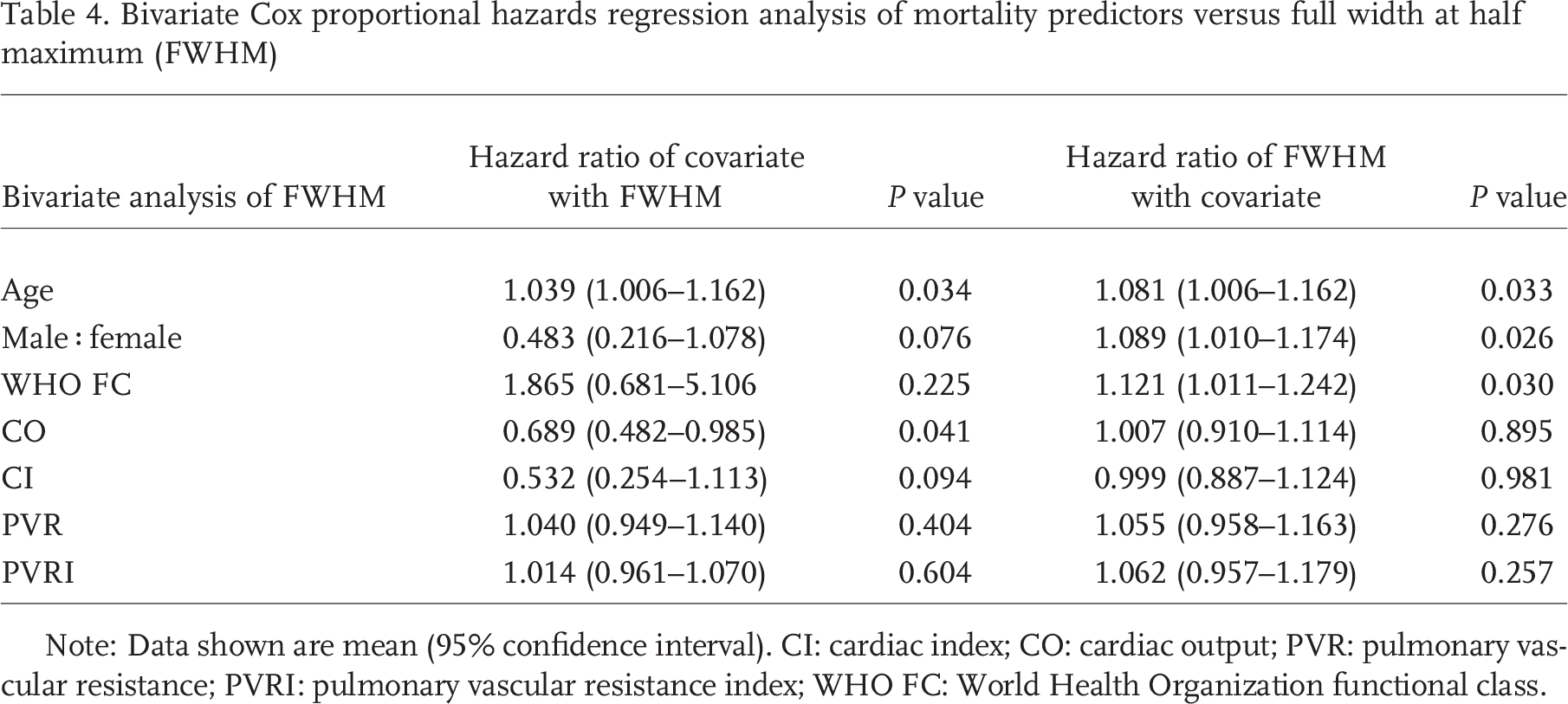
Note: Data shown are mean (95% confidence interval). CI: cardiac index; CO: cardiac output; PVR: pulmonary vascular resistance; PVRI: pulmonary vascular resistance index; WHO FC: World Health Organization functional class.
Bivariate Cox proportional hazards regression analysis of mortality predictors versus pulmonary transit time (PTT)
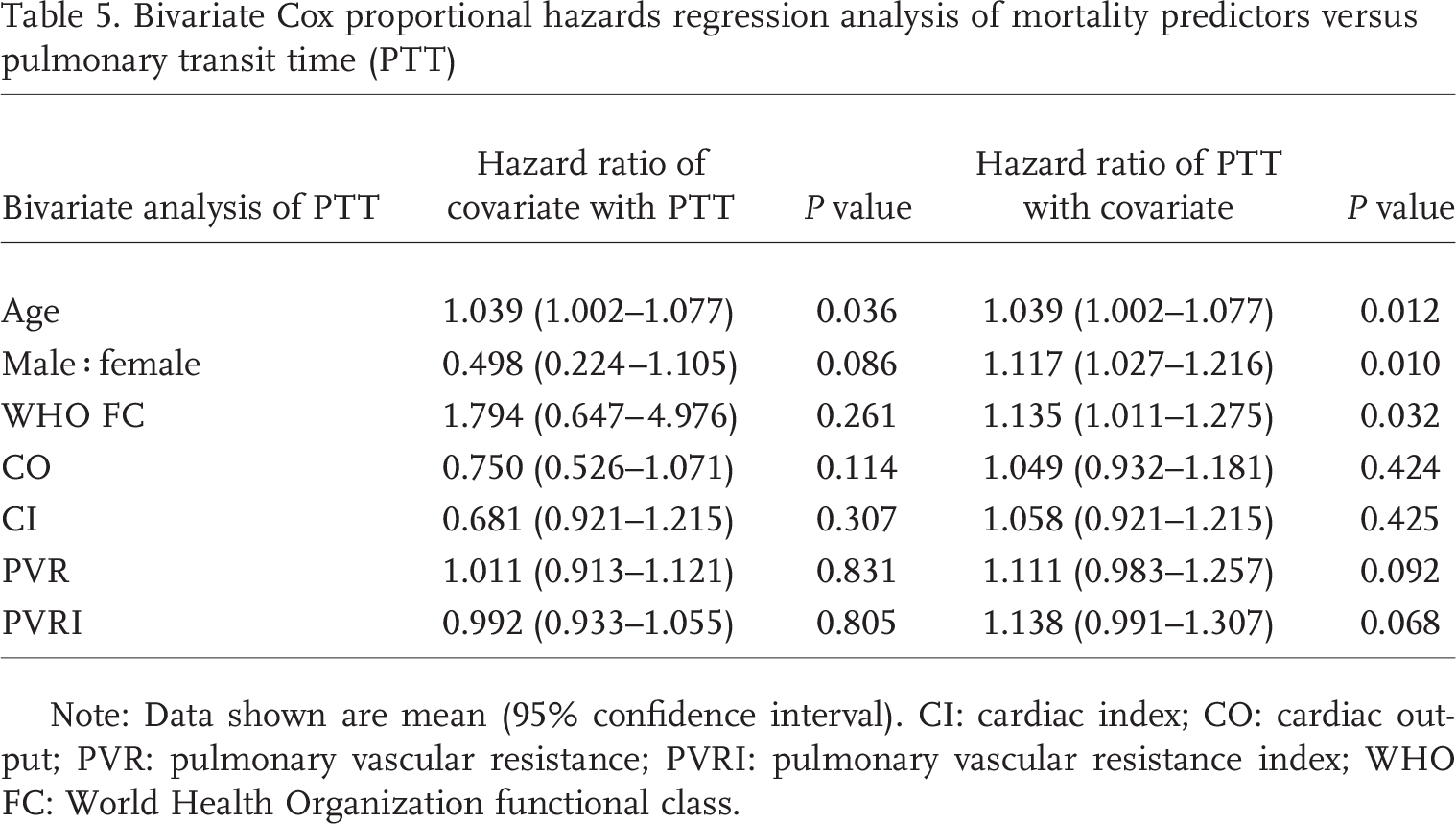
Note: Data shown are mean (95% confidence interval). CI: cardiac index; CO: cardiac output; PVR: pulmonary vascular resistance; PVRI: pulmonary vascular resistance index; WHO FC: World Health Organization functional class.
Kaplan-Meier analysis. Table 6 presents the Kaplan-Meier analysis results for demographic, hemodynamic, and DCE-MR indexes. Patients with PTT greater than the median value (>6.5 s) were associated with a poor outcome when compared to those with PTT ≤6.5 s (P = 0.004). FWHM >8 s was also associated with poor outcome (P = 0.014) at Kaplan-Meier analysis (Fig. 5).
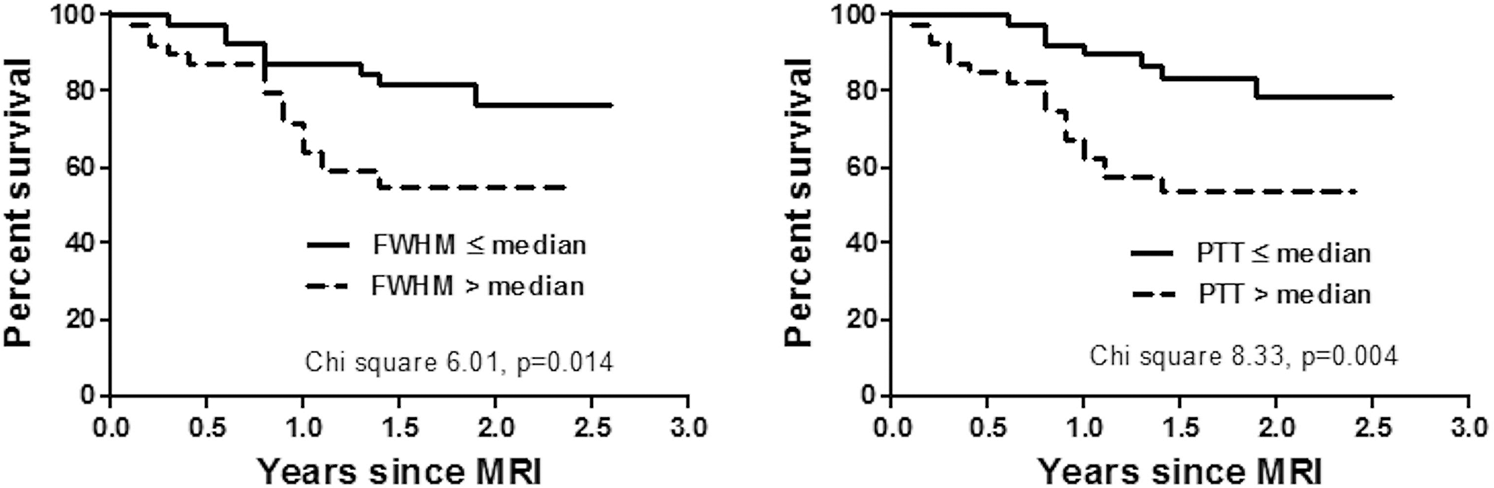
Kaplan-Meier plots for pulmonary transit time (PTT) and full width at half maximum (FWHM) above and below median values. MRI: magnetic resonance imaging.
Kaplan-Meier analysis
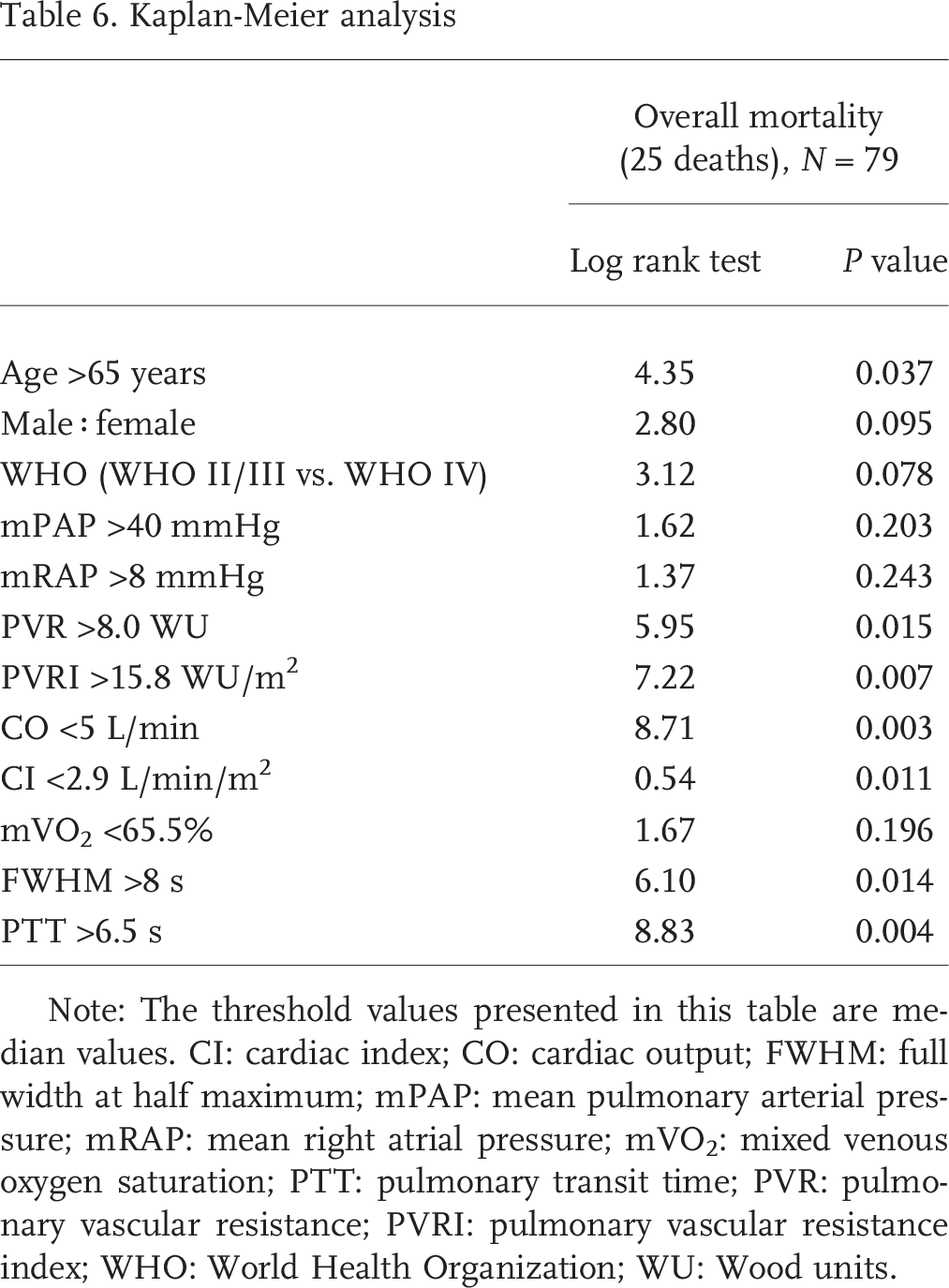
Note: The threshold values presented in this table are median values. CI: cardiac index; CO: cardiac output; FWHM: full width at half maximum; mPAP: mean pulmonary arterial pressure; mRAP: mean right atrial pressure; mVO2: mixed venous oxygen saturation; PTT: pulmonary transit time; PVR: pulmonary vascular resistance; PVRI: pulmonary vascular resistance index; WHO: World Health Organization; WU: Wood units.
Reproducibility
The DCE parameters FWHM and PTT both showed good interobserver reproducibility (intraclass correlation coefficient [ICC] 0.993 [range 0.979–0.997] and 0.960 [0.902–0.984], respectively). Bland-Altman plots showed good interobserver variability; measurement of FWHM had a bias of 0.3 s (SD 0.6), with limits of agreement of −1.3 to 0.8 s. PTT showed a bias of 0.1 s (SD 0.9), with limits of agreement of −1.8 to 1.7 s. In addition, good intraobserver variability was demonstrated for both FWHM and PTT (ICC 0.982 [0.957–0.993] and 0.993 [0.983–0.997], respectively).
DISCUSSION
This study assessed the value of DCE-MR TTs in the prognostic assessment of patients with PAH. PTT and FWHM were found to be most strongly associated with established invasive hemodynamic prognostic markers, namely, CI and PVRI. Prolonged transit of contrast measured by PTT and FWHM predicted mortality. Specifically, patients with a PTT >6.5 s and FWHM >8 s were at higher risk of adverse outcome.
Our study demonstrates a strong and significant inverse relationship between CI and DCE TTs in patients with PAH. Furthermore, FWHM and PTT measurements were highly accurate for detecting reduced CI (AUC at ROC analysis 0.91 and 0.92, respectively). The definition of TT of a contrast bolus from dilution theory is equal to the volume distribution of the contrast divided by the blood flow. 18 The volume distribution of contrast will be influenced by the volume of the vessels and heart but also the volume of contrast injected. Large right-sided cardiac volume and low CO are associated with longer TTs, a situation that will be encountered in patients with severe PAH. Given that low CO3,4 and large RV volume 19 have both been linked with poor outcome in PAH, the prognostic significance of DCE TTs is not entirely unexpected. Figure 6 shows example signal-time curves from a patient with severe pulmonary hypertension with prolonged transit and a patient with rapid transit of contrast.
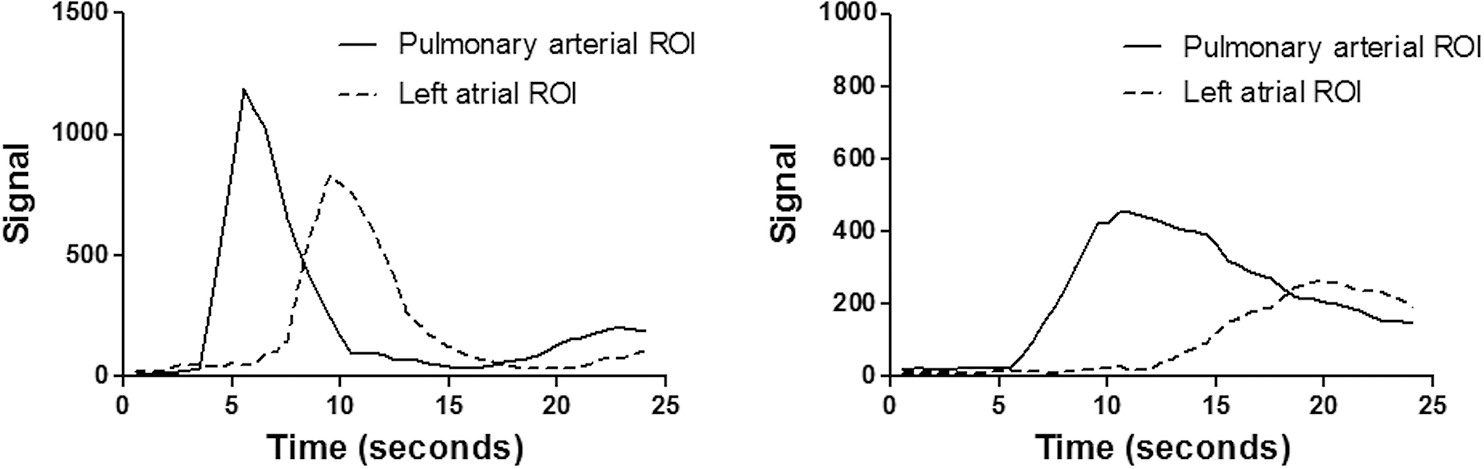
Time intensity curves from a patient with severe pulmonary hypertension (right) showing prolonged transit of contrast through both the pulmonary artery and the left atrium. The left-hand image shows signal-time curves from a patient without pulmonary hypertension with relatively rapid contrast transit (mean pulmonary arterial pressure <25 mmHg). ROI: region of interest.
The results have shown a significant linear relationship between DCE TT measurements and PVR, supporting work that has been performed previously.13,14 In addition, established predictors of mortality mRAP and mVO2 also correlated significantly with DCE data. At bivariate analysis, DCE TT measurements were found to predict mortality independent of age, gender, and WHO functional class. However, the prognostic significance of DCE indexes were not independent of hemodynamic indexes of PVRI and CI, which are important variables in assessment of disease severity and risk stratification for patients with PAH. 20 One explanation is the similar nature of the information derived from DCE TTs and hemodynamic indexes, masking the prognostic significance of both TT and hemodynamic indexes at bivariate analysis.
Alterations in DCE TTs have been shown in patients with chronic obstructive pulmonary disease; worsening spirometric indexes and increasing severity of emphysema have been shown to relate to prolonged pulmonary contrast TTs, 21 presumably as a result of increased vascular resistance from capillary damage combined with increased resistance associated with surrounding airway hyperinflation. Sergiacomi et al. 14 studied a cohort of patients with PH associated with combined emphysema and interstitial lung disease. DCE TT values were measured using ROIs placed in the distal main PAs. The authors identified good linear correlations between DCE TTs and both CI and PVR. In contrast, weaker correlation between MR contrast TTs versus mPAP has been found in this study. Hence, we recommend the use of DCE bolus hemodynamics in the assessment of disease severity and risk stratification rather than for the identification of PAH on the basis of the results of our study. Quantitative DCE parameters of blood flow and blood volume have been derived in previous studies.8,9,13 These parameters were calculated using indicator dilution theory for intravascular contrast agents where the arterial input function of the contrast bolus is determined. Other studies have focused solely on the first-pass contrast bolus TTs without deconvolution of the arterial input function.5,14,15 The association of DCE-MR TTs with left ventricular (LV) function has been studied,5,22 and cardiopulmonary TTs have been shown to be significantly prolonged in heart failure, correlating directly with LV end-diastolic and end-systolic volumes and inversely with LV ejection fraction. 5 Skrok et al. 15 studied the association of DCE TTs with RV function and invasive pulmonary hemodynamics, suggesting that such measurements may prove useful markers in the prognostic evaluation of patients with PAH. This study corroborates that hypothesis, demonstrating that DCE TTs are of prognostic value in PAH. Other MR measurements associated with adverse outcome in PAH include a large right ventricle, small left ventricle, and low stroke volume measured using dynamic cardiac MR imaging. 19 In addition MR imaging–derived PA pulsatility23,24 and PA black blood imaging have also proved useful in the prognostic evaluation of patients with pulmonary hypertension. 25
Our DCE TT measurements were found to be reproducible. FWHM was the more reproducible of the two measurements, with higher ICC and narrower limits of agreement. One explanation for this is that FWHM requires only a single ROI measurement, whereas PTT requires ROI placement in both the PA and the LA. The addition of the second measurement is one explanation for the reduced reproducibility. A second explanation for the lower reproducibility of PTT could be the shape of the left atrial signal-time curve, having lower signal intensity and a broader peak; thus, identification of the exact time of the peak signal is potentially more difficult. Both parameters can be automatically generated by parametric pixel-pixel curve fitting (e.g., Gamma variate), and the robustness of this automated process is currently being assessed for future routine clinical practice.
The lack of ionizing radiation and the noninvasive nature of DCE measurements are potential advantages over invasive prognostic indicators in the prognostic assessment of patients with PAH. This study has not shown the incremental prognostic value of DCE measurements, likely owing to the similarity of the information derived from both invasive hemodynamic measurements. Another advantage is that these measurements can be acquired in addition to the standard sequences recommended as part of an MR imaging protocol for assessment of patients with PAH, adding weight to the role of MR imaging as the future “one-stop shop” for the assessment of treatment response and failure. An MR imaging protocol including assessment of the cardiac morphology and function and pulmonary flow analysis, with the addition of DCE imaging, may provide a robust noninvasive alternative in the clinic.
Limitations
This was a single-center study, the results of which should be reproduced in other centers to validate the prognostic value of the DCE hemodynamic measurements. A bivariate model was used to test the independent prognostic significance of DCE indexes. Due to the small number of deaths, multivariate Cox proportional hazards regression analysis has been performed. All images were acquired at inspiratory breath hold. It is known that the phase of respiration and lung inflation affects the rate of passage of contrast medium through the pulmonary vasculature. Fink et al. 26 demonstrated shorter TTs and greater perfusion in expiration than in inspiration. Furthermore, pulmonary DCE hemodynamic measurements could be significantly affected if the subject performed a Valsalva maneuver during the acquisition of the MR imaging scan; the resultant increase in intrathoracic pressure would reduce venous return to the heart and alter contrast flow dynamics.27,28 It is therefore important to educate the patient for lung inflation by practicing breathing maneuvers prior to scanning and provide adequate instruction during the MR scan. Standardization of contrast injection protocol for intersite comparisons is required, and further work to determine the utility of dynamic contrast measurements in patient follow-up would be desirable.
Conclusions
DCE-MR TTs are closely related to the CI and the PVRI and have prognostic significance in patients with PAH.
